
Common Bottlenose Dolphin
Tursiops truncatus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Tursiops truncatus – (Montagu, 1821)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – DELPHINIDAE – Tursiops – truncatus
Common Names: Common Bottlenose Dolphin, Bottlenosed Dolphin, Bottlenose Dolphin, Bottlenosed Dolphin (English), Stompneusdolfyn (Afrikaans), Afalina (Turkish), Dauphin souffleur, Grand dauphin, Souffleur (French), Delfin Geddumu Qasir (Maltese), Delfin I Madh (Albanian), Delfin Kabir (Arabic), Delfín Mular (Spanish; Castilian), Dobri dupin (Croatian), Golfinho-roaz (Portuguese), Grosser Tümmler (German), Tursiope (Italian), Velika pliskavka (Slovenian), Ρινοδέλφινο (Rinodélfino) (Greek, Modern (1453-)
Synonyms: Delphinus truncatus Montagu, 1821; Tursiops nuuanu Andrews, 1911
Taxonomic Note:
The taxonomy of the genus Tursiops remains unresolved. Consequently, there is confusion in the literature between Tursiops species, not only in respect of taxonomy, but also natural history. In many regions across its distribution two forms of bottlenose dolphins have been described: an offshore form and a coastal form. Although many of their characteristics overlap, morphological and mitochondrial differences between these two regional forms have been recognised (Ross 1977; Hoelzel et al. 1998). In South African waters, Ross (1977) described two allopatric species based on morphological differences: the smaller, coastal form, T. aduncus, and the larger offshore form, T. truncatus. Hoelzel et al. (1998) compared mitochondrial and nuclear genetic markers between nearshore and offshore types of bottlenose dolphins in a range of geographic locations. Although, Hoelzel et al. (1998) described a clear distinction between the coastal and offshore forms of bottlenose dolphins in the western Pacific, no such distinction was identified between the larger offshore form (T. truncatus) and the smaller inshore form (sometimes referred to as T. aduncus) off southern Africa. Hoelzel et al. (1998) suggest that the separation between these forms off Africa may be comparatively recent, or some degree of gene flow between the populations may still exist. Additionally, Ross and Cockcroft (1990) suggest that the two forms should not be delineated as separate species. Generally, where they occur in the same geographic areas T. truncatus has a longer body, larger skull and less teeth than T. aduncus (Ross 1977; Gao et al. 1995). Both species show sexual dimporphism, with males larger than females (Cockcroft & Ross 1990; Hale et al. 2000). In this assessment, we consider only the offshore T. truncatus, while T. aduncus is assessed separately. There is likely to be further fine scale resolution of the genus in future, as research progresses.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Probert, R.1, & da Silva, J.2
Reviewer: Plön, S.1
Institutions: 1University of Cape Town, South Africa, 2South African National Biodiversity Institute
Previous Assessor: Plön, S.
Previous Reviewer: Cockcroft, V.
Previous Contributors: Findlay, K., Oosthuizen, H., Meyer, M., Elwen, S., Relton, C., Child, M.F. & Atkins, S.
Assessment Rationale
The Common Bottlenose Dolphin is widespread and abundant throughout its range and regular sightings and strandings within the assessment region suggest that there is no major population decline and no immediate major threats are suspected. However, shifts in climatic conditions over the years may be of concern, although there is insufficient data to support this thus far. In contrast to T. aduncus, which is commonly accepted as the coastal resident population of bottlenose dolphins, T. truncatus is considered to occur largely offshore. Anthropogenic disturbance in the form of boat traffic, fisheries and ecotourism, as well as pollution (including noise, plastic debris and persistent organic pollutants) are recognised as minor threats to this species. Common Bottlenose Dolphins are currently not considered a conservation priority and are therefore listed as Least Concern in line with the global listing.
Regional population effects: The Common Bottlenose Dolphin exhibits seasonal movements, often following the seasonal migrations of sardine off South Africa’s south and east coasts. There are no barriers to dispersal, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citations: Probert R & da Silva JM. 2025. A conservation assessment of Tursiops truncatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Globally, bottlenose dolphins are widely distributed, found throughout tropical and temperate regions, only absent in the extreme high latitudes (Skinner & Chimimba 2005). Two forms are often described, where one is commonly restricted to coastal areas and estuaries, while the other is associated with open ocean areas, usually regions of upwelling and high productivity, such as shelf edges and sea mounts. Across the entire assessment region, the range of the Common Bottlenose Dolphins extends from the Orange River mouth to Kosi Bay, present both in continental shelf waters and inshore, seldomly found in waters shallower than 50m.
A common assumption is that inshore records in the Indian Ocean belong to T. aduncus, while T. truncatus is only found further offshore (Best 2007). Findlay et al. (1992) describes the presence of T. truncatus offshore on the south and southeast coast, as well as inshore on the west coast of South Africa.
