Chacma Baboon
Papio ursinus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Papio ursinus – (Kerr, 1792)
ANIMALIA – CHORDATA – MAMMALIA – PRIMATES – CERCOPITHECIDAE – Papio – ursinus
Common Names: Chacma Baboon (English), Bobbejaan (Afrikaans), Ifene, Indwangula, yaKapa (Ndebele), Tshwêne (Sepedi, Sesotho, Setswana), Imfene (Swati, Xhosa, Zulu), Mfenha (Tsonga), Pfene (Venda) Babouin chacma (French), Papión Chacma (Spanish; Castilian)
Synonyms: Simia hamadryas ssp. ursinus Kerr, 1792
Taxonomic Note:
Although up to eight Chacma Baboon forms have been suggested in the literature (Hill 1970), only three are commonly accepted today (Jolly 1993, Groves 2001): Papio. ursinus. ursinus, P. u. griseipes, and P. u. ruacana. Papio u. ursinus, the typical Chacma, is a large baboon with black nape fringes, dark brown body hair, black hair on hands and feet, and a relatively short tail. This variant occurs in the more southern and western parts of the Chacma range, including South Africa and parts of Botswana. It includes ursinus, orientalis, and occidentalis subspecies as classified by Hill (1970). Papio ursinus griseipes, the Grey-footed Baboon, has a more fawn-coloured coat and is found in southwestern Zambia, Zimbabwe, Mozambique south of the Zambezi, parts of the Limpopo Province in South Africa, and the Okavango Delta in Botswana (Jolly 1993). This variant is smaller than P. u. ursinus and has grey hands and feet matching their limb colour, and a longer tail. Hill’s (1970) griesipes, ngamiensis, chobiensis and jubilaeus subspecies are included. Jubilaeus has since been clarified as a Yellow Baboon, and not a Chacma. Papio ursinus ruacana is a small, black-footed baboon that is darker than P. u. griseipes and smaller than P. u. ursinus. It is found in Namibia and southwestern Angola (Groves 2001).
Mitochondrial genetic data confirm at least two distinct lineages within Chacma Baboons, dividing the species into northern (griseipes) and southern (ursinus) populations (Sithaldeen et al. 2009; Zinner et al. 2009). These populations have persisted in geographical and genetic isolation since the Early Pleistocene and exhibit additional genetic structure at the population level, accompanying by adaptive behaviours likely driven by aridification (Sithaldeen et al. 2015). A third genetic clade may potentially represent orientalis (Keller et al. 2010). There is currently no conclusive genetic data identifying a distinct ruacana clade.
