Eland
Tragelaphus oryx

2025 Red list status
Least Concern
Regional Population Trend
Increasing
change compared
to 2016
No Change
Overview
Tragelaphus oryx – (Pallas, 1766)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Tragelaphus – oryx
Common Names: Eland (English, Afrikaans), Common Eland (English), Impofu (Swati, Xhosa, Zulu), Phofu, Phokhu (Sepedi), Phôhu, Phôfu (Sotho, Tswana), Mhofu (Tsonga), Impophi, Imphofu (Swati), Phofu (Venda)
Synonyms: Taurotragus oryx (Pallas 1766)
Taxonomic Note:
Mitochondrial DNA (Essop et al. 1997; Gatesy et al. 1997; Matthee & Robinson 1999) and allozyme (Georgiadis et al. 1990) data, as well as evidence of hybridisation between the Common Eland and the Greater Kudu, Tragelaphus strepsiceros (Boulineau 1933; Jorge et al. 1976; Van Gelder 1977a, 1977b), has led to the classification of this species within the genus Tragelaphus rather than Taurotragus (IUCN SSC Antelope Specialist Group 2016). Additionally, Lorenzen et al. (2010) report a significant difference between the mtDNA lineages of Common Eland in East and Southern Africa. Three subspecies of Common Eland have been recognised, though their validity has been in dispute (Thouless 2013):
- Tragelaphus o. livingstonii (Sclater 1864; Livingstone’s Eland): also called kaufmanni, niediecki, selousi and triangularis. It is found in the Central Zambezian Miombo woodlands i.e. south-central Africa (Angola, Zambia, Democratic Republic of the Congo, Zimbabwe, Mozambique and Malawi). Livingstone’s Eland has a brown pelt with up to twelve stripes.
- Tragelaphus o. oryx (Pallas 1766; Cape Eland): also called alces, barbatus, canna and oreas. This subspecies is found south of the Zambezi River (South Africa, Botswana and Namibia). The fur is tawny, and adults lose their stripes.
- Tragelaphus o. pattersonianus (Lydekker 1906; East African Eland or Patterson’s Eland): also called billingae. It is found in east Africa extending into the Somali arid areas, hence its common name. Its coat can have up to 12 stripes.
- In addition, Tragelaphus o. derbiabianus (Gray 1847; Giant or Lord Derby’s eland): This is found in central and western Africa. It is regarded as having more massive horns than other subspecies and a dark neck and dewlap and is one of the most prized hunting trophies.
Tragelaphus o. oryx occurs throughout the larger part of South Africa, but the far northern Limpopo Province bordering Zimbabwe is regarded as a transitional zone between T. o. oryx and T. o. livingstonii or an area where they overlap. This argues the case that they should rather be described as ecotypes (in ecotypes, it is common for continuous, gradual geographic variation to impose analogous phenotypic and/or genetic variation; this situation is called cline.).
Red List Status: C – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Venter, J.1 & da Silva, J.2
Reviewer: Anderson, J.3,4
Institutions: 1Nelson Mandela University, 2South African National Biodiversity Institute, 3IUCN SSC Antelope Specialist Group, 4International Conservation Services
Previous Assessors: Buijs, D., Venter, J. & Parrini, F.
Previous Reviewer: Child, M.F.
Previous Contributors: Selier, J. & Relton, C.
Assessment Rationale
Within the assessment region, this species is widespread and common, occurring in numerous protected areas across its range. Overall, it was estimated that a mature population size of 34,925–45,648 (using a 70% mature population structure), of which 17,219–27,853 (49–61%) mature animals occur in formally protected areas (based on the 2016 assessment). Although local declines are apparent, the overall population is increasing, especially on private land. Using a sample of 23 formally protected areas with adequate long-term data, the population was estimated to have increased by between 2.5% and 30.1% over three generations (1992–2015). There are no major threats that could cause rapid population decline and thus the Least Concern listing remains. Local declines may be at least partly compensated for by the continued growth of its numbers on private farms and conservancies. However, research should determine whether private subpopulations are eligible for Red List inclusion based on whether they are intensively or captive managed. Recent data received for this species was not robust enough to update population estimates, hence this assessment was largely based on the data used in the 2016 assessment. However, any change in population number will not affect the status of the species at present.
Throughout Africa, the total population of Common Eland has been estimated at c. 136,000, with about half occurring within protected areas, and the other half on private land. Eland moves seasonally in response to rainfall and forage availability and thus require large areas. However, human settlement and agricultural expansion will result in contraction of the distribution and size of free-ranging populations, which will make subpopulations more vulnerable to bushmeat poaching, drought, high tick burdens and disease. This highlights the need for transfrontier initiatives, such as the Kgalagadi and Greater Limpopo Transfrontier Parks.