In the southwest Atlantic a coastal population of T. truncatus occurs off Namibia, usually found in waters less than 10 m deep. It has been reported from waters between Cape Cross and Walvis Bay, but the geographical limits of its range remain largely uncertain (Best 2007). This is the only resident coastal population of this species in southern Africa (Namibia, South Africa and Mozambique); elsewhere off the South African coast this species is usually found in waters less than 100 m deep, as well as at depths of between 500 m and 1,000 m (Best 2007).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): NA
Elevation Upper Limit (in metres above sea level): NA
Depth Lower Limit (in metres below sea level): 1000 m
Depth Upper Limit (in metres below sea level): 0 m
Depth Zone: (Not specified)
Map
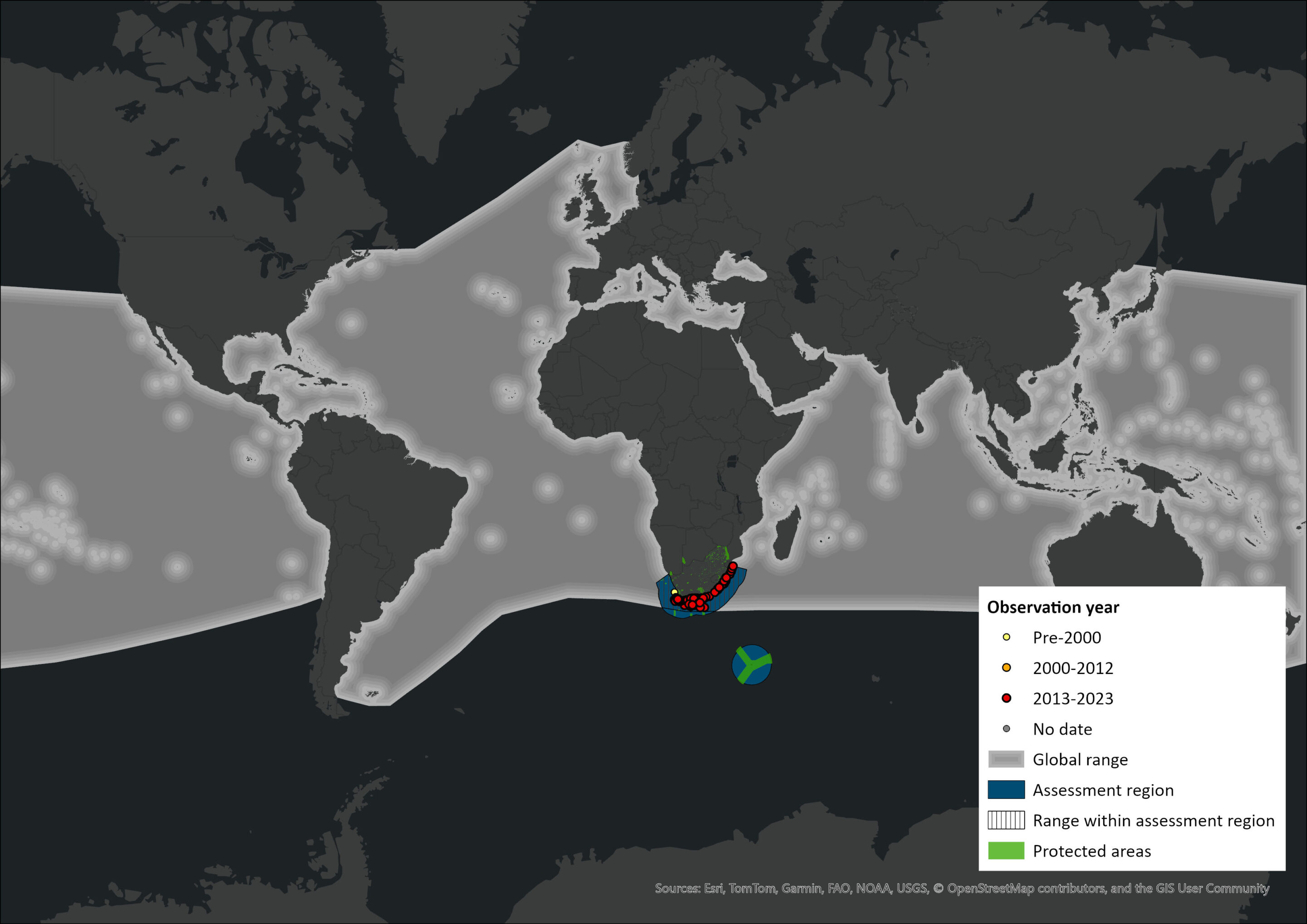 Figure 1. Distribution records for Common Bottlenose Dolphin (Tursiops truncatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Common Bottlenose Dolphin (Tursiops truncatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Australasian, Indomalayan, Nearctic, Neotropical, Oceanian, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Albania | Extant | Native | – | Resident |
| Algeria | Extant | Native | – | Resident |
| American Samoa | Extant | Native | – | Resident |
| Angola | Extant | Native | – | Resident |
| Anguilla | Extant | Native | – | Resident |
| Antigua and Barbuda | Extant | Native | – | Resident |
| Argentina | Extant | Native | – | Resident |
| Aruba | Extant | Native | – | Resident |
| Australia | Extant | Native | – | Resident |
| Bahamas | Extant | Native | – | Resident |
| Bahrain | Extant | Native | – | Resident |
| Bangladesh | Extant | Native | – | Resident |
| Barbados | Extant | Native | – | Resident |
| Belgium | Extant | Native | – | Resident |
| Belize | Extant | Native | – | Resident |
| Benin | Extant | Native | – | Resident |
| Bermuda | Extant | Native | – | Resident |
| Bonaire, Sint Eustatius and Saba | Extant | Native | – | Resident |
| Bosnia and Herzegovina | Extant | Native | – | Resident |
| Brazil | Extant | Native | – | Resident |
| British Indian Ocean Territory | Extant | Native | – | Resident |
| Brunei Darussalam | Extant | Native | – | Resident |
| Bulgaria | Extant | Native | – | Resident |
| Cabo Verde | Extant | Native | – | Resident |
| Cambodia | Extant | Native | – | Resident |
| Cameroon | Extant | Native | – | Resident |
| Canada | Extant | Native | – | Resident |
| Canada -> Newfoundland I | Extant | Vagrant | – | – |
| Cayman Islands | Extant | Native | – | Resident |
| Chile | Extant | Native | – | Resident |
| China | Extant | Native | – | Resident |
| Christmas Island | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Cocos (Keeling) Islands | Extant | Native | – | Resident |
| Colombia | Extant | Native | – | Resident |
| Comoros | Extant | Native | – | Resident |
| Congo | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Congo, The Democratic Republic of the | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Cook Islands | Extant | Native | – | Resident |
| Costa Rica | Extant | Native | – | Resident |
| Croatia | Extant | Native | – | Resident |
| Cuba | Extant | Native | – | Resident |
| Curaçao | Extant | Native | – | Resident |
| Cyprus | Extant | Native | – | Resident |
| Côte d’Ivoire | Extant | Native | – | Resident |
| Denmark | Extant | Native | – | Resident |
| Djibouti | Extant | Native | – | Resident |
| Dominica | Extant | Native | – | Resident |
| Dominican Republic | Extant | Native | – | Resident |
| Ecuador | Extant | Native | – | Resident |
| Egypt | Extant | Native | – | Resident |
| El Salvador | Extant | Native | – | Resident |
| Equatorial Guinea | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Eritrea | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Falkland Islands (Malvinas) | Extant | Native | – | Resident |
| Faroe Islands | Extant | Native | – | Resident |
| Fiji | Extant | Native | – | Resident |
| France | Extant | Native | – | Resident |
| French Guiana | Extant | Native | – | Resident |
| French Polynesia | Extant | Native | – | Resident |
| Gabon | Extant | Native | – | Resident |
| Gambia | Extant | Native | – | Resident |
| Georgia | Extant | Native | – | Resident |
| Germany | Extant | Native | – | Resident |
| Ghana | Extant | Native | – | Resident |
| Gibraltar | Extant | Native | – | Resident |
| Greece | Extant | Native | – | Resident |
| Grenada | Extant | Native | – | Resident |
| Guadeloupe | Extant | Native | – | Resident |
| Guam | Extant | Native | – | Resident |
| Guatemala | Extant | Native | – | Resident |
| Guernsey | Extant | Native | – | Resident |
| Guinea | Extant | Native | – | Resident |
| Guinea-Bissau | Extant | Native | – | Resident |
| Guyana | Extant | Native | – | Resident |
| Haiti | Extant | Native | – | Resident |
| Honduras | Extant | Native | – | Resident |
| Hong Kong | Extant | Native | – | Resident |
| India | Extant | Native | – | Resident |
| Indonesia | Extant | Native | – | Resident |
| Iran, Islamic Republic of | Extant | Native | – | Resident |
| Iraq | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Ireland | Extant | Native | – | Resident |
| Isle of Man | Extant | Native | – | Resident |
| Israel | Extant | Native | – | Resident |
| Italy | Extant | Native | – | Resident |
| Jamaica | Extant | Native | – | Resident |
| Japan | Extant | Native | – | Resident |
| Jersey | Extant | Native | – | Resident |
| Jordan | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Kenya | Extant | Native | – | Resident |
| Kiribati | Extant | Native | – | Resident |
| Korea, Democratic People’s Republic of | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Korea, Republic of | Extant | Native | – | Resident |
| Kuwait | Extant | Native | – | Resident |
| Lebanon | Extant | Native | – | Resident |
| Liberia | Extant | Native | – | Resident |
| Libya | Extant | Native | – | Resident |
| Macao | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Madagascar | Extant | Native | – | Resident |
| Malaysia | Extant | Native | – | Resident |
