
Dusky Pipistrelle
Pipistrellus hesperidus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Pipistrellus hesperidus – (Temminck, 1840)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Pipistrellus – hesperidus
Common Names: Dusky Pipistrelle, African Pipistrelle, Kuhl’s Pipistrelle (English), Kuhl se Vlermuis, Kuhlvlermuis (Afrikaans)
Synonyms: Vespertilio hesperida (Temminck, 1840), Vesperugo subtilis (Sundevall, 1847), Vesperugo hesperida (Fitzinger, 1870), Miniopterus hesperida (von Heuglin, 1877), Vesperus humbloti (A. Milne-Edwards, 1881) (Mammal Diversity Database 2025).
Subspecies: Pipistrellus hesperidus (Sundevall, 1846) ssp. subtilis; Pipistrellus hesperidus Roberts, 1948 ssp. broomi; Pipistrellus hesperidus Temminck, 1840 ssp. hesperidus; Pipistrellus hesperidus Thomas, 1901 ssp. fuscatus
Taxonomic Note:
Previously included within Pipistrellus kuhlii (Kuhl, 1817), this species was later reclassified as a distinct species, P. hesperidus, based on morphological and karyotype evidence (Kock 2001, Kearney et al. 2002). Five African subspecies were listed by Hayman and Hill (1971), including P. h. broomi (Roberts, 1948) from KwaZulu-Natal and subtilis from other regions of southern Africa and Madagascar (ACR 2024); three of these subspecies may require elevation to species level (Koubínová et al. 2013). Chromosomal and molecular genetic data suggests that the species constitutes a species complex (Koubínová et al. 2013; Monadjem et al. 2021). It is evident that further research is required to disentangle the evolutionary relationships among cryptic taxa.
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Howard, A.1, da Silva, J.M. 2, Balona, J. 3
Reviewers: Moir, M. 4 & Richards L.R. 5
Institutions: 1University of Free State; 2South African National Biodiversity Institute; 3Gauteng and Northern Regions Bat Interest Group; 4Centre for Epidemic Response and Innovation, Stellenbosch University; 5Durban Natural Science Museum
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., Cohen, L., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Relton, C., Raimondo, D. & Nicholson, S.K.
Assessment Rationale
Listed as Least Concern given its wide distribution (estimated extent of occurrence in the assessment region alone is 703,188 km2), its occurrence in multiple protected areas (including Great Limpopo Transfrontier Park, Greater Mapungubwe Transfrontier Conservation Area and iSimangaliso Wetland Park), its tolerance of disturbed habitats, its relative abundance compared to other species, and because there are no major identified threats that could cause widespread population decline. Further taxonomic resolution is required through molecular research.