Regional population effects: Current migratory routes between Botswana and South Africa have been observed in a number of regions along the border into the Limpopo and North West provinces, and well as into the Kgalagadi Transfrontier Park. (The movement across the Malopo in the region of probably no longer exists).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Venter J & da Silva JM. 2025. A conservation assessment of Tragelaphus oryx. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The former range of this species extended throughout the savannah woodland habitats of southern and eastern Africa, extending into the arid savannah and scrubland regions, and high-altitude grasslands of southern Africa’s Karoo and Kalahari habitats (IUCN SSC Antelope Specialist Group 2016). Expansion of human settlements has resulted in a severe constriction of their former range, and since the 1970s, the repercussions of civil wars in regions such as Mozambique, Angola, Rwanda, Uganda and Burundi (where they are now extinct; East 1996) have resulted in dramatic declines in population numbers. Although their historic distribution encompassed the majority of Botswana, Common Eland are now absent from much of the developed regions of the eastern sector and the Okavango Delta. Similarly, their range in Zimbabwe has been restricted to the northwest and southeastern parts of the country (Skinner & Chimimba 2005). Indigenous populations in Eswatini have been eradicated, but Eland have been successfully reintroduced into Malolotja Nature Reserve (Skinner & Chimimba 2005). Eland are occasional visitors into parts of Lesotho (for example, the Sehlabathebe National Park) from the Drakensberg, KwaZulu-Natal (Lynch 1994; Skinner & Chimimba 2005). The presence of true free-roaming, natural subpopulation between South Africa and Botswana has been suggested due to the presence of 28 individuals on a farm on the Molopo River, approximately 140 km from Molopo Nature Reserve. However, it is uncertain whether these individuals are escapees from Molopo Nature Reserve, as the game fence is not regarded as Eland-proof (D. Buijs pers. comm. 2014; Power 2014).
Common Eland have been reintroduced extensively onto many game ranches and private game farms in southern Africa within their natural range, most predominantly in South Africa, in order to boost abundance. They are now widespread throughout all provinces of South Africa, especially on private lands. Additionally, due to its commercial value, this species has been introduced widely into areas outside of their natural range; for example, into game ranches in southern and central Namibia (East 1999).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 4900
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
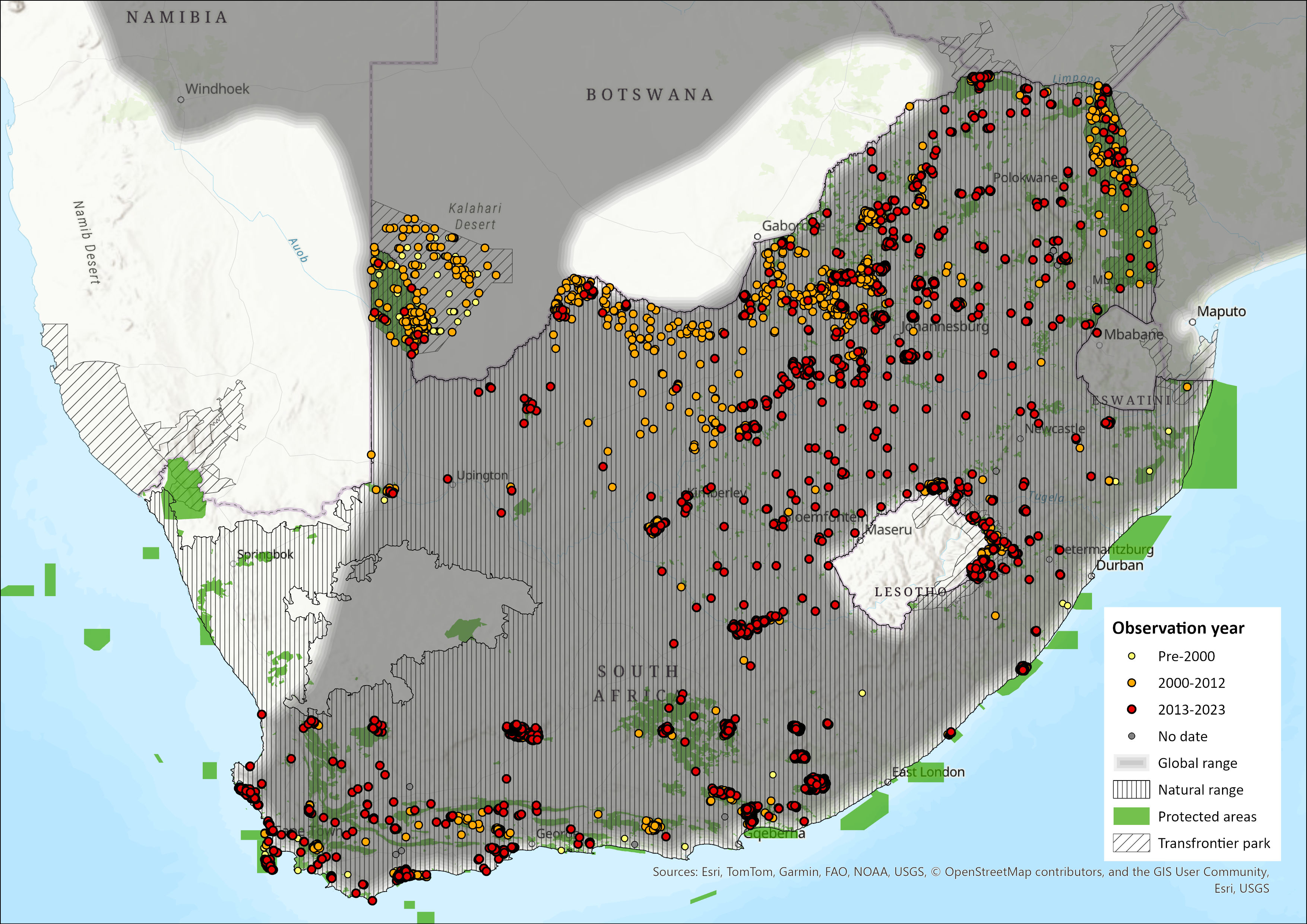
Map
Figure 1. Distribution records for Eland (Tragelaphus oryx) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Possibly Extinct | Native | - | - |
| Botswana | Extant | Native | - | - |
| Burundi | Extant | Native | - | - |
| Congo, The Democratic Republic of the | Extant | Native | - | - |
| Eswatini | Extant | Native | - | - |
| Ethiopia | Extant | Native | - | - |
| Kenya | Extant | Native | - | - |
| Lesotho | Extant | Vagrant | - | - |
| Malawi | Extant | Native | - | - |
| Mozambique | Extant | Native | - | - |
| Namibia | Extant | Native | - | - |
| Rwanda | Extant | Native | - | - |
| South Africa | Extant | Native | - | - |
| Sudan | Extant | Native | - | - |
| Tanzania, United Republic of | Extant | Native | - | - |
| Uganda | Extant | Native | - | - |
| Zambia | Extant | Native | - | - |