| Maldives | Extant | Native | – | Resident |
| Malta | Extant | Native | – | Resident |
| Marshall Islands | Extant | Native | – | Resident |
| Martinique | Extant | Native | – | Resident |
| Mauritania | Extant | Native | – | Resident |
| Mauritius | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Mayotte | Extant | Native | – | Resident |
| Mexico | Extant | Native | – | Resident |
| Micronesia, Federated States of | Extant | Native | – | Resident |
| Monaco | Extant | Native | – | Resident |
| Montenegro | Extant | Native | – | Resident |
| Montserrat | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Morocco | Extant | Native | – | Resident |
| Mozambique | Extant | Native | – | Resident |
| Myanmar | Extant | Native | – | Resident |
| Namibia | Extant | Native | – | Resident |
| Nauru | Extant | Native | – | Resident |
| Netherlands | Extant | Native | – | Resident |
| New Caledonia | Extant | Native | – | Resident |
| New Zealand | Extant | Native | – | Resident |
| Nicaragua | Extant | Native | – | Resident |
| Nigeria | Extant | Native | – | Resident |
| Niue | Extant | Native | – | Resident |
| Norfolk Island | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Northern Mariana Islands | Extant | Native | – | Resident |
| Norway | Extant | Vagrant | – | – |
| Oman | Extant | Native | – | Resident |
| Pakistan | Extant | Native | – | Resident |
| Palau | Extant | Native | – | Resident |
| Panama | Extant | Native | – | Resident |
| Papua New Guinea | Extant | Native | – | Resident |
| Peru | Extant | Native | – | Resident |
| Philippines | Extant | Native | – | Resident |
| Pitcairn | Extant | Native | – | Resident |
| Portugal | Extant | Native | – | Resident |
| Puerto Rico | Extant | Native | – | Resident |
| Qatar | Extant | Native | – | Resident |
| Romania | Extant | Native | – | Resident |
| Russian Federation | Extant | Native | – | Resident |
| Réunion | Extant | Native | – | Resident |
| Saint Barthélemy | Extant | Native | – | Resident |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | Resident |
| Saint Kitts and Nevis | Extant | Native | – | Resident |
| Saint Lucia | Extant | Native | – | Resident |
| Saint Martin (French part) | Extant | Native | – | Resident |
| Saint Pierre and Miquelon | Extant | Native | – | Resident |
| Saint Vincent and the Grenadines | Extant | Native | – | Resident |
| Samoa | Extant | Native | – | Resident |
| Sao Tome and Principe | Extant | Native | – | Resident |
| Saudi Arabia | Extant | Native | – | Resident |
| Senegal | Extant | Native | – | Resident |
| Seychelles | Extant | Native | – | Resident |
| Sierra Leone | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Singapore | Extant | Native | – | Resident |
| Sint Maarten (Dutch part) | Extant | Native | – | Resident |
| Slovenia | Extant | Native | – | Resident |
| Solomon Islands | Extant | Native | – | Resident |
| Somalia | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Spain | Extant | Native | – | Resident |
| Sri Lanka | Extant | Native | – | Resident |
| Sudan | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Suriname | Extant | Native | – | Resident |
| Syrian Arab Republic | Extant | Native | – | Resident |
| Taiwan, Province of China | Extant | Native | – | Resident |
| Tanzania, United Republic of | Extant | Native | – | Resident |
| Thailand | Extant | Native | – | Resident |
| Timor-Leste | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Togo | Extant | Native | – | Resident |
| Tokelau | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Tonga | Extant | Native | – | Resident |
| Trinidad and Tobago | Extant | Native | – | Resident |
| Tunisia | Extant | Native | – | Resident |
| Turks and Caicos Islands | Extant | Native | – | Resident |
| Tuvalu | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Türkiye | Extant | Native | – | Resident |
| Ukraine | Extant | Native | – | Resident |
| United Arab Emirates | Extant | Native | – | Resident |
| United Kingdom of Great Britain and Northern Ireland | Extant | Native | – | Resident |
| United States of America | Extant | Native | – | Resident |
| Uruguay | Extant | Native | – | Resident |
| Vanuatu | Extant | Native | – | Resident |
| Venezuela, Bolivarian Republic of | Extant | Native | – | Resident |
| Viet Nam | Extant | Native | – | Resident |
| Virgin Islands, British | Extant | Native | – | Resident |
| Virgin Islands, U.S. | Extant | Native | – | Resident |
| Wallis and Futuna | Extant | Native | – | Resident |
| Western Sahara | Extant | Native | – | Resident |
| Yemen | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: Benguela Current and Agulhas Current LMEs
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 21. Atlantic – northwest | Extant | Native | – | – |
| 27. Atlantic – northeast | Extant | Native | – | – |
| 31. Atlantic – western central | Extant | Native | – | – |
| 34. Atlantic – eastern central | Extant | Native | – | – |
| 37. Mediterranean and Black Sea | Extant | Native | – | – |
| 41. Atlantic – southwest | Extant | Native | – | – |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 51. Indian Ocean – western | Extant | Native | – | – |
| 57. Indian Ocean – eastern | Extant | Native | – | – |
| 61. Pacific – northwest | Extant | Native | – | – |
| 67. Pacific – northeast | Extant | Native | – | – |
| 71. Pacific – western central | Extant | Native | – | – |
| 77. Pacific – eastern central | Extant | Native | – | – |
| 81. Pacific – southwest | Extant | Native | – | – |
| 87. Pacific – southeast | Extant | Native | – | – |
Climate change
