Desert Pygmy Mouse
Mus indutus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Mus indutus – (Thomas, 1910)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Mus – indutus
Common Names: Desert Pygmy Mouse (English), Woestyndwergmuis (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note: Recent phylogenetic analyses support the classification of Mus indutus as a discrete monophyletic species of the subgenus Nannomys (Lamb et al. 2014). Four subspecies of Mus indutus were previously suggested by Meester et al. (1986) based largely on distribution. These included M. i. indutus, with a range extending across the Northern Cape into Botswana and western Zimbabwe; M. i. valschensis (Roberts 1926) from the Free State; M. i. pretoriae (Roberts 1926), which is distributed across the Limpopo, Mpumalanga, Gauteng and North West provinces; and finally M. i. sybilla (Thomas 1918) from Damaraland in Namibia north into Angola. However, more recently, sybilla has been reclassified as the distinct species Mus sybilla (Crawford-Cabral 1998; Lamb et al. 2014).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessor: Patel, T1. & da Silva, J. M.2
Reviewer: Smith, C 1.
Institutions: 1Endangered Wildlife Trust,2South African National Biodiversity Institute
Previous Assessors & Reviewers: Watson, J, Britton-Davidian, J., Monadjem, A. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern because it ranges extensively within the assessment region and at times has been recorded as extremely abundant due to occasional population explosions. The species previously lost part of its extent of occurrence in the North West due to habitat loss. However, because it is able to exist in agricultural habitats and wildlife ranching is expanding in the North West Province, we retain the Least Concern listing. Due to confusion with M. minutoides in the past, the distribution of this species has not been resolved. Further vetting of museum records and field surveys to determine occupancy are required to resolve the uncertainty around this species.
Regional population effects: There is presumably dispersal across contiguous habitat from Botswana and Namibia into Kgalagadi Transfrontier Park and surrounding areas.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recomended citation: Patel T & da Silva J M. 2025. A conservation assessment of Mus indutus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The geographic extent of this species’ range is largely uncertain, owing to the confusion with M. minutoides (Lamb et al. 2014). However, based on molecular grounds, it has been confirmed from three widely separated localities across South Africa and Botswana, suggesting that it has a wide distribution across the arid and semi-arid regions of southern Africa (Lamb et al. 2014). Although its range was previously suggested to extend into northeastern Namibia, southwestern Zambia, western Zimbabwe and southern Angola, no records of this species were confirmed from these regions by Lamb et al. (2014). It is possible that the previous records from southwestern Angola and Namibia belong exclusively to the species M. sybilla (previously recognised as a subspecies of M. indutus; Meester et al. 1986).
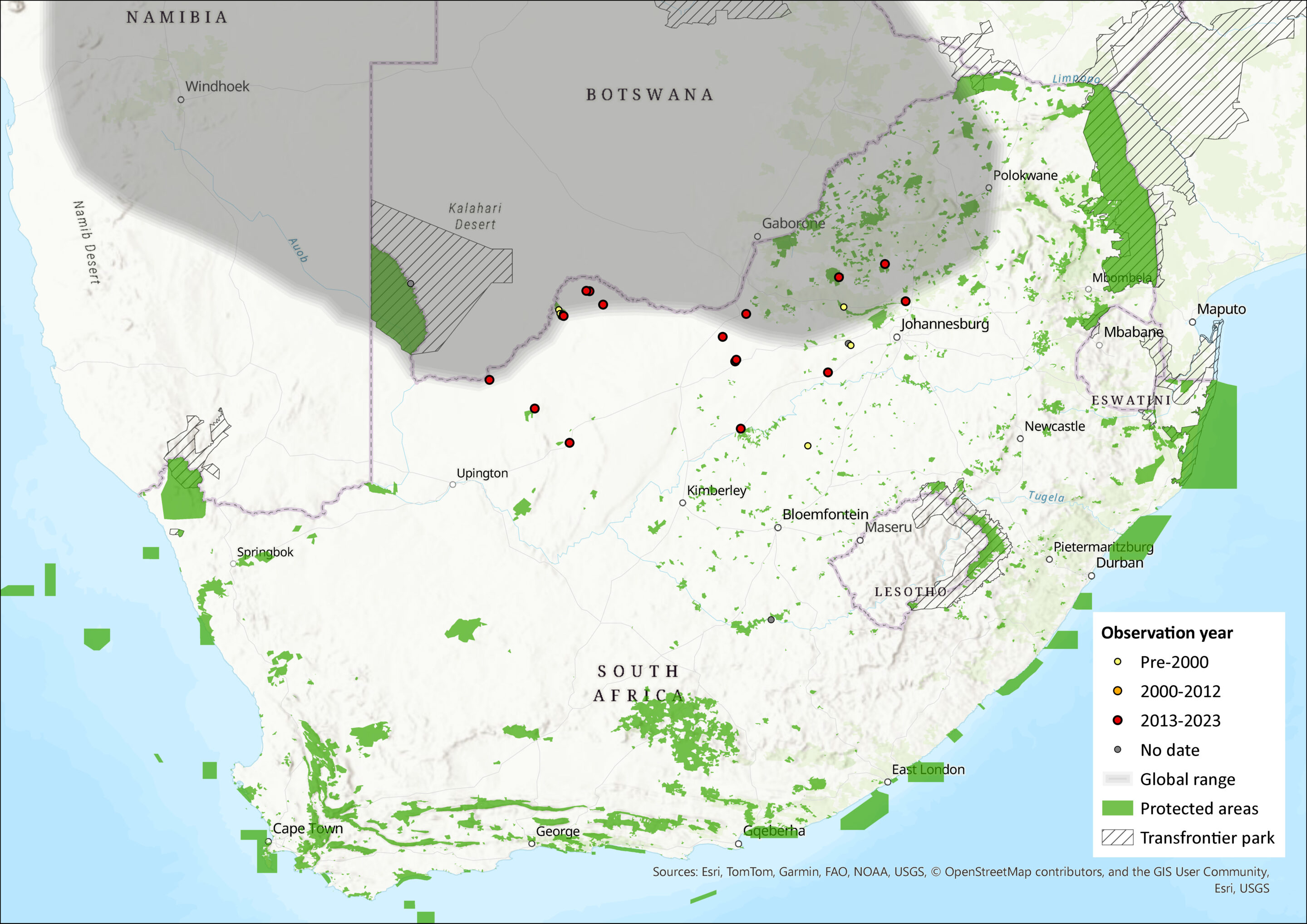
Within the assessment region, its range is believed to include the Northern Cape, North West, Gauteng and Free State provinces of South Africa. Within the North West Province, this tiny mouse species was found on a few occasions throughout the province, and was more often recorded than the nationally more common Pygmy Mouse, M. minutoides (Power 2014). Additionally, specimens from Tussen die Riviere in the Free State have recently been assigned to M. indutus using molecular markers and thus the range of the species should be extended south to 30°29’ S (Veyrunes et al. 2004) (Figure 1), which is reflected in Monadjem et al. (2015). Finally, the presence of this species has also been confirmed in the Sandveld Nature Reserve (near Bloemhof Dam in the Free State) on the basis of their karyotype (2n = 36) (F. Veyrunes & J. Britton-Davidian unpubl. data). Further vetting of museum records is required to delimit its distribution comprehensively.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 1600
