Caracal
Caracal caracal

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Caracal caracal – (Schreber, 1776)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – FELIDAE – Caracal – caracal
Common Names: Caracal, African Caracal, Asian Caracal, Desert Lynx (English), Rooikat, Lynx (Afrikaans), Indabutshe (Ndebele), Thwane (Setswana), Thooane (Sotho), Nandani (Tsonga), Thwani (Venda), Ingqawa, Ngada (Xhosa), Indabushe (Zulu)
Synonyms: Felis caracal Schreber, 1776
Taxonomic Note:
The Caracal has been classified variously with Lynx and Felis in the past, but molecular evidence supports a monophyletic genus. It is closely allied with the African Golden Cat (Caracal aurata) and the Serval (Leptailurus serval), having diverged around 8.5 mya (Janczewski et al. 1995, Johnson and O’Brien 1997, Johnson et al. 2006).
Seven subspecies have been recognised in Africa (Smithers 1975), of which two occur in southern Africa: C. c. damarensis from Namibia, the Northern Cape, southern Botswana and southern and central Angola; and the nominate C. c. caracal from the remainder of the species’ range in southern Africa (Meester et al. 1986). According to Stuart and Stuart (2013), however, these subspecies should best be considered as geographical variants.
Red List Status:LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Drouilly, M.1, Serieys L.E.K. 1, Leighton, G.R.M.2 & da Silva, J.M.3
Reviewers: Do Linh San, E.4 & Ehlers Smith, Y.5,6
Institutions: 1Panthera, 2Urban Caracal Project, 3South African National Biodiversity Institute, 4Sol Plaatjie University, 5Ezemvelo KZN Wildlife, 6University of KwaZulu-Natal
Previous Assessors & Reviewers: Avenant, N., Drouilly, M., Power, J., Thorn, M., Martins, Q., Neils, A., du Plessis, J.
Previous Contributors: Child, M.F. & Nicholson, S.
Assessment Rationale
Caracals are widespread within the assessment region. They are considered highly adaptable and, within their distribution area, are found in virtually all habitats except the driest part of the Namib. They also tolerate high levels of human activity, including near urban areas, and persist in most small-stock areas in southern Africa, despite continuously high levels of persecution over many decades. In some regions it is even expected that Caracal numbers are suspected to have increased. Thus, the Least Concern listing remains. The use of blanket control measures over vast areas, the illegal use of poisons, and the uncontrolled predation management efforts over virtually the total assessment region are, however, of concern (Drouilly et al. 2023). In the North West and Limpopo provinces, concerns have also been raised about hunting and live removals. Ongoing monitoring, education efforts, and the continuous propagation of mitigation measures such as exclusion and precautionary techniques, the removal of proven damage-causing animals (DCAs) and sustaining sufficient levels of natural prey diversity and biomass on farmlands, should be a priority to prevent possible national declines.
Regional population effects: Namibia, Botswana and South Africa offer the most important stronghold for Caracal worldwide. Radio-collared Caracals have been documented moving between Namibia and South Africa across the western fence line of the Kgalagadi Transfrontier Park (KTP) (see Melville et al. 2004), and they have been camera trapped along the Molopo River along the South Africa–Botswana border (see Power 2014). Hence, these countries’ borders are no doubt permeable to Caracal transboundary movements, which may include both individual forays as well as actual dispersals. In the other transfrontier conservation areas around and inside South Africa, it is also plausible that some individuals will move between Mozambique, South Africa and Zimbabwe, and between South Africa and Lesotho; this has, however, not been documented.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Drouilly M, Serieys LEK, Leighton GRM & da Silva JM. 2025. A conservation assessment of Caracal caracal. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Caracals are widely distributed across Africa, Central Asia, and southwest Asia into India (Avgan et al. 2016). The historical range of the Caracal mirrors that of the Cheetah (Acinonyx jubatus), and both coincide with the distribution of several small desert gazelles (Sunquist & Sunquist 2002).
Within the assessment region, although Caracals were amongst the species that were persecuted heavily by colonial settlers and then impacted further by crop and livestock farmers in the early part of the 20th century (Pringle & Pringle 1979; Stadler 2006; Bothma et al. 2009; Inskip & Zimmermann 2009), Pringle and Pringle (1979) mentioned increasing numbers of Caracals in eastern areas from c. 1970.
Currently, the species occurs in all South African provinces, as well as in Lesotho and Eswatini. Historically, Caracals were rare in the western Kalahari (Lloyd & Millar 1983), and apparently absent from the Highveld grasslands of the North West Province (Rautenbach 1978). Caracals have responded well to game farming in especially the latter province, and today are present on over 95% of farms in the bushveld landscapes, and on at least three quarters of farms on grasslands in the North West Province (see Thorn et al. 2011; Power 2014). Caracals are also doing well in small-livestock farms devoted of large predators in the Central Karoo (Drouilly et al. 2019a), despite high levels of persecution (Drouilly et al. 2023). Accordingly, there has been an increase in area of occupancy (AOO) of the Caracal throughout the North West and western parts of the Limpopo Province (Thorn et al. 2011), while its extent of occurrence (EOO) has, from 2000 to 2010 (Thorn et al. 2011), and even up to 2013 (Power 2014), remained unchanged.
The species was absent or present in low densities in the KwaZulu-Natal Drakensberg during the 1960s, and individuals were brought in from the then Cape Province (see Barnes 1991). Since then, Caracal numbers have seemingly increased throughout the whole KwaZulu-Natal Province (Barnes 1991) The eastern coastal belt was once considered to be excluded from their distribution range, but has since been colonised and the species have proliferated within the Indian Ocean Coastal Belt (Ehlers Smith et al. 2019).
