Damaraland Mole-rat
Fukomys damarensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Fukomys damarensis – (Ogilby, 1838)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – BATHYERGIDAE – Fukomys – damarensis
Common Names: Damaraland Mole-rat, Damaraland Blesmol (English), Damaralandse Vaalmol, Swart Blesmol (Afrikaans)
Synonyms: Bathyergus damarensis (Ogilby, 1838), Cryptomys damarensis (Ogilby, 1838, Gray 1864), Coetomys damarensis (Ingram et al. 2004)
Taxonomic Note: South Africa
Although originally included in the genus Cryptomys, molecular phylogenies based on both mitochondrial and nuclear genes suggest that the species is evolutionary divergent from other Cryptomys species (Faulkes et al. 2004, Ingram et al. 2004). Thus, the species has been assigned to the genus Fukomys (Kock et al. 2006).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Bennett, N.C.1, Finn, K.T.1 & da Silva, J.M.2
Reviewer: Faulkes, C.G.3
Institutions:1University of Pretoria,2South African National Biodiversity Institute,3Queen Mary University of London
Previous Assessors and Reviewers: Bennett, N.C., Thomas, H. & Jarvis, J.
Previous Contributors: Relton, C., Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern, the Damaraland Mole-rat is present within a number of protected areas within the assessment region, including Tswalu Game Reserve, the Kgalagadi Transfrontier Park and Molopo Nature Reserve. There are no major threats to this species; consequently, its population is unlikely to be declining. It is locally common and is frequently found at high population densities.
Regional population effects: This species is naturally fragmented, but no distinct barriers to dispersal have been identified, and thus a rescue effect is possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Bennett NC, Finn KT & da Silva JM. 2025. A conservation assessment of Fukomys damarensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
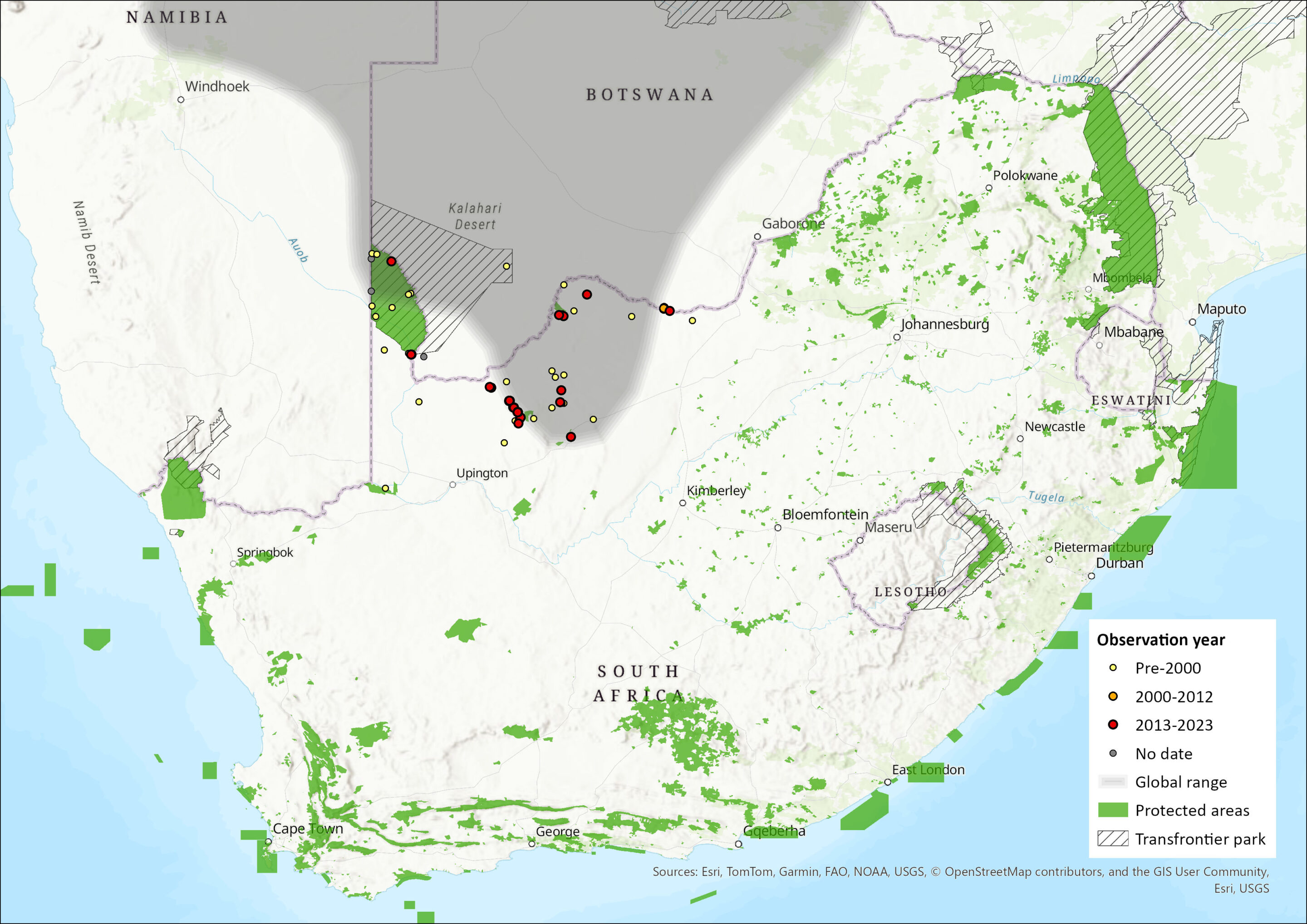
Endemic to sub-Saharan Africa, this species is widespread across the central regions of southern Africa, occurring from central and northern Namibia, across western Zambia, and throughout the majority of Botswana (with the exception of the extreme east), into western Zimbabwe. The habitat of this species is contiguous, although naturally fragmented. The southern portion of its range extends into the Northern Cape and North West provinces of South Africa, where it occurs in the Kgalagadi Transfrontier Park, Tswalu Game Reserve, Hotazel, Blackrock and Winton (Figure 1). Its distribution is associated with red Kalahari arenosols, but it also occurs in coarse sandy soils (Bennett 2013).
The species is said to be sympatric with the Mahali Mole-rat (Cryptomys hottentotus mahali) where soil sandiness ensures local niche differentiation (Skinner and Chimimba 2005), which may be the case in the North West Province where such conditions exist (Power 2014). Power (2014) surmises that the Mafikeng Bushveld vegetation type is a zone of sympatry, and that the record 100 km west of Mafikeng marks their most eastern distribution in South Africa and thus their range boundary should be redrawn accordingly.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Hairy Damaraland Mole-rat (Fukomys damarensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: (Not specified)