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Stone, O.1, Slater, K.2, Chowdhury, S.3 & da Silva, J.4
Reviewer: Barrett, A.2
Institutions: 1UNSW Sydney, 2University of South Africa, 3Brooklyn College, New York, 4South African National Biodiversity Institute
Previous Assessors & Reviewers: Hoffman, T.S., Beamish, E., Kaplan, B., Lewis, M., O’Riain, J. & Sithaldeen, R.
Previous Contributors: Davies-Mostert, H., Pfab, M. & Child, M.F.
Assessment Rationale
This species is listed as Least Concern due to its wide distribution and perceived abundance within the assessment region. However, several local subpopulations are threatened with extinction. In KwaZulu-Natal Province (KZN), the population is estimated at less than 10% of its expected size, emphasizing the need for further population-level spatial and demographic data to accurately assess its conservation status. The primary threat to this species is human–wildlife conflict, with baboon-specific electric fencing recommended as a key intervention to mitigate this issue.
Regional population effects: Given that Chacma Baboons are listed as Least Concern, there is no scope for downlisting this species to a lower extinction risk category. However, their adaptability and wide distribution afford them a substantial rescue effect.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Stone O, Slater K, Chowdhury S & da Silva JM. 2025. A conservation assessment of Papio ursinus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
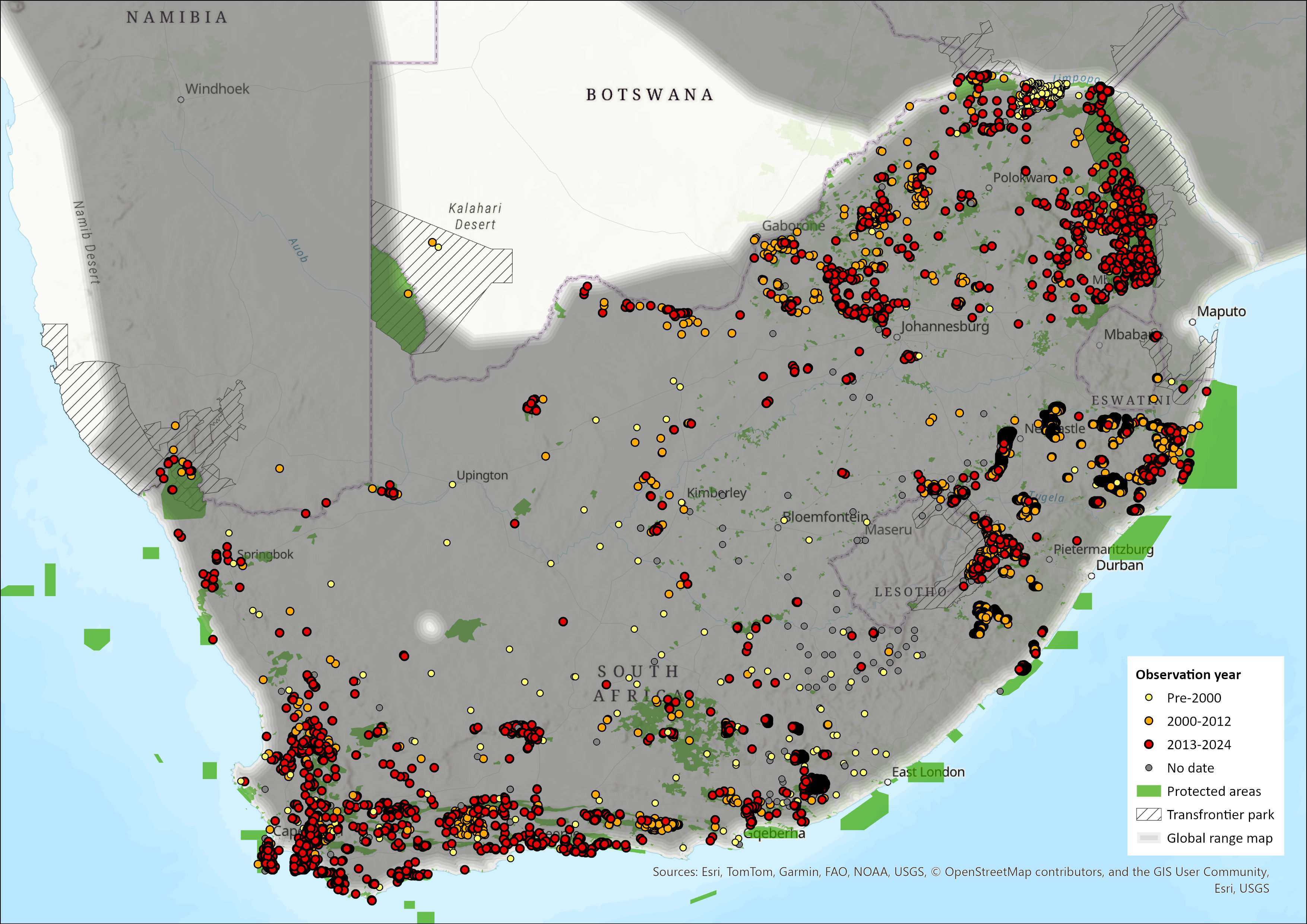
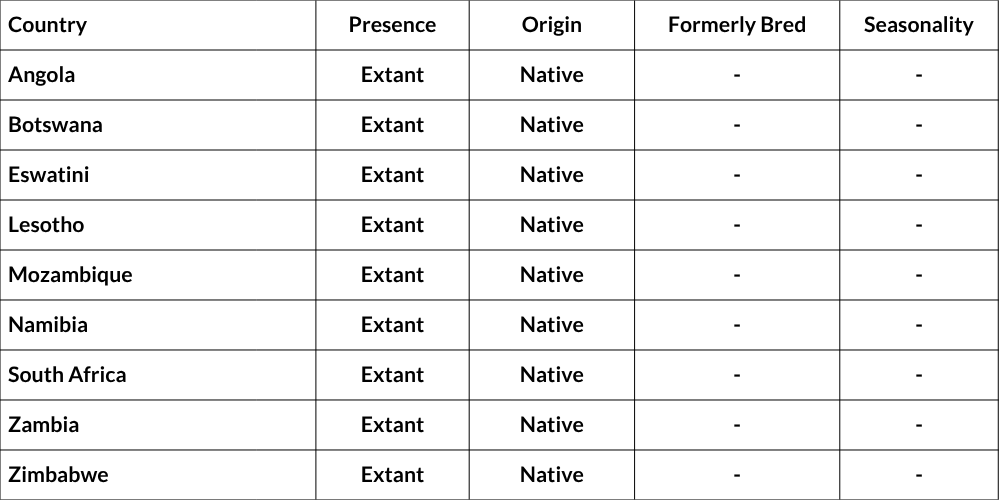
Regional Distribution and occurrence
Geographic Range
Chacma Baboons are widely distributed across southern Africa, including South Africa, Eswatini, Lesotho, Mozambique, Botswana, Namibia, Zimbabwe, southern Zambia, and southern Angola. Their dietary, ecological, and behavioural flexibility (Bronikowski & Altmann 1996; Swedell 2011) allows them to occupy a diverse range of habitat types, including deserts, savannahs, grasslands, forests, and fynbos (Altmann & Altmann 1970; Swedell 2011). They are, however, notably absent in the dune fields of the Kalahari and Namib deserts.
Recent studies of specific subpopulations indicate that, despite their wide distribution, Chacma Baboons exhibit landscape selectively (Henzi et al 2011; Hoffman & O’Riain 2012a). Furthermore, extensive habitat transformation in South Africa has altered their distribution patterns (Stone et al. 2012).
