
Cohen’s Horseshoe Bat
Rhinolophus cohenae

2025 Red list status
Vulnerable
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Rhinolophus cohenae – Taylor, Stoffberg, Monadjem, Schoeman, Bayliss & Cotterill, 2012
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – cohenae
Common Names: Cohen’s Horseshoe Bat (English)
Synonyms: No Synonyms
Taxonomic Note:
In previous Red List assessments (2008), this species was included within Rhinolophus hildebrandtii. Taxonomic changes, based on differences in morphology, echolocation call frequencies, biogeography and some support through molecular systematics, have taken place within the R. hildebrandtii complex. R. hildebrandtii was revised in 2012 (Taylor et al. 2012) and R. cohenae recognised as one of the four newly described species from this species complex.
Red List Status: VU – Vulnerable, C2a(ii) + D1 (IUCN version 3.1)
Assessment Information
Assessors: Cohen, L.1, Taylor, P.2, Balona, J.3 & da Silva, J.M.4
Reviewers: Howard, A.2 & Bastian, A.5
Institutions: 1Mpumalanga Tourism and Parks Agency, 2University of the Free State, 3Gauteng and Northern Regions Bat Interest Group, 4South African National Biodiversity Institute, 5University of KwaZulu-Natal
Assessors and Reviewers: Cohen, L., Taylor, P., Jacobs, D., Kearney, T., MacEwan, K., Monadjem, A., Richards, L.R., Schoeman, C. & Sethusa, T.
Previous Contributors: Roxburgh, L., Raimondo, D. & Child, M.F.
Assessment Rationale
This species is endemic to South Africa and known from the Mpumalanga Escarpment, and from a few records in the south-east of Limpopo Province with an estimated extent of occurrence of 12,393 km². There are inferred to be fewer than 1,000 mature individuals (and certainly fewer than 10,000) in the population. Colonies are usually small, numbering only a few individuals. The greatest number of mature individuals counted at a single site was ± 40. All recorded colonies are suspected to comprise the same subpopulation. An ongoing decline is inferred to be taking place as a result of loss of habitat due to poor land-use management practices, mining activities, agricultural intensification as well as infestation by alien invasive plant species. Further field surveys and vetting of museum records are needed to more accurately delimit the distribution range of the species. Currently, we list the species as Vulnerable under criterion C2a(ii) and D1.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Cohen L, Taylor P, Balona J & da Silva JM. 2025. A conservation assessment of Rhinolophus cohenae. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species has been recorded so far only from the two northernmost provinces of South Africa making it an endemic species. Known from the Mpumalanga escarpment from Mariepskop, Abel Erasmus Pass to Barberton with the southernmost known locality between Badplaas and Machadodorp, its distribution falls within an elevational range of 457 m to 1,698 m asl. As such, the main centre of its geographical range currently lies in the Mpumalanga Province with distribution just crossing into the Limpopo Province. The type locality for this species is from the Barberton Mountainlands Nature Reserve in Barberton, Mpumalanga Province at an elevation of 690 m asl (Taylor et al. 2012). This species appears to inhabit the Grassland and Savannah Biomes and further surveys and specimen reappraisal of existing museum material, previously referred to as R. hildebrandtii (ACR 2024), will probably reveal a wider distribution range.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 739 m asl (Mondajem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,431 m asl (Mondajem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