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change in the Kalahari associated with lowered rainfall and an increase in ambient temperatures could affect the food resources (geophytes) of this species and in turn affect its population size and distribution. Additionally, dry soils become difficult to excavate, thus significantly enhancing the energetic costs associated with burrowing (Jarvis et al. 1998).
Population Information
The number of subpopulations of this species is currently unknown, but the Damaraland Mole-rat is considered locally abundant in suitable habitats. Population densities may reach more than 380 individuals / km² in grasslands, thornscrub and wheat fields (J.U.M. Jarvis & N.C. Bennett unpubl. data). However, abundance of subterranean mammals is difficult to estimate (Bennett 2013).
Although the population is not suspected to be declining, it should be established whether there is a continuing loss in the area of occupancy due to climate change, changes in land-use, and poor farming practices.
Population Information
Current population trend: Stable
Continuing decline in mature individuals: No
Continuing decline in number of subpopulations: No
Extreme fluctuations in the number of subpopulations: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
All individuals in one subpopulation: No
Number of subpopulations: Unknown, but many
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: None
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: None
Probability of extinction in the wild within 100 years: Yes
Population Genetics
Refer to Mynhardt et al. 2021 study (https://bmcecolevol.biomedcentral.com/articles/10.1186/s12862-021-01950-2).
- strong signals of population genetic structure, with high levels of relatedness within colonies.
- it seems plausible that gene flow is well maintained at distances of up to 800 m
- male-biased dispersal
- during periods of sustained rainfall, this constraint relaxes significantly, and the softened sands stimulate increased dispersal by subordinates of both sexes
Habitats and ecology
This species occupies in semi-arid regions consisting of red Kalahari sands and sandy soils, occupying habitats including grassland, savanna, thornscrub and woodland (Bennett 2013). The annual rainfall in these areas is usually low to intermittent (200–400 mm). The Damaraland Mole-rat prefers coarse Kalahari sands, and loose unconsolidated alluvial sands. It is diurnal, subterranean, and lives in colonies averaging 16 individuals. It is one of a growing number of eusocial species of mole-rat emerging from recent research, and as many as 41 individuals have been identified in a single colony (Burland et al. 2002). Colonies consist of a single breeding female, one or more breeding males and their subsequent litters that do not leave the natal group (Bennett and Jarvis 1988, Jarvis and Bennett 1993). The non-reproductive members of the colony take up roles of caring for the young and burrow maintenance (Jarvis and Bennett 1993). A recent study reveals that the body shape of the reproductive female is significantly more elongate when compared to nonbreeding (Young and Bennett 2010).
