Dwarf Mongoose
Helogale parvula

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Helogale parvula – (Sundevall, 1847)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – HERPESTIDAE – Helogale – parvula
Common Names: Common Dwarf Mongoose, Dwarf Mongoose (English), Dwergmuishond (Afrikaans), Iduha (Ndebele), Motswitswane (Sesotho), Lefswekete, Leswêkêtê, Leswekete, Mothibitshwane (Setswana), Matswi (Venda), Mangouste naine (French), Zwergichneumon (German)
Synonyms: No Synonyms
Taxonomic Note:
Coetzee (1977) recognised three sections and a total of 13 subspecies of Helogale parvula, but Creel (2013) highlighted that the subspecific taxonomy is confused and in severe need of revision. According to the current state of knowledge, H. p. parvula is the only subspecies present in the assessment region.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Nicholson, S.K.1 & da Silva, J.M.2
Reviewer: Do Linh San, E.3 & Patel, T.1
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3Sol Plaatjie University
Previous Assessors & Reviewers: Sharpe, L., Kern, J., Stuart, C., Stuart, M. & Do Linh San, E.
Previous Contributor: Madikiza, Z.
Assessment Rationale
The Common Dwarf Mongoose (hereafter Dwarf Mongoose) is listed as Least Concern as it is common across its range, sometimes attaining high densities (from 20–42 individuals / km2 in some study sites in Limpopo) and is present in several protected areas. However, its distribution is restricted to the northeast of the assessment region. There are no major threats that could cause a range-wide population decline. Accidental persecution through poisoning and controlled burning may lead to local declines, whilst wildlife ranching may have a positive effect by conserving more suitable habitat and connecting subpopulations. Changing rainfall patterns associated with climate change could affect insect abundance and have an adverse effect on this species, but this would require further investigation.
Regional population effects: Dispersal across regional borders is suspected as the range is continuous between South Africa and Eswatini, Mozambique, Zimbabwe and Botswana, respectively; and the species is not constrained by fences.
Red List Index
Red List Index: No change
Recommended Citation: Nicholson SK & da Silva JM. 2025. A conservation assessment of Helogale parvula. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
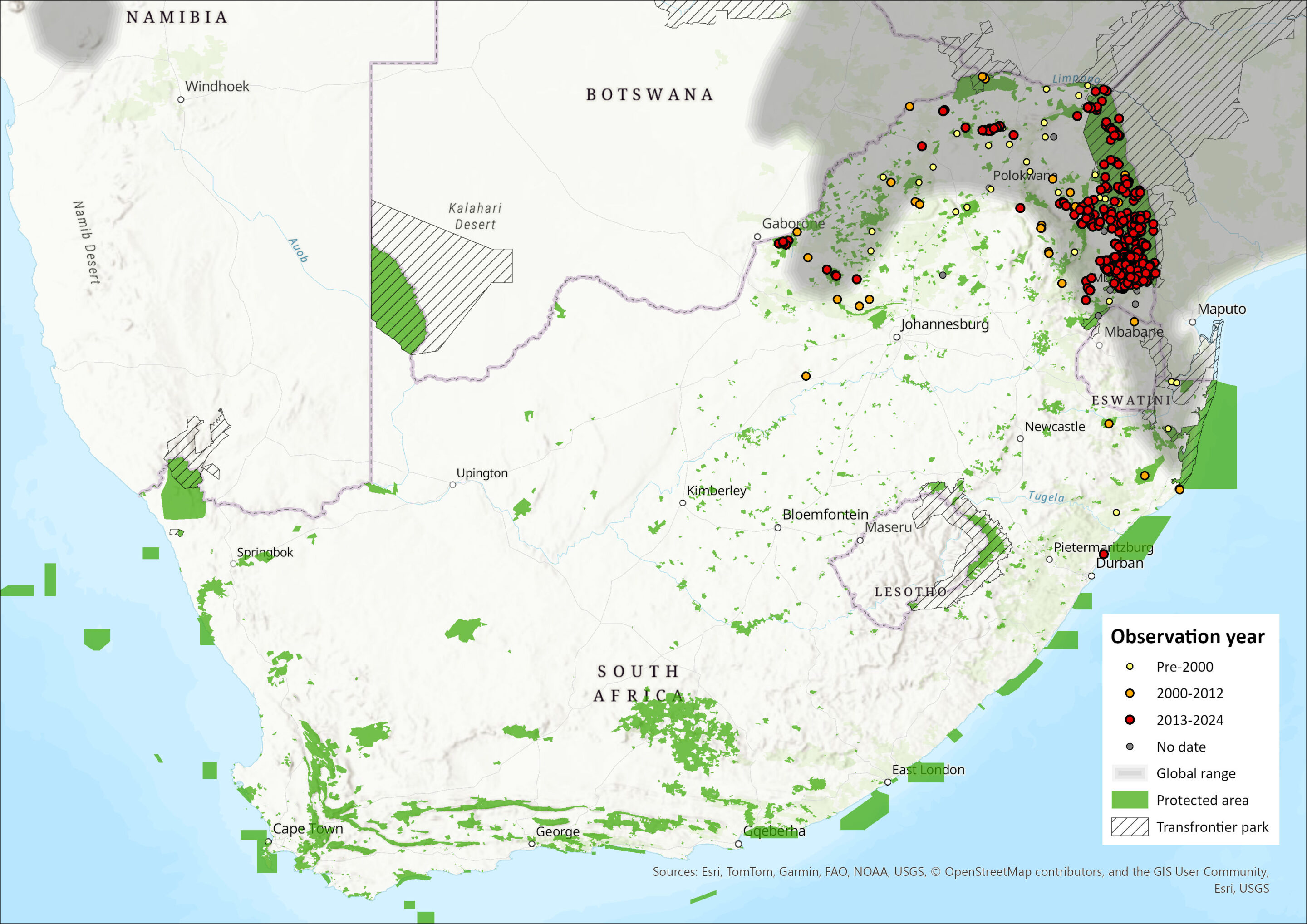
