Damara Horseshoe Bat
Rhinolophus damarensis

2025 Red list status
Vulnerable
Regional Population Trend
Unknown
Change compared
to 2016
Uplisted
Overview
Rhinolophus damarensis – Roberts, 1946
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – damarensis
Common Names: Damara Horseshoe Bat (English)
Synonyms: Rhinlophus darlingi ssp. damarensis Roberts, 1946
Taxonomic Note:
Previously considered a subspecies of Rhinolophus darlingi (Monadjem et al. 2010), where R. d. damarensis was known from Namibia and the Northern Cape (Meester et al. 1986). Jacobs et al. (2013) presented genetic evidence that R. darlingi (sensu lato) is polyphyletic, comprising two cryptic species corresponding to the western populations that occur in arid habitats and the populations occurring in central and eastern southern Africa. Specifically, R. damarensis occurs in Angola, Namibia, and northwestern South Africa, while R. darlingi occurs in eastern South Africa, Mozambique, Eswatini, Zimbabwe, Botswana, Zambia and Malawi.
Red List Status: VU – Vulnerable A4c (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.1, Lotter, C.2, & da Silva, J.M.3
Reviewers: Howard, A.1, Richards, L.4, Naidoo, T.4 & Bastian, A.5
Contributors: Patel, T.6, Roxburgh, L.6 & Raimondo, D.3
Institutions: 1University of the Free State, 2Inkululeko Wildlife Services (Pty) Ltd, 3South African National Biodiversity Institute, 4Durban Natural Science Museum, 5University of KwaZulu-Natal, 6Endangered Wildlife Trust
Previous Assessors and Reviewers: Jacobs, D., Taylor, P., Cohen, L., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Monadjem, A.
Previous Contributors: Raimondo, D. & Child, M.F.
Assessment Rationale
The Damara Horseshoe Bat has been uplisted from Least Concern to Vulnerable under Criterion A4c based on the following: Currently, the population size of this is inferred to be between 2,000 and 5,000 individuals (based on 20–50 colonies consisting of <100 individuals each). Within its estimated Extent of Occurrence (EOO) of 173,453 km2 this species depends predominantly on caves, abandoned mines, and similar habitats for roosting (Monadjem et al. 2020). The Area of Occupancy (AOO) is 800 km2, based on a grid size of 2 by 2 km, and all known records of the species, and 5,000 km2 based on a grid size of 5 by 5 km. In addition, it is suspected that the species within the assessment region may decline by 30% or more within the foreseeable future based on: i) anticipated loss of roosts or roosting habitat with new, expanding, and intensifying diamond and other mining activities along the Orange River and surrounds, as well as due to closure of old mine tunnels as gazetted by the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021); ii) extensive approved renewable (solar and wind) energy development in the Northern Cape, particularly within this species’ EOO (such as around Alexander Bay, Aggenys, Upington, Kenhart, and other towns; DFFE 2024); iii) a considerable predicted reduction in the range of this species based on future climate models (Taylor et al. 2024); and iv) the low number of reported locations for this species (Inkululeko Wildlife Services unpubl. data). Field surveys and monitoring are required to more accurately estimate population size and trend. Re-assessment will be necessary once more comprehensive population data are available.
Regional population effects: Habitat is contiguous with Namibia and thus dispersal is assumed to be occurring. However, with the predicted reduction in this species’ distribution range due to future climate change (Taylor et al. 2020), dispersal between South Africa and Namibia is expected to become highly constrained.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: Uplisted, due to an inferred population decline of up to 30% in the foreseeable future based on habitat loss from mining, energy development and climate change.