Comparisons of current and historical distribution records do not indicate major shifts in overall Chacma Baboon distribution patterns. However, neither dataset is spatially comprehensive, and differences in data collection protocols limits direct comparability.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): >3200
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map Status
Figure 1. Distribution records for Chacma Baboon (Papio ursinus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Countries of Occurrence
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Based on species distribution models constructed under current and future scenarios, Hill and Winder (2019) predicted that Chacma Baboons are likely to experience substantial losses of optimal and suitable habitats due to climate change, with mean temperature of the driest quarter identified as the most influential factor. Stone et al. (2016) found that the Chacma clades (Southern or Cape, Grey-Footed, and Ruacana) exhibit differing responses to various environmental conditions. The study showed cyclic fluctuations among the clades, with each flourishing or declining under specific circumstances. The Cape Baboon was identified as the most environmentally resilient, while the Grey-Footed Baboon, presently benefiting from existing conditions, was considered the most susceptible to environmental change.
A study of a population of Cape Baboons revealed significant differences in physiology and behaviour associated with seasonality, with higher glucocorticoid “stress” hormones, reduced sociality, and changes in activities observed in winter, when low temperatures and high rainfall likely pose energetic and thermoregulatory challenges for the baboons (Chowdhury et al. 2021). With ongoing climate change in the Cape Peninsula and South Africa—characterised by rising temperatures, shifting seasonality and rainfall, and increasing drought severity—the physiological impacts on baboons may intensify. Furthermore, drought-associated wildfires destroy baboon habitats and food sources, potentially compounding the adverse impacts on their physiology and behaviour.
Stone et al. (2016) suggest that the clade predicted to be most affected by climate change and rising temperatures is the Cape Baboon. Although this clade is anticipated to be the most resilient, it is currently facing severe challenges due to anthropogenic impacts, with many local populations at risk of extinction (Uys, 2011a; Uys 2011b; Uys 2012). In contrast, the Ruacana Baboon may potentially benefit from increasing temperatures.
Population information
To date, there has been no attempt to conduct a census of the overall population size of Chacma Baboons in the assessment region, and only a few studies since 2004 have observed or estimated subpopulation sizes (Beamish 2010). For example, Stone et al. (2012) estimated a meagre population of approximately 11,000 baboons in KZN, with a density in occupied areas of c. 1.8 animals / km2. Local subpopulations in KZN have also been shown to be at risk of extinction (Uys 2011a, 2011b, 2012), highlighting the vulnerability of these populations. Stone et al. (2012) reported that the baboon population in KZN was less than 10% of the expected size, highly fragmented, and largely reliant on protected areas.
Extrapolating subpopulation data to estimate total population size or trends is challenging and contentious. Difficulties arise due to factors such as the behavioural adaptability of baboon troops, large size variation among troops (even those ranging within the same subpopulation), demographic structure (for example, the ratio of mature individuals to immature individuals (Beamish 2010)), and diverse ranging patterns (Hoffman & O’Riain 2012b). In the absence of broader empirical data from across the mammal assessment region, it is not currently possible to provide an accurate estimate of overall population size or trends.
Current population trend: Unknown but suspected to be stable.
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified).
Population genetics
To date, two distinct mitochondrial lineages within Chacma have been identified separating the species into northern (griseipes) and southern (ursinus) groups (Sithaldeen et al. 2009; Zinner et al. 2009). These lineages have persisted in geographical and genetic isolation since the Early Pleistocene and exhibit differentiation comparable to that of other recognised baboon species (Sithaldeen et al. 2015), Moreover, these genetic lineages are accompanied by notably different adaptive behavioural traits in their respective regions (e.g. seafood foraging in low nutrient environments for ursinus, and persistence into the snow-line of the high alpine Drakensberg Mountains for grispies). This supports the formal separation at the species level into Papio ursinus (southwest) and Papio griseipes (northeast) (Sithaldeen et al. 2015).
Within each of these two lineages, additional population-level genetic structure has been identified. Specifically, three genetically distinct clusters are associated with P. u. ursinus (PU1-3), and two clusters are associated with P. u. grispies (PG1 & 2) (see Figure 5 in Sithaldeen et al. 2015). PU2 and PU3 are both bounded by the Kalahari in the north, suggesting that this landform has consistently acted as a barrier to successful dispersal and gene flow. Both PU2 and PU3 seem to isolate from PU1, which seems better associated as a montane refuge.
The high diversity of PG1 in the east along the Kalahari mirrors that of PU2 and PU3, suggesting that the arid interior also played an important role in shaping its genetic diversity and structure. The PG2 clade is centred on a region bounded to its west by the Drakensberg-Maluti Mountains and characterised by dense coastal forests in the north. The high diversity observed at the easternmost boundary of this clade might reflect a colonisation event from montane refugia in the Drakensberg.
Currently, no estimates have been made regarding the effective population size within each of these genetically distinct populations.
