Chestnut Climbing Mouse
Dendromus mystacalis

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Dendromus mystacalis s.l.- (Heuglin, 1863)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – NESOMYIDAE – Dendromus – mystacalis
Common Names: Chestnut African Climbing Mouse, Chestnut Climbing Mouse (English), Roeskleurklimmuis (Afrikaans)
Synonyms: Dendromys mystacalis von Heuglin, 1863; Dendromus ansorgei Thomas & Wroughton, 1905; Dendromus acraeus Wroughton, 1909; Dendromus jamesoni Wroughton, 1909; Dendromus whytei Wroughton, 1909; Dendromus nairobae Osgood, 1910; Dendromus ochropus Osgood, 1910; Dendromus whytei pallescens Osgood, 1910; Dendromus lineatus Heller, 1911; Dendromus whytei capitis Heller, 1912; Dendromus jamesoni pongolensis Roberts, 1931; Dendromus pumilio uthmoelleri Bohmann, 1939; Dendromus pumilio lineatus Setzer, 1956; Dendromus mystacalis ansorgei Hayman, 1963. According to the Mammal Diversity Database (2025), the taxonomic status of all other names historically associated with this species, currently retained under it on a provisional basis, remains unresolved, except for D. lineatus, which is formally recognised as a synonym of D. mystacalis.
Taxonomic Note: This widespread species complex includes many named subspecies with unconfirmed relationships between the groups (Monadjem et al. 2015). Chromosomal analysis by Solano et al. (2014) revealed potential species splits, and additional research was required to clarify their taxonomic relationships. Recent phylogenetic analyses of the genus Dendromus have led to a significant revision of the known distribution of D. mystacalis. Voelker et al. (2021) and more recently Mulualem et al. (2024) demonstrated populations previously assigned to D. mystacalis across sub-Saharan Africa represent distinct lineages, with D. mystacalis s.s. (type locality: Baschlo region, Ethiopia) reportedly geographically restricted to the Ethiopian Highlands. This necessitates a re-evaluation of all historical records of D. mystacalis from assessment region, as the evolutionary history and phylogenetic status of populations within southern Africa remain unresolved. In the absence of confirmed taxonomic assignment, the population/s within the assessment region are referenced as D. mystacalis s.l..
Historically, two subspecies were recognised within the southern African subregion (Meester et al. 1986). Dendromus m. jamesoni Wroughton, 1909, exhibited a broad distribution extending from the Western Cape across the eastern and northeastern regions of South Africa (including Eswatini), and reaching the southern parts of Mozambique and Zimbabwe. Dendromus m. whytei Wroughton, 1909 was reported from Botswana, with the type locality being Malawi.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1 Naidoo, T.1 & da Silva, J.M.2
Reviewer: Patel, T.3
Institutions:1Durban Natural Science Museum, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust
Previous Assessors & Reviewers: MacFadyen, D., Shoeman, C. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern as this species of Climbing Mouse is widespread across the eastern regions of the assessment region, occurs in multiple protected areas (including Kruger National Park), is common and sampled consistently in different vegetation types and has a documented recent range expansion in the North West Province. It exhibits a degree of habitat tolerance, and there are no known threats that could cause widespread population decline. However, the impacts of grassland loss on this species should be monitored, particularly land cover change from mining and agriculture synergising with climate change. Low numbers were recorded in the Highveld Grasslands of Gauteng and Mpumalanga provinces. This species should be reassessed following taxonomic resolution.