Red List Index
Red List Index: Uplisted
Recommended citation: Taylor P, Lötter C & da Silva JM. 2025. A conservation assessment of Rhinolophus damarensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The species is restricted to the arid, warm western parts of southern Africa, with records reported from Angola, Namibia and western South Africa (Jacobs et al. 2013). Within the assessment region, it has been recorded predominantly from the Northern Cape Province and marginally in the North-West Province. A record from the Western Cape Province (ACR 2015) was erroneous. The most recent call recordings of this species were made at Blinkklip/Gatkoppies, Soetfontein Cave, and Hopefield Mine (near Postmasburg) in February 2023, and at Koegelbeen Cave (near Grikwastad) in February 2024 (Inkululeko Wildlife Services unpubl. data). The estimated extent of occurrence is 173,453 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 30 m asl (Mondajem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,910 m asl (Mondajem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
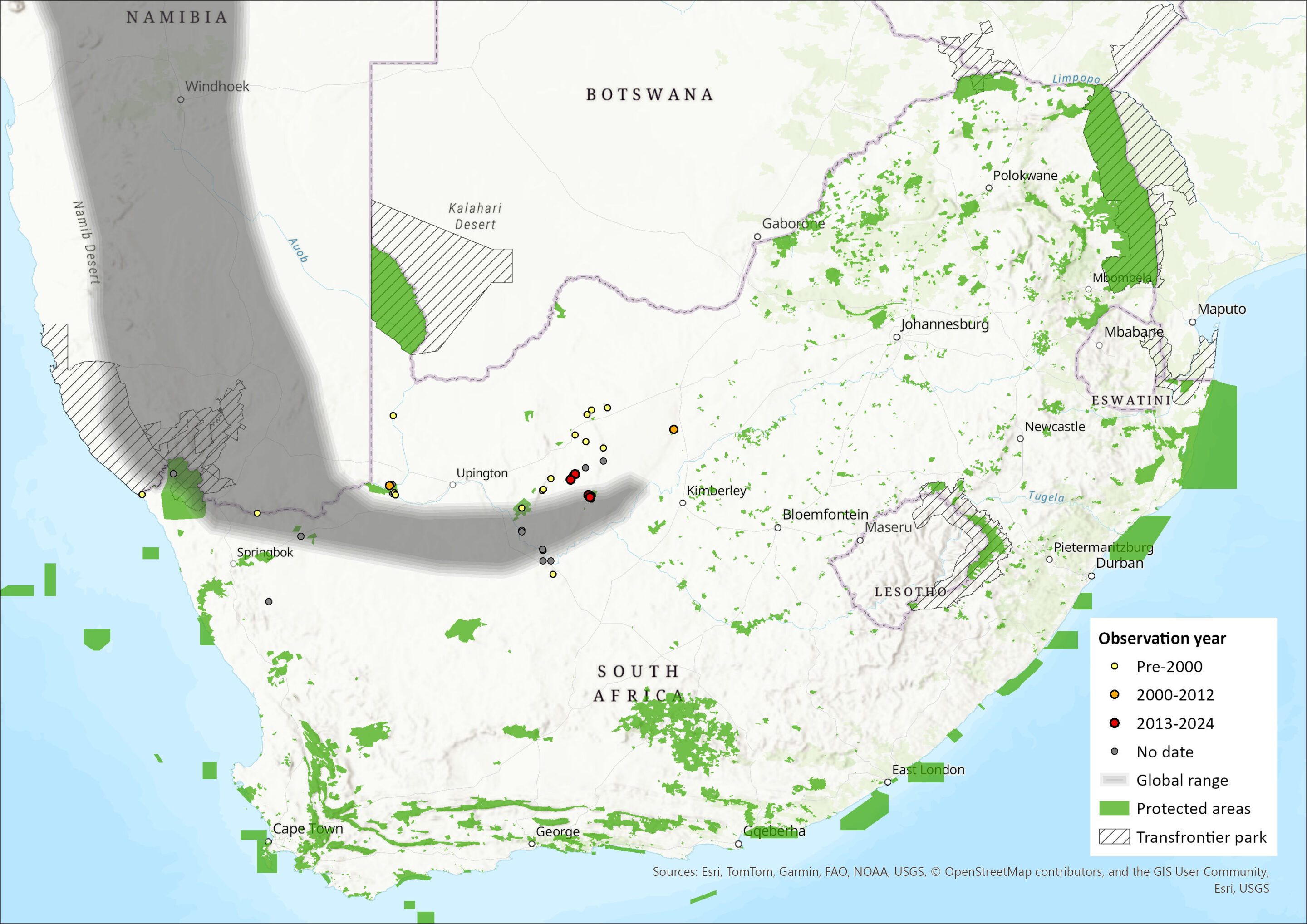 Figure 1. Distribution records for Damara Horseshoe Bat (Rhinolophus damarensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Damara Horseshoe Bat (Rhinolophus damarensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: (Not specified)
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | Resident |
| Namibia | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Sherwin et al. (2012) identified several risk factors for bats encompassing three broad aspects applicable for this species: 1) biogeography – small range size, and a range occupying a geographic area likely to become water stressed; 2) foraging niche –species restricted to aerial hawking; and 3) dispersal ability – species with restricted dispersal behaviour. These identified risk factors suggest that R. damarensis may be impacted by climate change more significantly than congeners which have larger distribution ranges and/or which occupy more temperate or mesic areas that are less prone to droughts (such as R. acrotis). Taylor et al. (2024) indeed reported a considerable reduction in the range of this species under future climate models. The modelling indicated that precipitation of the wettest month was the most important environmental variable for this arid-adapted Rhinolophus species.
