
False Killer Whale
Pseudorca crassidens

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Pseudorca crassidens – (Owen, 1846)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – DELPHINIDAE – Pseudorca – crassidens
Common Names: False Killer Whale (English), Valsmoordvis (Afrikaans), Falsa Orca (Spanish; Castilian), Pseudorca (Italian), Pseudorque (French)
Synonyms: Phocaena crassidens Owen, 1846
Taxonomic Note:
As the only species of its genus, the False Killer Whale was initially described from a sub-fossil skeleton found on the British Isles by Owen in 1846 (Odell & McClune 1999). No subspecies have been described, and the initial proposal of a distinction between northern and southern forms of False Killer Whales (Leatherwood et al. 1991) was later discredited based on the investigation of adult skeletons (Stacey et al. 1994). However, analyses of skull and dental morphology have revealed some degree of regional differentiation and evidence that this species occurs as several disjunct populations across the globe (Kitchener et al. 1990; Ferreira 2008). Genetic variation is not uncommon in cetaceans (Kitchener et al. 1990; Connor et al. 2000), and is likely attributed to changes in water temperature and prey distribution. Results exhibiting geographic variation in body size were found between Japanese and southern African populations, where Japanese specimens were significantly larger in comparison (Ferreira 2008), confirming previous suggestions that Southern Hemisphere populations are typically smaller and reach sexual maturity at shorter body lengths, compared to those of the northern hemisphere (Purves & Pilleri 1978; Kasuya 1986). Using mitochondrial DNA (mtDNA) control region sequence data Chivers et al. (2007) describe a demographically isolated population of False Killer Whales in the waters off Hawaii, in the eastern North Pacific.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Nadin, C.E.1, & da Silva, J. M.2
Reviewer: Purdon, J.3,4
Institutions: 1Sea Search Research and Conservation, 2South African National Biodiversity Institute, 3TUT Nature Conservation, 4The Whale Unit, University of Pretoria
Previous Assessors: Taylor, B.L., Baird, R., Barlow, J., Dawson, S.M., Ford, J., Mead, J.G., Notarbartolo di Sciara, G., Wade, P. & Pitman, R.L.
Previous Reviewer: Photopoulou, T.
Previous Contributors: Relton, C., Elwen, S., Findlay, K., Plön, S., Oosthuizen, H. & Meyer, M.
Assessment Rationale
Global and regional population trends and abundance data is unavailable for this species, and it is considered rare in the waters of the assessment region. Although, occasional mass stranding events have been documented in South Africa, it is suspected that these are accredited to natural causes, rather than anthropogenic activities. No major threats that may cause substantial population depletion, have been identified, resultantly, this species is listed as Least Concern, in line with the global assessment. However, considering the rarity and low reproductive potential of the False Killer Whale, it may be particularly vulnerable to minor threats, including fisheries bycatch (especially longline fisheries) and persecution, competition for prey resources, climate change and anthropogenic pollution. Continued research into potential risks, population abundance and distribution, as well as the identification of critical habitats may be necessary.
Regional population effects: The False Killer Whale is a wide-ranging pelagic cetacean, with a continuous distribution and no obvious barriers to dispersal, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citations: Nadin CE & da Silva JM. 2025. A conservation assessment of Pseudorca crassidens. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
False Killer Whales are widely distributed across the globe, predominantly occurring within deep tropical and warm temperate regions (usually in waters more than 25°C), but unlike the Killer Whale (Orcinus orca), they only very occasionally roam into colder waters (below 20°C) (Mitchell 1975). Their worldwide range is thought to extend from 50°N to 50°S (Odell & McClune 1999). Within southern African waters, this species has been documented off the coast of Lüderitz, Namibia, and from St Helena Bay to the north coast of KwaZulu-Natal (Findlay 1989). A pod of six was recorded off the coast of KwaZulu-Natal, at 29°02’ S; 32°02’ E (Bruyns 1969). This species has been frequently sighted in association with large groups of Common Bottlenose Dolphins (Tursiops truncatus) in the waters of Plettenberg Bay, Eastern Cape.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
