
Egyptian Fruit Bat
Rousettus aegyptiacus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Rousettus aegyptiacus – (É. Geoffroy, 1810)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – PTEROPODIDAE – Rousettus – aegyptiacus
Common Names: Egyptian Fruit Bat, Egyptian Rousette (English), Rossetto egiziano (Italian), Rousette d’ Égypte (French)
Synonyms: Pteropus egyptiacus É. Geoffroy, 1810; Rousettus egyptiacus (É. Geoffroy, 1810)
Taxonomic Note:
Two geographically isolated subspecies of R. aegyptiacus occur in sub-Saharan Africa (Monadjem et al. 2020). Both subspecies occur in the southern African region: unicolor (Gray, 1870) in Angola and leachii (A. Smith, 1829) in the rest of the region (Monadjem et al. 2020). Additional subspecies: princeps is endemic to Principe Island in the Gulf of Guinea and tomensis is endemic to Sao Tome (Korine 2016). The taxonomic relationship with Rousettus leschenaultii requires further review.
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Lötter, C.1, Richards, L.R.2, Balona, J.3 & da Silva, J.M.4
Reviewer: Richardson, E.J.5
Institutions: 1Inkululeko Wildlife Services (Pty) Ltd, 2Durban Natural Science Museum, 3Gauteng and Northern Regions Bat Interest Group, 4South African National Biodiversity Institute, 5Independent Consultant at Richardson & Peplow Environmental
Previous Assessors and Reviewers: Markotter, W., MacEwan, K., White, W., Cohen, L., Jacobs, D., Monadjem, A., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.J.
Previous Contributors: Raimondo, D., Nicholson, S.K. & Child, M.F.
Assessment Rationale
This species is listed as Least Concern as it is widely distributed and reasonably common within the assessment region (estimated extent of occurrence is 862,963 km2), occurs in multiple protected areas (including Kruger National Park, Table Mountain National Park, and iSimangaliso Wetland Park), and can occur in modified habitats. However, as this species is dependent on caves for roosting in large numbers, protecting key cave roosts is important. Additionally, the rapidly growing number of wind farms especially along the coastline of South Africa’s Western and Eastern Cape provinces, may represent a significant threat for this species. Monitoring wind farm fatalities, cave health, and colony sizes of this species is thus required as the results may necessitate re-assessment of this species’ conservation status.
Regional population effects: Wing-loading of this species is very high (Norberg & Rayner 1987), so its dispersal capacity is good. Due to its patchy distribution, however, rescue effects are questionable.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Lötter C, Richards LR, Balona J & da Silva JM. 2025. A conservation assessment of Rousettus aegyptiacus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species has a disjunct distribution in Africa. It is restricted to areas with fruiting trees and caves, but there are records in all biotic zones, except Mediterranean Coastal and Sahel Savannah. It is patchily distributed across sub-Saharan Africa and North Africa. It also ranges outside of Africa through southwest Asia to Iran and Pakistan; and also in Cyprus (Benda et al. 2004). It occurs from sea level to ~4,000 m above sea level. The subspecies R. a. leachii is found in in Ethiopia, Sudan, Democratic Republic of the Congo (DRC), Uganda, Kenya, Tanzania, Zambia, Malawi, Zimbabwe, Mozambique, and the east and south of South Africa. Although Korine (2016) states that this species occurs in both Eswatini and Lesotho, there are no available records to support this. While there is a tentative record from close to the Lesotho border (ACR 2024), neither Eswatini nor Lesotho have voucher specimens for this species (ACR 2024). Similarly, Skinner and Chimimba (2005) state that the species is absent from Lesotho and Eswatini, and Monadjem et al. (2020) state that there is a gap in this species’ distribution in Eswatini and southern Mozambique, with records reappearing in northern South Africa, through Zimbabwe, northern Mozambique, southern Zambia, Malawi and the southern DRC (Monadjem et al. 2020). The subspecies, R. a. unicolor occurs in western Angola northwards through western DRC, Gabon, Cameroon, Nigeria, Ghana, Togo, Côte d’Ivoire, Liberia, Senegal and the Gambia (Korine 2016).