| Zimbabwe | Extant | Native | - | - |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change in South Africa may result in habitat changes that will favour arid landscape antelope, such as the Eland. More research is needed on the effect of climate change on this species.
Population information
Population density estimates of Common Eland in African regions where this species is considered relatively common have been reported to range from 0.05–0.4/ km² (East 1999). Although higher population density estimates of 0.6–1.0 individuals / km² have been recorded by aerial counts and ground surveys in certain regions (Thouless 2013; IUCN SSC Antelope Specialist Group 2016). A global population estimate of 136,000 individuals was reported by (East 1999), with a number of countries, including South Africa, Namibia, Botswana, Zimbabwe, Malawi and possibly also Tanzania, hosting stable or increasing populations of Common Eland. In general, population trends are variable across protected areas, but are mostly increasing on private lands and decreasing in other regions (IUCN SSC Antelope Specialist Group 2016).
Within the assessment region, there are estimated to be between 24,470 and 39,790 animals on 116 formally and privately protected areas in South Africa (counts between 2013 and 2016), where the large range is due to seasonal differences in abundance in the Kgalagadi Transfrontier Park and dispersal between Botswana and South Africa. Assuming a 70% mature population structure, this yields 17,219–27,853 mature animals. Additionally, there are an estimated 25,423 animals occurring on 617 wildlife ranches across the country (counts between 2013 and 2014), which brings the total to 49,893–65,213 animals (34,925–45,648 mature animals) between 2013 and 2016. However, the number of private subpopulations eligible for inclusion in the Red List (wild and free-roaming) is unknown and further research should seek to ascertain this number by quantifying the management intensity on wildlife ranches. For example, in the North West Province, Eland populations are thought to be mostly free-roaming and self-sustaining in extensive wildlife ranches, but may be fed during drought conditions (Power 2014) and encouraged to use mineral licks that apply an acaricide (J.Anderson pers comm.). Such management regimes should be quantified.
Generation length is estimated as 7.8 years (IUCN SSC Antelope Specialist Group 2016), which yields a 22 year three-generation period (1992–2015). The overall population is estimated to have increased over three generations by between 2.5% and 30.1%. Using a sample of 23 protected areas with adequate long-term data, the population is estimated to have increased by 2.5% over three generations. However, using only those protected areas that have long-term data exactly over three generations, there is estimated to have been a 30.1% increase in abundance. In Free State provincial protected areas alone, the population has increased by 51% in eight protected areas since the previous assessment in 2004, from 1,023 to 1,546 individuals (2004 and 2014 respectively) (E. Schulze unpubl. data). Local declines have occurred in Kruger National Park (Harrington et al. 1999; Ferreira et al. 2013) and several protected areas in North West Province (Nel 2015), such as Madikwe Game Reserve where they have declined from 850 to 9 between 1995 and 2013. This decline is suggested to be attributed to the location of Madikwe, which is on the edge of the Eland’s seasonal range (Power 2014). Alternatively, this decline may be a direct consequence of high predation pressure on this small reserve, as Eland populations are thriving on farms adjacent to Madikwe, where Lions (Panthera leo) are absent. However, 70% of sampled subpopulations are increasing (for example, Bissett et al. 2016a, 2016b).