There have been significant shifts in both the Benguela current (Hutchings et al. 2009; Moloney et al. 2013; Jarre et al. 2015) and Agulhas current (Mead et al. 2013; Asdar et al. 2022). Large Marine Ecosystems have been documented over the years, including changes in wind patterns, upwelling regimes, and temperatures. These changes can have both direct and indirect effects on Common Bottlenose Dolphin populations. The average sea surface temperature (SST) in the Agulhas Bank increased by 1.5 °C from the 1980s to the early 2000s (Mead et al. 2013) and is estimated to further increase by 2 °C between 2020 and 2100 (Asdar et al. 2022). The Benguela current is warming at a faster rate of 0.15-0.22 °C per decade (Sweijd and Smit, 2020). Common Bottlenose Dolphins have been reported in waters between 10 and 32 °C. As T. truncatus is highly mobile and inhabits waters with a wide thermal range, it is unlikely that they will be severely impacted by increasing SST, although these changes can impact the distribution across their range.
Climate change can also exacerbate existing threats to T. truncatus such as habitat loss, prey availability, risks of bycatch, and pollutant concentrations. Increasing SST is linked to coral bleaching and has been documented along the South African coast (Celliers & Schleyer 2002), causing loss of reef habitats which are important for the species. Prey species favourable to T. truncatus have varying sensitivities to climate change. Mullet, a preferred prey item, is extremely sensitive to shifts in sea temperatures causing a change in their distribution along the coastline (James et al. 2016). These shifts, paired with those caused by increased fishing pressures, may decrease prey available to bottlenose dolphins. However, T. truncatus are generalist feeders with varying degrees of dietary and feeding plasticity (Neri et al. 2022). Further investigation into the effects of climate change on prey availability and prey choice is needed to fully understand the magnitude of the overall effect on Common Bottlenose Dolphin populations in the various complex ecosystems in South African waters. Furthermore, a decrease in fish biomass can result in an increase in fishing efforts, increasing the risk of dolphin bycatch and entanglement. Common Bottlenose Dolphins are at risk of pollutant contamination, particularly persistent organic pollutants (POPs), which are known to cause reproductive impairment and immune system suppression in other dolphin species (Borrell & Aguilar 2005). Increased sea temperatures have been shown to influence the metal toxicity in marine species (Langston 1990), although it has not been directly tested in dolphins.
Overall, climate change can have significant impacts, both direct and indirect, on Common Bottlenose Dolphins, impacting their physical habitat, habitat ranges and distribution, the abundance and distribution of prey, and their general and reproductive health. Shifts in species and prey distribution should be assessed concurrently with the changes in the various ecosystems in South African waters to further understand the effects of climate change on T. truncatus within this region.
Population
Globally, there are estimated to be more than 600,000 Common Bottlenose Dolphins (Hammond et al. 2012). Groups of several tens of T. truncatus are frequently sighted, along with False Killer Whales (Pseudorca crassidens), in the Plettenberg Bay area. This species is sighted regularly in South African waters, and thus, despite frequent stranding events, no population decline is expected
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | – | – |
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Current population trend: Stable
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic studies have been undertaken on T. truncatus in the assessment region. While the species is currently considered a single population in the area, it is possible subpopulations (genetic structure) is present as has been found for this species in other regions around the world (e.g., Sellas et al. 2005; Segura et al. 2006). Fine scale genomic studies would be useful to confirm this.
Thus far, there are no population size estimates for T. truncatus within the assessment region, however there are regular sightings of this species in South African waters indicating no apparent population decline. Investigation into population size and genetic differentiation are necessary for a robust population assessment within this region.
Habitats and ecology
Tursiops truncatus is commonly accepted as the open water form of the bottlenose dolphin. However, there are many exceptions to this rule, and this species may also be frequently located within shallower waters, nearer to the coast. Presumably though, T. truncatus generally makes use of deeper reefs further offshore, whereas T. aduncus is restricted to shallower inshore areas (Hale et al. 2000). Studies off the coast of North America found that this species is generally associated with waters exhibiting surface temperatures between 10°C and 32°C (Wells & Scott 1999). Common Bottlenose Dolphins form schools of between 3–100 individuals, with a general average of approximately 22, and are often associated with other cetacean species, for example the Long-finned Pilot Whale (Globicephala melas) and the False Killer Whale (Skinner & Chimimba 2005).
The stomach contents of two T. truncatus individuals from the Eastern Cape revealed that the dominant prey species was squid (Oregoniateuthis), with hake (Merluccius spp.) and Buttersnoek (Lepidpus caudatus) making up lesser proportions (Ross 1977, 1984). The results from these studies indicate that T. truncatus feeds further offshore when compared to T. aduncus, at least off the Eastern Cape coast (Ross 1984). Off the Western Cape, Sekiguchi et al. (1992) recorded that the diet of T. truncatus comprised of cephalopods (mostly the Cape Hope Squid, Loligo vulgaris reynaudii), and a wide variety of fish (dominated by Southern Mullet, Liza richardsonii, and Cape Horse Mackerel, Trachurus trachurus capensis). However, an updated investigation into the diet of Common Bottlenose Dolphins in various regions of the assessment area is needed, with focus on fishery target species in each respective area, as well as feeding strategies used. Leatherwood (1975) describes the high degree of plasticity associated with feeding behaviour of Tursiops spp. along the west coast of North America, including echolocation techniques, cooperative hunting, and the exploitation of anthropogenic fishing activities (such as depredation).