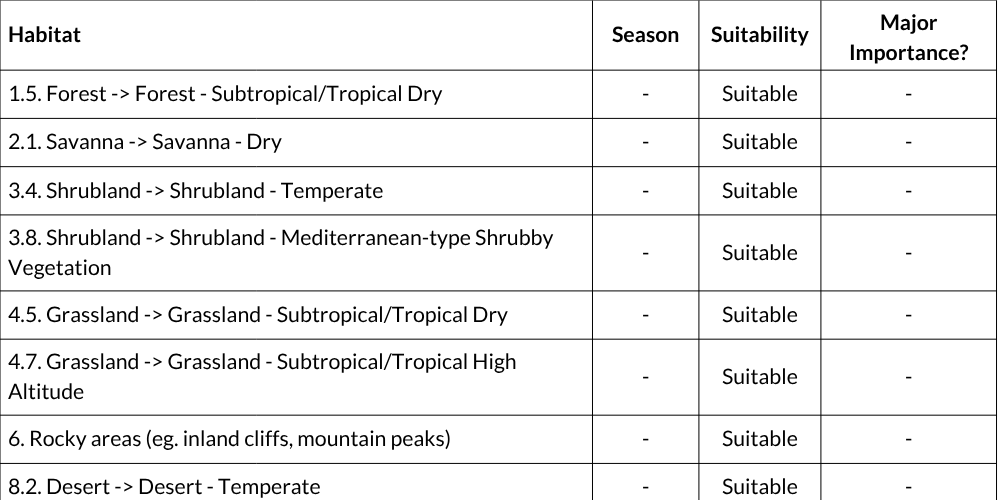
Habitats and ecology
Chacma Baboons occur across all biomes and bioregions within the assessment region, with the possible exception of the Namaqualand Sandveld bioregion, indicating that their distribution is largely unrestricted by habitat type. They inhabit a diverse range of niches, occupying mesic and arid savannas, low and high grasslands, and coastal and montane forests (Altmann & Altmann 1970; Cowlishaw & Davies 1997; Barrett et al. 2002; Codron 2003; Henzi et al. 2003; Sithaldeen et al. 2009; Swedell 2011). Chacma Baboons are also able to thrive in anthropogenically modified environments including urban and agricultural areas, particularly where these environments provide high quality, easily accessible food sources near water and suitable sleeping sites (Hoffman & O’Riain 2012a, 2012b). This adaptability to human-modified habitats is a major factor driving human-baboon conflict (Hoffman & O’Riain 2012c; Kaplan 2013), which has been associated with negative effects on baboon physiology and behaviour (Chowdhury et al. 2020).
Chacma Baboons are not limited by particular soil types or mean annual precipitation levels provided they have access to permanent water sources such as aquifers, seeps, or windmill pumps drawing ground water to the surface (Hamilton III 1986). The highest mean annual rainfall recorded in baboon inhabited areas is approximately 1,555 mm / year (Stone et al. 2013). Baboons are also capable of surviving at some of the highest altitudes (c. 3,280 m) in South Africa (Stone et al. 2013). However, they are unlikely to survive winter conditions at such elevations due to the harsh environmental conditions these pose, including restricted food availability and thermoregulatory constraints (Whiten et al. 1987). Winters in the Cape Peninsula, characterised by low temperatures and high precipitation, have been found to be physiologically challenging for baboons, indicating potential limits to their adaptability due to thermoregulatory needs (Chowdhury et al. 2021).
Reflecting their broad ecological range, Chacma Baboons are omnivores with generalist diets, though they selectively consume specific plant parts (Norton et al. 1987; Byrne et al. 1993; Altmann 1998; Alberts et al. 2005). Their diet primarily consists of fruits, leaves, grasses, forbs, nuts, seeds, and subterranean items, with flowers and animal matter constituting a smaller proportion of their diet.
Due to their behavioural adaptability and dietary breadth and flexibility, Chacma Baboons demonstrate marked ecological variation at the inter- and intra-subpopulation level. Troop sizes range from as few as seven individuals (Beamish 2010) to as many as 115 (Hoffman & O’Riain 2012b), with home range spanning from 1.5–37.7 km2. These range extremes have been recorded within a single subpopulation in the Cape Peninsula (Hoffman & O’Riain 2012b). Density varies widely, from 0.95 baboons / km2 in the Drakensberg Mountains (Whiten et al. 1987) to 16.8 baboons / km2 in the Okavango Delta (Hamilton III et al. 1976). This variation correlates with troop size and resource availability, as larger troops typically occupy larger home ranges at lower densities, while troops with access to high quality resources have smaller home ranges at higher densities (Hoffman & O’Riain 2012b).
Ecosystem and cultural services: As generalist omnivores with a largely plant-based diet, baboons have the potential to contribute significantly to seed dispersal for various plant species they consume (Slater & du Toit 2002; Tew et al. 2018). Studies indicate that baboons are important seed dispersers for certain species, such as Ziziphus mucronata, within southern African savanna environments (Slater & du Toit 2002). While their role as seed dispersers in other habitats remains untested, findings from these studies alongside research from other African regions (Lieberman et al. 1979; Kunz & Linsenmair 2008), suggest that baboons could play an important role in dispersing and propagation a range of plant species. Additionally, baboons contribute to zoogeomorphic processes by overturning rocks while foraging for invertebrates (Mare et al. 2019) and consume soil at geophagy sites, impacting soil composition (Pebsworth et al. 2012).