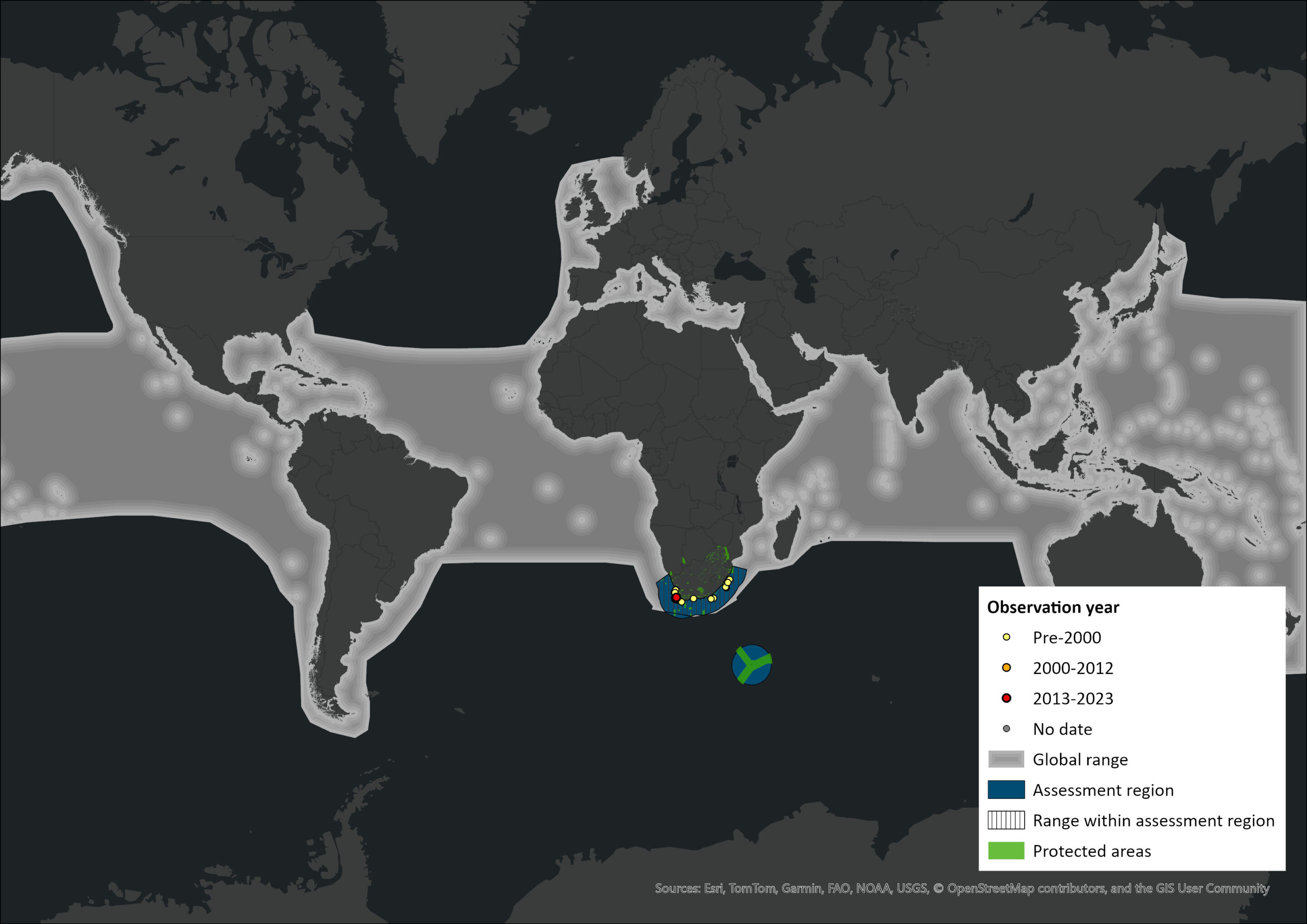 Figure 1. Distribution records for False Killer Whale (Pseudorca crassidens) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for False Killer Whale (Pseudorca crassidens) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Australasian, Indomalayan, Nearctic, Neotropical, Oceanian, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| American Samoa | Extant | Native | – | – |
| Angola | Presence Uncertain | Native | – | – |
| Anguilla | Extant | Native | – | – |
| Antigua and Barbuda | Extant | Native | – | – |
| Argentina | Extant | Native | – | – |
| Aruba | Extant | Native | – | – |
| Australia | Extant | Native | – | – |
| Bahamas | Extant | Native | – | – |
| Bangladesh | Extant | Native | – | – |
| Barbados | Extant | Native | – | – |
| Belgium | Presence Uncertain | Native | – | – |
| Belize | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Bermuda | Extant | Native | – | – |
| Bonaire, Sint Eustatius and Saba | Extant | Native | – | – |
| Bosnia and Herzegovina | Presence Uncertain | Native | – | – |
| Brazil | Extant | Native | – | – |
| British Indian Ocean Territory | Extant | Native | – | – |
| Brunei Darussalam | Extant | Native | – | – |
| Cabo Verde | Extant | Native | – | – |
| Cambodia | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Canada | Extant | Native | – | – |
| Cayman Islands | Extant | Native | – | – |
| Chile | Extant | Native | – | – |
| China | Extant | Native | – | – |
| Christmas Island | Presence Uncertain | Native | – | – |
| Cocos (Keeling) Islands | Extant | Native | – | – |
| Colombia | Extant | Native | – | – |
| Comoros | Presence Uncertain | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Cook Islands | Extant | Native | – | – |
| Costa Rica | Extant | Native | – | – |
| Croatia | Extant | Native | – | – |
| Cuba | Extant | Native | – | – |
| Cyprus | Presence Uncertain | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Denmark | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Dominica | Extant | Native | – | – |
| Dominican Republic | Extant | Native | – | – |
| Ecuador | Extant | Native | – | – |
| Egypt | Extant | Native | – | – |
| El Salvador | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Presence Uncertain | Native | – | – |
| Fiji | Extant | Native | – | – |
| France | Extant | Native | – | – |
| French Guiana | Extant | Native | – | – |
| French Polynesia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Germany | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Gibraltar | Extant | Native | – | – |
| Greece | Extant | Native | – | – |
| Grenada | Extant | Native | – | – |
| Guadeloupe | Extant | Native | – | – |
| Guam | Extant | Native | – | – |
| Guatemala | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Guyana | Extant | Native | – | – |
| Haiti | Extant | Native | – | – |
| Honduras | Extant | Native | – | – |
| Hong Kong | Extant | Native | – | – |
| India | Extant | Native | – | – |
| Indonesia | Extant | Native | – | – |
| Iran, Islamic Republic of | Extant | Native | – | – |
| Iraq | Presence Uncertain | Native | – | – |
| Ireland | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Italy | Extant | Native | – | – |
| Jamaica | Extant | Native | – | – |
| Japan | Extant | Native | – | – |
| Jordan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Kiribati | Extant | Native | – | – |
| Korea, Democratic People’s Republic of | Presence Uncertain | Native | – | – |
| Korea, Republic of | Presence Uncertain | Native | – | – |
| Kuwait | Extant | Native | – | – |
| Lebanon | Presence Uncertain | Native | – | – |
| Liberia | Extant | Native | – | – |
| Libya | Presence Uncertain | Native | – | – |
| Madagascar | Extant | Native | – | – |
| Malaysia | Extant | Native | – | – |
| Maldives | Extant | Native | – | – |
| Malta | Extant | Native | – | – |
| Marshall Islands | Extant | Native | – | – |
| Martinique | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mauritius | Presence Uncertain | Native | – | – |
| Mayotte | Presence Uncertain | Native | – | – |
| Mexico | Extant | Native | – | – |
| Micronesia, Federated States of | Extant | Native | – | – |
| Monaco | Presence Uncertain | Native | – | – |
| Montenegro | Presence Uncertain | Native | – | – |
| Montserrat | Presence Uncertain | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Myanmar | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nauru | Presence Uncertain | Native | – | – |
| Netherlands | Extant | Native | – | – |
| New Caledonia | Extant | Native | – | – |
| New Zealand | Extant | Native | – | – |
| Nicaragua | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Niue | Extant | Native | – | – |
| Norfolk Island | Presence Uncertain | Native | – | – |
| Northern Mariana Islands | Extant | Native | – | – |
| Norway | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Palau | Extant | Native | – | – |