Similar to the other eusocial species of mole-rat, the Naked Mole-rat (Heterocephalus glaber) of East Africa, F. damarensis, is found in areas of low, irregular rainfall. Infrequent rainfall results in high energetic costs associated with foraging and burrowing (Lovegrove 1991, Jarvis et al. 1994). Cooperative reproduction, burrowing and foraging is suggested to be an adaptive technique associated with areas of unpredictable food availability and restricted opportunities for independent breeding and dispersal (Jarvis et al. 1994). Feeding mostly on geophytes (Bennett and Jarvis 1988, Bennett & Faulkes, 2000), its main food in the Kalahari is the Gemsbok Cucumber (Acanthosicyos naudinianus) (Jarvis et al. 1998). In arid areas, geophytes tend to be clumped or widely dispersedand are therefore not always spatially predictable in their availability.
Up to four litters of an average of two young (up to a maximum of six) may be produced each year and, should the breeding female die, the colony will separate. On average, Damaraland Mole-rats live for three years; however, some have been recorded up to 11 years of age (Schmidt et al. 2013). Sex ratios of captured colonies range from 0.8 to 2.1 in favour of males. Mean body mass of individuals in the colony may vary from 103–202 g in males, and 88–145 g in females, depending upon the ages of the adult non-reproductive animals in the colony (Jacobs et al. 1991).
Ecosystem and cultural services: Generally, mole-rats are good subterranean ecosystem engineers. Not only do they consume underground vegetation, but they also alter the soil both physically and chemically (Hagenah and Bennett 2013). Their extensive excavations and the associated impacts generate a dynamic mosaic of nutrients and soil conditions that promote species diversity and maintain disturbance-dependent components of plant communities (Reichman and Seabloom 2002).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: 1 to 1.5 years
Age at Maturity: Male: 1 to 1.5 years
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 15 years
Average Reproductive Age: depends on reproductive status
Maximum Size (in cms): 300g
Size at Birth (in cms): 8 to 10 g
Gestation Time: 78-92 days
Reproductive Periodicity: 4 litters per annum
Average Annual Fecundity or Litter Size: 3 (1-6)
Natural Mortality: mole snakes and mongoose
Does the species lay eggs? no
Does the species give birth to live young: yes
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: Above and below ground dispersal
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form. Some individuals are taken from the wild for research purposes but this does not appear to have a negative impact upon the population. Individuals are also used for zoo exhibits to educate the public about their unique range of mating strategies.
Local Livelihood: No
National Commercial Value: No
International Commercial Value: No
End Use: None other than research
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species. As they occupy arid habitats, there is little conflict with agriculture (Bennett 2013). However, optimal conditions for foraging and burrowing are greatly restricted by the unpredictable and sporadic rainfall associated with the semi-arid habitats occupied by this species (Jarvis and Bennett 1991).
Conservation
The Damaraland Mole-rat is present within a number of protected areas throughout its range. These include the Kgalagadi Transfrontier Park, Tswalu Game Reserve and Molopo Nature Reserve, within the assessment region, as well as a number of protected areas in northern Botswana. Currently, no interventions are needed for this species within the assessment region. However, more research is needed on the possible effects of climate change on this species (Bennett and Jarvis, 2004).
Recommendations for land managers and practitioners:
- The establishment of a breeding programme of captive Damaraland Mole-rats for laboratory research practices would reduce the removal of wild caught colonies from their natural habitat.
Research priorities: Currently the Mammal Research Institute (University of Pretoria) is attempting to unravel the mechanism of sociality-induced infertility that is operational in this species.
- Research into the conservation of this species, and the effects of habitat loss and enhanced unpredictability of rainfall on population size, may be necessary in light of the current predicted effects of regional climate change.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP). However, due to their subterranean lifestyle, citizen sightings may be rare.