IUCN Habitat Classification Scheme
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Non-human primates, including Papio species, have been widely used as models for human medical research (Bailey 2005). Historically, wild Chacma Baboons were sourced for medical research within South Africa. However, this practice is now being discouraged, with captive-reared baboons preferred for such research.
Across their global range, Chacma Baboons are also utilised for bushmeat and traditional medicine (Minhós et al. 2013), although the extent and trends of this use are not well documented or well understood. They have been found in bushmeat markets in Johannesburg (Whiting et al. 2011) and the Eastern Cape (Simelane & Kerley 1998) and are commonly traded in markets across KZN (Ngwenya 2001). Within the assessment region, we suspect neither the bushmeat nor traditional medicine trade significantly impacts the population negatively.
Baboons are also hunted for both trophy and recreational purposes, with minimal hunting restrictions. According to the CITES trade database, an average of 334 ± 67 baboons were hunted annually from 2002–2012. While this level of hunting is not suspected to negatively impact the overall population, it may contribute to local subpopulation declines if not properly regulated.
National Commercial Value: No
International Commercial Value: No
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Anthropogenic impacts, including direct human activity, roads, infrastructure, and habitat degradation, are the most concerning influences on current populations (Stone et al. 2012). Time spent in human-occupied and impacted areas, where baboons are exposed to increased rates of human interaction and conflict, has been associated with negative changes in both stress physiology and behaviour of baboons, potentially affecting their long-term health and welfare (Chowdhury et al. 2020). Many Chacma populations in South Africa inhabit commercial plantations, and when these areas are cleared, baboons experience displacement, loss of foraging ground, food resources, and sleeping sites. Moreover, baboons are known to strip the bark from pine trees in these plantations, leading to damage and even death of the trees, which results in economic loss. Mitigation of this issue has often involved the culling of baboons. There are no other major threats to this species; however, problem animals may be shot or culled due to being classed as problematic (e.g. for crop damage). This practise has significantly contributed to the endangerment or extermination of local populations, potentially limiting genetic diversity (Stone et al. 2015; Stone et al. 2012). Baboons are also hunted locally, with parts of dead individuals offered for sale in markets in South Africa for traditional medicinal use, but this is not considered a major threat.
Habitat trend: Stable.
Urbanisation and agriculture can force Chacma Baboons into increasingly marginalised, and often high-lying and rugged, natural habitat (Hoffman & O’Riain 2012a). Additionally, baboons are attracted to the high density and quality of food resources in agricultural areas. Together these “push and pull” factors may drive high levels of human–baboon conflict, which is evident throughout the subregion. Provincial reports show land alteration rates are increasing (Goodman 2010), leaving large areas of land uninhabitable for baboons (Stone et al. 2012).
Conservation
The greatest hindrance to Chacma Baboon conservation is conflict with humans, particularly in the agricultural sector. People experiencing this conflict have attempted to mitigate it through various methods, including lethal removal, translocation, herding, a variety of deterrents, and food provisioning or diversionary feeding (outside of human areas; this should not be confused with supplementary feeding). While few studies quantify the efficacy of these measures, the interventions most likely to succeed in areas with high-quality concentrated resources (for example, vineyards and citrus farms) are, in order of long-term effectiveness: baboon-specific electric fencing, dynamic noise (for example, bear bangers), and dynamic pain aversion (for example, paintball markers), especially when paired with the presence of baboon caretakers or field rangers (van Doorn 2009; Kaplan 2013).
It is critical to note that, when dealing with an animal as adaptable and intelligent as a baboon, any intervention will fail unless it is implemented conscientiously and adaptively. Where lethal control is practiced—either on individual damage-causing animals or whole populations—best practice demands that the impacts of the removal on the sustainability of the population are assessed. Factors driving the conflict must be addressed to reduce the need for long-term lethal control. It is also imperative that damage is correctly ascribed to particular individuals or troops if lethal control is to be effective and not adversely impact the broader population.
A rare but alternative intervention to lethal control of baboons is translocation. This is a costly method that requires extensive financial and professional resources, long-term post-translocation monitoring, and suitable available habitat in an area where conflict is expected to be negligible and competition with conspecifics limited. An example of a successful translocation of a baboon troop is documented for Papio anubis, a close relative of the Chacma Baboon (Strum 2005).
Recommendations for land managers and practitioners:
- Currently there are no national or provincial management plans for Chacma Baboons; existing management plans are specific to certain areas (for example, the Cape Peninsula).
- To conserve baboons across the assessment region, efforts should focus on minimising human–wildlife conflict, the primary threat to their conservation. Achieving this requires (a) restricting baboons’ access to resources (food, water and sleeping sites) in human-modified areas, and (b) preserving sufficient natural habitat with necessary suitable resources (food, water and sleeping sites) to sustain baboon populations in undeveloped areas (Hoffman 2011; Kaplan 2013).