| Palestine, State of | Presence Uncertain | Native | – | – |
| Panama | Extant | Native | – | – |
| Papua New Guinea | Extant | Native | – | – |
| Peru | Extant | Native | – | – |
| Philippines | Extant | Native | – | – |
| Pitcairn | Extant | Native | – | – |
| Poland | Presence Uncertain | Native | – | – |
| Portugal | Extant | Native | – | – |
| Puerto Rico | Extant | Native | – | – |
| Qatar | Extant | Native | – | – |
| Russian Federation | Presence Uncertain | Native | – | – |
| Réunion | Presence Uncertain | Native | – | – |
| Saint Barthélemy | Extant | Native | – | – |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | – |
| Saint Kitts and Nevis | Extant | Native | – | – |
| Saint Lucia | Extant | Native | – | – |
| Saint Martin (French part) | Extant | Native | – | – |
| Saint Pierre and Miquelon | Extant | Native | – | – |
| Saint Vincent and the Grenadines | Extant | Native | – | – |
| Samoa | Extant | Native | – | – |
| Sao Tome and Principe | Extant | Native | – | – |
| Saudi Arabia | Presence Uncertain | Native | – | – |
| Senegal | Extant | Native | – | – |
| Seychelles | Presence Uncertain | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Singapore | Extant | Native | – | – |
| Sint Maarten (Dutch part) | Extant | Native | – | – |
| Slovenia | Presence Uncertain | Native | – | – |
| Solomon Islands | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Spain | Extant | Native | – | – |
| Sri Lanka | Extant | Native | – | – |
| Sudan | Presence Uncertain | Native | – | – |
| Suriname | Extant | Native | – | – |
| Sweden | Presence Uncertain | Native | – | – |
| Syrian Arab Republic | Extant | Native | – | – |
| Taiwan, Province of China | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Thailand | Extant | Native | – | – |
| Timor-Leste | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Tokelau | Extant | Native | – | – |
| Tonga | Extant | Native | – | – |
| Trinidad and Tobago | Extant | Native | – | – |
| Tunisia | Presence Uncertain | Native | – | – |
| Turkmenistan | Extant | Native | – | – |
| Turks and Caicos Islands | Extant | Native | – | – |
| Tuvalu | Presence Uncertain | Native | – | – |
| United Arab Emirates | Extant | Native | – | – |
| United Kingdom of Great Britain and Northern Ireland | Extant | Native | – | – |
| United States of America | Extant | Native | – | – |
| Uruguay | Extant | Native | – | – |
| Vanuatu | Extant | Native | – | – |
| Venezuela, Bolivarian Republic of | Extant | Native | – | – |
| Viet Nam | Extant | Native | – | – |
| Virgin Islands, British | Extant | Native | – | – |
| Virgin Islands, U.S. | Extant | Native | – | – |
| Wallis and Futuna | Extant | Native | – | – |
| Western Sahara | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 21. Atlantic – northwest | Extant | Native | – | – |
| 27. Atlantic – northeast | Extant | Native | – | – |
| 31. Atlantic – western central | Extant | Native | – | – |
| 34. Atlantic – eastern central | Extant | Native | – | – |
| 37. Mediterranean and Black Sea | Extant | Native | – | – |
| 41. Atlantic – southwest | Extant | Native | – | – |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 51. Indian Ocean – western | Extant | Native | – | – |
| 57. Indian Ocean – eastern | Extant | Native | – | – |
| 61. Pacific – northwest | Extant | Native | – | – |
| 67. Pacific – northeast | Extant | Native | – | – |
| 71. Pacific – western central | Extant | Native | – | – |
| 77. Pacific – eastern central | Extant | Native | – | – |
| 81. Pacific – southwest | Extant | Native | – | – |
| 87. Pacific – southeast | Extant | Native | – | – |
Climate change
No modelling of potential climate impacts has been conducted on animals within the assessment region, however, climate stressors – including sea level rise and ocean acidification – are the most rapidly increasing stressors within this species’ range (Purdon et al. 2020a). In regions where studied, False Killer Whales have been suggested to be adversely affected by ocean acidification (Lawler et al. 2007), whilst predicted CO2 level rises may seriously affect epipelagic squid abundance (Fabry et al. 2008), a vital food source for the False Killer Whale. Oceanic CO2 (pCO2 sw) partial pressures are positively correlated with temperature, which is increasing in the Agulhas ecosystem, and the Agulhas eddies is likely a primary contributor to the rapid acidification of the South Atlantic Central Water (Orselli et al. 2019). Increases in oceanic temperature, with almost 99% of the whole combined area of African Large Marine Ecosystems having warmed (Sweijd and Smit 2020), may also influence the distribution of pelagic fish species which are also important prey for False Killer Whales.