Population information
While not much is known about abundance or population trends of this species, it occurs in moderately small colonies of generally <100 individuals per colony (D. Jacobs unpubl. data), but sometimes up to ca. 160 individuals (Churchill et al. 1997 and Monadjem et al. 2008 cited in Monadjem et al. 2020). While it is difficult to determine population size from acoustic data, this species contributed 0.3-27% of all bat calls recorded during surveys undertaken at Blinkklip (3 of 11 calls for 30 minutes by day), Soetfontein Cave (36 of 255 calls recorded for 1.5 hours after sunset), Hopefield Mine (3 of 53 calls for 30 minutes at sunset), and Koegelbeen Cave (17 of 6435 calls recorded from sunset to sunrise over two nights in February 2024). From this call data, the population size of this species is inferred to be between 2,000 and 5,000 individuals (based on 20–50 colonies consisting of < 100 individuals each). Systematic monitoring is necessary to more accurately estimate population size.
Current population trend: Currently unknown, a decline is suspected in future.
Number of subpopulations: Unknown, but 20-50 colonies.
Continuing decline in mature individuals? (Not specified)
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Rhinolophus damarensis has been investigated in a phylogenetic context (Jacobs et al. 2013, Demos et al. 2019; Taylor et al. 2024) and two clear lineages have been identified: northern (Namibian) and southern (South African) (Jacobs et al. 2013; Taylor et al. 2024). These two lineages, however, are not supported by nuclear intron DNA (Benda et al. 2024; Taylor et al. 2024).
Based on available information, it would appear as though individuals within the assessment region form a single population; however, this structure may not persist into the future according to climatic models (See Taylor et al. 2024). Projections show a greatly reduced and isolated distribution with the species, which could result in isolated subpopulations.
Based on current estimates of approximately 20,000 mature individuals within the region, the Ne for this species is estimated at approximately 2000-6000 individuals.
Habitats and ecology
It has been recorded from arid savannah and shrubland habitats within the Nama-Karoo Biome (Jacobs et al. 2013). It has specifically been recorded from the Eastern Kalahari Bushveld, Kalahari Duneveld, Bushmanland, Gariep Desert, Southern Namib Desert, Richtersveld and the Upper Karoo vegetation types. The species predominantly roosts in natural caves. However, it has been observed and recorded in old gold and asbestos mines (Monadjem et al. 2008).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | Yes |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Yes | |
| 8.2. Desert -> Desert – Temperate | – | Suitable | – |
Life History
Generation Length: Average age 5-6 years; possibly 10 years (presumably for an arid Rhinolophus species).
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Sexes combined: Forearm mean = 4.91 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: No reproductive information is available (Monadjem et al. 2020)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown (likely to be limited).