The population today is probably contiguous and more widely spread over most of the assessment region. Skead (1980) reports that the southern Cape populations were, during dry years, regularly augmented from influxes from Karoo populations, so there may also be significant interchange at the biome level; however, the anthropogenic development in and around the Cape Town Peninsula is increasingly limiting Caracal movements (Serieys et al. 2023, Kyriazis et al. 2023). Caracals, like most felids, do not migrate, but young individuals can disperse over extensive areas (Norton & Lawson 1985; S. Hanekom pers. comm. 1990; Nattrass et al. 2020a), which seems to also be confirmed by the first insights into the genetic population structure of Caracals in South Africa that shows a possibly panmictic population between the Karoo, the Northern Cape and the Eastern Cape (Tensen et al. 2019).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 3300
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
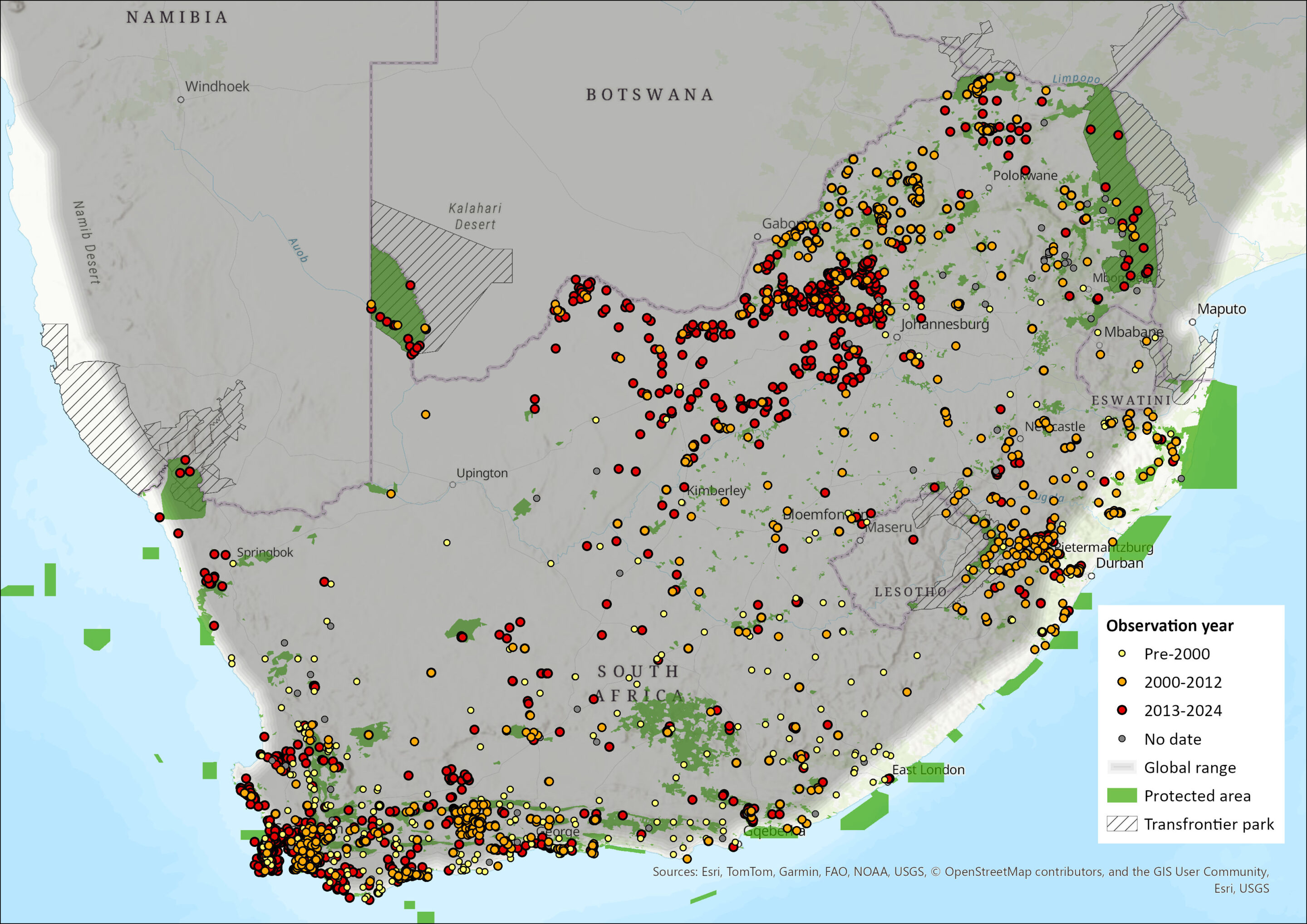
Map
Figure 1. Distribution records for Caracal (Caracal caracal) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Afghanistan | Extant | Native | – | – |
| Algeria | Extant | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Presence Uncertain | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Presence Uncertain | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Egypt | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| India | Extant | Native | – | – |
| Iran, Islamic Republic of | Extant | Native | – | – |
| Iraq | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Jordan | Extant | Native | – | – |
| Kazakhstan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Kuwait | Extant | Native | – | – |
| Lebanon | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Libya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Syrian Arab Republic | Extant | Native | – | – |
| Tajikistan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Tunisia | Extant | Native | – | – |
| Turkmenistan | Extant | Native | – | – |
| Türkiye | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| United Arab Emirates | Extant | Native | – | – |
| Uzbekistan | Extant | Native | – | – |
| Western Sahara | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
A review of how drylands mammals will respond to climate change was conducted by Fuller et al. (2021). The review showed that due to climate change, mammals inhabiting arid regions are confronting not only escalating heat stress but also dwindling water and food resources. Insufficient water availability impedes evaporative cooling, resulting in heightened core body temperatures during hot periods, while food scarcity diminishes the ability to regulate core temperature during cold nights. The combination of limited food and water will narrow the temperature range within which core body temperature is maintained relatively constant, thereby escalating the risk of physiological dysfunction and mortality. Adaptations in behaviour, such as adjusting activity patterns between day and night or seeking out thermally stable microclimates, may enable individuals to remain within a suitable temperature range for their body to function normally, but may incur costs such as decreased foraging efficiency or heightened competition and anthropogenic risks, ultimately affecting fitness. Caracals mostly live in drylands and partly depend, like their prey, on waterholes maintained by farmers and reserve managers (especially lactating females; M. Drouilly, pers. obs. 2013-2015). With rising temperatures, vast landscapes currently occupied by Caracals might be abandoned by struggling farmers, having a direct effect on availability of water and food resources for Caracals and their prey. To comprehensively assess how dryland mammals such as Caracals will cope with rising heat stress amidst water and food shortages, it is imperative to gain a deeper understanding of the sensitivities and reactions of these mammals to both the direct and indirect impacts of climate change.
An exploratory assessment from the University of Pretoria (van Jaarsveld et al. 2000) on the vulnerability and adaptation of South African animal taxa to a climate scenario involving the doubling of atmospheric CO2 concentrations, which was predicted to lead to an increase of average temperatures by approximately 2°C, predicted that Caracals would experience up to 37% range loss and one of their mainprey species, the Rock Hyrax (Procavia capensis), up to 38% range contraction. Although this predicted change is not as alarming as for other species, the preliminary nature of the study and the fact that it was conducted more than 20 years ago underscore the need for further research on the topic.