Salinity and chlorophyll a levels have been shown to be the most influential variables determining the distribution of False Killer Whales in South African waters, with salinity contributing to 63% of the predicted occurrence (Purdon et al. 2020b). General declines in chlorophyll-a concentrations have been documented, with statistically significant rates apparent in the Agulhas (Hernandez et al, 2017), which could negatively impact False Killer Whales in the assessment region if declining trends in chlorophyll-a concentration continue.
Although False Killer Whales face increasing climate threats, the level of protection of this species in the study region has increased from 2% to 7% since the approval of more Marine Protected Areas (MPAs) and most of their offshore habitat is now protected by recently approved MPAs in the Cape Canyon, Namaqua National Park and around Robben Island. The Natal bioregion, which offers an ideal habitat for cephalopod prey species and has a high predicted occurrence for False Killer Whales, now has three MPAs protecting this continental shelf edge (Purdon et al. 2020b). These MPAs will help to prevent over-fishing in these ecologically important areas for False Killer Whales in Southern African waters, which has been suggested to have negative impacts on cetaceans when combined with climate stressors.
Population
There are no global estimates of abundance available for this species, however approximately 39,800 (CV = 64%) individuals have been estimated in the eastern tropical Pacific (Wade & Gerrodette 1993), about 16,000 (CV = 26%) within the coastal waters of China and Japan (Miyashita 1993), and around 1,038 (CV = 71%) in the northern Gulf of Mexico (Mullin & Fulling 2004).
There are no estimates of abundance for the assessment region, as very little sighting data exists. However, eight mass stranding events have been documented on South Africa’s west coast, with the first one being documented in 1928 with over 100 stranded and the most recent one in 2009 with 55 stranded individuals.
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
To date, a comprehensive population genomic study of P. crassidens across its entire distribution has not been undertaken to identify population structure and connectivity. Most studies have been local in scope (e.g., Tezanos-Pinto et al. 2024) and no population genetic study has been conducted on animals within the assessment region. Despite this, population genetic structure is expected given the fact that considerable morphological divergence in skull morphology has been found between animals from different regions (e.g., Kitchener et al. 1990; Baird et al. 2009; Ferreira et al. 2014). With that said, only a single subpopulation of the species is thought to exist within the assessment region.
Research including populations inhabiting the eastern North Pacific Ocean (ENP), the central, western and eastern North Pacific, Indian, and Atlantic oceans does infer ocean-basin-scale population structure, with distinct haplotype sets identified in each region, notably the 10 base pair minimum difference documented between Indo-Pacific Ocean and Atlantic Ocean haplotypes (Chivers et al., 2007; Martien et al. 2014).). Where studied, False Killer Whales display low genetic diversity and high phylogeographic concordance, associated with their matrilineal social structures and limited female dispersal (Chivers et al. 2007; Martien et al. 2014; 2019; Palmer et al. 2023; Tezanos-Pinto 2024).
There are no estimates of population size for these species in the assessment region, preventing an estimate of effective population size being calculated. However, a low effective population size is expected in this species, linked to high fidelity to natal social groups, and an effective population size of 58 individuals has been documented in more studied, isolated populations in Hawaii (Martien et al., 2019). This population size is nonetheless concerning as lethal or semi-lethal genetic traits begin to be displayed when the population size declines to approximately 50 individuals.
Of noting, P. crassidens can hybridise with the Bottlenose Dolphin (Tursiops truncatus) and produce fertile offspring called “wholphins” (e.g., Baird et al. 2009).
Habitats and ecology
Very little is known about this pelagic species, and much of what has been described has been opportunistically collected during mass stranding events or from captive individuals (Ferreira 2008). More commonly located in deep, open waters exceeding 1,000 m, this species only sporadically enters shallower regions of the continental shelf or the waters around oceanic islands (Baird 2016).
False Killer Whales are considered gregarious, exhibiting cohesive social structures and long-term associations between individuals (Baird et al. 2008). Occurring in pods of between 20 and 100 individuals, subdivided into smaller family groups (Bruyns 1969; Baird 2002), individuals regularly interact with one another (Baird 2009). In South African waters, Findlay et al. (1992) recorded groups up to about 50, with an average of 16, but documented one incidental sighting of 68 individuals. False Killer Whales have a tendency to share prey resources amongst members of the same group, and will transport prey resources in their mouths for prolonged periods (Baird et al. 2008).
This species is exceptionally active during the day, usually hunting within surface waters (Baird 2013). Although little information is available documenting the diving behaviour of False Killer Whales, dive data from individuals tagged off Hawaii revealed that they spend a large proportion of time near the surface, however during infrequent deep dives, one individual reached a depth of over 1,000 m (Baird 2013). They are considered opportunistic hunters, and depending on their range, False Killer Whales primarily prey upon a variety of squid and fish, including Dorado (Coryphaena hippurus), tuna (Alonso et al. 1999; Odell & McClune 1999) and sailfish. Ross (1984) assessed the stomach contents of an individual caught in southern African waters, which contained the remains of cephalopods, mostly Todarodes angolensis. Additionally, Sekiguchi et al. (1992) found that the stomachs of 13 individuals contained a range of cephalopod species, but no fish remains. While, around the Hawaiian Islands, they feed predominantly on large commercially and recreationally harvested game fish (Gilman et al. 2007). There are also rare records of False Killer Whales feeding on smaller cetaceans (Odell & McClune 1999).