Regional population effects: Present within transfrontier conservation areas where resident populations seemingly have a continuous distribution with those of Zimbabwe and possibly Mozambique (Monadjem et al. 2020). However, it has low wing loading (Schoeman & Jacobs 2008) and thus rescue effects are uncertain. Additionally, there is evidence of genetic structuring within populations across the assessment region indicates limited dispersal between northern and northeastern populations and those situated further east (Moir et al. 2020b).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Howard A, da Silva JM & Balona J. 2025. A conservation assessment of Pipistrellus hesperidus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Dusky Pipistrelle’s distribution extends over much of sub-Saharan Africa, ranging from the Cape Verde Islands in the extreme northwest, eastwards through Liberia, Côte d’Ivoire, Nigeria and Cameroon to Ethiopia and Somalia. Its range covers much of central and East Africa southwards to eastern South Africa, Eswatini and central western Madagascar (Bates et al. 2006; Monadjem et al. 2020; Piraccini 2016). Within the assessment region, the species occurs widely in the eastern parts of the region, having been recorded from the Eastern Cape, north through KwaZulu-Natal, Swaziland and northern South Africa (Figure 1; Skinner & Chimimba 2005; Monadjem et al. 2020), to Zimbabwe, Zambia, Malawi and northern Mozambique (Monadjem et al. 2020). In the Limpopo Province, the species occurs in the Soutpansberg and Pafuri regions southwards into Mpumalanga (Skinner & Chimimba 2005). Single records from the Okavango, northern Botswana, and from Boshof in the western Free State indicate that this species probably has a broader distribution than previously thought (Monadjem et al. 2020). However, some records require vetting as this species is extremely difficult to distinguish in the field from other Pipistrellus, Hypsugo or Neoromicia species (Kearney 2005). Estimated extent of occurrence in the assessment region alone is 703,188 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 5 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 3,869 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
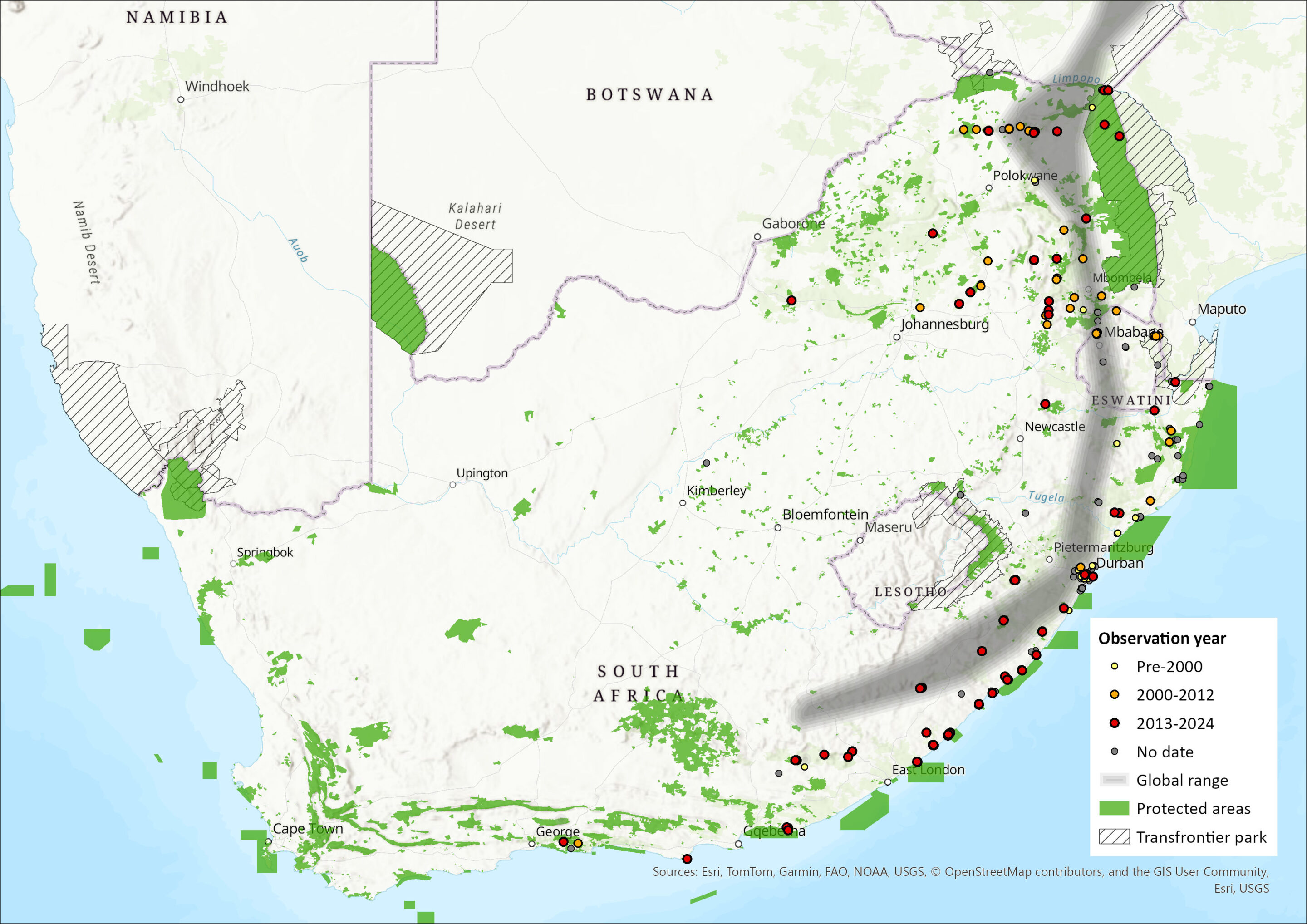 Figure 1. Distribution records for Figure 1. Distribution records for Suni (Pipistrellus hesperidus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Figure 1. Distribution records for Suni (Pipistrellus hesperidus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cabo Verde | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Equatorial Guinea -> Bioko | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Madagascar | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Spain -> Canary Is. | Presence Uncertain | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
No formal study on the direct effects of climate change in this species has been conducted. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). For example, Babiker and Salata (2012) demonstrated that species distribution is strongly correlated with annual precipitation within the assessment region, with areas receiving ≤50 mm per year generally unsuitable for the species. Consequently, increased temperatures combined with reduced annual rainfall could significantly alter the species distribution pattern within the region.
Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability, with increasing global aridity, which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020).