The Dwarf Mongoose is widely distributed from southern Somalia and Ethiopia south to northeast KwaZulu-Natal in South Africa and westwards to southeastern Democratic Republic of the Congo, southwest and central Angola, and northern Namibia (Creel 2013). It is usually found up to elevations of around 2,000 m asl.
Within the assessment region, it occurs within northern KwaZulu-Natal (southernmost range), Mpumalanga and Limpopo eastern Lowveld and Limpopo Valley into North West Province. However, it is rare in the North West, only occurring in the northern two districts (Power 2014).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Dwarf Mongoose (Helogale parvula) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Djibouti | Presence Uncertain | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Based on 11 years of observation (L. Sharpe unpubl. data), population levels fluctuate considerably depending on insect abundance, which in turn is dependent on rainfall. Thus, changing rainfall patterns associated with climate change could have an adverse impact on this species. This would require further investigation.
Population
It has been reported as the most abundant small carnivore in areas of open woodland or wooded savannah, with densities reaching as high as 31 individuals / km2 in Serengeti grasslands (Rood 1983) though estimates of around 5 individuals / km2 are more typical (Waser et al. 1995). Within the assessment region, recorded density is often much higher; for example, 20 individuals / km2 at Phuza Moya Private Game Reserve, near Hoedspruit, Limpopo Province (L. Sharpe unpubl. data), or even a record density of 42 individuals / km2 at the start of the breeding season in Sorabi Rock Lodge Reserve (Hoffmann et al. 2014). Additionally, it is the most frequently seen diurnal carnivore along the length of the Soutpansberg range in Limpopo Province (C. Stuart & M. Stuart pers. obs. 1985–2013). Generation length is estimated to be four years (Sharpe et al. 2015).
Population size is difficult to extrapolate, but there are certainly over 1,000 mature individuals and probably over 10,000 in the assessment region. More monitoring is needed to determine population size and trends accurately, but it is likely that the overall population is stable due to lack of major threats. Local declines may be caused by accidental persecution and controlled burning, while at the same time wildlife ranching is likely to be expanding favourable habitat for this species (see Use and Trade).