False Killer Whales have been recorded reaching speeds of approximately 30 km/hr for short durations (Williams 2009), and frequently approach ships, engaging in bowriding behaviour. In comparison to Pygmy Killer Whales (Feresa attenuata), with which they are often confused, False Killer Whales often engage in high-speed travel, while Pygmy Killer Whales are considered fairly lethargic in comparison. During a surface sighting, the most obvious difference between the two species is the size of the dorsal fin relative to its back, as Pygmy Killer Whales have proportionately larger dorsal fins (Baird et al. 2010).
This species is considered the most aggressive cetacean in captivity, and will attack other cetaceans or equipment (Defran & Pryor 1980). In the wild, they have been documented attacking dolphins around purse-seine tuna fisheries in the eastern Pacific (Perryman & Foster 1980). Additionally, reports of False Killer Whales damaging Japanese long-line fisheries are not unusual (Mitchell 1975). Non-aggressive associations between False Killer Whales and Common Bottlenose Dolphins are common in the wild, and a number of unsuccessful instances of hybridisation between the two species has occurred in captivity.
As a long-lived, slow-maturing species, P. crassidens have low reproductive potential. Results of a study conducted on False Killer Whales stranded on South Africa’s west coast revealed that females reach sexual maturity at an age of between 9 and 10.5 years, at lengths of approximately 3.25 m, which is shorter by 30 cm than individuals from Japanese waters (3.59 m) (Ferreira 2008). Males have been reported to reach sexual maturity several years older than females (Ferreira 2008), and at lengths ranging from 3.96 to 4.57 m (Skinner & Chimimba 2005). Additionally, a recent study found that South African False Killer Whales from a stranded group had lower fecundity than Japanese false killer whales harvested during a drive fishery (Photopoulou et al. in review). Some degree of seasonal reproduction is supported by the presence of significant numbers of calves in summer (Purves & Pilleri 1978), but no evidence of seasonality in conception was found in stranded South African individuals (Ferreira 2008). Calves are born at lengths of between 1.73 and 1.83 m, following a gestation period of just over 15 months (Purves & Pilleri 1978). Male and female lifespan has been estimated at 57 years and approximately 62 years, respectively (Photopoulou et al. in review; Kasuya 1986). Along with pilot whales (Globicephala spp.), Killer Whales and common dolphins (Delphinus spp.), False Killer Whales are commonly involved in mass stranding events, although the explanation for these phenomena remains unclear (Ferreira 2008).
Ecosystem and cultural services: As top-level predators on a wide variety of fishes and squids, False Killer Whales concentrate contaminants through bioaccumulation and integrate broadly across the ecosystem in terms of exposure to environmental impacts.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
| 10.2. Marine Oceanic -> Marine Oceanic – Mesopelagic (200-1000m) | – | Suitable | Yes |
| 10.3. Marine Oceanic -> Marine Oceanic – Bathypelagic (1000-4000m) | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Marine
General Use and Trade Information
There is no contemporary trade or use of this species in South Africa.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
– |
– |
– |
National Commercial Value: Yes
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
true |
– |
– |
|
7. Fuels |
true |
true |
– |
– |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
No major threats to this species have been identified within the assessment region, however considering that this species is naturally rare in most regions and some evidence of lower reproductive rates in the southern African region compared to other areas, even low levels of mortality and serious injury, could cause detrimental impacts to local populations. In South Africa, seasonal strandings (between August and December) have been recorded since 1920, all before anthropogenic sounds became a threat to marine animals. As a deep-water species, shallow water may cause disorientation, leading to stranding. Given these facts, it is highly probable that the strandings are not the result of anthropogenic activity and are more likely attributed to natural causes, for example, confusing acoustic reflection within sea canyons. A number of minor threats have been identified:
Fisheries bycatch: Studies have found that this cetacean is more vulnerable than any other to dangerous interactions with Hawaiian long-line fisheries (Forney & Kobayashi 2007). They take fish off long-line hooks and may be incidentally caught, leading to drowning, injury and/or subsequent death when hooks are lodged inside the mouth or gullet (Forney & Kobayashi 2005). In 2005, average rates of depredation of swordfish by False Killer Whales was estimated at 2,999–4,804 in the Atlantic, 509-2,706 in the Indian, and 114–348 in the Pacific Oceans (Ramos -Cartelle & Mejuto 2008). A number of individuals in the coastal waters off Hawaii have scars consistent with wounds inflicted by long-line fishing equipment (Baird & Gorgone 2005). Additionally, this behaviour often leads to persecution and shootings by fishermen, as seen in Killer Whales, in order to decrease depredation and limit economic loss (Ramos-Cartelle & Mejuto 2008).
Competition with fisheries: Overexploitation of large fish species, such as tuna and swordfish by fisheries, causing a decline in prey biomass and size, has been recognised as an additional and increasing threat to this species in Hawaii (Oleson et al. 2010). It is likely that this threat may be extrapolated to other regions of this species’ range, because False Killer Whales target many of the same fish and squid species as commercial fisheries (Ramos-Cartelle & Mejuto 2008).