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Desert Pygmy Mouse (Mus indutus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Namibia | Presence Uncertain | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Presence Uncertain | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The species occurs in the Northern Cape, North West, Gauteng and Free State provinces. They have a high habitat tolerance, so it is uncertain how they would respond to the 1.5-4°C increase in temperature predicted under various climate change scenarios (Engelbrecht et al. 2024). Rainfall is predicted to decrease in its range. These decreases will result in a more arid and drought-prone habitat, possibly affecting resource availability to the species.
Population Information
Although the population status of this species is uncertain, they are considered to be one of the most abundant species in the Kalahari. Additionally, this species may experience periods of population explosion when conditions are favourable and food availability increases. Their population numbers rise rapidly, followed by a population crash to very low levels (Smithers 1971). Within the assessment region, very few are trapped, possibly because of their small body size. However, they occur in disturbed habitats, and we suspect there are over 10,000 mature individuals. This species occurs sympatrically and has regularly been confused with M. minutoides (Chevret et al. 2014), thus caution should be taken when conducting population surveys of this species.
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: It is suspected that there may be more than 10,000 mature individuals within South Africa.
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No, can occur in agricultural habitats.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
The genus Mus has been studied extensively within a phylogenetic context (e.g., Bryja et al. 2014, Lamb et al. 2014), however no population genetic studies have been undertaken on this species. While Meester (1983) provisionally identified subspecies, no mention of any discrete distinctions were provided; simply geographic designations for each. In the absence of any corroborating evidence, it is assumed the species exists as a metapopulation.
While population estimates are not known, it is expected that at least 10,000 mature individuals exist within the assessment region. Even though the 10 000 value is not an actual census count (Nc), the quantity can still be used to infer effective population size (Ne) by applying a Ne/Nc conversion ratio of 0.1-0.3. This yields an Ne estimate between 1,000-3,000, which exceeds the Ne 500 threshold indicative of genetically healthy and stable populations. Given that dispersal across contiguous habitats is expected between Botswana, Namibia and South Africa, Ne is likely to far exceed these values.
It is highly recommended a population genetic study be undertaken to more accurately assess the genetic structure and diversity within the species.
Habitats and ecology
The Desert Pygmy Mouse has an extensive habitat tolerance throughout semi-arid savannahs (Lamb et al. 2014), although it typically avoids open microhabitats. They have been recorded from the arid scrub savannah of the Kalahari to the wetter regions of the Okavango Delta (Skinner & Chimimba 2005). In general, their range includes areas with an average annual precipitation of 200–700 mm.