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | - | based on data from 2016 assessment |
Current population trend: Increasing, based on 2016 assessment
Severely fragmented: Yes. Most subpopulations are fenced.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Uncertain
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Uncertain
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
The species is thought to exist as a single ESU, with two genetically differentiated management units identified that separate the species in the eastern parts of it range (Ethiopia, Uganda, Kenya, Tanzania) from the southern parts (Zambia, Zimbabwe, Botswana, Namibia, Angola, South Africa, Mozambique) (Lorenzen et al. 2010). No estimate of effective population size exists for the two groups; however, given that Eland is extremely prominent in the wildlife ranching industry, it is extensively translocated, and population numbers are large, suggesting that Ne is likely to exceed 500.
Habitats and ecology
Common Eland are highly adaptable ruminants, formerly inhabiting large proportions of the Nama- and Succulent Karoo biomes, as well as grasslands, Acacia savannahs, Miombo woodlands, semi-deserts and alpine moorlands (to altitudes of 4,900 m asl.) (Thouless 2013). Additionally, their range extended somewhat into the Fynbos Biome in southwestern South Africa (Skinner & Chimimba 2005). This species is generally absent from true deserts, dense forests, and entirely open grasslands; however, they are occasionally present in grasslands with substantial herb cover (Thouless 2013). Only anecdotal information is available on Eland water dependency: these animals can survive (and thrive) in arid regions (such as the Central Kalahari Game Reserve in Botswana) where surface water is absent, thus it is likely that they are able to meet much of their water requirements from their diet (Pappas 2002), but they will drink regularly when surface water is available (Kingdon 1997; Estes 1999; Pappas 2002).
Eland extensively utilise forb-rich montane grasslands of the South African Highveld (Rowe-Rowe 1983); observations in Mountain Zebra National Park (Eastern Cape, South Africa) also confirmed that vegetation type selection is mostly based on the presence and density of favoured browse species (Watson & Owen-Smith 2000). Seasonal habitat use by Eland is, therefore, supposedly driven by changes in forage quality and abundance conditions, and a number of studies suggested that Eland in savannah areas move from woodland to open grassland during the early wet season to forage on new growing grasses (Lamprey 1963; Hillman 1988; Buys 1990; Fabricius & Mentis 1990; Watson & Owen-Smith 2000). Similarly, Eland of the Drakensberg mountain range moves from montane forest and sub-alpine scrubland into grassland at the onset of rains (Skinstad 1972). In the Kgaswane Mountain Reserve (North West Province) Eland are found on open grassland even during the mid-dry season, where they forage on the available green grasses (D’Ammando et al. 2015). Eland shifted their diets and home ranges to feed primarily on grass in the wet summer months and towards green dicot browse in the drier months (Parrini et al. 2019).
As large animals with high metabolic rates, Eland require high quality forage across all seasons, often selecting broad-leaf savannah woodlands and forb-rich grasslands, which provide green forage throughout the year (Owen-Smith 2002). Thus, they travel long distances in search of ephemeral food sources (IUCN SSC Antelope Specialist Group 2016). Eland have been described as browsers, grazers and mixed feeders preferring grasses, according to various studies which tried to estimate the grazing and browsing components of their diet (Lamprey 1963; Kerr et al. 1970; Hofmann 1973, 1989; Field 1975; Buys 1990; Gagnon & Chew 2000; Watson & Owen-Smith 2000; Cerling et al. 2003; Codron et al. 2005, 2007; Wallington et al. 2007; D’Ammando et al. 2015). They feed on the leaves instead of lignified plant parts (Kerr et al. 1970), while selecting for the greenest and lowest in fibre content forage types (Watson & Owen-Smith 2000, 2002). Although Gagnon & Chew (2000) reported that the diet of the Eland is composed of 50% grasses, the proportion of grass in the diet is extremely variable among different study sites (Cerling et al. 2003). In southern Africa, Eland have usually been considered browsers consuming small proportions of grasses during the wet season (Kerr et al. 1970; Buys 1990; Watson & Owen-Smith 2000, 2002; Sponheimer et al. 2003; Codron et al. 2005, 2007; Wallington et al. 2007). Only Underwood (1975) reported that Eland of the Loskop Dam Nature Reserve consumed grass proportions comparable to those recorded for east African populations. In Suikersborand Nature Reserve, diet remained unaltered between the wet and the dry season, with very low proportions of C4 plants (Wallington et al. 2007). Similarly, Watson & Owen-Smith (2000) found that grasses contributed only 5.7% to the Eland’s annual diet in Mountain Zebra National Park, Eastern Cape, which comprised of mostly shrubs and dwarf shrubs. In the coastal grassland areas of the Eastern Cape forbs, and not grasses, form a significant proportion of their diet (Mivuyo 2014). In contrast, results from feeding site surveys in the Kgaswane Mountain Reserve showed that Eland in this area tend to forage extensively on green grasses during the mid-dry season (D’Ammando et al. 2015). It is possible that Eland may be able to adapt to local forage conditions, widening their diet in order to include the highest-quality plant species available, modifying the acceptance frequencies of grasses versus browse. This extreme versatility in feeding habits could explain the wide geographic distribution and the quite catholic habitat preferences of this ungulate (Thouless 2013). Faecal analyses have shown that the nitrogen level of Eland faeces is consistently higher than that of grazers like Gemsbok (Oryx gazella) and Black Wildebeest (Connochaetes gnou) during the winter, which reflects a higher protein intake by Eland (Buys 1987).