Population information
The Caracal is common in South Africa and southern Namibia, where removed individuals seem to be quickly replaced by other individuals. No published data exist, however, to say that the species is common outside of parts of southern Africa; in fact, in comparison with the situation in the assessment region, Caracals are considered rare throughout most of their range (Avgan et al. 2016).
Early scientifically gathered information on density estimates is virtually non-existent and makes comparison with newly gathered information, using relatively modern techniques, difficult. Benchmark information useful for future comparison were only gathered for the first time in South Africa’s northern provinces (Thorn et al. 2011; Power 2014). This information suggests that, at least in the North West Province, the species’ AOO has regionally increased (when compared to the information put forward in Rautenbach 1978), though, owing to differences in methodology (i.e. camera traps in later years), the significance of this is called into question. Notwithstanding, Caracal records shared with the IUCN between 2000-2012 in this province are less numerous than for the period 2013-2023. It is also fact that records of this species were only forthcoming from this part of the Highveld grasslands well after the late 1990s (Transvaal Provincial Administration Records; Newbery 1995). Elsewhere, hearsay information and the proliferation of records do also support an eastward range expansion in KwaZulu-Natal. This was first observed by Rowe-Rowe (1992) when he compared his data with the earlier records of Pringle (1977) and Rowe-Rowe (1978); today, further expansion can be noticed when recent MammalMap records are compared with those indicated in Rowe-Rowe (1992). In the Central Karoo, a questionnaire survey of small-livestock farmers also reported an expansion of Caracal range, with cumulative sightings increasing by 16% between before 1994 and 2014. Caracal numbers were also reported by 44% of the interviewees to have increased between 2004 and 2014. Only a handful of farmers thought that Caracal numbers had declined, and they attributed this to an increase in the Black-backed Jackal (Lupulella mesomelas) population (Drouilly et al. 2019c).
Caracal densities (as inferred from home range size) can vary markedly between habitats, depending on environmental variables such as the size, type, density and composition of prey available, habitat characteristics, and the degree of persecution by humans (Avenant 1993). For example, home ranges of males in Postberg Nature Reserve (PNR; part of the West Coast National Park) (Avenant & Nel 1998) were larger than those of males in the Mountain Zebra National Park (MZNP) (Moolman 1986), but smaller than those of males on farms around the MZNP (Moolman 1986). In turn, the home ranges in all three of these study areas were markedly smaller than that of a single male tracked in the mountains in the Western Cape (Norton & Lawson 1985). Similar differences were observed in female home ranges, with home ranges in a farming area, southwest of the Western Cape (Stuart 1982), significantly larger than at both MZNP (Moolman 1986) and PNR (Avenant & Nel 1998). The smaller home range size of females in the PNR could reflect the high density of rodent prey, the most common item in Caracal scats and the only prey group whose density and biomass significantly correlated with its percentage volume in Caracal scats at PNR (Avenant & Nel 1998). Fitting into the normal felid pattern, male home ranges are larger than those of females, and typically overlap with a number of female home ranges (Moolman 1986; Avenant 1993; Stuart & Stuart 2013). While sexual dimorphism, and the fact that the larger males may prefer larger prey species, which are less densely spaced than the smaller prey species, are still debated as a possible reason for this observation, Avenant (1993) found, within each of the four study areas mentioned above (Stuart 1982; Norton & Lawson 1985; Moolman 1986; Avenant 1993), strong positive correlations between home range size and standardised metabolic needs (SMN, where SMN = body weight0.75).
Avenant and Nel (1998) estimated a density of 0.23–0.47 Caracal / km² in PNR, while Moolman (1986) estimated a density of 0.38 Caracal / km² for MZNP. A density of 0.3 Caracal / km² is thus a reasonable estimate for a high-density population, should blanket extrapolations be required. Large differences may, however, occur on farmland where Caracals are actively hunted, and territoriality and social structure may differ from that in protected populations (du Plessis et al. 2015). Furthermore, in areas where Caracals and Black-backed Jackals co-occur, Caracal densities may be markedly lower in some habitats where they are excluded by Black-backed Jackals and vice versa (Ferreira 1988); however, a recent study in the Karoo (Woodgate et al. 2023) showed that Caracal and Black-backed Jackal did not exclude each other, but rather that their spatiotemporal overlap was high, largely independent of land-use, and positively influenced by the presence of the other species. Current information shows that, in areas where both species co-occur, Caracal seems to be the dominant species in more rocky and mountainous terrain and Black-backed Jackal more dominant on open plains areas (Drouilly et al. 2018a); this situation may, however, be impacted by the type and combination of prey items, as well as the persecution history of the area (see du Plessis 2013). Compensatory breeding is a factor that may explain the Caracal’s resilience to persecution (Avenant & du Plessis 2008; du Plessis 2013), but this has not been confirmed for this species.
Considering the possibility of such varying density estimates, a robust population estimate would be difficult to attain based on the current lack of data. However, given their wide distribution in South Africa, that Caracals seem to prefer rocky or mountainous terrain (such as at PNR and MZNP; Drouilly et al. 2018a; Drouilly et al. 2019c), but are very adaptable and can occur in many different vegetation types (e.g. they also occur in the Kalahari), the total Caracal population in the assessment region is likely over 20,000 individuals, depending on local densities (0.15–0.5 individual / km2) and occupancy.
Population Information
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: Unknown
Justification: It is not currently possible to determine the extent or number of subpopulations. Caracal distribution is regarded to be continuous, with a notable exception being the population on the Cape Peninsula that is cut off from the “mainland” population by the relatively dense and vast urban area (Serieys et al. 2023).
Continuing decline in number of subpopulations: Probably not
All individuals in one subpopulation: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Few population genetic studies have been conducted on Caracals, with the majority conducted in South Africa, with samples originating primarily from the Western Cape (Tensen et al. 2018, Tensen 2019, Kyriazis et al. 2024). Tensen et al. (2018, 2019) found high genetic diversity and low genetic differentiation (single genetic cluster) among animals (from the Central Karoo of the Western Cape, Northern Cape, Eastern Cape, Mpumalanga), indicating they all comprise a single metapopulation. However, sampling was limited and hence additional molecular work was needed to validate these results.
Using more comprehensive sampling and higher resolution molecular markers, Kyriazis et al. (2024) found four genetic clusters within the sampled region of the species corresponding to the Cape Peninsula, the Greater Cape Town region, the Central Karoo, and Namaqualand. With the exception of the Central Karoo, the sampled regions were not included in the Tensen studies. Consequently, this structure was likely missed from the earlier studies.