Bibliography
Bennett, N.C. 2013. Cryptomys damarensis Damaraland Mole-rat. In: Happold, D.C.D. (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, pp. 651–653. Bloomsbury Publishing, London.
Bennett, N.C. and Jarvis, J.U.M. 1988. The reproductive biology of the Cape mole-rat, Georychus capensis (Rodentia, Bathyergidae). Journal of Zoology 214: 95–106.
Bennett, N.C. & Faulkes, C.G. 2000. African Mole-rats: ecology and eusociality (Cambridge University Press) pp.273.
Bennett, N.C. & Jarvis, J.U.M. 2004. The Damaraland mole-rat, Cryptomys damarensis. Mammalian Species 756:1-5.
Burland, T.M., Bennett, N.C., Jarvis, J.U.M. & Faulkes, C.G. (2002). Eusociality in African mole-rats: new insights from patterns of genetic relatedness in the Damaraland mole-rat Proceedings of the Royal Society of London.269: 1025-1030.
Faulkes, C.G., Verheyen, E., Verheyen, W., Jarvis, J.U.M. and Bennett, N.C. 2004. Phylogeographical patterns of genetic divergence and speciation in African mole-rats (Family: Bathyergidae). Molecular Ecology 13(3): 613-629.
Gray, J.E. 1864. Notes on the species of sand-moles (Georychus). Proceedings of the Zoological
Society of London, 1864, 123–125.
Hagenah, N. and Bennett, N.C. 2013. Mole rats act as ecosystem engineers within a biodiversity hotspot, the Cape Fynbos. Journal of Zoology 289: 19-26.
Ingram, C.M., Burda, H., Honeycutt, R.L. 2004. Molecular phylogenetics and taxonomy of the African mole-rats, genus Cryptomys and the new genus Coetomys Gray, 1864. Molecular Phylogenetics and Evolution 31: 997-1014
Jacobs, D.S., Bennett, N.C., Jarvis, J.U.M. and Crowe, T.M. 1991. The colony structure and dominance hierarchy of the Damaraland mole-rat, Cryptomys damarensis (Rodentia: Bathyergidae), from Namibia. Journal of Zoology 224: 553–576.
Jarvis, J.U., Bennett, N.C. and Spinks, A.C. 1998. Food availability and foraging by wild colonies of Damaraland mole-rats (Cryptomys damarensis): implications for sociality. Oecologia 113: 290–298.
Jarvis, J.U., O’Riain, M.J., Bennett, N.C. and Sherman, P.W. 1994. Mammalian eusociality: a family affair. Trends in Ecology and Evolution 9: 47–51.
Jarvis, J.U.M. and Bennett, N.C. 1991. Ecology and behaviour of the family Bathyergidae. In: Sherman, P.W., Jarvis, J.U.M. and Alexander, R.D. (eds), The Biology of the Naked Mole-rat, pp. 66–96. Princeton University Press, Princeton, New Jersey.
Jarvis, J.U.M. and Bennett, N.C. 1993. Eusociality has evolved independently in two genera of bathyergid mole-rats, but occurs in no other subterranean mammal. Behavioral Ecology and Sociobiology 33: 253-260.
Kock, D., Ingram, C.M., Frabotta, L.J., Honeycutt, R.L., Burda, H. 2006. On the nomenclature of Bathyergidae and Fukomys n. gen. (Mammalia: Rodentia). Zootaxa 1142: 51-55.
Lovegrove, B.G. 1991. The evolution of eusociality in molerats (Bathyergidae): a question of risks, numbers, and costs. Behavioral Ecology and Sociobiology 28: 37–45.
Mynhardt, S., Harris, L., Bloomer, P. & Bennett, N.C. 2021. Social and ecological factorsdriving reproductive skew in the Damaraland mole-rat, Fukomys danarensis. 21: 221 1-17.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Reichman, O.J. and Seabloom, E.W. 2002. The role of pocket gophers as subterranean ecosystem engineers. Trends in Ecology and Evolution 17: 44–49.
Schmidt, C.M., Jarvis, J.U.M. and Bennett, N.C. 2013. The long-lived queen: reproduction and longevity in female eusocial Damaraland mole-rats (Fukomys damarensis). African Zoology 48: 193-196.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Young, A.J. and Bennett, N.C. 2010. Morphological divergence of breeders and helpers in wild Damaraland Mole-rat societies. Evolution 64: 3190–3197.