Breeding and calving seasons have not been clearly identified, and probably vary from one population to another (Kingdon 1982; Pappas 2002), with the high numbers of births coinciding with the rainy season. The calf is dependent on its mother only for the first two weeks of life, during which it is hidden in thick bush (Kingdon 1982; Estes 1999). When the grass lignifies and nutritive quality decreases at the end of the summer most calves are already a few months old and are less dependent on the females for feeding and protection, allowing the Eland to resume their browsing habits (Buys 1987).
Ecosystem and cultural services: Eland, through their browsing and grazing activities, play a facilitating role for more selective smaller sized ungulates (Venter et al. 2014). Eland serves as a food source for the larger predators, such as lion (Hayward & Kerley 2005). Eland also serves as a source of bushmeat in certain impoverished areas (Lindsey et al. 2012). In some provinces in South Africa Eland meat is donated to neighbouring communities as a token of goodwill from government nature reserves and parks.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | - | Suitable | - |
| 2.1. Savanna -> Savanna – Dry | - | Suitable | - |
| 3.4. Shrubland -> Shrubland – Temperate | - | Suitable | - |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | - | Suitable | - |
| 3.7. Shrubland -> Shrubland – Subtropical/Tropical High Altitude | - | Suitable | - |
| 4.4. Grassland -> Grassland – Temperate | - | Suitable | - |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | - | Suitable | - |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | - | Suitable | - |
Systems
System: Terrestrial
General Use and Trade Information
The Common Eland is hunted for food, sport, and other purposes. It is sold at live animal auctions and used extensively for trophy hunting in the private sector. Trade is unlikely to have any negative effect on the population. Their meat is highly prized and each animal provides a large quantity of meat, so they are particular targets of illegal hunters (IUCN SSC Antelope Specialist Group 2016).
The foundation of game ranching in South Africa was laid when the first game-proof fence on private land was erected near Dwaalboom in 1945. This was also the first time that game that had disappeared from an area was reintroduced. Eland were transported to the area by mule cart. Presently, Eland occur commonly on private lands across the country and many of these include Livingstone’s Eland (T. o. livingstonii) (for example, in North West; Power 2014), which are considered particularly valuable in the wildlife ranching market and are bred in smaller, controlled environments. Livingstone’s Eland are often released onto more extensive systems and often interbred with “pure” Cape Eland (T. o. oryx), especially because they are able to escape easily.
Common Eland have also been widely domesticated in Zimbabwe, South Africa and Kenya, as well as in Russia and the Ukraine due to a high yield of nutritious, ‘long life’, antibacterial milk; their ability to be tamed and herded; their long life expectancy in captivity; and their ability to survive in arid regions (low water requirements) (Thouless 2013). However, management practices such as high food supplement costs, confining them at night and herding them during the day are likely to negate their advantages over cattle in many environments (IUCN SSC Antelope Specialist Group 2016).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat | Yes | Meat, skins, trophies and live animal trade |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | - | - |
| 12. Handicrafts, jewellery, etc. | - | true | - | - |
| 13. Pets/display animals, horticulture | - | true | - | - |
| 15. Sport hunting/specimen collecting | - | true | true | - |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Meat, skins, trophies and live animal trade. Captive breeding for trophies
Threats
Habitat loss (due to expanding human settlements) and poaching for its superior meat have resulted in drastic reductions in population abundance of Eland throughout Africa (IUCN SSC Antelope Specialist Group 2016). However, within the assessment region, there are few major threats, especially now that wildlife ranching is providing more habitat for this species and is reintroducing it to former parts of its range. Past habitat loss from settlement and agricultural expansion has curtailed available habitat (Driver et al. 2012; GeoTerraImage 2015). As available habitat declines further, its large area requirements and wandering habitats may make populations increasingly vulnerable to subsidiary threats suchit as bushmeat poaching, drought and disease (Thouless 2013).
Hybridisation, both between species and between ecotypes, may be a minor threat to this species. For example, there are two well-documented records of hybrid male offspring between Common Eland and Greater Kudu, where one was known to be sterile and the other unknown (Jorge et al. 1976; Van Gelder 1977a). This threat can only be managed by identifying “pure” populations genetically if markers are available, as is the case for Bontebok (Damaliscus pygargus pygargus) hybrids. In order to prevent the risk of hybridisation, wild populations (e.g. those in Kgalagadi Transfrontier Park) should not be supplemented from game ranches/farms.