It is not surprising the Cape Peninsula represents a unique genetic population given that it is isolated from other larger subpopulations by approximately 800 km2 (Serieys et al. 2023). Inbreeding was notably high in the Cape Peninsula, although interestingly, moderate inbreeding was evident in both the Central Karoo and Namaqualand subpopulations (Kyriazis et al. 2024), both of which are intensively lethally managed (Drouilly et al. 2023; Teichman et al. 2023). These findings indicate that, although the Central Karoo and Namaqualand populations are not isolated by geographic barriers, lethal management may interfere with social structure and dispersal patterns like for other felid species (see Naude et al. 2020 for leopard (Panthera pardus) in lethally managed populations.
The effective population sizes for only the Cape Peninsula and Greater Cape Town regions were quantified (Ne = 28 and 92, respectively). Given Ne >500 represents a genetically healthy and stable population, it is concerning how low Ne is for both subpopulations, in particular the Cape Peninsula, which also show high levels of inbreeding.
Applying the estimated Ne values and the known Ne/Nc ratio for the species of approximately 0.49, the ancestral Ne was quantified to be 35,265. This large estimated ancestral effective population size likely reflects the broader caracal population in South Africa, which was historically large and connected by a high degree of migration (hence previously a single population). However, the consequence of population fragmentation and declines has resulted in genetic isolation and four current genetically distinct subpopulations (Kyriazis et al. 2024).
Additional population genetic work covering the full extent of the species range are needed to fully understand the population genetic structure of the species within the assessment region.
Habitats and ecology
The Caracal occupies a wide variety of habitats, from semi-desert to relatively open savannah and scrubland to moist woodland and thicket, evergreen forest, montane grassland, and arid mountains. It typically ranges up to 2,500 m and above 3,000 m asl in Lesotho (Avenant & du Plessis 2012; du Plessis et al. 2014) and the Ethiopian Highlands (Ray et al. 2005), and it occurs along the Drakensberg (Rowe-Rowe 1992) and Maluti (Avenant 2004) ranges. The species is absent from tropical forests and true deserts, and cover is needed wherever it occurs (Skinner & Chimimba 2005). However, the species increasingly observed in forests within the Indian Ocean Coastal belt forest (Ehlers Smith et al. 2019)
In the Kalahari, Caracals showed a definite selection for dune slope aspect in relation to specific types of behaviour (Melville 2004). They did not select dune crests and dune slopes for specific activities more than expected, and the dune crests were selected less than expected except when killing prey. In the Cape Peninsula, however, caracals selected for dunes while moving across the landscape (Serieys et al. 2023). On rangelands where Caracals co-occur with Black-backed Jackals, the former are more common in the rocky areas than on open plains (du Plessis 2013, Drouilly et al. 2018a). In the North West Province, Caracals occur in all vegetation types, but they generally prefer wooded vegetation types, especially mountain bushveld, and were found to have a local preference for the Gold Reef Mountain Bushveld (Power 2014). Caracals also occur in mountain and coastal fynbos, Strandveld, and in the various Nama and Succulent Karoo vegetation types (Drouilly et al. 2019a). In the Cape Peninsula, habitat selection analyses found that Caracals select areas with Fynbos and Renosterveld vegetation, wetlands, coastal areas, and proximity to urban areas, particularly at night (Serieys et al. 2023). Also in the Cape Peninsula, while foraging, caracals selected for proximity to urban areas, vineyards, and the coast. Gentler slopes and greener areas (higher NDVI) were also selected. The availability of vegetative cover was also important to their foraging ecology, particularly near urban areas (Leighton et al. 2022a). Cape Peninsula Caracals exhibit a strong positive functional response to the urban edge at foraging sites, selecting for urban edge habitats more as they became more available. This reveals that Caracals are able to modify their foraging habitat-selection patterns depending on the landscape in which they find themselves (Leighton et al. 2022a). In the southern Cape Peninsula, where Caracals are selectively foraging along the coast (Leighton et al. 2022a), marine prey is an important part of their diet, particularly Cape Cormorants (Phalacrocorax capensis), and to a lesser extent, African Penguins (Spheniscus demersus). This sustained use of marine subsidies expands their dietary niche (Leighton 2024); however, routine consumption of seabirds is linked with higher pollutant burdens, particularly metals (Parker et al. 2023).
Caracals are less frequently observed in Kruger National Park and adjoining private game reserves, where they appear to be more common in the mixed Combretum spp. woodlands on the granite landscapes. In such protected areas with large carnivores, they may be susceptible to interference competition. One particular competitor is the Leopard, with which they share a similar ecological role (see for example Norton & Lawson 1985; Braczkowski et al. 2012; Power 2014; Muller et al. 2022). Although spatial overlap between these species has been recorded (Jansen 2016: Namaqua National Park and surrounding farms; Cape Leopard Trust unpubl. data: Gouritz Corridor, Boland Mountains; Muller et al. 2022: Cederberg Mountains; M. Drouilly et al. 2018a, 2019a: Anysberg Nature Reserve), interspecific killing of Caracals by Leopards has also been documented (Martins 2010) and Caracals are thus likely to avoid areas where Leopards are prevalent although there are no data to support this claim. In areas where large predators have been extirpated, such as on rangelands and in the Table Mountain National Park, the Caracal often assumes the role of apex predator (du Plessis 2013; Pohl 2015; Drouilly et al. 2018b, 2019a, Leighton et al. 2022a, Serieys et al. 2023, Serieys et al. 2024) ). On Free State farmland, Ferreira (1988) reported that Black-backed Jackal and Caracal numbers inversely fluctuated in some habitats where they co-occur, suggesting that these species may actively limit each other’s numbers in certain areas. However, Caracals marginal probability of occupancy was higher on farmland (where persecution and jackal numbers are high, but apex predators are absent) than in a neighbouring protected area (Woodgate et al. 2023). Caracal and Black-backed Jackal diets do not only overlap to a large extent (Drouilly et al. 2018b), but they have been reported predating on each other’s young (Ferreira 1988; Pohl 2015), and adult Caracal even kill and eat adult Black-backed Jackal (Melville 2004; Q. Martins unpubl. data).
