
Egyptian Slit-faced Bat
Nycteris thebaica

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Nycteris thebaica – É. Geoffroy, 1818
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – NYCTERIDAE – Nycteris – thebaica
Common Names: Egyptian Slit-faced Bat, Cape Slit-faced bat, Cape Long-eared Bat, Common Slit-faced Bat, Wood’s Slit-faced Bat, Wood’s Long-eared Bat (English), Wood se Speetneusvlermuis, Wood-spleetneusvlermuis, Woodse Langoorvlermuis, Gewone-spleetneusvlermuis (Afrikaans)
Synonyms: Nycteris geoffroyi A. G. Desmarest, 1821; Nycteris affinis A. Smith, 1829; Nycteris capensis A. Smith, 1829; Nycteris thebaius Lesson, 1836 [incorrect subsequent spelling]; Nycteris albiventris J. A. Wagner, 1840; Nycteris discolor J. A. Wagner, 1840; Nycteris fuliginosa W. C. H. Peters, 1851; Nycteris labiata von Heuglin, 1861; Nycteris g. senegalensis Hartmann, 1868; Nycteris angolensis W. C. H. Peters, 1871; Nycteris damarensis W. C. H. Peters, 1871; Nycteris revoilii Robin, 1881; Nycteris adana (Andersen, 1912); Petallia d. brockmani (Andersen, 1912); Petallia d. media (Andersen, 1912); Petalia (Nycteris) thebaica aurantiaca (De Beaux, 1923); najdiya Nader & Kock, 1982 (see ACR 2024, Mammal Diversity Database 2025).
Taxonomic Note:
South Africa
Meester et al. (1986) recognised three subspecies in the region. However, these three populations do not appear to occupy geographically isolated areas and because they represent stages in a gradual cline in characters from east to west where individuals in the east tend to be darker and smaller, while those from the Northern Cape and Namibia are significantly larger and distinctly paler (Monadjem et al. 2020). Demos et al. (2019) report multiple deeply divergent lineages within African lineage and suggest the widespread N. thebaica to be a species complex. Individuals from southern Africa (South and Malawi) formed a clade (N. cf. thebaica) distinct from other African samples. Further integrative systematic studies are required to determine the taxonomic status of the reported lineages, inclusive of N. cf. thebaica.
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Richards L.R.1 & da Silva, J.M.2
Reviewer: Smith, C.3
Institutions: 1Durban Natural Science Museum, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust
Previous Assessors: Monadjem, A., Cohen, L., Jacobs, D., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Raimondo, D., Nicholson, S.K. & Relton, C.
Assessment Rationale
This species has a large range (estimated extent of occurrence is 1,580,895 km2) within the assessment region, occurring in most regions (besides much of the Highveld grasslands) including many protected areas and modified habitats. It is presumed to be abundant, with roosts containing hundreds of individuals, and faces no major threats. Hence it is listed as Least Concern.