Population Information
In southern Africa, colonies occur in small groups of about 12 bats (Skinner & Chimimba 2005). However, it can be locally plentiful. For example, in a study in the Durban region, this species was one of the most commonly recorded of 16 species along the Umbilo River (Naidoo et al. 2011). It was regularly detected in forests across KwaZulu-Natal and Eastern Cape Provinces (Moir et al., 2020a). At another site in eastern Soutpansberg, it was the most frequently captured species (J. Balona, unpubl. data, 2008). It is relatively well represented in museums, with over 100 specimens examined in Monadjem et al. (2020).
Current population trend: Stable
Continuing decline in mature individuals? Unknown
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
A phylogenetic examination of African pipistrelloid bats identified two clades within P. hesperidus in the assessment region, specifically within Eswatini (Taylor et al. 2022). Samples from South Africa fell within a single clade, suggesting a single ESU; however, a fine-scale study incorporating more comprehensive sampling across the species range would be beneficial to assess whether further sub-structuring (discrete populations) exists. Two population genetic studies have been conducted for the eastern region of the assessment region. Moir et al. 2020b, employed mitochondrial markers and found high population structure estimates for the study region, with northern localities (Limpopo, Mpumalanga and Eswatini) exhibiting notable divergence from more southern localities in the Eastern Cape and KwaZulu-Natal. Additionally, there was relatively low-level structuring between localities within the Eastern Cape (Moir et al. 2020b). The second study employed microsatellite markers and found high gene flow and low population structure across the Eastern Cape and KwaZulu-Natal provinces (Moir et al., 2020c). The land use between sampled localities did not have a detectable effect on genetic differentiation between populations and the extent of anthropogenic development between sites did not affect gene flow. Waterbodies and wetlands were the only land cover shown to facilitate gene flow (Moir et al. 2020c).
Given that the abundance of this species across much of its range is poorly known, it is not possible to estimate the effective population size for the species.
Habitats and ecology
It is associated with woody habitats, such as riparian vegetation and forest patches (Happold et al. 1987), especially in the proximity of water (Skinner & Chimimba 2005; Monadjem & Reside 2008). This species has been sampled along the polluted Umbilo River in the Durban region in 2008 (Naidoo et al. 2011). However, this may have been influenced by the presence of nearby Paradise Valley Nature Reserve, which possibly provided roosting sites such as tree cavities (Naidoo et al. 2011). There is a general lack of information on the types of diurnal roosting sites utilised by this species. However, they have been recorded in narrow cracks in rocks and under the loose bark of dead trees (Smithers 1971; Skinner & Chimimba 2005). There is sexual dimorphism in the species, with females being, on average, slightly heavier than the males (Monadjem et al. 2010). Little is known about its reproductive ecology, but in KwaZulu-Natal, a pregnant female with two foetuses was located in October (Taylor 1998), suggesting that young are born during the warm wet season, similar to other insectivores. The species is a clutter-edge forager, with a diet including Coleoptera, Hemiptera, Diptera and Lepidoptera (Schoeman 2006; Monadjem et al. 2020).
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations that damage crops (Boyles et al. 2011; Kunz et al. 2011), such as stinkbugs (Taylor et al. 2013, 2018).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: Mean forearm length = 3.24 ±0.13 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Female: Mean forearm length = 3.29 ±0.11 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Same as above
Longevity: Unknown
Average Reproductive Age: possibly 1-2 years
Maximum Size (in cms): Male maximum forearm length = 3.50 cm; Female maximum forearm length = 3.50 cm (Monadjem et al. 2020)
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Exhibit seasonal monoestry with breeding/births take place during the austral summer months, with pregnant females recorded in October and December (Taylor 1998; Moir et al. 2020a).
Average Annual Fecundity or Litter Size: Typically produces twins, though occasionally only one pup (Rautenbach 1982; Kearney 2013).
Natural Mortality: Many natural predators have been reported including avian raptors and snakes (Kearney 2013).
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: No available information
Congregatory: Reported to roost in groups of 12 individuals (Kearney 2013). Also known to forage in groups (Rautenbach 1982; Kearney 2013) and may engage in ‘swarming’ behaviour.
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region.
Local Livelihood: None reported
National Commercial Value: Known to assist with pest insect suppression activities in agroecosystems such as macadamia (Taylor et al. 2013, 2018) and found foraging within mixed crop orchards (e.g. banana and macadamia) along the southern KwaZulu-Natal coast (M. Moir unpublished data).
International Commercial Value: Plays a role in suppressing insect pests in internationally exported crops such as macadamias (Taylor et al. 2013; 2018).
End Use: None reported
Is there harvest from captive/cultivated sources of this species? None reported
Harvest Trend Comments: None reported
Threats
No major threats have been identified for this species at present, but further research is required to delineate the distributional limits of this species, its taxonomic status, roosting behaviour, as well as potential threats. Ongoing habitat loss from agricultural transformation, especially in KwaZulu-Natal (Jewitt et al. 2015), may reduce the insect prey base for this species. The impacts of climate change should also be investigated (sensu Sherwin et al. 2013).