Caracal prey mainly on small- to medium-sized mammals, from small murids to antelope up to > 50 kg, but they also take birds, reptiles up to the size of a large Rock Monitor (Varanus albigularis), and invertebrates (Stuart & Stuart 2013; Pohl 2015; Jansen 2016; ; Drouilly et al. 2018b; Drouilly et al. 2019b; Leighton et al. 2020; Parchizadeh et al. 2023). They are highly opportunistic and take a wide range of species (Leighton et al. 2020; Parchizadeh et al. 2023) and thus have a wide trophic niche (Leighton et al. 2024), even in arid, anthropogenic landscapes (Drouilly et al. 2018b). On the Cape Peninsula, > 70 prey species have been recorded, mostly wild prey, with some exotic and synanthropic species (Leighton et al. 2020), whereas > 20 species were recorded in the diet of Caracal in the arid Karoo, including domestic sheep and goat (Drouilly et al. 2018b) Very little plant material is ingested, and then considered mostly accidental; larger quantities have been found in scats, but then together with scorpion remains (Avenant 1993). They are known to kill and eat other carnivores, including Black-backed Jackal, Aardwolf (Proteles cristata), Bat-eared Fox (Otocyon megalotis), Cape Fox (Vulpes chama), Water Mongoose (Atilax paludinosus), Cape Grey Mongoose (Herpestes pulverulentus), Yellow Mongoose (Cynictis penicillata), Polecat (Ictonyx striatus), African Wildcat (Felis silvestris), Small-spotted Genet (Genetta genetta), Large-spotted genet (Genetta tigrina) and even conspecifics have been listed (e.g. Stuart 1982; Bekker 1994; Avenant 1993; Kok 1996; Melville 2004; Braczkowski et al. 2012; Pohl 2015; Drouilly et al. 2018b, 2019b; Serieys et al. 2023, Leighton et al. 2020). Caracals sometimes scavenge (Mills 1984; Avenant 1993; Nowell & Jackson 1996; Drouilly et al. 2019b; Serieys unpubl.data); in the PNR, however, these instances could be traced to non-territorial cats and not the dominant males or females (Avenant 1993), but in the Central Karoo, adult, territorial male and female Caracals were recorded to scavenge on greater kudu (Tragelaphus strepsiceros), Nguni cow and sheep (4.7% of sites investigated as part of a study on diet through the identification of 107 GPS clusters, Drouilly et al. 2019b). Like Leopards, they are known to hoist their kills into trees (see Mills 1984; Davies 1997), and may also return to carcasses; for example, in the PNR, a female with young has been documented to return to a carcass for up to four nights (Avenant 1993).
Relative to their small size, Caracals can readily capture prey larger than themselves, such as Springbok (Antidorcas marsupialis) (e.g. Mills1984; Avenant 1993; Pohl 2015), Mountain Reedbuck (Redunca fulvorufula) (Moolman 1984; Pohl 2015), Grey Rhebok (Pelea capreolus) (Pringle & Pringle 1979; Palmer & Fairall 1988; Stuart 1982; Stuart & Hickman 1991; Bekker 1994), Southern Bushbuck (Tragelaphus sylvaticus) (Stuart 1982), sheep (e.g. Stuart 1982; Moolman 1984; Ferreira 1988; Brand 1989; Bekker 1994; Gunter 2008; Strauss 2009; van Niekerk 2010; Pohl 2015; Drouilly et al.2018b, 2019b), goats (Moolman 1984; Brand 1989; Gunter 2008; Blaum et al. 2009; van Niekerk 2010; Badenhorst 2014; Jansen 2016; Leighton et al. 2020; Drouilly et al. 2018b, 2019b), and bontebok (Damalisus pygargus) (Leighton et al. 2020). Together with Black-backed Jackal, Caracal is the major damage-causing species on small-livestock farms (du Plessis 2013; Drouilly et al. 2021, 2023) but Caracals always selected wild prey species over domestic livestock on farmland in the Central Karoo (Drouilly et al. 2018b). More recently, Caracals have also been implicated as causing significant damage to the cattle (Thorn et al. 2012; Badenhorst 2014) and game farming (Power 2014; Schepers 2016) industries. They capture the young of virtually all game that are stocked, some of which (scarce species and colour variants) may be extremely valuable. In the case of game farms, there generally is adequate small prey, as farmers protect buffer prey species (Drouilly et al. 2018a), but game populations are often small, so even modest levels of Caracal predation may have a significant impact to a landowner (Thorn et al. 2011; Power 2014; Schepers 2016). On the other hand, small-stock farmers in the central parts of South Africa often complain that they do not get support from the game ranches and nature reserves in controlling Caracal (and Black-backed Jackal) as these areas are seen as sources where both species multiply (Drouilly et al. 2019c). In the small-livestock and cattle industries, the Caracal’s direct impact is mostly on the younger individuals, but some adults are also killed (Drouilly et al. 2019b). Some of these individuals (e.g. breeding stock) carry a higher-than-average financial value and, again, their loss is a major cause forthe negative perceptions farmers display towards Caracals (Drouilly et al. 2021). A few instances of surplus killing of small-stock have also been documented (Skinner 1979; Stuart 1986; Brand 1989; Drouilly et al. 2019b). In the field of conservation, concerns of Caracal predation upon threatened colonies of African Penguins (Spheniscus demersus) in the Western Cape have recently been raised (Leighton et al. 2024). We now also know that some Caracals kill domestic cats in golf estates (Nattrass and O’Riain, 2020b) and when domestic cats enter natural areas (Leighton et al. 2020), in the same province. However, around Cape Town, where Caracals are selectively foraging on the urban edge (Leighton et al. 2022a), domestic animals (including domestic cats) comprise < 4% of Caracal diet (Leighton et al. 2020).