Mother and calf associations may last as long as 3 to 4 years (Bearzi et al. 1997), which may be a general reflection of the inter-birth interval exhibited by female Common Bottlenose Dolphins. Although females usually only breed every 3–6 years, Connor et al. (2000) described intervals of 2 years off the coast of Florida. Model-based estimates of generation time are 21.1 years (Taylor et al. 2007).
Ecosystem and cultural services: This is the archetype of dolphins and, since most South Africans are unaware of the variety of delphinids, this is typically what they envisage when “dolphins” are mentioned.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Marginal | – |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 9.10. Marine Neritic -> Marine Neritic – Estuaries | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
| 13.4. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Coastal Brackish/Saline Lagoons/Marine Lakes | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: 5-12 (population dependent)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Marine
General Use and Trade Information
There is no trade of this species within South Africa, although there were three, but currently two pure T. truncatus at uShaka Marine World, KwaZulu-Natal, but this has no effect on the wild populations of this species.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | – | – | – |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 2. Food – animal | true | true | – | – |
| 13. Pets/display animals, horticulture | – | true | true | – |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Around the world, Common Bottlenose Dolphins are vulnerable to both accidental and intentional catch, climate change, habitat degradation (Curry & Smith 1997), as well as disturbance and harassment (often due to ecotourism activities). Within the assessment region, this species is not expected to be at risk of any significant population decline; however, a number of minor threats have been identified, and the combination of these threats may become a cause for concern in the future.
Anthropogenic disturbance: Although no known tourism targets this species in South Africa, tourism, boat traffic and ‘swim-with’ programmes are known to influence the natural movements (Constantine et al. 2004; Lusseau 2005), social behaviours (Nowacek et al. 2001; Bejder et al. 2006b; Rocha et al. 2023), energy budgets and geographic ranges (Bejder et al. 2006a) of bottlenose dolphin species. For example, a long-term study in New Zealand found an increase in dolphin avoidance of swimmers, and a decrease in dolphin interaction with humans over time. Additionally, cetaceans have shown additional avoidance behaviours in response to other forms of anthropogenic disturbance (Finley et al. 1990; Kruse 1991; Janik & Thompson 1996; Bejder et al. 1999), which may in turn affect natural foraging, resting and socialising behaviour (Constantine 2001; Constantine et al. 2004). Continued disruption of feeding, resting and social activities of Common Bottlenose Dolphins could have detrimental impacts on reproduction rates (Stensland & Berggren 2007; Dans et al. 2008) and calf survival (Bejder et al. 2006a; Stensland & Berggren 2007).
Collision with boats: Vessel-related physical injury of bottlenose dolphins has been documented across a number of regions (e.g. Nowacek et al. 2001; van Waerebeek et al. 2007; Bechdel et al. 2009; Dwyer et al. 2014), where collision with propellers and hulls may result in injuries ranging from minor lacerations and blunt force trauma to death. For example, photo-identification data captured off the coast of Florida showed that 6.0% of the distinctly marked population of bottlenose dolphins had injuries attributed to motorised vessels (Bechdel et al. 2009), and in the Gulf of Guayaquil, nearly 2% of bottlenose dolphins had propeller-related scars and injuries (van Waerebeek et al. 2007). Three fatally injured bottlenose dolphins off western Florida showed a range of injuries, including a completely severed tail and substantial bruising (Morgan & Patton 1990). In the Southern Hemisphere, van Waerebeek et al. (2007) found that habituation of dolphins to boats appears to be a contributing factor in dolphin-vessel collision events.
Fisheries bycatch: Accidental bycatch of Common Bottlenose Dolphins occurs throughout the species’ range in both commercial and recreational fisheries, as well as shark nets, but incidental reports are poorly documented (Wells & Scott 1999). As shark nets are deployed in shallow, coastal waters in South Africa, bycatch of offshore T. truncatus is not a problem within the assessment region. However, depredation (or the act of stealing or damaging prey captured in fishing gear) occurs in offshore commercial and local fishing areas and can lead to serious physical injury or death of cetaceans through entanglement or ingestion. Additionally, continued and learned behaviour associated with depredation impacts natural activity patterns (Cox et al. 2003; Lauriano et al. 2004; Brotons et al. 2008; Sigler et al. 2008; Powell & Wells 2011). Furthermore, dolphins often cause substantial economic impacts for fishermen, including net damage and a reduction in overall fish catch (Buscaino et al. 2009), leading to negative responses towards dolphins.
Competition: Depredation behaviour is likely a direct response to increased competition for forage resources between cetaceans and humans. Loss of prey availability and biomass as a result of overfishing and environmental degradation is an increasing threat to this species in large parts of its range.
Noise pollution: Cetaceans depend on auditory stimuli for navigation, communication and hunting, thus are commonly considered sensitive to anthropogenic noise pollution (Finneran et al. 2000). Noise associated with ships, seismic exploitation, marine construction, demolition and sonars affect the movements and diving patterns of cetaceans and disrupts their communicative pathways and behaviour (Buckstaff 2004; Branstetter et al. 2018; Kragh et al. 2019), and may result in negative physiological responses, such as increased stress (Nowacek et al. 2007; Koper & Plön 2012).
Environmental contaminants: Xenobiotic chemicals and their toxic effects threaten the reproductive potential and immune system of this species. Bioaccumulation of persistent organic pollutants (POPs) within the body tissues of top marine predators is common and is documented for this species (Yordy et al. 2010a, 2010b).
Climate change: Climate change has direct and indirect effects on marine species. Shifts in climatic conditions can alter the habitat range and distributions of dolphins, as well as prey species, and can exacerbate existing threats, such as habitat loss, bycatch, and pollutant concentrations.
Current habitat trend: Declining in quality due to ongoing coastal development and poor agricultural practices upstream of watersheds.