Population Information
Current population trend: Probably stable
Continuing decline in mature individuals: Unknown, but probably not
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: It is not currently possible to determine the extent or number of subpopulations.
Severely fragmented: No. Favourable habitat is relatively well connected across this species’ range.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While no population genetic study has been conducted across the species’ entire range, a localised study with the assessment region has provided great insight into the life-history and structuring within the species (Arbon et al. 2024).
As with other mongoose species, H. parvula are obligate cooperative breeders, with breeding pairs unable to reproduce successfully without help from non-breeders. The species is typically structured in small groups of related individuals and movement between groups can be observed; however, such movement does not prevent inbreeding (Keane et al. 1996; Arbon et al. 2024). This inbreeding likely explains the substantial genetic structuring observed within a private game reserve in Limpopo Province, South Africa (Arbon et al. 2024). However, the dominant breeding individuals within each group were less related than under random mating scenarios. Moreover, a high degree of heterozygosity was detected. This evidence suggests the existence of an effective mate avoidance mechanism within dwarf mongooses.
While the Arbon et al. (2024) study provided great insight into the structure and diversity within part of H. parvula‘s range, a more comprehensive population genetic study across the entire assessment region and into neighbouring countries would help understand if such genetic structuring is common throughout the range or variable, with some areas showing less genetic differentiation.
Even though data is limited, H. parvula is considered one of the as the most abundant small carnivore, with a wide distribution. No subpopulations are thought to have gone extinct, therefore, it can still be possible to quantify the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM). This indicator would receive a value of 1.0, reflecting all known populations/subpopulation remaining.
While no census (Nc) or effective population size (Ne) data exists, it is estimated that within the assessment region there are between 1,000-10,000 individuals. Applying a Ne/Nc conversion ratio of 0.1-0.3, these estimates amount to an Ne of between 100-3000 for the whole species, which cradles the Ne 500 ratio for a genetically stable population. However, given that there are dominant breeders in any group, these conversion ratios may be too high. Considering this and given the existence of genetic sub structuring, it is difficult to estimate the GBF’s headline genetic indicator – proportion of populations with Ne > 500. It could be that the entire population within the assessment region falls below Ne 500 (as indicated by Ne = 100 calculated above, or even lower if a different conversion ratio applies) or perhaps some populations far exceed the Ne 500 threshold based on increased connectivity between certain groups/countries.
Habitats and ecology
Dwarf Mongooses inhabit open woodlands, thickets and wooded savannahs, particularly where there are termitaria, rock outcroppings or crevices, or hollow logs and trees for use as dens (Hiscocks & Perrin 1991; Creel 2013; Hoffmann et al. 2014). Waser et al. (1995) noted that the most important determinant of habitat preference in this species is the density of suitable dens, but this might not necessarily be the case (Hoffmann et al. 2014). It is not found in highly arid areas or in dense riparian vegetation.
Helogale parvula is almost entirely insectivorous, feeding primarily on beetles, beetle larvae and termites, although it will occasionally prey on small vertebrates (Creel 2013). In Phuza Moya Private Game Reserve, this small carnivore very rarely consumed termites (despite living in termite mounds), whereas grasshoppers, spiders, crickets and scorpions were common prey items (L. Sharpe pers. obs. 2006–2016).