Regional population effects: The range of this species extends across most of southern Africa, and dispersal is likely between South Africa and Namibia, Zimbabwe and Mozambique. Although it has low wing-loading (Monadjem et al. 2020), it is capable of dispersing great distances. For example, a banded individual was recovered 100 km from where it was originally captured by Monadjem (2005, 2006), however this movement could not be explained by post-natal dispersal.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR & da Silva JM. 2025. A conservation assessment of Nycteris thebaica. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Broadly distributed across savannah and riparian zones, this species is predominantly found in sub-Saharan Africa. However, it has also been recorded in Morocco, Libya, Egypt (primarily down the Nile River valley, but also into Sinai) and the Middle East (Israel, Palestine and Jordan). Its altitudinal range extends from sea level to about 2,000 m asl (ACR 2024). It is absent from the open semiarid grassland regions of southern Botswana. It is common in the assessment region, occurring in all provinces of South Africa, as well as in Swaziland but not Lesotho. It is common in the North West, Gauteng, Mpumalanga, Free State and KwaZulu-Natal provinces, but is considered rare in the Northern, Western and Eastern Cape (Skinner and Chimimba 2005), and is absent from much of the plateau grasslands (Monadjem et al. 2010). Its estimated extent of occurrence is 1,580,895 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 3,869 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
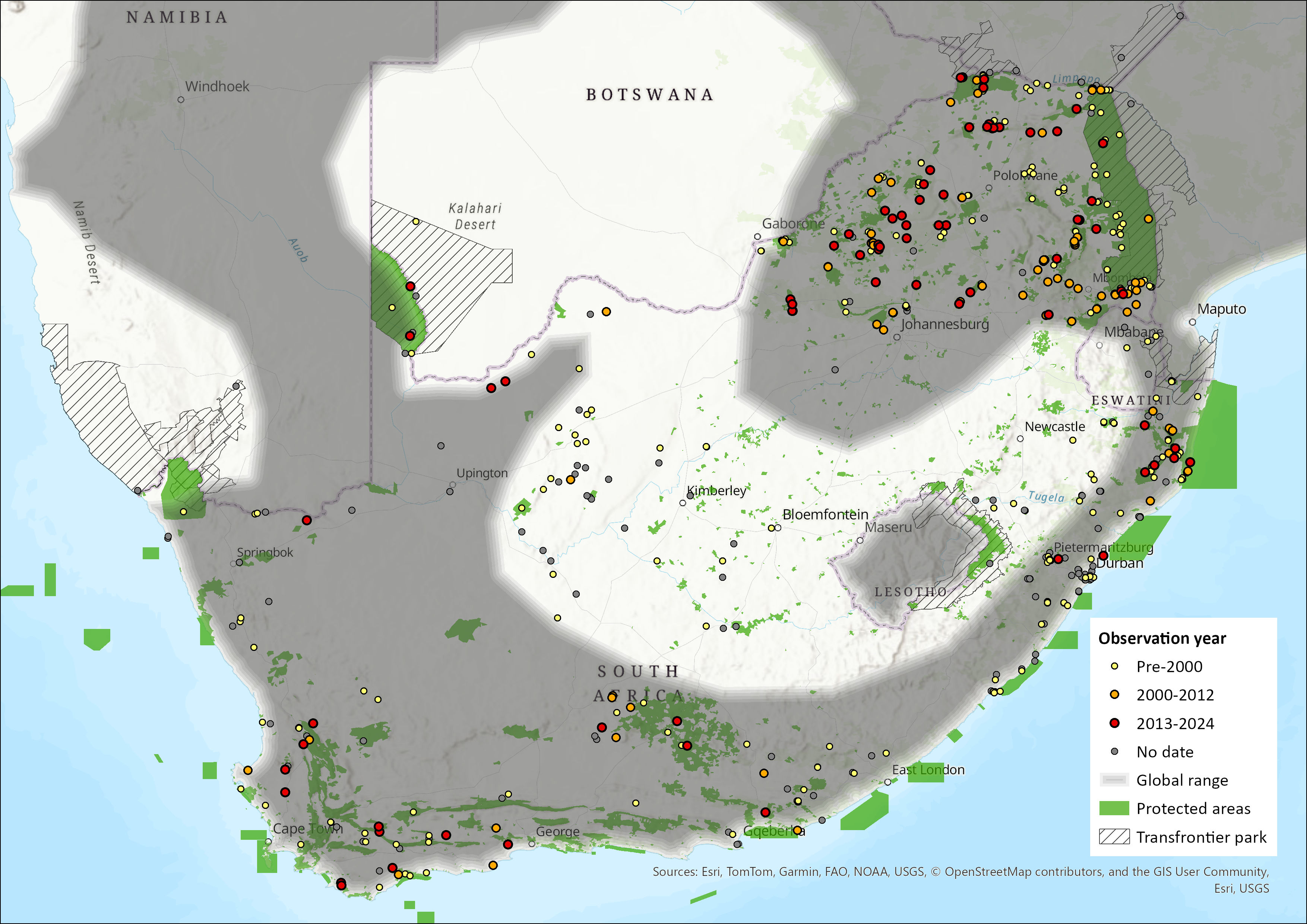 Figure 1. Distribution records for Egyptian Slit-faced Bat (Nycteris thebaica) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Egyptian Slit-faced Bat (Nycteris thebaica) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Egypt | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Jordan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lebanon | Presence Uncertain | Native | – | – |
| Lesotho | Presence Uncertain | Native | – | – |
| Liberia | Presence Uncertain | Native | – | – |
| Libya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Presence Uncertain | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Palestine, State of | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
Climate change and the associated increase in ambient temperature has been identified as a threat for bat species, and specifically in N. thebaica water stress and aerial hawking are considered factors at risk due to climate change (Sherwin et al. 2013).