Home range studies have mostly been conducted in South Africa and Namibia (see Bothma & Le Riche 1994; Moolman 1986; Avenant 1993; Avenant & Nel 1998; Marker & Dickman 2005). Environmental variables such as the size, density, composition and distribution of available prey, the type and density of sympatric predators, habitat characteristics (including the amount of cover), and the degree of persecution by humans have all been indicated as having a marked impact on Caracal home range size and use (see du Plessis 2013). Consequently, relatively large home ranges were observed in more arid areas (Bothma & Le Riche 1994: one male in the KTP had a range size of c. 300 km²; Van Heezik & Seddon 1998; Marker & Dickman 2005: three males on Namibian rangeland, averaging 316 km²), mountainous terrain (Norton & Lawson 1985: a single male tracked in mountains in the Western Cape, 65 km²; Q. Martins unpubl. data: three males in the Cederberg mountains, averaging 184 km², and one female, 44 km²) and on farmland (Moolman 1986: male and female home ranges larger on farms around the MZNP than inside the Park; also Marker & Dickman 2005, see above; Drouilly et al. unpubl. data: two males on Central Karoo farms, averaging c. 56 km²). In the Cape Peninsula where 25 Caracals were tracked with GPS collars, the average adult male (n =9) home range size was 74.1 km², while adult female (n = 7) home ranges were on average 17.4 km². Subadult male (n = 7) home ranges were considerably smaller than adult male home ranges and averaged 33.0 km2 (Serieys et al. 2021). Inside the PNR and the MZNP, home ranges were relatively small: two male home ranges averaged 26.9 km² in PNR (Avenant & Nel 1998), while seven males both in and adjacent the MZNP had home ranges of between 15 and 19 km² (Moolman 1986). Female home ranges are considerably smaller than those of males, as is the case with most solitary felids, and can be attributed to the larger males’ higher energy requirements (SMNs), the fact that they may select for larger prey, or the males’ social needs, where one male’s home range typically overlaps with those of a number of females (Avenant 1993). In the isolated Cape Peninsula, adult male home ranges were observed to overlap extensively (Leighton et al. 2022a).
Little is known about the reproduction and maternal behaviours of Caracals, particularly in free-ranging systems. In the Cape Peninsula, isolated by the City of Cape Town, observations were made of four GPS-collared female caracals which birthed their young during September and December, suggesting a birth pulse in the Southern Hemisphere spring and summer (Serieys et al. 2024). Sightings observations of caracals with young in the Cape Peninsula and the Central Karoo support the timing of this birth pulse (Serieys and Leighton, unpubl.data; Drouilly, unpubl. data). Seven primary den sites were documented for the four females, although two of the four females were disturbed during denning and possibly moved their den sites as a result of attempted investigations of the dens while in use. Two females that were undisturbed used a single den site. All females intensively denned for a period of roughly 6 weeks, after which they utilised short-term auxiliary dens (used on average for four days). For the only female for which complete GPS-collar data was available, she abandoned the use of auxiliary dens after 91 days post-parturition indicating the use of auxiliary dens for potentially six weeks after leaving the primary den site. The four females exhibited central place foraging while denning home ranges restricted to an average of 31% of their size using their full dataset. Trips away from the dens averaged 12.3 hours, with an average maximum time away of 41 hours. The average distance travelled away from the den sites was 1135 m, with an average maximum distance of 2639 m. Utilising 3-hour GPS-data, Serieys et al. (2024) calculated that the average distance females travelled when away from the dens was 8299 m. While these observations fill in knowledge gaps about Caracal reproductive ecology, data from other regions are needed.
Ecosystem and cultural services: Caracals have a wide and almost uninterrupted distribution in South Africa, where they feed opportunistically on a wide variety of prey, ranging from invertebrates, reptiles and birds to sympatric carnivores and mammals of up to > 50 kg. They, therefore, serve as key regulators in the ecosystem, suppressing both competing predators and regulating prey populations, and are therefore important for the conservation of biodiversity (see e.g. Avenant 1993; du Plessis 2013; Pohl 2015; Tambling et al. 2018). The importance of this role increases in the different regions of South Africa, such as the Central Karoo, and large areas of the Western Cape, Gauteng and the Free State, where Caracals fill (mostly together with Black-backed Jackal) the role of apex predator (Tambling et al. 2018). The exclusion of Caracals from, or their severe suppression in, ecosystems will almost certainly have direct negative impacts, such as smaller-predator release, an eruption of prey numbers, an overexploitation of associated species, and a potential decrease in vertebrate and invertebrate species. Many of these competing predator and prey species can themselves be damage-causing to human activities: e.g. rodents destroying crops, Rock Hyrax competing for forage with sheep, mole-rat tunnels causing damage to tractors and ploughs, carnivores (e.g. Yellow Mongoose and Black-backed Jackal) and rodents carrying diseases, and carnivores that are predating on livestock or poultry (e.g. Black-backed Jackal, many of the mongooses, genets and otters) (see du Plessis 2013). Indirectly, the exclusion of Caracal may potentially also start a cascade effect leading to an overall decrease in biodiversity and healthy ecosystem functioning, but research on the ecological role of Caracal still needs to be conducted to confirm those hypotheses.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
| 8.2. Desert -> Desert – Temperate | – | Suitable | – |
Life History
(reported information found in Veals et al. 2020)
Generation Length: Estimated 3 years
Age at Maturity: Female or unspecified: Between 7 and 12 months depending on areas.
Age at Maturity: Male: Between 9 and 14 months depending on areas.
Size at Maturity (in cms): Female: 71-102 cm + 21-34 cm for the tail
Size at Maturity (in cms): Male: 75-108 cm + 21-34 cm for the tail
Longevity: (Not specified)
Average Reproductive Age: Females likely do not begin mating until 14 months of age and can remain reproductive until 18 years of age.
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 78-81 days. Reproduction is timed with the phenology of prey species, peaking when vegetation, and therefore prey, production is highest. Thus, timing of reproduction varies by geographic location.
Reproductive Periodicity: Births are typically separated by 1 year, but consecutive litters can be spaced approximately 10 months apart.
Average Annual Fecundity or Litter Size: Litters average 2–4 kittens but up to six kittens may be born.
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: No
Systems
System: Terrestrial
General Use and Trade Information
Caracals can be trophy-hunted in South Africa and Namibia, for a price varying from US$650–1,500, depending on the hunting company, the type of hunt (with a pack of hounds or not), and the type of weapon used (rifle or bow). In some provinces, such as the Eastern Cape, Limpopo and Free State provinces, only a hunting license is necessary to do so if the Caracal hunt takes place on own property, but a permit is needed when hunting on someone else’s property; in the Western Cape and North West provinces, hunters now have to apply for a special Caracal hunting permit, even if hunting on their own property.
In their struggle to combat stock losses, some farmers legally and illegally hunt Caracal themselves (Drouilly et al. 2023), make use of damage-causing animal (DCA) or “specialist” professional hunters to remove Caracal (and Black-backed Jackal) from their properties, or they send some of their workers for DCA courses, which deal with a fairly large variety of precautionary and removal techniques (van Niekerk 2010; du Plessis 2013; Badenhorst 2014; Schepers 2016). These training and hunting businesses have now become small industries, and advertisements/banners on the internet and hunters’ vehicles have become commonplace. When making use of hunters, a farmer will typically cover the hunter’s fuel (km) tariff plus pay an amount per DCA removed; the cost for one Caracal removed is in the order of R1400/individual plus a night fee or cost for the fuel and mileage (about R5/ km) (M. Drouilly, pers. comm. April 2024). When a Caracal is caught or hunted, the carcass is often available for the farm workers to skin and eat, or it is simply discarded in the veld.