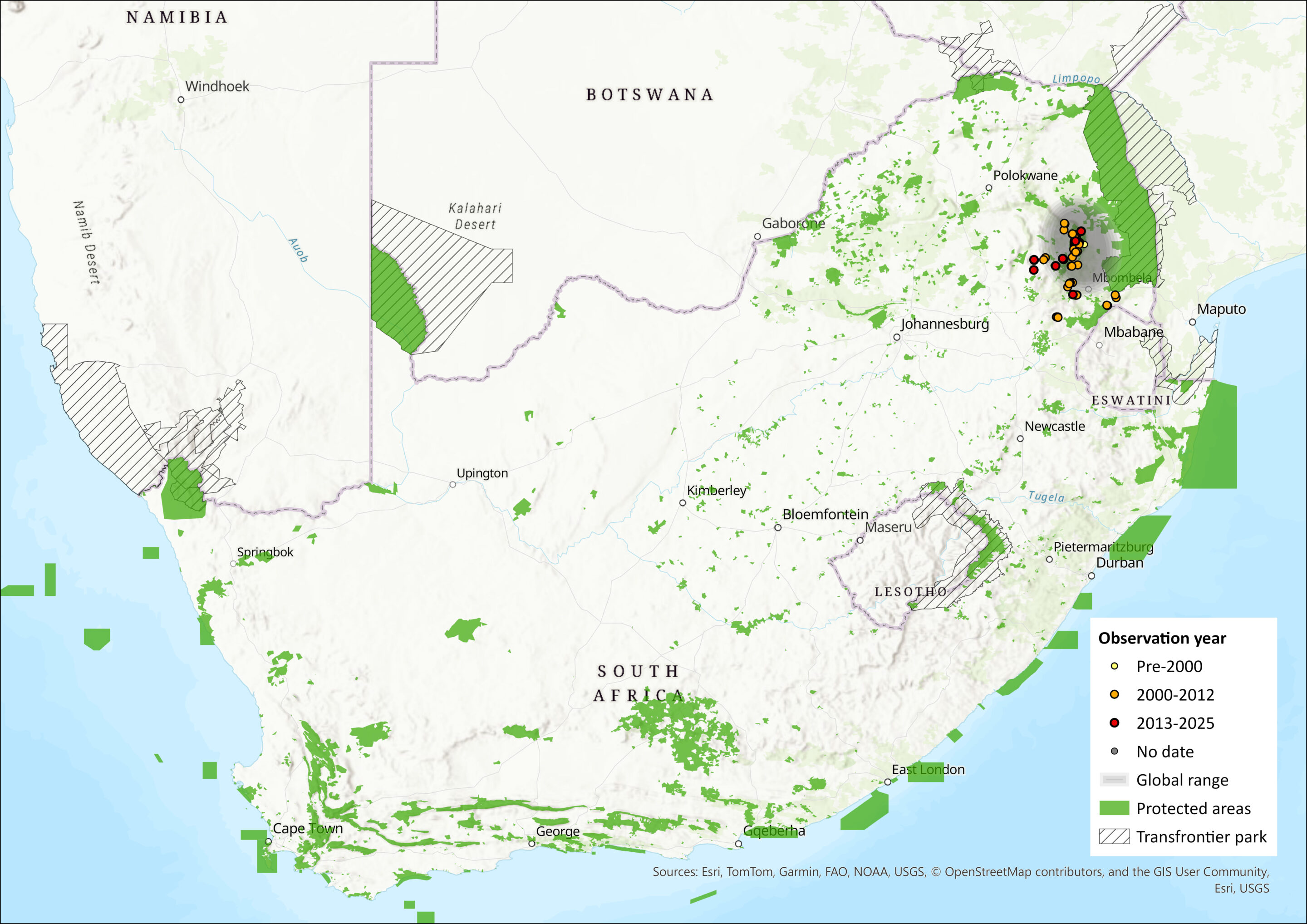 Figure 1. Distribution records for Cohen’s Horseshoe Bat (Rhinolophus cohenae) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Cohen’s Horseshoe Bat (Rhinolophus cohenae) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change may influence micro-climate distribution. This species is very dependent on suitable subterranean environments for roosting and maternity requirements as well as on associated natural habitats for foraging (Cohen et al. 2017). These sites are limited throughout its distribution range and beyond. The effects of climate change can severely impact on the survival of this species if the above is not provided for and not adequate for habitation anymore.
Population information
In total, 240 individuals have been counted in surveys, but this is an underestimate. The total population is thus inferred to be fewer than 1,000 mature individuals and thus certainly fewer than 10,000 mature individuals, as this species is encountered in small groups of which around 40 individuals was the highest number counted at a single site. All recorded colonies are suspected to be part of one subpopulation.
Current population trend: Declining, inferred.
Continuing decline in mature individuals: Yes, inferred.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: Yes
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Please refer to section on Population. No specific counts have been conducted since the last assessment, and no population genetic studies has yet been conducted on this species.
Habitats and ecology
The species mainly occurs in the Mesic Highveld Grassland Bioregion but also occurs in the Lowveld Bioregion and the Central Bushveld Bioregion of South Africa. Key roosting sites include subterranean habitats like caves and old mine adits. They have been recorded day-roosting in rock crevices and fissures and are occasionally observed in old buildings. Thus, its occurrence is in most cases subject to suitable sheltered and/or subterranean habitats. Regions with rock habitats that form cavities are of utmost importance for the survival of this species. Artificially created habitat such as abandoned mine or prospecting shafts/tunnels within various vegetation types also serve as important/essential habitat sanctuaries. Rhinolophus cohenae requires suitable natural habitat outside roost sites for foraging. Individuals are normally encountered in very small groups of which around 40 was the highest number counted at one locality consisting of a maze of old mine tunnels. No reproductive information is known of this species (Monadjem et al. 2020).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.2. Savanna -> Savanna – Moist | Resident | Suitable | Yes |
| 4.4. Grassland -> Grassland – Temperate | Resident | Suitable | Yes |
| 6. Rocky areas (e.g. inland cliffs, mountain peaks) | Resident | Suitable | Yes |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | Resident | Suitable | Yes |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified) Unknown
Reproductive Periodicity: Unknown
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: Unknown
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The Mpumalanga Tourism and Parks Agency (MTPA) mapped all development applications received at a cadastral scale over a 14-year period (2000-2014), which showed that greatest pressure for land-use change has come from prospecting applications (54% of the land surface area) and mining (25% of land surface area) (Lötter et al. 2014). A major threat within this species’ range is mining (legal, illegal and recommissioning of old mines). Future developments at the above rates or even higher are likely to cause further detriment towards natural ecosystems and processes and in particular, disturb or destroy foraging grounds and roosting and maternity sites, or alter key micro-climates needed by the species. Additionally, loss of natural habitat around roost sites through poor land-use management practices, (such as inappropriate burning regimes, overgrazing and alteration of vegetation structure negatively affect foraging areas and prey base), land development activities including agricultural intensification (Driver et al. 2012), and alien invasive plant infestations are causing a decline in available habitat for foraging.