Population Information
It is fairly common through most of its African range and is particularly common throughout the assessment region. Both day and night roosts may contain hundreds of individuals (Monadjem 1998), thus we infer a large population size. Overall, it is well represented in museums, with over 1,200 specimens examined by Monadjem et al. (2020). Monadjem (2001) found the adult sex ratio to be highly skewed and seasonally variable in this species, with females usually outnumbering males by 2.8:1 between July and December.
Current population trend: Stable
Continuing decline in mature individuals? Not suspected
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: No evidence
All individuals in one subpopulation: Presumed so
Number of mature individuals in largest subpopulation: >10,000 individuals
Number of Subpopulations: Presumed to be one metapopulation, although phylogeographic and landscape genetic studies are needed to confirm this.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unlikely
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unlikely
Probability of extinction in the wild within 100 years: Unlikely
Population Genetics
A phylogenetic study on slit‐faced bats (Chiroptera: Nycteridae) has uncovered deeply divergent African lineages within the family (Demos et al. 2019). Regarding N. thebaica, a species complex was identified with individuals from South Africa clustering outside the main N. thebaica clade and grouping with individuals from Malawi (N. cf. thebaica 1: Demos et al. 2019), possibly warranting their designation as a separate species.
The species appears to be widespread throughout the assessment region and may comprise a single metapopulation. Considering individual roosts may contain hundreds of individuals, it is expected that the number of mature individuals for this species (metapopulation) exceeds 5000 individuals. Consequently, the effective population size (Ne) is expected to exceed the Ne 500 threshold. To verify these assumptions, a comprehensive population genomic study is recommended.
Habitats and ecology
A savannah species with wide habitat tolerance, the Egyptian Slit-faced Bat occurs in moist and dry savannah, ranging into desert, arid rocky areas, and riparian strips. It appears to occur throughout the savannah and Karoo biomes but avoids open grasslands (Monadjem et al. 2020), possibly because it needs tree cover. For example, it forages by flying low above the ground, listening for prey scurrying on the surface, which may make it susceptible to predation by owls, hence the requirement for tree cover (Monadjem et al. 2020). It has, been recorded from mixed grassland habitats associated with the Mpumalanga highveld (de Jong et al. 2025).
This is essentially a cave-roosting species, but it may also roost during the day in mine adits, Aardvark (Orycteropus afer) holes, rock crevices, culverts under roads, roofs, basements, and hollow trees, typically in open savannah woodland (Smithers and Lobao-Tello 1976; Taylor 1998). It is a clutter forager, and its insectivorous diet varies seasonally, with Orthoptera, Coleoptera and Lepidoptera predominating (Monadjem et al. 2010). Arachnids, amphibians, fish and vegetative matter may also form small proportions of its diet (Skinner and Chimimba 2005).
This gregarious species roosts in colonies ranging in size of up to a few hundred individuals, which are loosely packed and in scattered groups (Skinner and Chimimba 2005). It has also been recorded roosting alongside other species, such as Miniopterus natalensis, Hipposideros caffer and Rhinolophus simulator (Skinner and Chimimba 2005). Although other species are known to hover during copulation, in N. thebaica copulation takes place in flight (Lindeque 1987). In KwaZulu-Natal, studies showed that young are born in November following a five-month gestation period, and lactation was found to last two months (Bernard 1982). In Eswatini, individuals travel an average of 1.1 km between day roosts and foraging areas where foraging home ranges are relatively small (12.9 ha) and overlap significantly with those of neighbouring bats (Monadjem et al. 2009). Records show that this species cannot be kept in captivity for longer than three days (Fenton et al. 1983).