Dwarf Mongooses are diurnal, obligate cooperative breeders that live in stable social groups of 3–30 individuals (Sharpe et al. 2016). Within their well-defended yet overlapping territories, group members forage together over distances of 0.5–3.5 km (J. Kern unpubl. data) and may utilise up to 30 different sleeping refuges (Rood 1983; Hoffman et al. 2014). Since individuals predominantly dig for invertebrate prey they are particularly vulnerable to predation while foraging and a sentinel is often posted. Territory size is similar in the Serengeti (0.25–0.35 km2; Rood 1978) and in Limpopo (0.3–0.4 km2; Sharpe et al. 2012) but is twice to thrice as large in the Taru Desert, Kenya (0.65–0.96 km2; Rasa 1987). Social groups normally consist of an alpha pair, their adult offspring and 1–3 immigrant males (Rood 1986). The dominant pair is responsible for 80% of pups (Keane et al. 1994) and their subordinate helpers of both sexes have a positive influence on reproductive success (Creel & Waser 1994). All adults take part in cooperative activities such as territory defence, sentinel behaviour, allo-grooming and pup care (Rood 1983). In South Africa, groups rear 2–3 consecutive litters during the summer wet season (October–April; Photo 1). Gestation is c. 50 days; inter-birth interval averages 60 days and mean litter size (at pup emergence) is 4.1 (N = 53 litters; L. Sharpe unpubl. data). Individuals reach sexual maturity by one year (Creel et al. 1992), but males usually delay dispersal until 2–3 years of age (Rood 1990). Females tend to remain in their natal group for life (queuing for the alpha position), and their reproduction is suppressed by the alpha female via endocrine mechanisms and infanticide (Creel et al. 1992). Dispersing females, unlike males, are unable to join established groups (Rood 1986) and so must found their own groups. Because group size is positively correlated with survival of pups, adults and groups (Rasa 1987; Rood 1990; Creel & Waser 1994), small, newly established groups are rarely successful (Rasa 1986; Rood 1986) and vacant territories often remain unoccupied for years (Rood 1986). The maximum lifespan of wild Dwarf Mongooses is 13 years (Rood 1990; L. Sharpe pers. obs. 2016).
Ecosystem and cultural services: Dwarf Mongooses share a co-dependent relationship with a number of bird species, for example hornbills of the genus Tockus (Rasa 1983) or Fork-Tailed Drongos (Dicrurus adsimilis; Sharpe et al. 2010). Forming mixed-species foraging parties, they provide the birds with additional foraging opportunities (i.e. flushed prey) while utilising the birds’ anti-predator vigilance and warning calls.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
Life History
Generation Length: 4 years
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not used in the bushmeat trade in South Africa, nor traditional medicine as far as we are aware. Its pelt has no commercial value. Pups are sometimes taken from the wild for pets, but they usually die of stress-related illness within about 12 months (G.J. Scheepers pers. comm. 2009).
Wildlife ranching and the private sector are suspected to be having a positive effect on this species by conserving more suitable habitat and possibly helping to connect subpopulations. However, more research needs to be done to confirm this.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
No |
– |
– |
– |
National Commercial Value: No
International Commercial Value: No
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Dwarf Mongooses do not currently face major threats. They are not persecuted directly by wildlife ranchers, but some individuals may be killed as bycatch in control programmes of damage-causing animals (DCAs), especially where poison baits are in use. The impact is however believed to be minimal.
Population levels are possibly locally reduced by controlled burning operations, either directly or indirectly through changes to habitat structure potentially lowering food availability and increasing predation risk. Numerous private wildlife ranches do not use burning, which may support higher populations. However, the impact of fire on this species has not yet been quantified.
Habitat trend: Stable or possibly increasing due to an increase in the wildlife ranching industry.
Conservation
Dwarf Mongooses are present in most protected areas across the assessment region, including Kruger and Mapungubwe national parks, Waterberg complex and Soutpansberg Biosphere Reserve. Since they attain high densities and have small territories, even small, protected areas and wildlife ranches or game farms can harbour large populations (Creel 2013).
Recommendations for land managers and practitioners:
- Minimise use of non-selective control methods (e.g. poison) for DCAs.
- Private landowners should ensure that they do not burn the land too frequently and that termite mounds are conserved.
- Create conservancies to protect and connect favourable habitat.
Research priorities: This is one of the best-studied African small carnivores (see review in Creel 2013), but the following research topics will assist in gathering conservation-relevant information:
- Long-term monitoring of (some) subpopulations.
- Evaluation of relative impact of wildlife ranching on habitat and populations.
- Documenting the degree to which controlled burning impacts on population levels.
There is an ongoing research project on this species located at Sorabi Rock Lodge near Hoedspruit in Limpopo Province. The project, which focuses on the species’ behavioural ecology, was established in 2011 and is run by Bristol University.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. This will help in improving this species’ distribution map.