Finally, bushmeat poaching and deteriorating habitat conditions have also been identified as localised threats, which could lead to local subpopulation declines. This has been observed in Dwesa-Cwebe Nature Reserve in the Eastern Cape, where bushmeat poaching has led to significant population declines (Hayward 2009). In the North West, Eland are one of the most targeted species for bushmeat poaching (Nel 2015).
Conservation
Protected areas that support major, resilient populations include the Kgalagadi Transfrontier Park (Botswana and South Africa) and the Maloti-Drakensberg Transfrontier Conservation and Development Area (South Africa and Lesotho). For example, the subpopulation in the Kalahari Gemsbok National Park (the South African side of the Kgalagadi Transfrontier Park) ranges seasonally from 1,081 to 16,401 animals (counts between 2012 and 2013) (Ellis & Herbst 2013). These large populations are crucial in conserving the species, as they reflect different bioregions.
Additionally, several smaller protected areas have large populations (all counts 2013–2014): Molopo Nature Reserve and Bloemhof Dam Nature Reserve in the North West Province host approximately 600 and 300 individuals respectively; Doornkloof Nature Reserve and Mokala National Park in the Northern Cape has over 450 and 700 animals respectively (Smit 2014; Bissett et al. 2016a); Golden Gate Highlands National Park in the Free State has over 1,000 animals (Bissett et al. 2016b); and Great Fish River Nature Reserve and Mpofu Nature Reserve in the Eastern Cape have 594 and 461 animals respectively (Peinke & Gibisela 2014). Relatively large numbers of Common Eland presently occur on private land in South Africa, Namibia and Zimbabwe, reflecting its value as a trophy animal. Transfrontier parks and conservancies should be expanded to facilitate resilient and self-sustaining subpopulations of this species. For example, the incorporation of Qwa-Qwa National Park into Golden Gate Highlands National Park in 2008 enabled the increase of the Eland subpopulation from 116 in 1994 to 1,054 in 2016 (Bissett et al. 2016b).
Reintroduction should be used to supplement existing subpopulations and establish former subpopulations within their natural range. For example, Eland once occurred on S. A. Lombard Nature Reserve in North West Province (Buys & Dott 1991), and reintroductions are thought to be feasible (Power 2014). The recommended capacity for the reserve is 30–40 animals (Buys & Dott 1991).
Additionally, regulation of translocation is required to prevent hybridisation with exotic species and various ecotypes. This can be achieved through translocation regulations and auditing of source populations.
Hillman (1979) concluded that Eland are inferior to cattle for meat production under normal farming conditions due to their high degree of mobility, low social cohesiveness and the species’ natural existence at very low densities. However, Eland are increasingly valuable in hot, semi-arid regions, where cattle are less suitable. Thus, Eland may be a valuable asset on game farms located in suitable environments when kept under as natural conditions as possible, and this species should continue to be sustainably utilised as it can form a key species within the wildlife-based, rural economy. However, a thorough knowledge of the basic ecology of Eland living in particular regions is essential (Buys 1987).
Eland from regions free of “Heartwater” (a rickettsial disease Ehretina rumenantium) are susceptible to infection and will die if introduced to a Heartwater area with Amblyomma ticks. Eland introduced from Namibia to Pilanesberg were “blocked” before free release by inoculating them with infected blood and then treated them with Oxytetracycline (A. Rough pers comm.). An attempt to introduce and breed Giant Eland near Malelane in the Lowveld was unsuccessful when all animals died from Theileriosis (J.P. Raath pers. comm.).
Recommendations for land managers and practitioners:
- Separation of ecotypes/species, such as the Livingstone’s Eland and the Giant Eland (T. derbianus) should be made, and records must be kept of hybridisations when translocating and selling them to keep track of genetic diversity (Power 2014).
- Develop this species as a keystone within the sustainable, wildlife-based rural economy. Provide incentives for landowners to provide cheap, low-carbon protein to local communities and to create conservancies where the benefits of this species are shared.
- Because of their high metabolism and energy requirements, this species is not suitable for smaller fenced-in areas without supplementary feeding, if production is required. The same applies to grassland ranches with limited browse available in winter. In truly extensive systems, Eland are nomadic and roam widely in search of the optimal food resources, which they are unable to do when fenced in, or may even leave the farm by leaping over the fence.
- Conservancies with Eland should reintroduce Red-billed Oxpeckers as Eland suffer from high tick burdens.
Research priorities:
- Factors contributing to local population declines, such as in the Kruger National Park (Harrington et al. 1999).
- Effects of wildlife ranching and methods of creating wildlife-based economies from this species and its efficacy as a source of protein for local communities.
- Genetic studies, and the effect of hybridisation between subspecies.
- Vulnerability of Eland to large predators in small fenced protected areas.
- The effects of movement restriction imposed by small, fenced reserves, and how his may exacerbate vegetation degradation, leading to detrimental effects on other herbivore species.
