Dassie Rat
Petromus typicus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Petromus typicus – A. Smith, 1831
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – PETROMURIDAE – Petromus – typicus
Common Names: Dassie Rat, Rock Rat (English), Dassierot (Afrikaans), Noki (Damara, Nama)
Synonyms: No Synonyms
Taxonomic Note: The Petromuridae family consists of a single species, Petromus typicus which is endemic to the southwestern arid region of Africa (Sénégas 2004; Monadjem et al. 2015). Several forms of this species have been described (Skinner & Chimimba 2005), but de Graaf (1981) proposed a revision of the Petromuridae family, due to the large number of subspecies described over a limited area. Additionally, Musser and Carleton (1993) proposed that many of these subspecies should be discredited. Further analyses are required to delineate the taxonomic structure of this genus.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Russo, I.1 & da Silva, J.M.2
Reviewer: Kraai, M.3
Institutions: 1Cardiff University,2South African National Biodiversity Institute,3Sol Plaatjie University
Previous Assessors and Reviewers: Wilson, B., Todd, S. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Mondajem, A., Palmer, G. & Taylor, P.
Assessment Rationale
This species is endemic to the arid regions of Namibia and South Africa, with most of the population confined to Namibia. It is locally common and inhabits inaccessible rocky areas, particularly granite koppies, which are unlikely to be rapidly transformed. There are no major identified threats, although climate change may represent an emerging threat as this species relies on moisture-rich vegetation. Additionally, parts of its range are ear-marked for wind and solar developments, resulting in potential habitat loss and fragmentation of subpopulations. The impacts of such renewable energy developments should be monitored. Presently, the species is listed as Least Concern with no evidence of decline and a relatively large extent of occurrence (96,705 km2) within the assessment region.
Regional population effects: Sporadic dispersal of this species across the Orange River between South Africa and Namibia is likely, thus rescue effects are considered possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Russo I & da Silva JM. 2025. A conservation assessment of Petromus typicus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
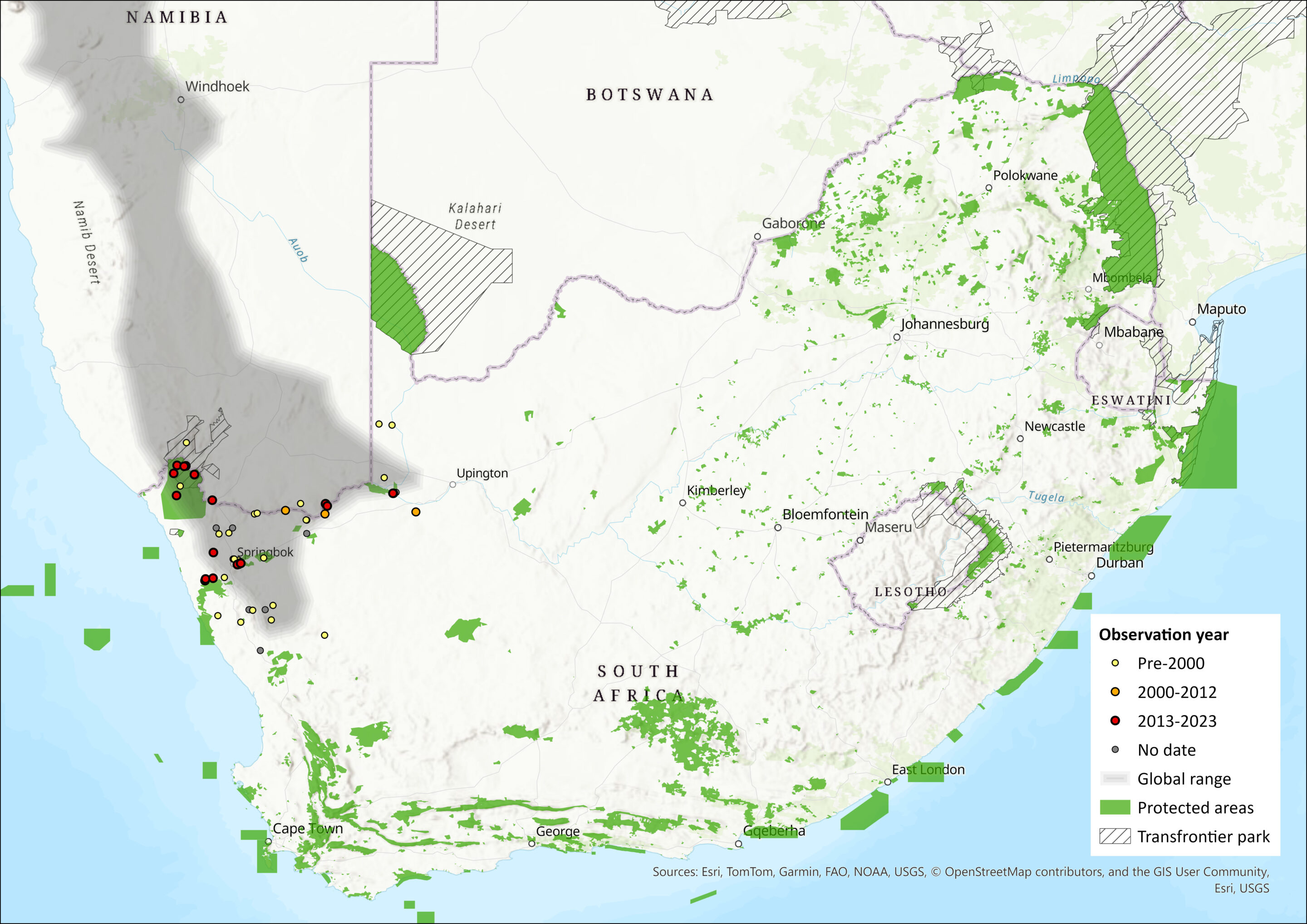
Dassie Rats are restricted to the southwestern arid, and semi-arid regions of Africa from southwestern Angola, southwards through the xeric, rocky regions of western Namibia and into the northwestern parts of South Africa. Monadjem et al. (2015), however, showed that the distribution is restricted to Namibia and does not extend into Angola. Their distributional limits, as described by Coetzee (2002) and range from 16°S 12°E in the north to 30°S 18°E in the south (Namaqualand, South Africa). Although they have been recorded at altitudes of up to 1,200 m asl, this is limited only to warm, dry regions, thus they are absent from the cloudy Eselfontein Plateau (Shortridge 1942), where it is considered too moist and cold (Coetzee 2002). Similarly, Dassie Rats are not present within the moist, semi-tropical savannas of western Angola (Coetzee 2002). The estimated extent of occurrence using all records is 96,705 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 1200
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Dassie Rat (Petromus typicus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Additionally, an increased severity and frequency of drought, associated with climate change, is projected to have contrasting effects on resource availability and distribution for small herbivorous species in the semi-arid Succulent Karoo (Hoffman et al. 2009). The apparent dependence of this species on advective fog or green plant material for moisture is likely to increase its vulnerability to climate-induced increases in aridity or the severity of droughts. Further research should be undertaken to assess the effects of climate effects on the species (water stress).