Bibliography
Arbon, J.J., Morris-Drake, A., Kern, J.M., Howell, G.M.K., Wentzel, J., Radford, A.N. & Nichols, H.J. 2024. Life-history and genetic relationships in cooperatively breeding dwarf mongoose groups. R. Soc. Open Sci. 11241125 http://doi.org/10.1098/rsos.241125
Coetzee, C.G. 1977. Order Carnivora. Part 8. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-42. Smithsonian Institution Press, Washington, DC, USA.
Creel SR, Creel N, Wild DE, Montfort DL. 1992. Behavioural and endocrine mechanisms of reproductive suppression in Serengeti dwarf mongooses. Animal Behaviour 43: 231–245.
Creel SR, Waser PM. 1994. Inclusive fitness and reproductive strategies in dwarf mongooses. Behavioral Ecology 5: 339–348.
Creel, S. 2013. Helogale parvula Dwarf Mongoose. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 368-373. Bloomsbury, London, UK.
Hiscocks, K. and Perrin, M.R. 1991. Den selection and use by dwarf mongooses and banded mongooses in South Africa. South African Journal of Wildlife Research 21: 119-122.
Hoffmann, M., Roberts, R.L. and Kern, J. 2014. Tree climbing and denning by Common Dwarf Mongoose Helogale parvula. Small Carnivore Conservation 49: 66-67.
Keane B, Creel SR, Waser PM. 1996 No evidence of inbreeding avoidance or inbreeding depression in a social carnivore. Behav. Ecol. 7, 480–89. doi:10.1093/beheco/7.4.480
Keane B, Waser PM, Creel SR, Creel NM, Elliott LF, Minchella DJ. 1994. Subordinate reproduction in dwarf mongooses. Animal Behaviour 47: 65–75.
Power, J.P. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rasa OAE. 1983. Dwarf mongoose and hornbill mutualism in the Taru Desert, Kenya. Behavioral Ecology and Sociobiology 12: 181–190.
Rasa OAE. 1986. Ecological factors and their relationship to group size, mortality and behaviour in the dwarf mongoose. Cimbebasia 8: 15–21.
Rasa OAE. 1987. The dwarf mongoose: a study of behavior and social structure in relation to ecology in a small social carnivore. Advances in the Study of Behavior 17: 121–163.
Rood JP. 1978. Dwarf mongoose helpers at the den. Zeitschrift für Tierpsychologie 48: 277–287.
Rood JP. 1990. Groups size, reproduction, and routes to breeding in dwarf mongooses. Animal Behaviour 39: 566–572.
Rood, J.P. 1983. The social system of the Dwarf Mongoose. In: J.F. Eisenberg and D.G. Kleiman (eds), Advances in the study of mammalian behaviour, American Society of Mammalogists (Special Publication 7) and Allen Press, Lawrence, KS, USA.
Rood, J.P. 1986. Ecology and social evolution in the mongooses. In: D. Rubenstein and R. Wrangham (eds), Ecological Aspects of Social Evolution, pp. 131-152. Princeton University Press, Princeton, USA.
Sharpe L, Kern J, Do Linh San E. 2015. Helogale parvula. The IUCN Red List of Threatened Species 2015.
Sharpe LL, Jooste MM, Cherry MI. 2012. Handstand scent marking in the dwarf mongoose (Helogale parvula). Ethology 118: 575–583.
Sharpe LL, Joustra AS, Cherry MI. 2010. The presence of an avian co-forager reduces vigilance in a cooperative mammal. Biology Letters 6: 475–477.
Sharpe LL, Rubow J, Cherry MI. 2016. Robbing rivals: interference foraging competition reflects female reproductive competition in a cooperative mammal. Animal Behaviour 112: 229–236.
Waser, P.M., Elliott, L.F., Creel, N.M. and Creel, S.R. 1995. Habitat variation and mongoose demography. In: A.R.E. Sinclair and P. Arcese (eds), Serengeti II: dynamics, management, and conservation of an ecosystem, pp. 421-447. University of Chicago Press, Chicago, IL, USA.