Regional population effects: This species is expected to disperse from Mozambique, Zimbabwe and Botswana into South Africa. This species may disperse where suitable habitat of coarse-grassed grassland is continuous.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR, Naidoo T & da Silva JM. 2025. A conservation assessment of Dendromus mystacalis s.l.. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
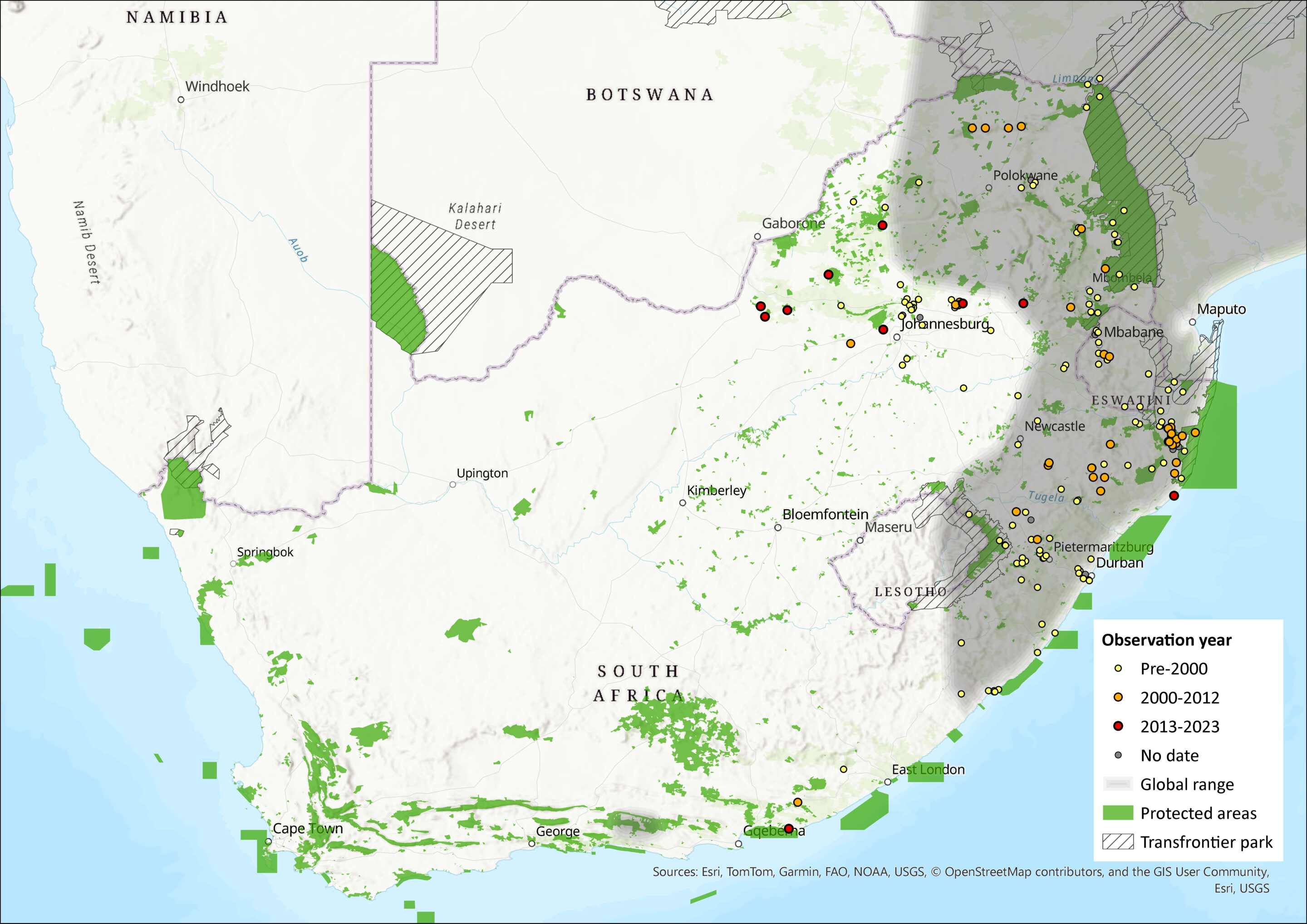
This species is widely, but discontinuously, distributed in low-lying savannah and grasslands (typically below 1,000 m) of southern and East Africa (Monadjem et al. 2015). Previously, it was reported to occur from Sudan and Ethiopia southwards to Kenya and Tanzania, then westwards to the Democratic Republic of the Congo, Ghana, Angola and Nigeria. Further south its range extends into Zimbabwe, Zambia, Malawi, parts of Mozambique and extensively across eastern South Africa. However, recent phylogenetic and integrative taxonomic studies recognise populations from Ethiopia, South Sudan and Ethiopia as D. mystacalis sensu stricto; the taxonomic affiliation of individuals from other countries, including countries in eastern and southern Africa, remain uncertain. As such, the precise limits of its distribution are unknown, and further taxonomic resolution will assist in delimiting its distributional range.