Research priorities:
- Despite Chacma Baboons being widely distributed, up to date distribution data across their full geographic range is lacking. Additionally, there is no national monitoring of population numbers, and only one province, KZN, conducts annual population counts, resulting in population data only being available at local scales. Threats to this species – which are potentially large – are also poorly monitored and documented. These factors hinder the accurate assessment of the species’ extinction risk.
Examples of research being undertaken:
- The Baboon Research Unit at the University of Cape Town has conducted research on the demography, ecology and management of Chacma Baboons in the Cape Peninsula. This research, now nearing completion, has significantly influenced made local baboon management and provided valuable insights for broader baboon management and conservation efforts.
- Dietmar Zinner at the German Primate Center, conducted extensive research on baboon phylogeny and distribution. A lot of his work focuses on the genetic diversity ang geographic distribution of baboon species across Africa, including the Chacma Baboon. He has explored their genetic diversity and geographic distribution, hybridisation, and their ecological adaptability.
More research is needed to address:
- The distribution, population numbers, and population trends of Chacma Baboons across their range.
- The impact of human persecution and human-wildlife conflict on Chacma Baboon populations.
- The extent and impact of baboon use in traditional medicine markets, both local and internationally.
Encouraged citizen actions:
- Restricting baboon access to human refuse in residential and recreational areas by using baboon-proof refuse bins.
- Restricting baboon access to human food sources, such as by removing or protecting vegetable gardens.
- Utilising deterrent methods to exclude baboons from human-modified environments (for example, crop fields or residential areas):
- Install baboon-specific electric fencing.
- Use adaptive noise aversion techniques, like bear bangers.
- Deploy paintball markers (non-lethal) as adaptive deterrents.
- Educate residents and tourists about responsible behaviour within or adjacent to, baboon habitats.
- People can also contribute to conservation efforts by collecting submitting distribution and demographic information about baboons to virtual platforms such as iNaturalist and MammalMAP. Doing this will improve our understanding of this species.
Bibliography
Alberts SC, Hollister-Smith JA, Mututua RS, Sayialel SN, Muruthi PM, Warutere JK, Altmann J. 2005. Seasonality and long-term change in a savanna environment. Pages 157–195 in Brockmann DK, van Schaik CP, editors. Seasonality in Primates: Studies of Living and Extinct Human and Non-Human Primates. Cambridge University Press, Cambridge, UK.
Altmann SA, Altmann J. 1970. Baboon Ecology, African Field Research. Bibliotheca Primatologica, No. 12. University of Chicago Press, Chicago, Illinois, USA.
Altmann SA. 1998. Foraging for Survival: Yearling Baboons in Africa. University of Chicago Press, Chicago, Illinois, USA.
Bailey J. 2005. Non-human primates in medical research and drug development: a critical review. Biogenic Amines 19: 235– 255.
Barrett L, Gaynor D, Henzi SP. 2002. A dynamic interaction between aggression and grooming reciprocity among female chacma baboons. Animal Behaviour 63: 1047–1053.
Beamish EK. 2010. Causes and consequences of mortality and mutilation in baboons of the Cape Peninsula, South Africa. M.Sc. Thesis. University of Cape Town.
Biryahwaho B. 2002. Community Perspectives Towards Management of Crop raiding Animals: Experiences of Care–DTC with Communities Living Adjacent to Bwindi Impenetrable and Mgahinga Gorilla National Parks, Southwest Uganda. Human– Wildlife Conflict: Identifying the problem and possible solutions.
Bronikowski AM, Altmann J. 1996. Foraging in a variable environment: weather patterns and the behavioral ecology of baboons. Behavioral Ecology and Sociobiology 39: 11–25.
Brown LR, Marais AJ, Barrett L, Henzi SP. 2006. Habitat structure, population characteristics and resource utilisation by chacma baboons in commercial forestry areas of the Eastern Mpumalanga Escarpment. Report to Global Forestry Products and Komatiland Forests, Sabie, South Africa.
Byrne RW, Whiten A, Henzi SP, McCulloch FM. 1993. Nutritional constraints on mountain baboons (Papio ursinus): implications for baboon socioecology. Behavioral Ecology and Sociobiology 33: 233–246.
Chowdhury S, Brown J, Swedell L. 2020. Anthropogenic effects on the physiology and behaviour of chacma baboons in the Cape Peninsula of South Africa. Conservation Physiology 8(1): coaa066. doi: 10.1093/conphys/coaa066.
Chowdhury S, Brown JL, Swedell L. 2021. Costs of seasonality at a southern latitude: Behavioral endocrinology of female baboons in the Cape Peninsula of South Africa. Hormones and Behavior 134: 105020. doi: 10.1016/j.yhbeh.2021.105020.
Codron DM. 2003. Dietary ecology of Chacma baboons Papio ursinus (Kerr, 1972) and Pleistocene Cercopithecoidea in Savanna environments of South Africa. M.Sc. Thesis. University of Cape Town.