Within the assessment region, R. a. leachii occurs from Cape Town, along the coast to KwaZulu- Natal (Monadjem et al. 2020) and through to Mpumalanga and Limpopo (Skinner & Chimimba 2005). It occurs in the moist, well-watered eastern parts of the region, but is absent from the dry west; which is possibly an indication of its reliance on fruiting trees (Jacobsen & du Plessis 1976; Monadjem et al. 2020). The type locality of R. a. leachii is from the Company Gardens, Cape Town (Monadjem et al. 2020). The estimated extent of occurrence for this species is 862,963 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 4000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
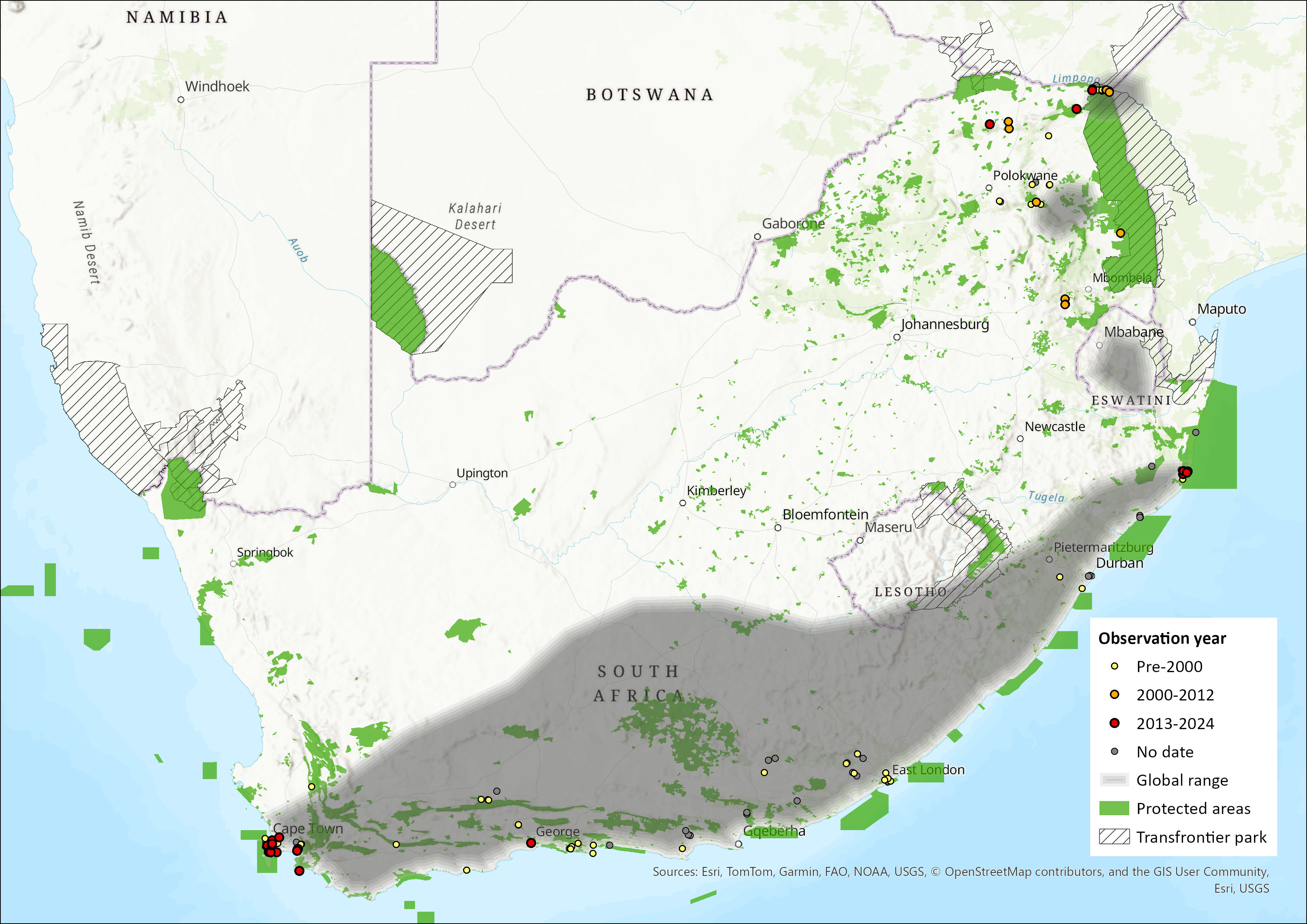 Figure 1. Distribution records for Egyptian Fruit Bat (Rousettus aegyptiacus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Egyptian Fruit Bat (Rousettus aegyptiacus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | Resident |
| Burundi | Extant | Native | – | Resident |
| Cameroon | Extant | Native | – | Resident |
| Congo | Extant | Native | – | Resident |
| Congo, The Democratic Republic of the | Extant | Native | – | Resident |
| Cyprus | Extant | Native | – | Resident |
| Côte d’Ivoire | Extant | Native | – | Resident |
| Egypt | Extant | Native | – | Resident |
| Equatorial Guinea | Extant | Native | – | Resident |
| Eritrea | Extant | Native | – | Resident |
| Ethiopia | Extant | Native | – | Resident |
| Gabon | Extant | Native | – | Resident |
| Gambia | Extant | Native | – | Resident |
| Ghana | Extant | Native | – | Resident |
| Guinea | Extant | Native | – | Resident |
| Iran, Islamic Republic of | Extant | Native | – | Resident |
| Israel | Extant | Native | – | Resident |
| Jordan | Extant | Native | – | Resident |
| Kenya | Extant | Native | – | Resident |
| Lebanon | Extant | Native | – | Resident |
| Liberia | Extant | Native | – | Resident |
| Libya | Extant | Native | – | Resident |
| Malawi | Extant | Native | – | Resident |
| Mozambique | Extant | Native | – | Resident |