This nocturnal species spends the daylight hours under cover, within crevices beneath debris, logs, bark and stones, and occasionally will dig small burrows into sandy soils, or utilise holes dug by other species (Skinner & Chimimba 2005). A colony of 35 individuals was documented during a period of favourable environmental conditions in the Kalahari Transfrontier Park, Northern Cape (Skinner & Chimimba 2005). They are not seasonal breeders, and give birth in round grass nests within shallow burrows (Smithers 1971).
This omnivorous species primarily feeds on grass seeds, seeds of Vachellia (previously Acacia spp.), dry pods, and the dry exterior layer of fruit from trees such as Ziziphus mucronata (Buffalo Thorn). They will also consume termites and small beetles, and have been documented exhibiting cannibalistic behaviour (Skinner & Chimimba 2005).
Ecosystem and cultural services: This species represents a valuable prey species for a number of predators, such as Suricates (Suricata suricatta) and Barn Owls (Tyto alba), and may also contribute to seed dispersal in the semi-arid and arid regions of southern Africa.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Marginal | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Unknown | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species does not appear to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
No major threats have been recognised for this species. However, Driver et al. (2012) document considerable loss of natural habitat in North West Province due to agricultural expansion, mining, and human settlement expansion. This species can exist in disturbed habitats and, given the proliferation of wildlife ranching within the province (Power 2014), should continue to be stable. While they are able to survive in degraded areas, hugely overgrazed areas and areas transformed into habitats with little or no biomass for forage and shelter would negatively impact this species (Hoffmann & Zeller 2005).
Current habitat trend: Declining. In North West Province, which comprises the bulk of its range, habitat loss between 1994 and 2010 amounted to 12% (NW READ 2014; Desmet & Schaller 2015).
Conservation
The Desert Pygmy Mouse occurs in many protected areas within the assessment region and no specific conservation interventions are necessary at present. However, further research is required to delineate the population dynamics, current and potential threats, as well as the general ecology and life history traits of this species. Should this species become a nuisance, due to population explosions, Barn Owls are a valuable means of biocontrol for nocturnal rodent species.
Recommendations for land managers and practitioners:
- Landowners and managers should breed Barn Owls to mitigate the species as an agricultural and human health threat.
Research priorities:
- Accurate distribution mapping and the identification of population size and trend estimates is necessary. This includes vetting museum records.
- Investigations into this species’ ability to survive in agricultural areas and on wildlife ranches.
- Population trends in disturbed areas.
Encouraged citizen actions:
- Accurate sighting reports of this species on virtual museum platforms is unlikely, considering the morphological similarities between this species and M. minutoides.
- Citizens can plant indigenous gardens, like Brenthurst Gardens in Johannesburg, and create corridors of natural vegetation between properties.
- Save electricity and fuel to mitigate CO2 emissions and hence the rate of climate change.
Bibliography
Bryja, J., Mikula, O., Šumbera, R. et al. 2014. Pan-African phylogeny of Mus (subgenus Nannomys) reveals one of the most successful mammal radiations in Africa. BMC Evol Biol 14, 256. https://doi.org/10.1186/s12862-014-0256-2
Chevret, P., Robinson, T.J., Perez, J., Veyrunes, F., Britton-Davidian, J. 2014. A Phylogeographic Survey of the Pygmy Mouse Mus minutoides in South Africa: Taxonomic and Karyotypic Inference from Cytochrome b Sequences of Museum Specimens. PLoS ONE 9(6): e98499.
Crawford-Cabral, J. 1998. The Angolan rodents of the superfamily Muroidea. An account on their distribution. Estudos, Ensaios e Documentos 161: 1-222.
Desmet PG, Schaller R. 2015. North West Biodiversity Sector Plan Technical Report. North West Department of Rural, Environment and Agricultural Development, Mahikeng, South Africa.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Engelbrecht, F.A., Steinkopf, J., Padavatan, J. and Midgley, G.F., 2024. Projections of future climate change in Southern Africa and the potential for regional tipping points. In Sustainability of Southern African Ecosystems under Global Change: Science for Management and Policy Interventions (pp. 169-190). Cham: Springer International Publishing.
Hoffmann, A. and Zeller, U. 2005. Influence of variations in land use intensity on species diversity and abundance of small mammals in the Nama Karoo, Namibia. Belgian Journal of Zoology 135: 91–96.
Lamb, J., Downs, S., Eiseb, S. and Taylor, P.J. 2014. Increased geographic sampling reveals considerable new genetic diversity in the morphologically conservative African Pygmy Mice (Genus Mus; Subgenus Nannomys). Mammalian Biology 79: 24-35.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
NW READ. 2014. North West Environment Outlook Report 2013. North West Provincial Government, Mahikeng, South Africa.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Veyrunes F, Catalan J, Sicard B, Robinson TJ, Duplantier J-M, Granjon L, Dobigny G, Britton-Davidian J. 2004. Autosome and sex chromosome diversity among the African pygmy mice, subgenus Nannomys (Murinae; Mus). Chromosome Research 12: 369–382.