Cowlishaw G, Davies JG. 1997. Flora of the Pro-Namib Desert Swakop River catchment, Namibia: community classification and implications for desert vegetation sampling. Journal of Arid Environments 36: 271–290.
Goodman PS. 2010. KwaZulu-Natal State of Biodiversity Report— 2009. Unpublished Report. Ezemvelo KZN Wildlife, Pietermaritzburg, South Africa.
Groves, C.P. 2001. Primate Taxonomy. Smithsonian Institution Press, Washington, DC, USA.
Hamilton III WJ, Buskirk RE, Buskirk WH. 1976. Defense of space and resources by chacma (Papio ursinusbaboon troops in an African desert and swamp. Ecology 57: 1264–1272.
Hamilton III WJ. 1986. Namib Desert chacma baboon (Papio ursinus) use of food and water resources during a food shortage. Madoqua 14: 397–407.
Henzi SP, Barrett L, Gaynor D, Greeff J, Weingrill T, Hill RA. 2003. Effect of resource competition on the long-term allocation of grooming by female baboons: evaluating Seyfarth’s model. Animal Behaviour 66: 931–938.
Hill CM. 2000. Conflict of interest between people and baboons: crop raiding in Uganda. International Journal of Primatology 21: 299–315.
Hill, W.C.O. 1970. Primates Comparative Anatomy and Taxonomy VIII Cynopithecinae. Edinburgh University Press, Edinburgh, Scotland.
Hoffman TS, O’Riain MJ. 2012a. Landscape requirements of a primate population in a human-dominated environment. Frontiers in Zoology 9: 1.
Hoffman TS, O’Riain MJ. 2012b. Troop Size and Human-Modified Habitat Affect the Ranging Patterns of a Chacma Baboon Population in the Cape Peninsula, South Africa. American Journal of Primatology 74: 853–863.
Hoffman TS, O’Riain MJ. 2012c. Monkey management: using spatial ecology to understand the extent and severity of human– baboon conflict in the Cape Peninsula, South Africa. Ecology and Society 17: 13.
Hoffman TS. 2011. The spatial ecology of chacma baboons (Papio ursinus) in the Cape Peninusula, South Africa: towards improved management and conservation strategies. Ph.D. Thesis. University of Cape Town.
Jolly CJ. 1993. Species, subspecies, and baboon systematics. Pages 67–107 in Species, Species Concepts and Primate Evolution. Springer, New York, USA.
Kansky R, Gaynor D. 2000. Baboon management strategy for the Cape Peninsula. Final report, Table Mountain Fund Project number ZA 568. Cape Town, South Africa.
Kaplan B. 2013. In Pursuit of a Panacea: Mitigating HumanBaboon Conflict in the Cape Peninsula, South Africa. Ph.D. Thesis. University of Cape Town.
Kaplan BS, O’Riain MJ, van Eeden R, King AJ. 2011. A low-cost manipulation of food resources reduces spatial overlap between baboons (Papio ursinus) and humans in conflict. International Journal of Primatology 32: 1397–1412.
Kaplan BS, O’Riain MJ. 2015. Shedding light on reflective prisms as potential baboon (Papio ursinus) deterrents in the Cape Peninsula, South Africa. African Journal of Wildlife Research 45: 117–121.
Katsvanga CAT, Mudyiwa SM, Gwenzi D. 2006. Bark stripping and population dynamics of baboon troops after chemical control in pine plantations of Zimbabwe. African Journal of Ecology 44: 413–416.
Keller, C., Roos, C., Groeneveld, L.F., Fischer, J. and Zinner, D. 2010. Introgressive hybridization in southern African baboons shapes patterns of mtDNA variation. American Journal of Physical Anthropology 142: 125–136.
Kingdon, J. 1997. The Kingdon Field Guide to African Mammals (first edition}. Academic Press, San Diego, California, USA.
Kunz BK, Linsenmair KE. 2008. The role of the olive baboon (Papio anubis, Cercopithecidae) as seed disperser in a savanna– forest mosaic of West Africa. Journal of Tropical Ecology 24: 235– 246.
Laurance WF, Lovejoy TE, Vasconcelos HL, Bruna EM, Didham RK, Stouffer PC, Gascon C, Bierregaard RO, Laurance SG, Sampaio E. 2002. Ecosystem decay of Amazonian forest fragments: a 22-year investigation. Conservation Biology 16: 605– 618.
Lieberman D, Hall JB, Swaine MD, Lieberman M. 1979. Seed dispersal by baboons in the Shai Hills, Ghana. Ecology 60: 65–75.
Mascarenhas A. 1971. Agricultural Vermin in Tanzania. Pages 259– 267 in Ominde SH, editor. Studies in East African Geography and Development. . University of California Press, California, USA.
Minhós, T., Wallace, E., da Silva, M.J.F., Sá, R.M., Carmo, M., Barata, A and Bruford, M.W. 2013. DNA identification of primate bushmeat from urban markets in Guinea-Bissau and its implications for conservation. Biological Conservation 167: 43–49.