Conservation
The species is listed in Appendix II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) and the Marine Living Resources Act (No. 18 of 1998). Mitigation measures designed to limit accidental cetacean bycatch in gillnet fisheries include spatiotemporal fishery closure regimes, marine protected areas, the use of acoustic alarms and other modifications of fishing equipment and techniques. Acoustic alarms often emit high frequency sounds, designed to deter cetaceans away from nets, or at least warn them of the barrier’s presence (Dawson et al. 1998). The use of high frequency alarms on gillnets along the US east coast were found to have only a subtle deterring effect on bottlenose dolphins and are unlikely to reduce dolphin bycatch to any significant degree (Cox et al. 2003). The use of pingers in artisanal fisheries around the Balearic Islands (western Mediterranean), reduced the level of interaction between bottlenose dolphins and bottom-set nets; however, the propensity for dolphin habituation calls for continued research into the long-term viability of acoustic deterrents, or the use of alternative mitigation efforts (Brotons et al. 2008). However, there is some evidence (Erbe et al. 2016) that pingers increase T. aduncus catch (no data for T. truncatus), and the potential for pingers to increase rather than decrease catch is a concern.
This species is likely to be impacted by offshore resource exploration (seismic surveys) and exploitation (for example, drilling and blasting), which has increased substantially in South African waters over the last decade. Working with environmental impact agencies to mitigate any impacts and applying pressure on governmental authorities to make accepted good practice mitigation measures obligatory during any exploration/exploitation, are important interventions.
Finally, in response to the increasing levels of negative impacts associated with the interaction between dolphins and the fishing industry, Buscaino et al. (2009) suggest a collaborative response towards sustainable exploitation of oceanic resources, a decrease in the intensity of marine extraction and the establishment of protected areas .
Recommendations for managers and practitioners:
- Further field surveys to delimit geographical boundaries and identify threats.
Research priorities:
- Continued research into the taxonomic relationships and the genetic variation between these southern African populations is necessary. Genetic analyses to assess potential differences in population structure of bottlenose dolphins between South Africa’s west and east coasts, as well as those off Namibia.
- Continued investigation into the response of bottlenose dolphins to anthropogenic sound. Including offshore petroleum exploration and exploitation.
- Physiological and behavioural effects of anthropogenic pollution, including bioaccumulation of toxins, noise pollution and plastic debris to Common Bottlenose Dolphins within the assessment region.
- Investigation into the direct and indirect impacts of climate change, particularly increasing sea temperatures, and how these can affect the population and distribution of Common Bottlenose Dolphins.
Encouraged citizen actions:
- Use information dispensed by the South African Sustainable Seafood Initiative to make good choices when buying fish in shops and restaurants, for example FishMS 0794998795.
- Buy fresh produce that has been grown in pesticide-free environments.
- Save electricity and fuel to mitigate CO2 emissions and hence, the rate of climate change.
- Buy local products that have not been shipped.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) to help with mapping geographical distribution, and report any stranded dolphins to your nearest museum, the Centre for Dolphin Studies or to relevant local authorities.
Bibliography
Asdar S, Jacobs ZL, Popova E, Noyon M, Sauer WH, Roberts MJ. 2022. Projected climate change impacts on the ecosystems of the Agulhas Bank, South Africa. Deep Sea Research Part II: Topical Studies in Oceanography 200:105092.
Bearzi, G., Notarbartolo-Di-Sciara, G. and Politi, E. 1997. Social ecology of bottlenose dolphins in the Kvarnerić (northern Adriatic Sea). Marine Mammal Science 13(4): 650-668. https://doi.org/10.1111/j.1748-7692.1997.tb00089.xhttps://doi.org/10.1111/j.1748-7692.1997.tb00089.x.
Bechdel SE, Mazzoil MS, Murdoch ME, Howells EM, Reif JS, McCulloch SD, Schaefer AM, Bossart GD. 2009. Prevalence and impacts of motorized vessels on bottlenose dolphins (Tursiops truncatus) in the Indian River Lagoon, Florida. Aquatic Mammals 35: 367.
Bejder L, Dawson SM, Harraway JA. 1999. Responses by Hector’s dolphins to boats and swimmers in Porpoise Bay, New Zealand. Marine Mammal Science 15: 738-750.
Bejder L, Samuels A, Whitehead H, Gales N. 2006a. Interpreting short-term behavioural responses to disturbance within a longitudinal perspective. Animal Behaviour 72: 1149-1158.
Bejder, L., Samuels, A., Whitehead, H., Gales, N., Mann, J., Connor, R.C., Heithaus, M.R., Watson-Capps, J.J., Flaherty, C., and Krützen, M. 2006. Decline in relative abundance of bottlenose dolphins exposed to long-term disturbance. Conservation Biology 20: 1791-1798.
Best, P.B. 2007. Whales and Dolphins of the Southern African Subregion. Cambridge University Press, Cape Town, South Africa.
Borrell A, Aguilar A. 2005. Differences in DDT and PCB residues between common and striped dolphins from the southwestern Mediterranean. Archives of Environmental Contamination and Toxicology 48:501-8.
Branstetter BK, Bowman VF, Houser DS, Tormey M, Banks P, Finneran JJ, Jenkins K. 2018. Effects of vibratory pile driver noise on echolocation and vigilance in bottlenose dolphins (Tursiops truncatus). The Journal of the Acoustical Society of America 143(1):429-39.
Brotons, J.M., Grau, A. and Rendell, L. 2008. Estimating the impact of interactions between bottlenose dolphins and artisanal fisheries around the Balearic Islands. Marine Mammal Science 24(1): 112-127.
Buckstaff KC. 2004. Effects of watercraft noise on the acoustic behavior of bottlenose dolphins, Tursiops truncatus, in Sarasota Bay, Florida. Marine Mammal Science 20: 709-725.
Buscaino, G., Buffa, G., Sara, G., Bellante, A., Tonello, Jr. A.J., Hardt, F.A.S., Cremer, M.J., Bonanno, A., Cuttitta, A. and Mazzola, S. 2009. Pinger affects fish catch efficiency and damage to bottom gill nets related to bottlenose dolphins. Fisheries Science 75: 537-544.
Celliers L, Schleyer MH. 2002. Coral bleaching on high-latitude marginal reefs at Sodwana Bay, South Africa. Marine Pollution Bulletin 44(12):1380-7.
Cockcroft, V.G., and Ross, G. J. B. 1990. Age, growth and reproduction of bottlenose dolphins Tursiops truncatus from the east coast of southern Africa. Fishery Bulletin 88: 289-302.
Connor, R. C., Wells, R. S., Mann, J., and Read, A. J. 2000. The bottlenose dolphin: social relationships in a fission-fusion society. In: J. Mann, R. C. Connor, P. L. Tyack and H. Whitehead (eds), Cetacean societies: field studies of dolphins and whales, pp. 91-126. University of Chicago Press, Chicago.