Ecosystem and cultural services: As this species is insectivorous, it contributes to controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Bats often prey on the insect species that destroy crops (Boyles et al. 2011; Kunz et al. 2011). Molecular DNA-sequence studies of insect exoskeletal remains in faecal pellets, confirmed that the species preys upon green vegetable stink bugs, an important insect pest responsible for an estimated R54 million annual harvest loss to the macadamia nut industry in South Africa (Taylor et al. 2013). Further molecular diet studies provided evidence that N. thebaica also consumes important agricultural pest moth species (Weier et al. 2019). Ensuring a healthy population of insectivorous bats can thus result in a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | – |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
Life History
Generation Length: Unknown, however, capture-mark-recapture studies using wing bands, indicate that animals can live for at least 7-8 years (Monadjem et al. 2006).
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean forearm length = 4.72 ±0.21 cm; mean total length = 10.90 ±1.01 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean forearm length = 4.61 ±0.26 cm; mean total length = 10.50 ±0.69 cm (Monadjem et al. 2020)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): Unknown, however the mean mass at birth is 6 g (Bernard 1980)
Gestation Time: Five month or 155-day gestation period (Bernard 1980, 1982)
Reproductive Periodicity: Seasonal breeding, with breeding occurring in June and young born between October to December (Bernard 1982; Monadjem et al. 1998).
Average Annual Fecundity or Litter Size: one pup (Bernard 1980, 1982; Enders et al. 2009)
Natural Mortality: Predation by nocturnal avian predators (e.g. owls and bat hawks).
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Radio-telemetry tracking studies indicate an average home range of 12.9 hectares, with individual bats venturing an average of 1.1 km between their day roost and foraging grounds (Monadjem et al. 2009).
Congregatory: Yes, some day and night roost can harbour one to several hundred individuals (Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region.
Local Livelihood: Unknown
National Commercial Value: Provides essential ecosystem services in the form of insect pest control.
International Commercial Value: Provides essential ecosystem services in the form of insect pest control, including those relating to exported crops (e.g. macadamias).
End Use: Unknown
Is there harvest from captive/cultivated sources of this species? Not that we know of.
Harvest Trend Comments: N/A
Threats
There are currently no major threats to this widespread species. Habitat destruction and degradation have been identified as threats in other regions of this species’ range, such as the Mediterranean (ACR 2024). As a cave-roosting species, N. thebaica may be threatened by human disturbance at roost sites as a result of traditional ceremonies and tourism, which frequently take place in caves. The loss of roosts in the form of old mine tunnels outside the presently known distribution within the assessment region from i) recommissioning of old mines that have been colonised by this species (pers. comm. C. Lötter) or ii) closure (sealing) of old mine tunnels, inter alia, to prevent their exploitation by illegal miners, (Government Notice 446, 21 May 2021), is considered a potential risk. Being a slow- and low-flying species, they are sometimes killed by light motor vehicles when traversing roadways (Collinson et al. 2015; MPTA records).
Conservation
The species occurs in more than 10 protected areas within the assessment region and is found in many large reserves including Kruger National Park, Kgalagadi Transfrontier Park, Mapungubwe National Park, Vhembe Biosphere Reserve, Hluhluwe-Imfolozi Game Reserve and iSimangaliso Wetland Park, thus is considered well protected. Continued protection of the roost sites outside protected areas is required. Additionally, cavities in large trees, such as Baobabs, provide critically important cool microhabitats for this species, and may assist in buffering increases in ambient temperature associated with climate change (Toussaint and McKechnie 2012). Thus, the protection and preservation of these trees is considered crucial for the prolonged local presence of this species.
Recommendations for land managers and practitioners:
- Protect and preserve large trees, which provide critically important cool, microhabitats for this species.
- Reduce pesticide use in agricultural landscapes.
- Farmers are encouraged to retain or rehabilitate natural vegetation and water sources within and adjacent to the confines of their farms, as such areas provide resident bat species with the necessary resources (e.g. roost sites) for their continued presence and associated pest control services Weier et al. 2019).