| Nigeria | Extant | Native | – | Resident |
| Oman | Extant | Native | – | Resident |
| Pakistan | Extant | Native | – | Resident |
| Palestine, State of | Extant | Native | – | Resident |
| Rwanda | Extant | Native | – | Resident |
| Saudi Arabia | Extant | Native | – | Resident |
| Senegal | Extant | Native | – | Resident |
| Sierra Leone | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Sudan | Extant | Native | – | Resident |
| Syrian Arab Republic | Extant | Native | – | Resident |
| Tanzania, United Republic of | Extant | Native | – | Resident |
| Togo | Extant | Native | – | Resident |
| Türkiye | Extant | Native | – | Resident |
| Uganda | Extant | Native | – | Resident |
| United Arab Emirates | Extant | Native | – | Resident |
| Yemen | Extant | Native | – | Resident |
| Zambia | Extant | Native | – | Resident |
| Zimbabwe | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Research suggests that climate change could result in negative demographic responses among Pteropodid Old World fruit bats (Festa et al. 2023). More specifically, the Egyptian Fruit Bat could be impacted by climate change due to changes in fruit and flower availability (Sherwin et al. 2013).
Arumoogum et al. (2019) used ensemble niche modelling to investigate the relative influence of abiotic and biotic variables on the potential current and future distributions of three fruit bat species in southern Africa. Suitable habitat of R. aegyptiacus was primarily mediated by temperature under current climatic conditions yet by freestanding fig distribution under both moderate and extreme future climate change scenarios.
Czenze et al. (2020) reported that this cave-roosting species has a physiological heat tolerance of approximately 38 °C. Unlike insectivorous bat species, South African Egyptian Fruit bats do not enter torpor when exposed to suboptimal ambient conditions (Barclay et al. 2017). This limit may limit their ability to employ physiological strategies in response to temperatures exceeding their thermal tolerance.