An international trade exists for Caracals to be kept as pets, especially in the United States of America (USA) Russia, Ukraine, Canada and the Netherlands (see advertisements openly available on the internet). Although the number of kittens exported is considered to be low, there are indications that this trade may be increasing (e.g. in the North West Province, no kittens were exported in 2015, 10 were exported in 2016, and within the first three months of 2017, four were already exported; North West Provincial Government records). Between January and June 2018, a single American wildlife trader illegally imported 18 Caracals and 12 Servals (Leptailurus serval), from a captive breeder in South Africa, according to court documents.
The average price for a kitten is US$1,500–2,000 but can go up to US$7,500 in the USA. In South Africa, Caracal kittens can be bought for as low as US$200. Some caracals come from legal breeding centres but with the increase of the black-market trade of wildlife online, more and more Caracals and other wild cat species seem to be traded illegally. Some provinces, including North West and the Free State, do confiscate such animals if owners do not have the correct permits to keep them
The capture and removal of wild Caracals for import into captivity and trade is currently considered a minor threat, but the situation is monitored. A typical fine for keeping an animal not protected by Threatened or Protected Species (TOPS) legislation is R750. On farms and ranches where predators are lethally controlled, it often happens that a female is killed, leaving the kittens on their own. These kittens are then sometimes adopted as pets (Moolman 1986; M. Drouilly pers. obs. 2012–2015; N.L. Avenant pers. obs. 1996–2016), and it is thus not uncommon for farming families to raise orphaned Caracals.
Apart from a few stories in the folklore (e.g. Greaves & Clement 1993), Caracals have apparently not been used for specific cultural purposes by the Basotho (Avenant 2004; Moffett 2010) or other people in South Africa (N.L. Avenant pers. obs. 1989–2016). They are, however, eaten by a number of cultural groups, including the Basotho, Xhosa and mixed farm workers in the Free State, Nama Karoo and Succulent Karoo (N.L. Avenant pers. obs. 1989–2016; M. Drouilly pers. obs. 2012–2015).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Source of meat on rangelands with predator control measures. | Yes | Trophy hunting. Pet trade. |
National Commercial Value: Yes
International Commercial Value: Yes
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Increasing in some provinces.
Threats
Threats to caracals are numerous and include habitat loss, persecution and lethal management, vehicle collisions, poaching, predation, disease, and poisoning. In southern Africa, particularly in South Africa and Namibia, caracals cause significant damage in the small livestock (van Niekerk 2010; also see du Plessis 2013, Drouilly et al. 2019c), cattle (Thorn et al. 2012; Badenhorst 2014) and game farming (Power 2014; Schepers 2016) industries, and they are thus are subject to persecution through hunting, trapping and, in some areas, even illegal poisoning (Drouilly et al. 2023). In the Central Karoo, interviews with 77 small-livestock farmers showed that 97.4% reported using lethal methods to prevent predation of their livestock by Caracal and Black-backed Jackal. Half of them also reported using poison either regularly (17%) or as a last resort (33%) for dealing with the most elusive and difficult-to-catch individuals (Drouilly et al. 2023). Stuart (1982) recorded that over the years 1931–1952, an average of 2,219 Caracals / year were killed in control operations in South Africa’s Karoo ecosystem. In a similar environment, Namibian farmers responding to a government questionnaire reported killing up to 2,800 Caracals in 1981 (Nowell & Jackson 1996). In the Karoo between April 2014 and April 2015, professional hunters and small-livestock farmers killed 168 Caracals during five blanket hunts that lasted on average 8.6 nights, which represents an annual persecution rate of 1.1 Caracals/100 km² without taking into account other individuals killed through routine predator control, such as trapping (Drouilly, unpublished data). Cattle farmers in the North West Province have indicated Caracal as a serious DCA (Badenhorst 2014), and game farmers in the Limpopo Province currently consider Caracal as one of the three major predators of game (Schepers 2016). In the Central Karoo, 63% of 77 small-livestock farmers interviewed about their relationships with predators ranked Caracal as the second predator responsible for the most livestock losses after the Black-backed Jackal. Yet, the percentage of lamb losses experienced by those farmers was not a significant variable explaining their tolerance for the felid (Drouilly et al. 2021). In the North West Province, around 50% (N = 198) of surveyed game farms complained about the Caracal as a problem animal (Power 2014). The annual persecution rate reported by farmers in the North West Province was 1.1 Caracal / 100 km² (Thorn et al. 2012), which compares favourably to the scale estimated for the then Cape Province of 1.6 Caracal / 100 km² (Brand 1989). As seen above, this rate was higher in the Karoo and may be higher in areas like the southern Free State where stock losses due to predation are reported to be amongst the highest in South Africa (van Niekerk 2010). In the Western Cape province of South Africa (and probably in other areas), snares are an important threat to Caracals. Since the Western Cape Snare Response Plan was launched as part of Snare Free initiative (https://snarefree.co.za), live Caracals caught in snares have been the species most responded to, with 12 incidents reported between May 2021 and March 2024. In 9 cases, the Caracal was found alive caught in a snare and in 4 cases, the Caracal carcass was found in a snare. These incidents were from the Cape Winelands, the Cape peninsula, Overberg / Bot Rivier, Worcester area, and around Mossel Bay and represent only the cases scientists are aware of and are thus an underestimation. Van Tonder (2024) conducted 309 interviews with farmers and conservation officials across the entire Western Cape between 2021 – 2022 and also found that snares were an important threat to wildlife, including Leopards and Caracals, with snares being reported on 55% of landowners’ properties.