Population Information
The Dassie Rat is considered relatively common through much of its range, which extends only marginally into the assessment region in the Northern Cape and Western Cape Provinces. No published population densities are available for this species. Their abundance is limited by the availability of shelter within rocky outcrops and by mean annual rainfall while vegetation composition is less significant (Coetzee 2002).
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While no population genetic study has been undertaken for this species, it is possible the species exists as a single metapopulation extending into Namibia. From this, one of the two genetic indicators in the Convention of Biological Diversity’s Global Biodiversity Framework can be quantified – the complementary indicator- proportion of populations maintained with a species with 1 of 1 populations remaining, resulting in a value of 1.0 for this indicator.
The GBF’s headline indicator – proportion of populations with Ne greater than 500 – cannot be quantified given no estimates on population sizes or density. It is recommended that a population genetic study be undertaken to better understand the population genetic structure and diversity within the species and to calculate the indicator values.
Habitats and ecology
Confined to the southwestern arid and semi-arid regions of Africa, the Dassie Rat is associated with rocky areas of mountainous habitats and inselbergs (de Graaff 1981; Skinner & Smithers 1990). This species lives among rocky outcrops, seeking shelter for nest sites in crevices and under large boulders (George 1981; Skinner & Chimimba 2005). In the Augrabies Falls National Park (AFNP) this species co-occurs with the Rock Hyrax, where they compete for rocky shelters. Dassie Rats have a flattened skull and are restricted to rocky microhabitats that are too small for Rock Hyraxes to enter (George & Crowther 1981).
Dassie Rats may live in colonies (Coetzee 2002) or form monogamous pairs (Rathbun & Rathbun 2006), defending territories comprised of rocky cracks and crevices for shelter, and open areas for predator surveillance and basking, which are close enough to forage resources (Skinner & Chimimba 2005). Having low energy and water requirements, Dassie Rats are well adapted to arid areas, and in the largest portion of their range (Namib Desert), they may acquire moisture from advective fog or from the consumption of succulent plants (Withers 1979). This species is diurnal, and peak activity occurs during the early mornings and late afternoons.
The herbivorous diet of this species is highly varied (Rathbun & Rathbun 2005). In AFNP their diet comprised mostly of monocotyledonous grasses, such as Enneapogon scaber, Cenchrus ciliarus and Triraphis ramossissima and to a lesser extentthe dicotyledons, such as Schotia afra and Hermannia stricta (George 1981). George (1981) described how Dassie Rats feed off the water-rich bases of grass stems, which contain three times more moisture than the top portion of grass stems. This species has also been recorded feeding on leaves of Tribulus spp., flowers and the fruit of Cucumis dinteri (Skinner & Chimimba 2005). As it is unable to survive on air-dried seeds in the laboratory (Withers et al. 1980), the consumption of moisture-rich vegetation may be critical for its survival in the wild.
The timing of breeding varies, but is closely related to rainfall, occurring predominantly in late spring and autumn (Coetzee 2002). Following a gestation period of about three months, between one and three (usually two) young are born in nests within rocky cracks (Coetzee 2002; Mess 2002).