Within the assessment region, D. mystacalis s.l. appears to be confined to the eastern and northeastern parts of country, including parts of the Eastern Cape, KwaZulu-Natal, Gauteng, Mpumalanga, and Limpopo provinces (Skinner & Chimimba 2005). Additionally, there has been a recent range expansion westwards into the North West Province, as Power (2014) confirms that specimens recently collected from wetlands in the Marico and Molopo rivers are the most westerly recorded (see Newbery 1995). In Gauteng and Mpumalanga provinces this species was recently recorded in low numbers in Rocky Highveld Grassland and Moist Degraded Grassland on Ezemvelo Nature Reserve (Gauteng) and Telperion Nature Reserve (Mpumalanga) (MacFadyen 2014).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 1500 m asl
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
Figure 1. Distribution records for Chestnut Climbing Mouse (Dendromus mystacalis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
The distributional range of the taxon remains uncertain, pending confirmation of taxonomic assignment of individuals from the southern African subregion. The below table provides the historical distribution of Dendromus mystacalis sensu lato.
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Possibly Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
To date, no formal studies have examined the potential impacts of climate change, along with associated bioclimatic and environmental shifts, on the biology, ecology, or physiology of this species. Although the species is found across a variety of habitats, it is more commonly associated with grass-dominated landscapes (Skinner and Chimimba 2005). Midgeley and Thuiller (2010) predicted a 30% replacement of grassland-dominated habitats by more arid-adapted or tolerant vegetation types (e.g. thorn-scrub savanna). This may result in the displacement or range contraction of grassland-associated species, that may include Dendromus Mystcalis s.l. within the assessment region. Rodent population declines tend to be more pronounced in habitats experiencing significant anthropogenic transformation or, alternatively, in high-altitude environments where the effects of climate change are likely to be intensified (Wan et al. 2022). Further research is required to assess the potential impacts of climate change on this species within the assessment region.
Population
This species is widespread across its range but is not usually collected in large numbers. Low capture success may be attributable to its small size that makes capture using traditional rodent live-traps difficult. Population numbers tend to fluctuate with reported low abundance during the austral wet season, and peaks in population numbers coinciding with the drier winter months (Monadjem 2013). Similarly, higher numbers have been recorded in autumn on the Highveld (MacFadyen 2014). Suitable habitat is often fragmented, and the population is concentrated towards the north and eastern parts of the country. MacFadyen (2014) estimated the population of this species in Rocky Highveld Grassland at 2 animals / ha, with a lower density of 1 animal / ha recorded in Moist Degraded Grassland. Monadjem (2013) reported similar densities from Eswatini (2.5 individuals / ha). Additionally, it was trapped by Rautenbach et al. (2014) at Phinda Private Game Reserve in a variety of habitats, and by Avenant and Kuyler (2002) in a cultivated area at Maguga Dam, Eswatini. These results are an indication that this species has the ability to inhabit a range of habitat types.
Population Information
Current population trend: Unknown, pending phylogenetic and phylogeographic studies
Continuing decline in mature individuals: Cannot be assessed/determined given the uncertainty concerning the taxonomy and distributional limits of the species.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: Unknown, pending phylogenetic and phylogeographic studies
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown, pending phylogenetic and phylogeographic studies
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Cannot be assessed/determined given the uncertainty concerning the taxonomy and distributional limits of the species.