Anthropogenic pollution: Plastic pollution is a widespread and increasing problem within all oceans. The ingestion of plastic debris has been documented in stranding records of False Killer Whales (Baird 2002) and is a fairly common phenomenon in similar species (Stamper et al. 2006), and those that commonly feed on cephalopods. Finally, the bioaccumulation of persistent organic pollutants (POPs) within the body tissues of False Killer Whales may put this species at risk of diminishing reproductive potential and immunosuppression (Oleson et al. 2010). As long-lived top predators, the risk of exposure to organic pollutants is increased, compared to other species that feed at lower trophic levels (Oleson et al. 2010). Aside from in Hawaii, there is limited data documenting the concentrations of POPs in False Killer Whales. Although reports of high concentrations of toxins contained within the blubber of False Killer Whales stranded off Canada (Jarman et al. 1996), Taiwan (Chou et al. 2004) and Japan (Haraguchi et al. 2006) have been recorded.
Current habitat trend: Declining, due to overexploitation of prey resources by commercial fisheries.
Conservation
The species is listed in Appendix II of the Convention on Internation Trade I Endangered Species of Wild Fauna and Flora (CITES) and protected by the Marine Living Resources Act (No. 18 of 1998) of the national legislation.
Considering the substantial interaction rate and vulnerability of this species to longline fisheries in Hawaiian waters, investigations into the bycatch associated with South Africa’s longline fisheries is imperative for this species. Unfortunately, bycatch is often discarded overboard and unrecorded, therefore hindering the documentation of abundance estimates, and the quantification of this threat. Sustainable mitigation of cetacean bycatch is only possible if accurate records regarding fishing techniques and equipment, geographic distribution, season and quantitative data of bycatch is recorded. Additionally, fatally injured individuals may be valuable for dissection in order to enhance the scientific study into the ecology and morphology of this poorly known species.
The current lack of abundance and distribution data for this species within the assessment region, currently prevents the implementation of species-specific mitigation actions, however, it is likely that this species may benefit from the development and expansion of marine protected areas developed with other cetaceans in mind, as they are frequently sighted in association with other cetacean species. The implementation of seasonal and geographic longline fishery exclusion zones of ‘critical habitat’ in areas of high cetacean concentration, such those developed for False Killer Whales in Hawaii since 1992, may reduce False Killer Whale mortality and injury associated with fishery interactions. Critical habitats should be carefully considered and associated with primary feeding and reproduction areas, which are protected from disturbance (Baird et al. 2012).
Recommendations for managers and practitioners:
- Accurate bycatch assessments in the longline fishery.
- Enforce regulations associated with deep water fisheries, including bycatch mitigation efforts.
- Sightings data should be recorded during systematic monitoring of other marine species.
Research priorities:
- Population size and trend estimates for the assessment region.
- Threats to this species in relation to long-line fisheries.
- Identification of high concentration areas, and critical habitats in South African waters, including distributional limits, seasonal movements and diving behaviour.
- Diet, reproduction and general biology.
- Cumulative impacts of anthropogenic influences, such as pollution, commercial fisheries and persecution.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNauralist and MammalMAP) to help with mapping geographical distribution.
- Use information dispensed by the South African Sustainable Seafood Initiative (SASSI) to make good choices when buying fish in shops and restaurants, e.g. wwfsa.mobi, FishMS 0794998795.
- Buy local products that have not been shipped.
- Avoid using plastic bags.
- Report any stranding reports to the relevant local authorities.
Bibliography
Alonso, M.K., Pedaza, S.N., Schiavini, A.C.M., Goodall, R.N.P. and Crespo, E.A. 1999. Stomach contents of false killer whales (Pseudorca crassidens) stranded on the coasts of the Strait of Magellan, Tierra del Fuego. Marine Mammal Science 15(3): 712-724.
Baird RW. 2013. False Killer whales around Kaua’i and Ni’ihau. Hawai’i Fishing News 39: 24-25.
Baird, R. W. 2002. False killer whale Pseudorca crassidens. In: W. F. Perrin;B. Wursig;J. G. M. Thewissen (ed.), Encyclopedia of Marine Mammals, pp. 411-412. Academic Press.
Baird, R.W. 2009. A review of false killer whales in Hawaiian waters: biology, status, and risk factors. Report prepared for the U.S. Marine Mammal Commission under Order No. E40475499. Cascadia Research Collective Olympia.
Baird, R.W. 2016. The Lives of Hawai’i’s Dolphins and Whales: Natural History and Conservation. University of Hawai’i Press, Honolulu, HI.
Baird, R.W. and Gorgone, A.M. 2005. False killer whale dorsal fin disfigurements as a possible indicator of long-line fishery interactions in Hawaiian waters. Pacific Science 59: 593-601.
Baird, R.W., Gorgone, A.M., McSweeney, D.J., Webster, D.L., Salden, D.R., Deakos, M.H., Ligon, A.D., Schorr, G.S., Barlow, J. and Mahaffy, S.D. 2008. False killer whales (Pseudorca crassidens) around the main Hawaiian Islands: long-term site fidelity, inter-island movements, and association patterns. Marine Mammal Science 24(3): 591-612.
Baird, R.W., Hanson, M.B., Schorr, G.S., Webster, D.L., McSweeney, D.J., Gorgone, A.M., Mahaffy, S.D., Holzer, D.M., Oleson, E.M., and Andrews, R.D. 2012. Assessment of range and primary habitats of Hawaiian insular false killer whales: informing determination of critical habitat. Endangered Species Research 18: 47-61.