Research priorities:
- Seasonal behaviour and movement patterns.
- Population size and trends, especially in the case of large, well-established colonies.
- Investigations into how climate change may affect this species.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report 2012. AfricanBats NPC Pretoria, South Africa.
Bernard, R.T.F. 1980. Female reproduction in five species of Natal cave-dwelling Microchiroptera. Ph.D. dissertation. University of Natal, Pietermaritzburg, South Africa, 289 pp.
Bernard, R.T.F. 1982. Female reproductive cycle of Nycteris thebaica (Microchiroptera) from Natal, South Africa. Zeitschrift für Säugetierkunde 47: 12–18.
Boyles, J.G., Cryan, P.M., McCracken, G.F., and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Collinson, W. J., D. M. Parker, R. T. F. Bernard, B. K. Reilly and H. T. Davies-Mostert. 2015. An inventory of Vertebrate roadkill in the Greater Mapungubwe Transfrontier Conservation Area, South Africa. African Journal of Wildlife Research, 45 (3): 301 – 311. doi: 10.3957/056.045.0301.
de Jong, G.J., Melville, H.I. and Richards, L.R. 2025. The Insectivorous Bat Assemblage in Telperion Nature Reserve, Mpumalanga, South Africa: Species Inventory and Call Library. Acta Chiropterologica, 26(2), pp.315-327.
Enders, A.C., Jones, C.J.P., Taylor, P.J. and Carter, A.M. 2009. Placentation in the Egyptian slit-faced bat Nycteris thebaica (Chiroptera: Nycteridae). Placenta, 30(9), pp.792-799.
Fenton, M.B., Gaudet, C.L. and Leonard, M.L. 1983. Feeding behaviour of the bats Nycteris grandis and Nycteris thebaica (Nycteridae) in captivity. Journal of Zoology (London) 200: 347–354.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Lindeque, M. 1987. Observations on mating behaviour in the common slit-faced bat Nycteris thebaica. Madoqua 15: 183–185.
Mammal Diversity Database. 2025. Mammal Diversity Database (Version 2.2). Zenodo. https://doi.org/10.5281/zenodo.15007505
Meester, J.A., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Monadjem, A. 2001. Sexual dimorphism, sex ratio and preliminary recapture rates of Nycteris thebaica (Nycteridae: Chiroptera) in Swaziland. Durban Museum Novitates 26: 49–52.
Monadjem, A. 2005. Survival and roost-site selection in the African bat Nycteris thebaica (Chiroptera: Nycteridae) in Swaziland. Belgian Journal of Zoology 135: 103–107.
Monadjem, A. 2006. Longevity and movement of the Common slit-faced bat Nycteris thebaica. African Bat Conservation News, 9: 7.
Monadjem, A., Reside, A., Cornut, J. and Perrin, M.R. 2009. Roost selection and home range of an African insectivorous bat Nycteris thebaica (Chiroptera, Nycteridae). Mammalia 73: 353–359.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, Second Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Sherwin, H.A., Montgomery, W.I., and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. and Lobao-Tello, J.L.P. 1976. Check list and atlas of the mammals of Mozambique. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Taylor, P. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., Bohmann, K., Steyn, J.N., Schoeman, M.C., Matamba, E., Zepeda-Mendoza, M., Nangammbi, T. and Gilbert, M.T.P. 2013. Bats eat pest green vegetable stinkbugs (Nezara viridula): diet analyses of seven insectivorous species of bats roosting and foraging in macadamia orchards at Levubu, Limpopo Province, South Africa. South African Macadamia Growers’ Association Yearbook, 21, pp.37-43.
Toussaint, D.C. and McKechnie, A.E. 2012. Interspecific variation in thermoregulation among three sympatric bats inhabiting a hot, semi-arid environment. Journal of Comparative Physiology B 182: 1129–1140.
Weier, S.M., Moodley, Y., Fraser, M.F., Linden, V.M., Grass, I., Tscharntke, T. and Taylor, P.J. 2019. Insect pest consumption by bats in macadamia orchards established by molecular diet analyses. Global Ecology and Conservation, 18, p.e00626.