Conservation
The species occurs in the Blyderivierspoort Nature Reserve and Barberton Mountainlands Nature Reserve. No specific conservation actions are directed towards this species at the moment. The MTPA has developed the Mpumalanga Biodiversity Sector Plan (MBSP) that indicates areas of high conservation value and is based on a systematic conservation plan which considers the distribution of all species and their habitat, sets quantitative targets for these and tries to find the most sufficient selection of areas to meet these targets. A few R. cohenae‘s localities fall within the boundaries of protected areas but most are situated on private land. The MBSP has categorised areas in term of its biodiversity value and R. cohenae localities located within the Protected Area and Critical Biodiversity and Ecological Support Areas will potentially receive the best protection measures from a land development perspective where certain activities will not be allowed or be restricted. With regards to all other areas, the MBSP land-use guidelines should also be followed and Environmental Impact Assessment legislative tools applied.
Recommendations for land managers and practitioners:
- Landowners should implement best landuse management practices to maintain sustainability and limit disturbance at roosting sites. To achieve this, ecological advisory services and training are needed.
Research priorities:
- Further taxonomic research is required as the species status has been challenged (Demos et al. 2019) on the grounds of <1% genetic differences in the mitochondrial marker in Taylor et al. (2012); see also Monadjem et al. (2020). Inclusion of nuclear DNA and non-coding markers is recommended.
- Systematic surveys to identify further colonies and assess population size and trend.
- Field surveys and niche modelling to more accurately delimit distribution range, and to identify key roost sites for protection.
- Further vetting of R. hildebrandtii specimens to more accurately delimit distribution range.
- Investigating patterns of movement to establish level of demographic and genetic exchange between colonies and quantifying the effects on transformation/fragmentation on such processes.
Encouraged citizen actions:
- Minimise disturbance to caves when visiting.
- Avoid or limit the use of pesticides/insecticides for agricultural and household purposes as this can potentially negatively affect the prey base and lead to poisoning of individuals of this species and bats in general.
- Deposit any dead specimens at your local conservation agency for identification.
- As this species occurs in urban and rural areas, citizens can report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
ACR. 2024. African Chiroptera Report 2013. AfricanBats, African Chiroptera Project, i-xix. Pretoria.
Avenant, N., MacEwan, K., Balona, J., Cohen, L., Jacobs, D., Monadjem, A., Richards, L., Schoeman, C., Sethusa, T. and Taylor, P. 2016. A conservation assessment of Cistugo lesueuri. In: M.F. Child, L. Roxburgh, E. Do Linh San, D. Raimondo, J., Selier & H.T. Davies-Mostert (ed.), The Red List of Mammals of South Africa, Swaziland and Lesotho, South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Demos, T.C., Webala, P.W., Goodman, S.M., Kerbis Peterhans, J.C., Bartonjo, M. and Patterson, B.D. 2019. Molecular phylogenetics of the African horseshoe bats (Chiroptera: Rhinolophidae): expanded geographic and taxonomic sampling of the Afrotropics. BMC Evolutionary Biology, 19(1), p.166.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-2. Available at: www.iucnredlist.org. (Accessed: 14 September 2017).
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Lötter, M.C. 2015. Technical Report for the Mpumalanga Biodiversity Sector Plan – MBSP. Mpumalanga Tourism & Parks Agency, Mbombela.
Lötter, M.C., Cadman, M.J. and Lechmere-Oertel, R.G. 2014. Mpumalanga Biodiversity Sector Plan Handbook. MTPA. Nelspruit.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Taylor, P.J., Stoffberg, S., Monadjem, A., Schoeman, M.C., Bayliss J. and Cotterill, F.P.D. 2012. Four new bat species (Rhinolophus hildebrandtii complex) reflect Plio-Pleistocene divergence of dwarfs and giants across an Afromontane archipelago. PLoS ONE 7(9): e41744. doi:10.1371/journal.pone.0041744.