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
A recent phylogenetic investigation of the genus Dendromus revealed D. mystacalis to have a significantly restricted distribution than previously recognised (Voelker et al. 2021, Mulualem et al. 2024). Based on the recent phylogentic studies, the species appears to be restricted to the Ethiopian Highlands, and therefore may not present within the assessment region. Further studies are required to resolve the sub-Saharan taxonomy, distribution and fine scale genetics.
Habitats and ecology
Found within grassland and savannah mosaic habitats, where it prefers tall, rank grassland; but can also inhabit riparian forests, Afromontane forests, Sand Forest, wetlands, drainage lines and thickets (Monadjem 2013; Rautenbach et al. 2014; Delcros et al. 2015). In the North West Province, Power (2014) collected three specimens in wetlands of the dolomitic eyes of both the Marico and Molopo rivers (in Phragmites australis reedbeds), and elsewhere at Batlako vlei in the Pilanesberg uplands, which represents new information on their use of wetland habitats. MacFadyen (2014) recorded individuals in short grass in moist degraded grassland at Telperion Nature Reserve (Mpumalanga) and Ezemvelo Nature Reserve (Gauteng). Although preferring tall, rank grassland, this species is also recorded in areas with a low basal cover, and does not appear to have any particular key vegetation type. Occupancy is predicted to increase with increasing grass biomass (Loggins et al. 2019). Monadjem (1998) recorded the species from “hilly landscapes” in Eswatini.
Interestingly, it was sampled, together with Suncus infinitesimus (together the two most uncommon species sampled), in Kikuyu (Pennisetum clandestinum) pastures in Umvoti Vlei Conservancy, KwaZulu-Natal Province, despite this habitat having low small mammal abundance overall (Fuller & Perrin 2001). Suspected to forage in pairs, as two individuals were routinely captured in the same rodent live-trap during sampling sessions in Eswatini (Monadjem et al. 2013).
This species is omnivorous. It will feed on seeds and a wide range of invertebrates, including termites, crickets, moths and small beetles (Monadjem 1997). Like other species of climbing mice, this nocturnal and largely terrestrial species accesses seeds and invertebrates in low bushes and grass stalks, using its prehensile tail for balance while climbing. Similarly, like other climbing mice, it constructs nests with multiple entrances, in low bushes and trees up to approximately 1-2 m off the ground (Monadjem 2013). These nests are used during the warm, wet seasons, primarily for rearing young and resting (Skinner & Chimimba 2005). However, they may also occupy disused burrows and weaver nests (Monadjem 2013). The species is of similar size to Dendromus melanotis, differentiated by the bright chestnut colouration and also has a similar dark band running from behind the shoulders to the base of the tail. It is solitary or found in pairs or small family groups. Population numbers appear to peak during the austral winter and early spring months (June to October) (Monadjem and Perrin 2003; Macfadyen 2014).
Ecosystem and cultural services: There are no known cultural services or folklores associated with this species. Similar to other small mammals, this species plays a role in regulating invertebrate numbers, seed dispersal, nutrient cycling, and is an important prey species for predators such as Barn Owls (Tyto alba; Coetzee 1972) and Black-shouldered Kites (Elanus caeruleus; Mendelsohn 1982).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean total length = 14.8 cm (Rautenbach 1982)
Size at Maturity (in cms): Male: Mean total length = 14.9 cm (Rautenbach 1982)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Female maximum total length = 17.1 cm; Male maximum total length = 16.9 cm (Rautenbach 1982)
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Breeding appears to coincide with the austral summer, with young appearing between January to March each year (Smithers and Wilson 1979). Pregnant females have been recorded from the former Transvaal region in March (Rautenbach 1982)
Average Annual Fecundity or Litter Size: 3-8 pups within a litter (Rautenbach 1982; Skinner and Chimimba 2005).
Natural Mortality: Climbing mice, in general, are preyed upon by Barn Owls (Coetzee 1972; Durban Natural Science Museum specimen records), with D. mystcalis skeletal remains also reported from the regurgitated Black-winged Kite (formely Black-shouldered Kite) (Mendelsohn 1982).