Baird, R.W., Schorr, G.S., Webster, D.L., McSweeney, D.J., Hanson, M.B. and Andrews, R.D. 2010. Movements and habitat use of satellite tagged false killer whales around the main Hawaiian Islands. DTIC Document.
Baird, R. W. 2009. “False Killer Whale: Pseudorca crassidens”. In Perrin, W. F.; Würsig, B.; Thewissen, J. G. M. (eds.). Encyclopedia of marine mammals. Academic Press. pp. 405–406. ISBN 978-0-08-091993-5.
Bruyns WFJM. 1969. Sight records and notes on the false killer whale, Pseudorca crassidens (Owen, 1846). Säugetierkundliche Mitteilungen 17: 351-356.
Chivers, S.J., Baird, R.W., McSweeney, D.J., Webster, D.L., Hedrick, N.M. and Salinas, J.C. 2007. Genetic variation and evidence for population structure in eastern North Pacific false killer whales (Pseudorca crassidens). Canadian Journal of Zoology 85: 783-794.
Chou CC, Chen YN, Li CS. 2004. Congener-specific polychlorinated biphenyls in cetaceans from Taiwan waters. Archives of environmental contamination and toxicology 47: 551-560.
Connor, R. C., Wells, R. S., Mann, J., and Read, A. J. 2000. The bottlenose dolphin: social relationships in a fission-fusion society. In: J. Mann, R. C. Connor, P. L. Tyack and H. Whitehead (eds), Cetacean societies: field studies of dolphins and whales, pp. 91-126. University of Chicago Press, Chicago.
Defran RH, Pryor K. 1980. The behaviour and training of cetaceans in captivity. In: Herman LM (ed.), Cetacean Behavior: Mechanisms and Functions, pp. 319-362. John Wiley, New York, USA.
Ferreira IM. 2008. Growth and reproduction in false killer whales (Pseudorca crassidens Owens, 1846). M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Ferreira, I.M., Kasuya, T., Marsh, H. and Best, P.B. 2014. False killer whales (Pseudorca crassidens) from Japan and South Africa: Differences in growth and reproduction. Mar Mam Sci, 30: 64-84. https://doi.org/10.1111/mms.12021
Findlay KP, Best PB, Ross GJB, Cockcroft VG. 1992. The distribution of small odontocete cetaceans off the coasts of South Africa and Namibia. South African Journal of Marine Science 12: 237-270.
Findlay KP. 1989. The distribution of cetaceans off the coast of South Africa and South West Africa/Namibia. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Forney KA, Kobayashi D. 2005. Updated estimates of mortality and injury of cetaceans in the Hawaii-based longline fishery, 1994- 2004. Southwest Fisheries Science Center, National Marine Fisheries Service. La Jolla, CA, USA.
Forney KA, Kobayashi DR. 2007. Updated estimates of mortality and injury of cetaceans in the Hawaii-based longline fishery, 1994- 2005. NOAA Technical Memorandum NMFS. Southwest Fisheries Science Center.
Gilman, E., Clarke, S., Brothers, N., Alfaro-Shigueto-J., Mandelman, J., Mangel, J., Piovano, S., Peterson, S., Watling, D. and Dalzell, P. 2007. Strategies to Reduce Shark Depredation and Unwanted Bycatch in Pelagic Longline Fisheries: Industry Practices and Attitudes, and Shark Avoidance Strategies. Western Pacific Regional Fishery Management Council, Honolulu, USA.
Haraguchi, K., Hisamichi, Y. and Endo, T. 2006. Bioaccumulation of naturally occurring mixed halogenated dimethylbipyrroles in whale and dolphin products on the Japanese market. Archives of Environmental Contamination and Toxicology 51: 135-141.
Hernandez, O., Jouanno, J., Echevin, V. and Aumont, O., 2017. Modification of sea surface temperature by chlorophyll concentration in the A tlantic upwelling systems. Journal of Geophysical Research: Oceans, 122(7), pp.5367-5389.
IUCN (International Union for Conservation of Nature). 2012. Pseudorca crassidens. The IUCN Red List of Threatened Species. Version 3.1. Available at: http://www.iucnredlist.org. Downloaded on 21 February 2016.
Jarman WM, Norstrom RJ, Muir DCG, Rosenberg B, Simon M, Baird RW. 1996. Levels of organochlorine compounds, including PCDDS and PCDFS, in the blubber of cetaceans from the west coast of North America. Marine Pollution Bulletin 32: 426-436.
Kasuya T. 1986. False Killer Whales. In: Tamura T, Ohsumi S, Arai S (ed.), Report of investigation in search of solution for dolphin-fishery conflict in the Iki Island area. Report prepared for the Japan Fisheries Agency. Pp 285. English translation by N. Inagaki.
Kitchener, D.J., Ross, G.J.B. and Caputi, N. 1990. Variation in skull and external morphology in the false killer whale, Pseudorca crassidens, from Australia, Scotland and South Africa. Mammalia 54: 120-135.
Leatherwood S, McDonald D, Prematunga WP, Girton P, Ilangakoon A, McBrearty D. 1991. Records of the “Blackfish” (killer, false killer, pilot, pygmy killer and melonheaded whales) in the Indian Ocean, 1772-1986. In: Leatherwood S, Donovan GP (ed.), Cetaceans and Cetacean Research in the Indian Ocean Sanctuary. Marine Mammal Technical Report 3, pp. 33-65. Nairobi-United Nations Environment Programme, Nairobi, Kenya.