Constantine R. 2001. Increased avoidance of swimmers by wild bottlenose dolphins (Tursiops truncatus) due to long-term exposure to swim-with-dolphin tourism. Marine Mammal Science 17: 689-702.
Constantine, R., Brunton, D. H., and Dennis, T. 2004. Dolphin-watching tour boats change bottlenose dolphin (Tursiops truncatus) behaviour. Biological Conservation 117: Biological Conservation.
Cox TM, Read AJ, Swanner D, Urian K, Waples D. 2003. Behavioral responses of bottlenose dolphins, Tursiops truncatus, to gillnets and acoustic alarms. Biological Conservation 115: 203-212.
Curry, B. E. and Smith, J. 1997. Phylogeographic structure of the bottlenose dolphin (Tursiops truncatus): stock identification and implications for management. In: A. E. Dizon, S. J. Chivers and W. F. Perrin (eds), Molecular genetics of marine mammals, pp. 227-247. The Society of Marine Mammalogy, Allen Press, Lawrence.
Dans SL, Crespo EA, Pedraza SN, Degrati M, Garaffo GV. 2008. Dusky dolphin and tourist interaction: effect on diurnal feeding behavior. Marine Ecology Progress Series 369: 287-296.
Dawson S, Barlow J, Ljungblad D. 1998. Sounds recorded from Baird’s beaked whale, Berardius bairdii. Marine Mammal Science 14: 335-344.
Dwyer SL, Kozmian-Ledward L, Stockin KA. 2014. Short-term survival of severe propeller strike injuries and observations on wound progression in a bottlenose dolphin. New Zealand Journal of Marine and Freshwater Research 48(2):294-302.
Erbe C, Wintner S, Dudley SF, Plön S. 2016. Revisiting acoustic deterrence devices: Long-term bycatch data from South Africa’s bather protection nets. In: Proceedings of Meetings on Acoustics (Vol. 27, No. 1). AIP Publishing.
Findlay KP, Best PB, Ross GJB, Cockcroft VG. 1992. The distribution of small odontocete cetaceans off the coasts of South Africa and Namibia. South African Journal of Marine Science 12: 237-270.
Finley, K. J., Miller, G. W., Davis, R. A. and Greene, C. R. 1990. Reactions of belugas Delphinapterus leucas and narwhals Monodon monoceros to ice-breaking ships in the Canadian High Arctic. Canadian Bulletin of Fisheries and Aquatic Sciences 224: 97–117.
Finneran JJ, Schlundt CE, Carder DA, Clark JA, Young JA, Gaspin JB, Ridgway SH. 2000. Auditory and behavioral responses of bottlenose dolphins (Tursiops truncatus) and a beluga whale (Delphinapterus leucas) to impulsive sounds resembling distant signatures of underwater explosions. The Journal of the Acoustical Society of America 108: 417-431.
Gao A, Zhou K, Wang Y. 1995. Geographical variation in morphology of bottlenose dolphins (Tursiops spp.) in Chinese waters. Aquatic Mammals 21: 121-135.
Hale, P. T., Barreto, A. S., and Ross, G. J. B. 2000. Comparative morphology and distribution of the aduncus and truncatus forms of bottlenose dolphin Tursiops in the Indian and Western Pacific Oceans. Aquatic Mammals 26: 101-110.
Hammond PS, Bearzi G, Bjørge A, Forney KA, Karkzmarski L, Kasuya T, Perrin WF, Scott MD, Wang JY, Wells RS, Wilson B. 2012. Tursiops truncatus. The IUCN Red List of Threatened Species 2012: e. T22563A17347397.
Hoelzel AR, Potter JW, Best P. 1998. Genetic differentiation between parapatric “nearshore” and “offshore” populations of the bottlenose dolphin. Proceedings of the Royal Society of London B 265: 1177-1183.
Hutchings L, Van der Lingen CD, Shannon LJ, Crawford RJ, Verheye HM, Bartholomae CH, Van der Plas AK, Louw D, Kreiner A, Ostrowski M, Fidel Q. 2009. The Benguela Current: An ecosystem of four components. Progress in Oceanography 83(1-4):15-32.
IUCN (International Union for Conservation of Nature). 2012. Tursiops truncatus. The IUCN Red List of Threatened Species. Version 3.1. Available at: http://www.iucnredlist.org. Downloaded on 21 February 2016.
James NC, Whitfield AK, Harrison TD. 2016. Grey mullet (Mugilidae) as possible indicators of global warming in South African estuaries and coastal waters. Marine Environmental Research 122:188-95.
Janik VM, Thompson PM. 1996. Changes in surfacing patterns of bottlenose dolphins in response to boat traffic. Marine mammal science 12: 597-602.
Jarre A, Hutchings L, Kirkman SP, Kreiner A, Tchipalanga PC, Kainge P, Uanivi U, van der Plas AK, Blamey LK, Coetzee JC, Lamont T. 2015. Synthesis: climate effects on biodiversity, abundance and distribution of marine organisms in the Benguela. Fisheries Oceanography 24:122-49.
Koper RP, Plön S. 2012. The potential impacts of anthropogenic noise on marine animals and recommendations for research in South Africa. EWT Research & Technical Paper No. 1. Endangered Wildlife Trust, South Africa.
Kragh IM, McHugh K, Wells RS, Sayigh LS, Janik VM, Tyack PL, Jensen FH. 2019. Signal-specific amplitude adjustment to noise in common bottlenose dolphins (Tursiops truncatus). Journal of Experimental Biology 222(23):jeb216606.
Kriesell HJ, Elwen SH, Nastasi A, Gridley T. 2014. Identification and characteristics of signature whistles in wild bottlenose dolphins (Tursiops truncatus) from Namibia. PloS One 9: e106317.
Kruse S. 1991. The interactions between killer whales and boats in Johnstone Strait, B.C. In: Pryor K, Norris KS (ed.), Dolphin Societies: Discoveries and Puzzles, pp. 149-159. University of California Press, Berkeley, CA, USA.
Lauriano G, Fortuna CM, Moltedo G, Notarbartolo di Sciara G. 2004. Interactions between common bottlenose dolphins (Tursiops truncatus) and the artisanal fishery in Asinara Island National Park (Sardinia): assessment of catch damage and economic loss. Journal of Cetacean Research and Management 6: 165-173.