Morris B. 2000. Wildlife depredations in Malawi. Pages 36–49 in Natural Enemies: People–Wildlife Conflicts in Anthropological Perspective. Routledge, London, UK.
Naughton-Treves L. 1996. Uneasy neighbors: wildlife and farmers around Kibale National Park, Uganda. Ph.D. Thesis. University of Florida.
Naughton-Treves L. 1998. Predicting patterns of crop damage by wildlife around Kibale National Park, Uganda. Conservation Biology 12: 156–168.
Ngwenya MP. 2001. Implications of the medicinal animal trade for nature conservation in KwaZulu-Natal, Report No. NA/124/04. Ezemvelo KZN Wildlife.
Norton GW, Rhine RJ, Wynn GW, Wynn RD. 1987. Baboon diet: a five-year study of stability and variability in the plant feeding and habitat of the yellow baboons (Papio cynocephalus) of Mikumi National Park, Tanzania. Folia Primatologica 48: 78–120.
Pebsworth PA, Bardi M, Huffman MA. 2012. Geophagy in chacma baboons: patterns of soil consumption by age class, sex, and reproductive state. American Journal of Primatology 74(1): 48-57. doi: 10.1002/ajp.21008.
Pepeh K. 1996. A preliminary assessment of crop damage by wild animals in and around Gashaka Gumti National Park. Unpublished report to Nigerian Conservation Foundation, World Wildlife Fund and National Park Service.
Simelane TS, Kerley GIH. 1998. Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research 28: 121–126.
Sithaldeen R, Ackermann RR, Bishop JM. 2015. Pleistocene Aridification Cycles Shaped the Contemporary Genetic Architecture of Southern African Baboons. PLOS ONE 10(5): e0123207. https://doi.org/10.1371/journal.pone.0123207
Sithaldeen, R., Bishop, J.M. and Ackermann, R.R. 2009. Mitochondrial DNA analysis reveals Plio-Pleistocene diversification within the chacma baboon. Molecular Phylogenetics and Evolution 53: 1042–1048.
Slater K, Toit JT Du. 2002. Seed dispersal by chacma baboons and syntopic ungulates in southern African savannas. South African Journal of Wildlife Research 32: 75–79.
Stone, O.M.L., Herries, A.I.R., Brink, J.S. & Laffan, S.W. (2016) The chacma baboon (Papio ursinus) through time: a model of potential core habitat regions during a glacial–interglacial cycle. Evolutionary Ecology, 1-28.
Stone OML, Laffan SW, Curnoe D, Herries AI. 2013. The spatial distribution of chacma baboon (Papio ursinus) habitat based on an environmental envelope model. International Journal of Primatology 34: 407–422.
Stone, O.M.L., Laffan, S.W., Curnoe, D. & Herries, A.I.R. (2015) Potential human impact on the environmental central niche of the chacma baboon. South African Journal of Science, 111
Stone OML, Laffan SW, Curnoe D, Rushworth I, Herries AI. 2012. Distribution and population estimate for the chacma baboon (Papio ursinus) in KwaZulu-Natal, South Africa. Primates 53: 337–344.
Strum SC. 1994. Prospects for management of primate pests. 49: 295–306.
Strum SC. 2005. Measuring success in primate translocation: a baboon case study. American Journal of Primatology 65: 117–140.
Swedell L. 2011. African papionins: diversity of social organization and ecological flexibility. Pages 241–277 in Campbell CJ, Fuentes A, MacKinnon KC, Bearder S, Stumpf R. Primates in Perspective. Oxford University Press, Oxford, UK.
Uys R. 2011a. Oribi Gorge baboon count. Unpublished report. Ezemvelo KZN Wildlife, Durban, South Africa.
Uys R. 2011b. Umtamvuna baboon count. Unpublished report. Ezemvelo KZN Wildlife, Durban, South Africa.
Uys R. 2012. Ongoye baboon count. Unpublished report. Ezemvelo KZN Wildlife, Durban, South Africa.
Warren Y, Buba B, Ross C. 2007. Patterns of crop-raiding by wild and domestic animals near Gashaka Gumti National Park, Nigeria. International Journal of Pest Management 53: 207–216.
Whiten A, Byrne RW, Henzi SP. 1987. The behavioral ecology of mountain baboons. International Journal of Primatology 8: 367–388.
Whiting MJ, Williams VL, Hibbitts TJ. 2011. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Journal of Zoology 284: 84–96.
Zinner D, Groeneveld LF, Keller C, Roos C. 2009. Mitochondrial phylogeography of baboons (Papio spp.) – Indication for introgressive hybridization? BMC Evolutionary Biology 9: 83.
van Doorn A. 2009. The interface between socioecology and management of chacma baboons (Papio ursinus) in the Cape Peninsula, South Africa. PhD Thesis. University of Cape Town.
van Oosten V. 2004. The Conflict Between Primates and the Human Population in a Protected Area in North-Cameroon. Programme d’Environnement et Développement, Centre des Études de l’Environnement (CML), Université de Leiden.