Martien, K.K., Taylor, B.L., Chivers, S.J., Mahaffy, S.D., Gorgone, A.M. and Baird, R.W., 2019. Fidelity to natal social groups and mating within and between social groups in an endangered false killer whale population. Endangered Species Research, 40, pp.219-230.
Mitchell, E. 1975b. Porpoise, dolphin and small whale fisheries of the world. IUCN Monograph 3: 1-129.
Miyashita, T. 1993. Abundance of dolphin stocks in the western North Pacific taken by the Japanese drive fishery. Reports of the International Whaling Commission 43: 417-437.
Mullin, K.D. and Fulling, G.L. 2004. Abundance of cetaceans in the oceanic northern Gulf of Mexico, 1996-2001. Marine Mammal Science 20(4): 787-807.
Odell, D. K. and Mcclune, K. M. 1999. False killer whale Pseudorca crassidens (Owen, 1846). In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Vol. 6: The second book of dolphins and the porpoises, pp. 213-244. Academic Press, San Diego.
Oleson, E.M., Boggs, C.H., Forney, K.A., Hanson, M.B., Kobayashi, D.R., Taylor, B.L., Wade, P.R., and Ylitalo, G.M. 2010. Status review of Hawaiian insular false killer whales (Pseudorca crassidens) under the Endangered Species Act. NOAA Technical Memorandum NMFS-PIFSC-22. U.S. Department of Commerce, National Oceanic and Atmospheric Administration, National Marine Fisheries Service, Pacific Islands Fisheries Science Center.
Orselli, I.B., Kerr, R., de Azevedo, J.L., Galdino, F., Araujo, M. and Garcia, C.A., 2019. The sea-air CO2 net fluxes in the South Atlantic Ocean and the role played by Agulhas eddies. Progress in Oceanography, 170, pp.40-52.
Palmer, C., Martien, K.K., Raudino, H., Robertson, K.M., Withers, A., Withers, E., Risk, R., Cooper, D., D’Cruz, E., Jungine, E. and Barrow, D., 2023. Evidence of resident coastal population (s) of false killer whales (Pseudorca crassidens) in northern Australian waters. Frontiers in Marine Science, 9, p.1067660.
Perryman WL, Foster TC. 1980. Preliminary Report on Predation by Small Whales, Mainly the False Killer Whale Pseudorca Crassidens, on Dolphins (Stenella Spp. and Delphinus Delphis) in the Eastern Tropical Pacific. Southwest Fisheries Center.
Photopoulou T, Ferreira IM, Kasuya T, Best PB, Marsh H. Submitted. Evidence for a postreproductive phase in female false killer whales Pseudorca crassidens. Frontiers in Zoology.
Purdon, J., Shabangu, F.W., Pienaar, M., Somers, M.J. and Findlay, K., 2020a. Cetacean species richness in relation to anthropogenic impacts and areas of protection in South Africa’s mainland Exclusive Economic Zone. Ocean & Coastal Management, 197, p.105292.
Purdon, J., Shabangu, F., Pienaar, M., Somers, M.J. and Findlay, K.P., 2020b. South Africa’s newly approved marine protected areas have increased the protected modelled habitat of nine odontocete species. Marine Ecology Progress Series, 633, pp.1-21.
Purves, P. E., and Pilleri, G. 1978. The functional anatomy and general biology of Pseudorca crassidens (Owen) with a review of the hydrodynamics and acoustics in Cetacea. Investigations on Cetacea 9: 67-230.
Ramos-Cartelle, A. and Mejuto, J. 2008. Interaction of the false killer whale (Pseudorca crassidens) and the depredation on the swordfish catches of the Spanish surface longline fleet in the Atlantic, Indian and Pacific Oceans. Report, International Commission for the Conservation of Atlantic Tunas (ICCAT), Collective Volume of Scientific Papers (SCRS/2007/025) 62: 1721-1783.
Ross, G. J. B. 1984. The smaller cetaceans of the south east coast of southern Africa. Annals of the Cape Provincial Museums (Natural History) 15: 173-410.
Sekiguchi, K., Klages, N.T.W. and Best, P.B. 1992. Comparative analysis of the diets of smaller odontocete cetaceans along the coast of southern Africa. South African Journal of Marine Science 12: 843-861.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stacey, P. J., Leatherwood, S. and Baird, R. W. 1994. Pseudorca crassidens. Mammalian Species 456: 1-6.
Stamper, M. A., Whitaker, B. R., and Schofield, T. D. 2006. Case study: morbidity in a pygmy sperm whale Kogia breviceps due to ocean-borne plastic. Marine Mammal Science 22: 719-722.
Sweijd, N.A. and Smit, A.J., 2020. Trends in sea surface temperature and chlorophyll-a in the seven African Large Marine Ecosystems. Environmental Development, 36, p.100585.
Tezanos-Pinto, G., Bohorquez, L., Zaeschmar, J. R., Stockin, K., Carroll, E. L., & Caballero-Gaitán, S. 2024. Population structure and genetic diversity of false killer whales (Pseudorca crassidens) in New Zealand waters: preliminary results. New Zealand Journal of Marine and Freshwater Research, 1–15. https://doi.org/10.1080/00288330.2024.2353208
Wade, P.R. and Gerrodette, T. 1993. Estimates of cetacean abundance and distribution in the eastern tropical Pacific. Reports of the International Whaling Commission 43: 477-493.
Williams TM. 2009. Swimming. In: Perrin WF, Wursig B, Thewissen JGM (ed.), Encyclopedia of Marine Mammals. Second edition, pp. 1140-1147. Academic Press, San Diego, California, USA.