- Determine optimal conditions for Eland on wildlife ranches and the reasons why many promising introductions have failed and how to mitigate these limiting factors.
- Climate change in South Africa may result in habitat changes that will favour arid landscape antelope. Modelling this may encourage more landowners to stock Eland.
- Determine whether private subpopulations are eligible for Red List inclusion based on whether they are intensively or captive managed.
- Eland venison is highly prized and creating a sought after market for Eland venison should increase the demand and value of Eland and ultimately increase the populations on wildlife ranches.
Encouraged citizen actions:
- Landowners should create conservancies for this species and engage local stakeholders to create sustainable, wildlife-based rural economies.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially of free-roaming herds outside protected areas.
Bibliography
Bissett C, Ferreira S, Bezuidenhout H, Daemane E, Smit I, Joubert D, Kotsoe K, Moolman L. 2016a. Mokala National Park Herbivore Off-take Recommendations 2016: An integrated approach combining local knowledge with data derived from animal census, herbivore models, vegetation field monitoring and satellite imagery. Internal Report 10/2016. Scientific Services, South African National Parks, Skukuza, South Africa.
Bissett, C., Ferreira, S., Bezuidenhout, H., Smit, I., Daemane, E., Mokoena, V. and Sikhosana, T. 2016. Golden Gate Highlands National Park herbivore off-take recommendations 2016: An integrated approach combining local knowledge with data derived from animal census, herbivore models, vegetation field monitoring and satellite imagery. Scientific Services, South African National Parks.
Boulineau P. 1933. Hybridations d’antelopides. La Terre et la Vie 3: 690-691.
Buys D, Dott HM. 1991. Population fluctuations and breeding of eland Taurotragus oryx in a western Transvaal nature reserve. Koedoe 34: 31-36.
Buys D. 1987. The ecology of eland (Taurotragus oryx) in the western Transvaal Highveld. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Buys D. 1990. Food selection by eland in the western Transvaal. South African Journal of Wildlife Research 20: 16-20.
Cerling TE, Harris JM, Passey BH. 2003. Diets of East African Bovidae based on stable isotope analysis. Journal of Mammalogy 84: 456-470.
Codron D, Codron J, Lee-Thorp JA, Sponheimer M, De Ruiter D, Sealy J, Grant R, Fourie N. 2007. Diets of savanna ungulates from stable carbon isotope composition of faeces. Journal of Zoology 273: 21-29.
Codron D, Codron J, Lee-Thorp JA, Sponheimer M, De Ruiter D. 2005. Animal diets in the Waterberg based on stable isotopic composition of faeces. South African Journal of Wildlife Research 35: 43-52.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
D’Ammando G, Parrini F, Attorre F, Boitani L. 2015. Observations on dry season grazing by eland in a Magaliesberg Nature Reserve, South Africa. African Journal of Ecology 53: 112-115.
East R. 1996. Antelope survey update. 2. IUCN SSC Antelope Specialist Group. Gland, Switzerland
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Ellis G, Herbst M. 2013. Kalahari Gemsbok National Park Aerial Census Report. Internal Field Report/2013. Scientific Services. South African National Parks.
Essop MF, Harley EH, Baumgarten I. 1997. A molecular phylogeny of some Bovidae based on restriction-site mapping of mitochondrial DNA. Journal of Mammalogy 78: 377-386.
Estes, R.D. 1999. The Safari Companion: A Guide to Watching African Mammals Including Hoofed Mammals, Carnivores, and Primates. Chelsea Green Publishing Company, Vermont.
Fabricius C, Mentis MT. 1990. Seasonal habitat selection by eland in arid savanna in southern Africa. South African Journal of Zoology 25: 238-244.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Summary Report: Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Field CR. 1975. Climate and the food habits of ungulates on Galana Ranch. African Journal of Ecology 13: 203-220.
Gagnon M, Chew AE. 2000. Dietary preferences in extant African Bovidae. Journal of Mammalogy 81: 490-511.
Gatesy J, Amato G, Vrba E, Schaller G, DeSalle R. 1997. A cladistic analysis of mitochondrial ribosomal DNA from the Bovidae. Molecular Phylogenetics and Evolution 7: 303-319.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Georgiadis NJ, Kat PW, Oketch H, Patton J. 1990. Allozyme divergence within the Bovidae. Evolution: 2135-2149.
Harrington R, Owen-Smith N, Viljoen PC, Biggs HC, Mason DR, Funston P. 1999. Establishing the causes of the roan antelope decline in the Kruger National Park, South Africa. Biological Conservation 90: 69-78.
Hayward MW, Kerley GI. 2005. Prey preferences of the lion (Panthera leo). Journal of Zoology 267: 309-322.
Hayward MW. 2009. Bushmeat hunting in Dwesa and Cwebe Nature Reserves, Eastern Cape, South Africa. South African Journal of Wildlife Research 39: 70-84.