Furthermore, the species exhibits notable physiological stress and a decline in body condition during the winter and early spring when food resources are limited or of poor quality (Korine et al. 1999). Females may be particularly susceptible to bioclimatic fluctuations as their lactation period, and consequently, pup survival is correlated with notable peaks in rainfall and fruiting trees (Penzhorn and Rautenbach, 1988).
The above suggests that the species will become increasingly threatened with future likely increases in temperatures, altered rainfall patterns and a loss of fig trees within its distribution range.
Population Information
Common and sometimes abundant near large caves. In Africa, it occurs in large colonies of up to 50,000 individuals (ACR 2023). This species is well represented in museums, with more than 200 specimens examined in Monadjem et al. (2020). Within the assessment region, only a few colonies have been counted, which range from small colonies of 15 individuals to colonies of 12,000 individuals. Abundance seems to vary greatly between seasons. For example, in the Mission Rocks caves in the Greater St Lucia Wetland Park, numbers fluctuate from over 5,000 to fewer than 300 in summer (Monadjem et al. 2020). Conversely, in caves in the Tzaneen area of Limpopo, numbers reached over 9,000 individuals at the Matlapitsi Cave in March–April (late wet season) and declined to just over 3,000 individuals in June–August (winter) (Jacobsen and du Plessis 1976), which suggests some movement between the two sites Indeed, a bat recorded in the Tzaneen area was subsequently recovered 500 km away at Cape Vidal in northern KwaZulu-Natal (Jacobsen & du Plessis 1976).
Current population trend: Unknown
Continuing decline in mature individuals? Unknown
Number of Subpopulations: Unknown
Extreme fluctuations in the number of subpopulations: Some fluctuations but noting extreme reported thus far.
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Unknown
Number of mature individuals in largest subpopulation: 12000
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Not likely
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 100 years: Unknown
Population Genetics
Limited molecular work has been undertaken on this species, often relating to the genus as a whole in a broad phylogenetic context (e.g. Hollar and Springer 1997). However, given that this species is known to migrate over hundreds of kilometres (Monadjem et al. 2020), it is likely that the species exists as a single metapopulation within South Africa, extending beyond into neighbouring countries. Additionally, since these bats are known to roost in large numbers (e.g., thousands) (Jacobsen and du Plessis 1976; Monadjem et al. 2020), this species’ effective population size is thought to greatly exceed the threshold of 500.
Habitats and ecology
This species has a broad habitat tolerance and is found in arid to moist tropical and subtropical biomes, so long as fruiting trees and appropriate roosting caves are available. It feeds on soft fruits (date, carob, mulberry, azedarach, fig, apricot, peach, mango and apple), flowers and occasionally eats leaves. These bats often forage in orchards, such as Litchi chinensis (Jacobsen & du Plessis 1976). They are also capable of foraging in urban habitats with fruiting trees (Barclay and Jacobs 2011). Other fruits recorded in its diet include Syzygium spp., Harpephyllum caffrum, Ekebergia capensis, Prunus africana and Diospyros senensis (Jacobsen & du Plessis 1976; Herzig-Straschil & Robinson 1978; Thomas & Fenton 1978). In Cape Town, Barclay et al. (2006) observed this species deliberately feeding on Scarabid beetles (Pachnoda sinuata).
It is a strictly cavernicolous species (Herzig-Straschil & Robinson 1978), roosting in moist natural caverns and artificial structures including underground irrigation tunnels (ghanats), ruins, mines, and open wells. The species often roosts with other bat species, such as Miniopterus natalensis, Rhinolophus capensis, M. fraterculus and possibly R. acrotis (ACR 2024). The presence of caves is suspected to influence their distribution more than vegetation associations (Monadjem et al. 2020), although see climate change section below. They are gregarious and roost in large numbers. They also migrate over hundreds of kilometres (Monadjem et al. 2020). Additionally, species of this genus do echolocate by generating sound through repetitive tongue clicks, making Rousettus an exception among fruit bats (Monadjem et al. 2020).