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: The movement patterns of this species remain largely unknown, however, several studies have reported a ‘disappearance’ of the species from areas where they have been previously recorded coinciding with the onset of the austral wet season (e.g. Macfadyen 2014; G. Muller and T. Mathoko pers. comm.). This apparent absence may relate to a change in foraging behaviour rather than a true disappearance from the study area (Monadjem 2013).
Congregatory: Reported to forage in pairs (Monadjem 2013).
Systems
System: Terrestrial
General Use and Trade Information
There is no known subsistence or commercial use of this species.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? None reported
Harvest Trend Comments: (Not specified)
Threats
Currently no major threats have been identified for this species, but grassland habitat loss from agricultural expansion (especially monocultures), forestry and mining developments and loss of habitat quality from overgrazing and incorrect fire management may also cause local declines. Climate change is also expected to reduce Afromontane grassland, further jeopardising the species similar to other Afromontane species (Taylor et al. 2016). The impact of these threats on the population remains to be quantified.
Conservation
This species occurs in several protected areas within its range in the assessment region, including Kruger National Park, Vhembe Biosphere Reserve, iSimangaliso Wetland Park, Maloti-Drakensberg Park, Telperion Nature Reserve (Mpumalanga), and Ezemvelo Nature Reserve (Gauteng). No direct interventions are necessary but protected area expansion to incorporate grassland habitats would certainly benefit the species, specifically to create habitat linkages where connectivity between natural habitat patches has been lost. Planners should prioritise previously cultivated areas for development instead of remaining natural areas. For example, previously ploughed areas now left fallow make up 8.9% of the Grassland Biome in Mpumalanga, and these areas should be prioritised for further development (Lotter et al. 2014). Grassland restoration projects through post-mining rehabilitation programs are also likely to benefit this species. Grasslands are restored in a number of ways, most importantly, the ground should be contoured, top soil replaced, and grass seed sowed. Seeds were successfully sowed and grassland rehabilitated on old lands on Telperion Nature Reserve (D. MacFadyen unpubl. data). It is the mining and agricultural industries’ responsibility to rehabilitate mined and ploughed areas, respectively. Additionally, landowners are responsible for managing the land and vegetation, ensuring over-utilisation of grasses is avoided.
Recommendations for land managers and practitioners:
- Landowners should maintain and/or expand corridors of grassland to offset the impacts of urbanisation and ensure movement and gene flow between populations, especially corridors of natural grassland in agricultural and mining areas.
- Employ correct land management practices to prevent overgrazing and indiscriminate burning.
- Protected areas expansion through provincial biodiversity stewardship programmes.
- Reduce over-stocking on cattle and game ranches.
Research priorities:
- This species complex requires an urgent taxonomic revision; the inclusion of samples from eastern and southern Africa in further phylogenetic or integrative taxonomic studies will hopefully clarify the taxonomic affinity of these populations.
- Conduct phylogeographic and landscape genetic analyses to elucidate the genetic structure of animal populations within the assessment region.
- Land cover maps and climate-based modelling must be used to project future decline in the Grassland Biome and potential impact on the distributional range of the taxon within the assessment.
- Quantifying habitat preferences and understanding local movements and dispersal mechanisms between habitats; quantifying the species’ habitat under formal protection.
- Research into the reproductive biology and physiology of the species and the potential impacts of climate change associated extreme fluctuations in temperature and precipitation seasonality on the species.
- Research into the contribution of this species complex to ecosystem functioning.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Deceased specimens should be submitted to the nearest accredited museum or biological repository for proper documentation and preservation.
- Plant indigenous grasses in urban and peri-urban gardens.
- Reduce use of insecticides and herbicides as much as possible.
- Proclaim private land nature reserves/protected areas under government stewardship programmes.
Bibliography
Avenant NL, Kuyler P. 2002. Small mammal diversity in the Maguga Dam inundation area, Swaziland. South African Journal of Wildlife Research 32: 101-108.
Coetzee, C.G., 1972. The identification of southern African small mammal remains in owl pellets. Cimbebasia 2: 53-64.