Conservation
This species occurs in many protected areas within the assessment region, having been recorded from Great Limpopo Transfrontier Park, Greater Mapungubwe Transfrontier Conservation Area, Loskop Dam Nature Reserve, Songimvelo Nature Reserve, Ndumo Game Reserve, iSimangaliso Wetland Park, Hluhluwe-iMfolozi Game Reserve, Sibudeni Nature Reserve, Nkandla Forest Reserve, Mome Nature Reserve, Krantzkloof Nature Reserve, Dwese-Cwebe Wildlife Reserve, Fort Fordyce Nature Reserve, Oribi Gorge Nature Reserve, Umtamvuna Nature Reserve, Mkambati Nature Reserve, Silaka Nature Reserve, Cape Morgan Nature Reserve, Woody Cape Nature Reserve, The Island Nature Reserve, Garden Route National Park, Dlinza Forest Reserve, Phinda Private Game Reserve, Mkuze Game Reserve, Hlatikulu Forest Reserve, Mlawula Nature Reserve, Lajuma Mountain Reserve and Blouberg Nature Reserve. No specific conservation interventions are necessary at present. However, outside of protected areas, it would benefit from holistic land management that reduces pesticide use and conserves buffer strips of natural vegetation to sustain insect biomass.
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes and maintain buffer strips of natural vegetation.
- Maintain and conserve water sources within the landscape
Research priorities:
- Additional taxonomic studies are needed to better define its range relative to other species of Pipistrellus (ACR 2024), as well as to resolve the status of the putative subspecies and documented cryptic species.
- Vetting of museum specimens is needed to more accurately delimit its range.
- Studies into its roosting behaviour, habitat selection and population status are needed.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution. However, it should be noted that this species is very difficult to distinguish from other Pipistrellus, Hypsugo or Neoromicia species. It is larger than P. rusticus (Monadjem et al. 2020).
- Deceased specimens should be submitted to the nearest accredited museum or biological repository for proper documentation and preservation.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Benda, P., Hanak, V., Andreas, M., Reiter, A. and Uhrin, M. 2004. Two new species of bats (Chiroptera) for the fauna of Libya: Rhinopoma hardwickii and Pipistrellus rueppellii. Myotis 41: 109–124.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Helbig, M.T., Datzmann, T., Mayer, F. and Fahr, J. 2010. Molecular phylogeny of African “pipistrelle” bats (Vespertilionidae) suggests new clades, rearrangement of genera, and extensive cryptic diversity within species. Pages 168–169 in Horácek I, Benda P, editors. 15th IBRC – the Conference Manual: Programme, abstracts, list of participants. Volume of abstracts of the 15th International Bat Research Conference. Prague, Czech Republic.
Kearney, T. 2013. Pipstrellus hesperidus Dusk Pipistrelle; pp 629 – 631 in Happold M., Happold D.C.D. (eds) Mammals of Africa: Volume IV, Hedgehogs, Shrews and Bats. Bloomsbury Publishing, London.
Koubínová, D., Irwin, N., Hulva, P., Koubek, P. and Zima, J. 2013. Hidden diversity in Senegalese bats and associated findings in the systematics of the family Vespertilionidae. Frontiers in Zoology 10: 48.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Mammal Diversity Database. 2025. Pipistrellus hesperidus (Temminck, 1840) Dusky Pipistrelle. https://mammaldiversity.org/taxon/1005620
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Moir, M., Richards, L.R.R., Rambau, R.V., Cherry, M.I. 2020a. Bats of Eastern Cape and southern KwaZulu-Natal forests, South Africa: diversity, call library and range extensions. Acta Chiropterologica, 22(2):365-381
Moir, M., Richards, L.R.R., Cherry, M.I., Rambau, R.V. 2020b. Demographic responses of forest-utilizing bats to past climate change in South Africa. Biological Journal of the Linnean Society, 130(4): 850-868.
Moir, M., Richards, L.R., Rambau, R.V., Wannenburgh, A., Cherry, M.I. 2020c. Fragmentation does not affect gene flow in forest populations of the dusky pipistrelle bat on the eastern seaboard of South Africa. Journal of Mammalogy 101(6):1587-1600.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J., 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Rautenbach, I.L. 1982. Mammals of the Transvaal. Ecoplan Monographs 1. Ecoplan, Pretoria.
Sherwin, H.A., Montgomery, W.I. and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. Natal University Press, South Africa
Taylor, P.J., Grass, I., Alberts, A.J., Joubert, E. and Tscharntke, T. 2018. Economic value of bat predation services–A review and new estimates from macadamia orchards. Ecosystem Services 30: 372-381.