Ecosystem and cultural services: This species has been recognised as a valuable prey resource for aerial predators, as well as Black Mongooses (Galerella nigrata; Rathbun & Cowley 2008). Rathbun and Rathbun (2005) suggested that the association between this species and Rock Hyraxes may be mutually beneficial, due to enhanced vigilance for predators.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 6. Rocky areas (eg. inland cliffs, mountain peaks) | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species does not appear to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
No major threats have been identified for this species, however, they may be preyed upon by domestic cats, where their range extends into urban areas. The rapidly expanding construction of major solar farms in the Northern Cape in the vicinity of the Upington corridor, as well as wind farms in the northwestern parts the Western Cape, may signify an emerging threat to this species as its range partially overlaps with these approved or in progress developments (van der Westhuizen 2013). Additionally, there is an increase in granite mining activities in Namaqualand which usually target similar areas to the preferred habitat of Petromys. This will result in some habitat loss and disturbance for this species, but as the extent of this activity is currently still low, this
would have a local impact only.
Current habitat trend: Stable. However, climate change may make habitat increasingly unsuitable for this species.
Conservation
The Dassie Rat is present within certain protected areas of northwestern South Africa, including AFNP, |Ai-|Ais/Richtersveld Transfrontier Park (B. Wilson unpubl. data), Goegap Nature Reserve and Namaqua National Park. No direct interventions are required at present. However, this species is likely to benefit from continued research into the potential threat of enhanced aridity associated with climate change, and how this may affect food resources for this species, as well as spatial research into potential habitat loss from renewable energy developments and granite mining.
Recommendations for land managers and practitioners:
- Systematic surveys are needed to gather information on population size and trends.
Research priorities:
- Population size, distribution and trend estimates.
- Associated threats to this species, specifically with regards to the perceived threats of climate change.
- Analysis of potential impacts of renewable energy developments, and granite mining.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Save electricity and fuel to mitigate CO2 emissions and hence the rate of climate change.
Bibliography
Coetzee, C.G. 2002. The distribution and breeding seasons of the dassie-rat, Petromus typicus (Peteromuridae, Rodentia). Folia Zoologica 51: 23-35.
De Graaff, G. 1981. The Rodents of Southern Africa. Butterworths, Durban, South Africa.
George W. 1981. The diet of Petromus typicus (Petromuridae, Rodentia) in the Augrabies Falls National Park. Koedoe 24: 159–167.
George, W. and Crowther, G. 1981. Space partitioning between two small mammals in a rocky desert. Biological Journal of the Linnean Society 15: 195-200.
Hoffman M.T., Carrick P.J., Gillson L, West A.G. 2009. Drought, climate change and vegetation response in the succulent karoo, South Africa. South African Journal of Science 105: 54–60.
Mess A. 2002. Petromus typicus: reproductive biology of a poorly known animal. Research Committee Newsletter: 38–40.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Musser, G.G. and Carleton, M.D. 1993. Family Muridae. In: D.E. Wilson and D.A. Reeder (eds), Mammal species of the world: A taxonomic and geographic reference, pp. 501-736. Smithsonian Institution Press, Washington, DC, USA.
Rathbun GB, Rathbun CD. 2005. Noki or dassie-rat (Petromus typicus) feeding ecology and petrophily. Belgian Journal of Zoology 135: 69–75.
Rathbun GB, Rathbun CD. 2006. Social monogamy in the noki or dassie-rat (Petromus typicus) in Namibia. Mammalian Biology-Zeitschrift für Säugetierkunde 71: 203–213.
Rathbun, G.B. and Cowley, T.E. 2008. Behavioural Ecology of the black mongoose (Galerella nigrata) in Namibia. Mammalian Biology 73(6): 444-450.
Rutherford MC, Powrie LW, Schulze RE. 1999. Climate change in conservation areas of South Africa and its potential impact on floristic composition: a first assessment. Diversity and Distributions 5: 253–262.
Shortridge, G.C. 1942. Field notes on the first and second expeditions of the Cape Museums mammal survey of the Cape Province, and descriptions of some new subgenera and subspecies. Annals of the South African Museum 3: 281-291.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skinner, J.D. and Smithers, R.H.N. 1990. The Mammals of the Southern African Subregion. University of Pretoria, Pretoria.
Sénégas F. 2004. A new species of Petromus (Rodentia, Hystricognatha, Petromuridae) from the early Pliocene of South Africa and its paleoenvironmental implications. Journal of Vertebrate Paleontology 24: 757–763.
Van der Westhuizen C. 2013. Determination of Development Potential: Department of Environmental Affairs National Wind and Solar PV Strategic Environmental Assessments – To facilitate the efficient and effective rollout of wind and solar PV energy in South Africa. Centre for Scientific and Industrial Research, Pretoria.
Withers PC, Louw GN, Henschel J. 1980. Energetics and water relations of Namib desert rodents. South African Journal of Zoology 15: 131–137.
Withers PC. 1979. Ecology of a small mammal community on a rocky outcrop in the Namib Desert. Madoqua 11: 229–246.