Ecosystem and cultural services: As this species is frugivorous, these bats play a crucial role in pollination and seed dispersal of many plant species, and thus plant community persistence and habitat regeneration (Herzig-Straschil and Robinson 1978; Monadjem et al. 2020; Kunz et al. 2011). This species has been recorded to pollinate Baobab Trees (Adansonia digitata) (Herzig-Straschil and Robinson 1978) and disperse the seeds of Ficus spp. (ACR 2015), which represent a keystone resource for numerous frugivores and other animals, and which are economically important trees in the African savannah.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.7. Forest -> Forest – Subtropical/Tropical Mangrove Vegetation Above High Tide Level | – | Suitable | – |
| 1.8. Forest -> Forest – Subtropical/Tropical Swamp | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | Yes |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | Yes |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
| 17. Other | – | Suitable | – |
Life History
Generation Length: 5
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean forearm length = 9.26 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean forearm length = 9.20 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: Unknown
Maximum Size (in cms): Unknown
Size at Birth (in cms): Unknown
Gestation Time: 105-107 days
Reproductive Periodicity: Parturition usually occurs in the wet summer months (Monadjem et al. 2020).
Average Annual Fecundity or Litter Size: One, or occasionally two offspring (Penzhorn and Rautenbach 1988).
Natural Mortality: Unknown
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: This species can migrate over hundreds of kilometres (Monadjem et al. 2020).
Congregatory: Colonies in Africa may comprise up to 50,000 individuals (ACR 2024), but in the assessment region, colonies range from only a few to 12,000 individuals (Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or utilised within the assessment region. However, it is hunted for food in some cave systems in Africa (Korine 2016).
National Commercial Value: No
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? The species is harvested for bushmeat elsewhere in Africa.
Harvest Trend Comments: Not known from the region.
Threats
Cave disturbance may be a significant threat to this species. Similarly, persecution is a problem in parts of its range. For example, this species has been shown to cause damage to litchi orchards in South Africa (Jacobsen & du Plessis 1976), which can result in localised persecution. Additionally, the rapidly growing number of wind farms in the Eastern Cape, including areas close to roost sites presents a serious threat to this species (Baerwald et al. 2008; Cryan & Barclay 2009; Rydell et al. 2010; MacEwan 2016). Bats that fly near to turbine blades generally collide directly with a blade, but may experience barotrauma, which is tissue damage caused by rapid changes in air pressure near the blades) (Baerwald et al. 2008; Cryan & Barclay 2009; Rydell et al. 2010). Growing numbers of fatalities of R. aegyptiacus from wind turbines have been recorded since 2015 and are likely to escalate (MacEwan 2016; Aronson 2022; Inkululeko Wildlife Services unpubl. data). The impact that these threats will have on the overall population in the assessment region is currently unknown and the species needs to be monitored. Furthermore, its dependency on fruit-bearing trees and long-distance dispersal habits makes this species vulnerable to removal of temporary roost sites and shifts in habitats and environmental gradients associated with climate change (Sherwin et al. 2013).
Conservation
This species is present, at least at times of the year, in a number of protected areas including: Kruger National Park, Legalameetse Nature Reserve, iSimangaliso Wetland Park, Hluhluwe-iMfolozi Park, Umlazi Nature Reserve, Addo Elephant National Park, Wolkberg Nature Reserve, Garden Route National Park, Keurboomrivier Nature Reserve, Kogelberg National Park, Gamkapoort Nature Reserve, Geelkrans Provincial Nature Reserve, Marloth Nature Reserve, Grootwinterhoek Wilderness Area, Jonkershoek Nature Reserve and Table Mountain National Park.