Hillman J. 1988. Home range and movement of the common eland (Taurotragus oryx Pallas 1766) in Kenya. African Journal of Ecology 26: 135-148.
Hillman JC. 1979. The biology of the eland (Taurotragus oryx Pallas 1766) in the wild. Ph.D. Thesis. University of Nairobi, Nairobi, Kenya.
Hofmann RR. 1973. The Ruminant Stomach: Stomach Structure and Feeding Habits of East African Game Ruminants. East African Literature Bureau, Nairobi, Kenya.
Hofmann RR. 1989. Evolutionary steps of ecophysiological adaptation and diversification of ruminants: a comparative view of their digestive system. Oecologia 78: 443-457.
IUCN SSC Antelope Specialist Group. 2016. Tragelaphus oryx. The IUCN Red List of Threatened Species 2016: e.T22055A50196938.
Jorge W, Butler S, Benirschke K. 1976. Studies on a male eland x kudu hybrid. Journal of Reproduction and Fertility 46: 13-16.
Kerr MA, Wilson VJ, Roth HH. 1970. Studies on the agricultural utilization of semi-domesticated eland (Taurotragus oryx) in Rhodesia. 2. Feeding habits and food preferences. Rhodesian Journal of Agricultural Research 8: 71-77.
Kingdon, J. 1982. East African Mammals: An Atlas of Evolution in Africa. Academic Press, London, UK.
Kingdon, J. 1997. The Kingdon Field Guide to African Mammals (first edition}. Academic Press, San Diego, California, USA.
Knight M, Mills G, Hes L. 1997. Eland Taurotragus oryx. In: Mills G, Hes L (ed.), The Complete Book of Southern African Mammals, pp. 287. Struik Publishers, Cape Town, South Africa.
Lamprey HF. 1963. Ecological separation of the large mammal species in the Tarangire Game Reserve, Tanganyika. African Journal of Ecology 1: 63-92.
Lorenzen ED, Masembe C, Arctander P, Siegismund HR. 2010. A long-standing Pleistocene refugium in southern Africa and a mosaic of refugia in East Africa: insights from mtDNA and the common eland antelope. Journal of Biogeography 37: 571-581.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Matthee CA, Robinson TJ. 1999. Mitochondrial DNA population structure of roan and sable antelope: implications for the translocation and conservation of the species. Molecular ecology 8: 227-238.
Mivuyo M. 2014. A stable carbon isotopic composition of diets of different ungulates in Mkambati Nature Reserve. Walter Sisulu University, Mtatha.
Nel, P. 2015. Population estimates for large herbivores and predators in protected areas in the North West Parks Board November 2015. North West Parks Board.
Owen-Smith RN. 2002. Adaptive herbivore ecology: from resources to populations in variable environments. Cambridge University Press, Cambridge, UK.
Parrini. F, G, D’Ammando, & J. P. Marshal. 2019. Seasonal shifts in habitat use and diet by eland confined in a small, fenced reserve. African Journal of Ecology 57 (3), 394-403
Pappas LA. 2002. Taurotragus oryx. Mammalian Species 689: 1-5.
Peinke DM, Gibisela Z. 2014. Game management recommendations for 2014. Eastern Cape Parks and Tourism Agency, East London.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rowe-Rowe DT. 1983. Habitat preferences of five Drakensberg antelopes. South African Journal of Wildlife Research 13: 1-8.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skinstad JR. 1972. Factors influencing the distribution and seasonal movements of the Cape Eland (Taurotragus oryx) in the Giant’s Castle Game Reserve. Natal Parks Board, South Africa.
Smit ZM. 2014. The ecological planning of Doornkloof Nature Reserve, Northern Cape Province. M.Sc. Thesis. University of the Free State, South Africa..
Thouless, C. R. 2013. Tragelaphus oryx. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa, Academic Press, Amsterdam, The Netherlands.
Underwood R. 1975. Social behaviour of the eland (Taurotragus oryx) on Loskop Dam Nature Reserve. University of Pretoria, Pretoria, South Africa.
Van Gelder RG. 1977a. An eland X kudu hybrid, and the content of the genus Tragelaphus. Lammergeyer 23: 1-6.
Van Gelder RG. 1977b. Mammalian hybrids and generic limits. American Museum Novitates : 2635.
Venter JA, Prins HH, Balfour DA, Slotow R. 2014. Reconstructing grazer assemblages for protected area restoration. PloS One 9: e90900.
Wallington BP, McKechnie AE, Owen-Smith RN, Woodborne S. 2007. Stable carbon isotope analysis of eland (Taurotragus oryx) diet in the Suikerbosrand Nature Reserve. South African Journal of Wildlife Research 37: 127-131.
Watson LH, Owen-Smith N. 2002. Phenological influences on the utilization of woody plants by eland in semi-arid shrubland. African Journal of Ecology 40: 65-75.
Watson LH, Owen-Smith RN. 2000. Diet composition and habitat selection of eland in semi-arid shrubland. African Journal of Ecology 38: 130-137.