G, Taylor PJ, Schoeman MC. 2015. Ecological correlates of small mammal assemblage structure at different spatial scales in the savannah biome of South Africa. Mammalia 79: 1–14.
Fuller JA, Perrin MR. 2001. Habitat assessment of small mammals in the Umvoti Vlei Conservancy, KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 31: 1–12.
Jewitt D, Goodman PS, Erasmus BFN, O’Connor TG, Witkowski ETF. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Loggins, A.A., Monadjem, A., Kruger, L.M., Reichert, B.E. and McCleery, R.A., 2019. Vegetation structure shapes small mammal communities in African savannas. Journal of Mammalogy, 100(4): 1243-1252.
Lötter MC, Cadman MJ, Lechmere-Oertel RG. 2014. Mpumalanga Biodiversity Sector Plan Handbook. Mpumalanga Tourism & Parks Agency, Mbombela, South Africa.
MacFadyen DN. 2014. The dynamics of small mammal populations in Rocky Highveld Grassland, Telperion, South Africa. Ph.D. Thesis. University of Pretoria, Pretoria, South Africa.
Mammal Diversity Database. 2025. Dendromus mystacalis. https://mammaldiversity.org/taxon/1003720 . Accessed 14 November 2025.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J., Baker, C.M. 1986. Classification of Southern African Mammals. Transvaal Museum Monographs, 5(1): 1-359.
Mendelsohn, J., 1982. The feeding ecology of the blackshouldered kite Elanus caeruleus (Aves: Accipitridae). Durban Museum Novitates, 13(8), pp.75-116.
Midgley, G.F. and Thuiller, W., 2011. Potential responses of terrestrial biodiversity in Southern Africa to anthropogenic climate change. Regional Environmental Change, 11(Suppl 1), pp.127-135.
Monadjem A. 1997. Stomach contents of 19 species of small mammals from Swaziland. South African Journal of Zoology 32: 23–26.
Monadjem A. 2013. Dendromus mystacalis Chestnut African Climbing Mouse. Pages 178–179 in Happold DCD, editor. Mammals of Africa. Volume III: Rodents, Hares and Rabbits. Bloomsbury Publishing, London, UK.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Newbery CH. 1995. Mammal Checklist of the Provincial Nature Reserves. North West Parks Board, Mafikeng, South Africa.
Mulualem G, Lavrenchenko, L.A, Kerbis Peterhans J.C, Mikula O, Bryjova A, Meheretu Y, Bryja J. 2024. Taxonomic revision and evolutionary history of the climbing mice in eastern Africa (Dendromus mystacalis clade): the role of elevation and geographical barriers in the speciation process. Organisms Diversity and Evolution 24: 573-598
Power RJ. 2014. The Distribution and Status of Mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Rautenbach, I.L. 1982. Mammals of the Transvaal. Ecoplan Monograph 1: 1-211.
Rautenbach A, Dickerson T, Schoeman MC. 2014. Diversity of rodent and shrew assemblages in different vegetation types of the savannah biome in South Africa: no evidence for nested subsets or competition. African Journal of Ecology 52: 30-40.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Solano E, Taylor PJ, Rautenbach A, Ropiquet A, Castiglia R. 2014. Cryptic speciation and chromosomal repatterning in the South African climbing mice Dendromus (Rodentia, Nesomyidae). PloS one 9: e88799.
Taylor PJ, Nengovhela A, Linden J, Baxter RM. 2016. Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia in press.
Voelker G, Huntley J.B, Denys C, Sumbera R, Demos T.C, Lavrenchenko L, Nicolas V, Gnoske T.P, Kerbis Peterhans, J.C. 2021. Molecualr systematics and biogeographic history of the African climbing-mouse complex (Dendromus). Molecular Phylogenetics and Evolution 161: 107166.
Wan, X., Yan, C., Wang, Z. and Zhang, Z. 2022. Sustained population decline of rodents is linked to accelerated climate warming and human disturbance. BMC Ecology and Evolution, 22(1), p.102.