Identification of key roost sites and subsequent protected area expansion in areas suffering from habitat loss would benefit this species. Additionally, the impact of wind farming should be monitored to determine its impact on this species. An effective and standardised method for quantifying the activity of fruit bats at proposed and operational wind farm sites is needed. Thermal binoculars may prove useful in this regard. To mitigate mortalities from turbine collisions, development of wind farms must avoid encroachment into the prescribed buffers around important habitat (e.g. forest, riverine woodland, large fruiting trees) and confirmed and potential roosts of this species (for buffer recommendations see MacEwan et al. 2020 or later). Turbine-fatalities of this species must be reduced below fatality threshold values (as calculated according to MacEwan et al. 2018 or later) with interventions such as curtailing turbines at low wind speeds during times of the year with known fruit bat fatalities (Baerwald et al. 2009; Berthinussen et al. 2010; Arnett et al. 2011). As high bat fatalities at wind farms cannot be easily offset (Aronson et al. 2018; Mark Botha pers. comm.), avoidance and minimisation of bat fatalities is critical. Improved conservation (protection, rehabilitation, and expansion) of forest, riverine, and savannah habitats would, however, be beneficial for this species.
Recommendations for land managers and practitioners:
- Development of wind farms must strictly avoid encroachment into the prescribed buffers around roosts and other important habitat for fruit bats, especially since current options for mitigating fatalities at wind farms are more limited for fruit bats compared to insectivorous bats.
- Turbine-fatalities of R. aegyptiacus must be reduced with interventions such as curtailing turbines at low wind speeds during times of the year with known fatalities.
- Data sharing by wind farm managers into a national database is needed to be able to calculate cumulative impacts and thereafter implement collaborative mitigation and management efforts.
- Farmers should be educated on the ecological benefits of having fruit bats around their orchards, and the importance of preserving natural habitat on their farms.
- Protect large fruit bearing trees.
Research priorities:
- The taxonomic relationship with R. leschenaultii requires further review and large-scale phylogeographic studies ought to be conducted to eludicate potential genealogical lineages across southern Africa, particularly in relation to previously described subspecies.
- Identify key roost sites and undertake systematic monitoring to determine subpopulation trends.
- A meta-analysis of all recorded R. aegyptiacus fatalities at wind farms in South Africa to date, to assess the cumulative impact of this industry on this species, and to identify any spatial, temporal, or turbine-specific trends, which may assist in devising effective fatality mitigation for future implementation.
- Research to assess the efficacy of different mitigation methods to reduce fruit bat mortality at wind farms. For example, it is not known whether turbine blade painting (to mitigate avian fatalities) is beneficial or detrimental for fruit bats.
- Wind farm carcasses of this species could be used to obtain greater insight into the biogeography, genetics, reproductive biology, ecology (e.g. diet), and ecosystem services of this species in the assessment region.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report 2023. Van Cakenberghe, V. and Seamark, E.C.J. (Eds.) African Chiroptera Project, Pretoria. i – xviii; 1 – 1210 pp.
Arnett, E.B., Huso, M.M., Schirmacher, M.R. and Hayes, J.P. 2011. Altering turbine speed reduces bat mortality at wind-energy facilities. Frontiers in Ecology and the Environment 9: 209–214.
Aronson, J. 2022. Current state of knowledge of wind energy impacts on bats in South Africa. Acta Chiropterologica 24: 221-238.
Arumoogum, N., Schoeman, M.C. and Ramdhani, S. 2019. The relative influence of abiotic and biotic factors on suitable habitat of Old World fruit bats under current and future climate scenarios. Mammalian Biology 98: 188-200.
Baerwald, E.F., D’Amours, G.H., Klug, B.J. and Barclay, R.M. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18: 695–696.
Baerwald, E.F., Edworthy, J., Holder, M. and Barclay, R.M. 2009. A large-scale mitigation experiment to reduce bat fatalities at wind energy facilities. Journal of Wildlife Management 73: 1077–1081.
Barclay, R.M., Barclay, L.E. and Jacobs, D.S. 2006. Deliberate insectivory by the fruit bat Rousettus aegyptiacus. Acta Chiropterologica 8: 549–553.Barclay, R.M. and Jacobs, D.S. 2011. Differences in the foraging behaviour of male and female Egyptian fruit bats (Rousettus aegyptiacus). Canadian Journal of Zoology 89(6):466-73.