Congregatory: Colonies of up to 160 individuals have been recorded (Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
Not known to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There is a considerable predicted reduction in the range of this species based on future climate models (Taylor et al. 2024). Roost disturbance and loss presents a serious threat, which should be closely monitored. New, expanding, and intensifying diamond and other mining activities along the Orange River and the surrounds, is likely to cause a loss of roosts and roosting habitat. Re-opening of old mines, and implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021), may threaten colonies that are roosting in old mine tunnels. Roost loss may also occur with disturbance during recreational or tourism activities. Furthermore, Rhinolophus bat species are sensitive to water quality changes (Laverty and Berger 2020) and mining may impact water quality. Therefore, systematic monitoring of colony sizes and trends is needed, and the status of this species must be checked every few years or more often. Extensive renewable (solar and wind) energy development in the Northern Cape, particularly within this species’ EOO (such as around Alexander Bay, Aggenys, Upington, Kenhart, and other towns; DFFE 2024) may also present a major threat to this species. Due to wing morphology and the clutter foraging habits of this species, it is regarded as having a Low risk of fatality from wind turbines (MacEwan et al. 2020). However, colonies could be compromised by destruction/disturbance/degradation of, and displacement from, roost sites, water sources, feeding areas, and movement paths due to e.g. clearing of vegetation for solar panels, noise and vibrations from construction activities, spinning turbines, and light pollution in the previously dark nighttime landscape of the Northern Cape. As a cavity-roosting species, R. damarensis could be particularly sensitive to light pollution. Indirect poisoning resulting from the use of insecticides, pesticides and similar chemicals, and light pollution may also decrease the natural prey base of this species within its arid range.
Conservation
Within the assessment region, the species is recorded from the formally protected Richtersveld National Park and the Augrabies Falls National Park. The South Africa Department of Mineral Resources and Energy should engage with the South African Bat Assessment Association with regards to future closure of old mine tunnels in the country, so as to minimise the impact of this on cavity-roosting bat species such as R. damarensis. Renewable energy developments must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (for buffer recommendations see MacEwan et al. 2020 or later). Monitoring of population trends in response to the threat of mine expansion and closure, and renewable energy development, is strongly recommended. This species would also benefit from holistic land management techniques that reduce the needs for pesticides.
Recommendations for land managers and practitioners:
- Identify and protect important roost sites for this species.
- The South Africa Department of Mineral Resources and Energy should engage with the South African Bat Assessment Association with regards to future closure of old mine tunnels in the country, so as to minimise the impact of this on cavity-roosting bat species such as R. damarensis.
- Renewable energy development must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (see MacEwan et al. 2020).
- Reduce pesticide use in agricultural landscapes.
Research priorities:
- Systematic surveys to identify further colonies and assess population size and trend.
- Research investigating the severity of identified threats, including the effects of climate change, and potential conservation interventions.
- Systematic assessment of unresolved structure in the clade, specifically in relation to Namibian lineages and potential introgression events with R. cervenyi.
Encouraged citizen actions:
- Minimise disturbance to caves when visiting, particularly within this species’ distribution range.
- Citizens can report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Demos, T.C., Webala, P.W., Bartonjo, M. and Patterson, B.D. 2018. Hidden Diversity of African Yellow House Bats (Vespertilionidae, Scotophilus): Insights From Multilocus Phylogenetics and Lineage Delimitation. Frontiers in Ecology and Evolution 6: https://doi.org/10.3389/fevo.2018.00086.
DFFE (Department of Forestry, Fisheries, and the Environment). 2024. South African Renewable Energy EIA Application Database (REEA). Version 2024_Q2. https://egis.environment.gov.za. Accessed September 2024.
Jacobs, D.S., Babiker, H., Bastian, A., Kerney, T. van Eeden, R. and Bishop, J.M. 2013. Phenotypic convergence in genetically distinct lineages of a Rhinolophus species complex (Mammalia, Chiroptera). Plos One 8(12): e82614.
Laverty, T. and Berger, J. 2020. Do bats seek clean water? A perspective on biodiversity from the Namib Desert. Biological Conservation 248. https://doi.org/10.1016/j.biocon.2020.108686.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association. South Africa.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem, A., Higgins, N., Smith, T., and Herrmann, E. 2008. Bats recorded from Koegelbeen cave and selected other sites in the Northern Cape, South Africa. African Bat Conservation News 18: 2–4.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2010. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Sherwin, H.A., Montgomery, W.I. and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Taylor, P.J., Kearney, T.C., Clark, V.R., Howard, A., Mdluli, M.V., Markotter, W., Geldenhuys, M., Richards, L.R., Rakotoarivelo, A.R., Watson, J., Balona, J., Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate‐driven diversification and a buffer against future climate change in bats. Global Change Biology, 30(6), p.e17344.