Leatherwood S. 1975. Some observations of feeding behavior of bottle-nosed dolphins (Tursiops truncatus) in the northern Gulf of Mexico and (Tursiops cf. T. gilli) off southern California, Baja California, and Nayarit, Mexico. Marine Fisheries Review 37: 10-16.
Lusseau, D. 2005. Residency pattern of bottlenose dolphins (Tursiops spp.) in Milford Sound, New Zealand, is related to boat traffic. Marine Ecology Progress Series 295: 265-272.
Mead A, Griffiths CL, Branch GM, McQuaid CD, Blamey LK, Bolton JJ, Anderson RJ, Dufois F, Rouault M, Froneman PW, Whitfield AK. 2013. Human-mediated drivers of change—impacts on coastal ecosystems and marine biota of South Africa. African Journal of Marine Science 35(3):403-25.
Moloney, C.L., Fennessey, S.T., Gibbons, M.J., Roychoudhury, A., Shillington, F.A. von der Heyden, B.P. and Watermeyer, K. 2013. Reviewing evidence of marine ecosystem change off South Africa. African Journal of Marine Science 35(3): 427-448.
Morgan MA, Patton GW. 1990. Human dolphin interactions on the west coast of Florida: Documentation from MML’s Marine Mammal Response Program. Mote Marine Laboratory Technical Report 191: 1-9.
Neri A, Sartor P, Voliani A, Mancusi C, Marsili L. 2022. Diet of bottlenose dolphin, Tursiops truncatus (Montagu, 1821), in the northwestern Mediterranean Sea. Diversity 15(1):21.
Nowacek SM, Wells RS, Solow AR. 2001. Short-term effects of boat traffic on bottlenose dolphins, Tursiops truncatus, in Sarasota Bay, Florida. Marine Mammal Science 17: 673-688.
Nowacek, D.P., Thorne, L.H., Johnston, D.W. and Tyack, P.L. 2007. Responses of cetaceans to anthropogenic noise. Mammal Review 37(2): 81-115.
Peters KJ, Parra GJ, Skuza PP, Möller LM. 2013. First insights into the effects of swim-with-dolphin tourism on the behavior, response, and group structure of southern Australian bottlenose dolphins. Marine Mammal Science 29: e484-e497.
Powell, J.R., and Wells, R.S. 2011. Recreational fishing depredation and associated behaviors involving common bottlenose dolphins (Tursiops truncatus) in Sarasota Bay, Florida. Marine Mammal Science 27: 111-129.
Reeves, R.R., McClellan, K. and Werner, T.B. 2013. Marine mammal bycatch in gillnet and other entangling net fisheries, 1990 to 2011. Endangered Species Research 20: 71-97.
Rocha D, Marley SA, Drakeford B, Potts J, Gullan A. 2023. Effects of dolphin-swim activities on the behaviour of an Indo-Pacific bottlenose dolphin population off the south coast of Mozambique. Biological Conservation 279:109949.
Ross GJB, Cockcroft VG. 1990. Comments on Australian bottlenose dolphins and the taxonomic status of Tursiops aduncus (Ehrenberg, 1832). In: Leatherwood S, Reeves RR (ed.), The Bottlenose Dolphin, pp. 101-128. Academic Press, New York, USA.
Ross GJB. 1977. The taxonomy of the bottlenosed dolphins, Tursiops species in South African waters, with notes on their biology. Annals of the Cape Provincial Museums. Natural History 11: 135-194.
Ross, G. J. B. 1984. The smaller cetaceans of the south east coast of southern Africa. Annals of the Cape Provincial Museums (Natural History) 15: 173-410.
Segura, I., Rocha-Olivares, A., Flores-Ramírez, S., & Rojas-Bracho, L. 2006. Conservation implications of the genetic and ecological distinction of Tursiops truncatus ecotypes in the Gulf of California. Biological Conservation, 133(3), 336-346.
Sekiguchi, K., Klages, N.T.W. and Best, P.B. 1992. Comparative analysis of the diets of smaller odontocete cetaceans along the coast of southern Africa. South African Journal of Marine Science 12: 843-861.
Sellas, A. B., Wells, R. S., & Rosel, P. E. 2005. Mitochondrial and nuclear DNA analyses reveal fine scale geographic structure in bottlenose dolphins (Tursiops truncatus) in the Gulf of Mexico. Conservation Genetics, 6, 715-728.
Sigler MF, Lunsford CR, Straley JM, Liddle JB. 2008. Sperm whale depredation of sablefish longline gear in the northeast Pacific Ocean. Marine Mammal Science 24: 16-27.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stensland E, Berggren P. 2007. Behavioural changes in female Indo-Pacific bottlenose dolphins in response to boat-based tourism. Marine Ecology Progress Series 332: 225-234.
Sweijd NA, Smit AJ. 2020. Trends in sea surface temperature and chlorophyll-a in the seven African Large Marine Ecosystems. Environmental Development 36:100585.
Taylor, B.L., Chivers, S.J., Larese, J. and Perrin, W.F. 2007. Generation length and percent mature estimates for IUCN assessments of cetaceans. NOAA, Southwest Fisheries Science Center Administrative Report LJ-07-01. La Jolla, California.
Wells, R. S. and Scott, M. D. 1999. Bottlenose dolphin Tursiops truncatus (Montagu, 1821). In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Vol. 6: The second book of dolphins and the porpoises, pp. 137-182. Academic Press, San Diego, CA, USA.
Yordy JE, Pabst D, McLellan WA, Wells RS, Rowles TK, Kucklick JR. 2010a. Tissue-specific distribution and whole-body burden estimates of persistent organic pollutants in the bottlenose dolphin (Tursiops truncatus). Environmental Toxicology and Chemistry 29: 1263-1273.
Yordy JE, Wells RS, Balmer BC, Schwacke LH, Rowles TK, Kucklick JR. 2010b. Life history as a source of variation for persistent organic pollutant (POP) patterns in a community of common bottlenose dolphins (Tursiops truncatus) resident to Sarasota Bay, FL. Science of the Total Environment 408: 2163-2172.
van Waerebeek K, Baker AN, Félix F, Gedamke J, Iñiguez M, Sanino GP, Secchi E, Sutaria D, van Helden A, Wang Y. 2007. Vessel collisions with small cetaceans worldwide and with large whales in the Southern Hemisphere, an initial assessment. Latin American Journal of Aquatic Mammals 6: 43-69.