Barclay, R.M., Jacobs, D.S., Harding, C.T., McKechnie, A.E., McCulloch, S.D., Markotter, W., Paweska, J. and Brigham, R.M. 2017. Thermoregulation by captive and free-ranging Egyptian rousette bats (Rousettus aegyptiacus) in South Africa. Journal of Mammalogy 98(2):572-8.
Benda, P., Hanak, V., Andreas, M., Reiter, A. and Uhrin, M. 2004. Two new species of bats (Chiroptera) for the fauna of Libya: Rhinopoma hardwickii and Pipistrellus rueppellii. Myotis 41: 109–124.
Berthinussen, A., Richardson, O.C. and Altringham, J.D. 2010. Bat Conservation: Global Evidence for the Effects of Interventions. Synopses of Conservation Evidence. Pelagic Publishing, Exeter.
Cryan, P.M. and Barclay, R.M. 2009. Causes of bat fatalities at wind turbines: hypotheses and predictions. Journal of Mammalogy 90: 1330–1340.
Czenze, Z. J., Smit, B., van Jaarsveld, B., Freeman, M. T. and McKechnie, A. E. 2022. Caves, crevices and cooling capacity: Roost microclimate predicts heat tolerance in bats. Functional Ecology, 36, 38–50.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Herzig-Straschil, B. and Robinson, G.A. 1978. On the ecology of the fruit bat, Rousettus aegyptiacus leachi (A. Smith, 1829) in the Tsitsikama Coastal National Park. Koedoe 21: 101–110.
Hollar, L.J and Springer, M.S. 1997. Old World fruitbat phylogeny: Evidence for convergent evolution and an endemic African clade. https://www.pnas.org/doi/10.1073/pnas.94.11.5716
Jacobsen, N.H.G. and du Plessis, E. 1976. Observations on the ecology and biology of the Cape Fruit Bat Rousettus aegyptiacus leachi in the eastern Transvaal. South African Journal of Science 72: 270– 273.
Korine, C., Izhaki, I. and Arad, Z. 1999. Is the Egyptian fruit-bat Rousettus aegyptiacus a pest in Israel? An analysis of the bat’s diet and implications for its conservation. Biological Conservation 88 (3): 301-306. https://doi.org/10.1016/S0006-3207(98)00126-8.
Korine, C. 2016. Rousettus aegyptiacus. The IUCN Red List of Threatened Species 2016.e.T29730A22043105.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
MacEwan, K.L. 2016. Fruit bats and wind turbine fatalities in South Africa. African Bat Conservation News 42: 3–5.
MacEwan, K., Aronson, J., Richardson, K., Taylor, P., Coverdale, B., Jacobs, D., Leeuwner, L., Marais, W. and Richards, L. 2018. South African Bat Fatality Threshold Guidelines. Edition 2. South African Bat Assessment Association, South Africa.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association, South Africa.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Simelane, F., Shapiro, J.T., Gumbi, B.C., Mamba, M.L., Sibiya, M.D., Lukhele, S.M. and Mahlaba, T.A. 2021. Using species distribution models to gauge the completeness of the bat checklist of Eswatini. European Journal of Wildlife Research 67(2):21.
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Penzhorn, B.L. and Rautenbach, I. 1988. Reproduction of the Egyptian fruit bat Rousettus aegyptiacus in the southern tropics. South African Journal of Wildlife Research 18(3):88-92.
Rydell, J., Bach, L., Dubourg-Savage, M-J., Green, M., Rodrigues, L. and Hedenström, A. 2010. Bat mortality at wind turbines in northwestern Europe. Acta Chiropterologica 12: 261–274.
Sherwin, H.A., Montgomery, W.I. and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Thomas, D.W. and Fenton, M.B. 1978. Notes on the dry season roosting and foraging behaviour of Epomophorus gambianus and Rousettus aegyptiacus (Chiroptera: Pteropodidae). Journal of Zoology 186: 403–406.