Brand (1989) found that Caracals were responsible for the loss of up to 5.3 domestic stock / 100 km² per year and recorded 0.02–1.6 Caracal(s) killed or captured / 100 km² per year in the former Cape Province of South Africa. In more recent surveys, livestock farmers have indicated that Caracal is responsible for between 9% (in Mpumalanga) and 36% (in the Western Cape Province) of small-stock predations (van Niekerk 2010) and 11% of cattle calf predation in the North West Province (Badenhorst 2014). Although these figures may need further investigation to ascertain actual predation accuracy, they provide a good reflection of livestock farmers’ perceptions that Caracal is an important DCA and is indicative of the danger of persecution that this species is likely facing (du Plessis et al. 2015). Severity of depredation by Caracal may be related to a number of factors, including the type, composition and density of prey, the geography of the specific area, the presence and types of husbandry techniques, and the season (i.e. the reproductive season of the livestock animals, natural prey and the Caracal themselves) (see Avenant & du Plessis 2008; Stuart & Stuart 2013; du Plessis 2013; Pohl 2015; Teichman et al. 2015). This information should be taken into account when farmers plan their husbandry and management programmes. For instance, the risk of losing small-stock to Caracal may increase when natural prey numbers are low – such as during the seasons when prey densities are naturally at their lowest, and or on properties where prey densities are low because of management practices. The risk may then further increase if this “lean” season overlaps with the lambing season and/or the period when Caracals have their own young, and or when the stock (lambs) spend more time in the Caracal’s preferred habitat (≈ rocky areas/“kliprantjies” and kopjes) (Avenant & du Plessis 2008). Other precautionary techniques, such as the use of livestock guarding dogs (LGDs), kraaling (where feasible), electric fencing, and a range of others, and even better when using a combination of these (see du Plessis 2013) – for example, livestock guarding dogs with herders (Drouilly et al. 2020) – can potentially decrease the number of stock and wildlife losses and therefore also the persecution on the Caracal population. Other long-term beneficial management actions may be to remove only the damage-causing individuals (with the help of e.g. poison collars) and leaving the non-damage-causing territorial cats to lessen the time that non-territorials cats spend (and feed) in the area (Avenant & du Plessis 2008; Avenant et al. 2009; du Plessis 2013).
With more farms in fewer (larger farmers’) hands, as well as with South Africa’s progressive labour legislation for farm workers that have led to fewer farmers being able to keep more employees on their farms (for cost and logistical reasons), fewer workers are patrolling the farms searching for predator signs, repairing fences and protecting livestock. This, together with many farmers’ hopeless perceptions that the stock-loss problem is just increasing (Drouilly et al. 2019c), contributes to the ongoing illegal practise of poisoning with, for example, Poison 1080 (Sodium Fluoroacetate) and Two-Step (carbamate insecticide found in the pesticide Temik) over alarmingly large areas (see van Niekerk 2010; du Plessis 2013; Drouilly et al. 2023). This and other blanket control methods are not only suspected to be ineffective in the long term, but also have severe detrimental impacts on the whole ecosystem (see du Plessis 2013). For example, in the Karoo, lethal control of Caracals and Black-backed Jackals was associated with increased livestock losses the following year, providing further evidence that lethal control of mesopredators in this context is probably counterproductive (Nattrass et al. 2020a).
On the Cape Peninsula, heavily influenced by urban activities, vehicle collisions comprise a significant portion of mortalities (>70 %). Other threats in the region include indiscriminate poaching, domestic dog attacks, lethal management surrounding African Penguin management (Serieys et al. 2023, Leighton et al. 2022a), and exposure to toxic anticoagulant rodenticides (Serieys et al. 2019) and other pesticides, including DDT (Leighton et al. 2022b). Additionally, this population has dietary exposure to PCBs (Leighton et al. 2022b) and metal pollution, including lead, arsenic, mercury and selenium (Parker et al. 2023). Exposure to these environmental pollutants is linked to use of human-transformed landscapes, including vineyards, roads, and urban areas. There is evidence of physiological effects of exposure, with increased measures of immunological response that may increase susceptibility to disease (Leighton et al. 2022; Parker et al. 2023). Most individuals were exposed to multiple pollutants and pesticides and their interactive effects are unknown. Disease may also threaten the Cape Peninsula population (Viljoen et al. 2020). Further, due to spatial and genetic isolation, the Cape Peninsula Caracal population is relatively inbred compared to populations elsewhere in the country (i.e., Greater Cape Town, Namaqualand and Central Karoo), and going forward extinction risk is elevated in scenarios of low migration and increased mortality from anthropogenic threats (Kyriazis et al. 2024). Given the rapid urbanisation of the Cape Town area and the associated increase in threats, such as habitat fragmentation, vehicle traffic, and pesticide use, this isolated population requires a higher level of protection to avoid local extinction.
Conservation
Caracal populations within their African range are included on Appendix II of CITES. In sub-Saharan Africa, the species is protected from hunting in about half of its range states (Nowell & Jackson 1996). In Namibia and South Africa, however, Caracals are considered DCAs, which permits landowners to kill the species, with varying levels of restrictions.
In the Western Cape, landowners need a specific permit that can be issued for six months by CapeNature to kill the species, only through the “call and shoot” method. No permits are issued for the use of helicopters, gin traps (which are illegal) and soft traps. In the other provinces of South Africa, an annual DCA permit is issued with unlimited species numbers to hunt at night and for the use of a helicopter. Methods include cage traps, dogs and gin traps. In the Free State and KwaZulu-Natal, property owners do not need a permit to hunt Caracals on their own property, though in the North West Province one does require one now (but this may prove hard to enforce). In the North West, Caracals are inadvertently caught in cage traps set for routine DCA control aimed at TOPS species (for example, Leopards) and many of them are simply relocated nearby.
Although Caracals are present in most national parks and provincial nature reserves, there are no protected areas specifically established for this species.
Caracals probably do respond well to reintroduction, as is evident in how they have seemingly colonised, or recolonised, the province of KwaZulu-Natal after introductions to the KwaZulu-Natal Drakensberg and Ithala Game Reserve (see Pringle 1977; Rowe-Rowe 1978; Barnes 1991; Rowe-Rowe 1992). Introduction is, however, not considered a priority intervention for this species in most of its range. However, on the Cape Peninsula, where the population is spatially and genetically isolated, genetic rescue combined with increased efforts to reduce anthropogenic threats will be required to ensure the long-term persistence of Caracals (Kyriazis et al. 2024).
Persecution on livestock and game farms remains the largest threat for Caracal in southern Africa. Proposed interventions and management recommendations are discussed below.
Recommendations for land managers and practitioners: There is currently no species management plan for this species, but many researchers and the Predation Management Forum of South Africa (PMF) are working towards a more sustainable and best practise plan (Avenant et al. 2006; de Waal 2009; PMF website; PMF pers. comm. 2017). A scientific assessment of livestock predation and its management in South Africa (Kerley, Wilson and Balfour 2018) was also published. Public awareness, education of landowners, increased collaborative research with landowners, conservation managers and researchers on board, and continuous feedback to especially livestock and wildlife owners, are all necessary to address the current predation conundrum. Livestock and wildlife owners can take cognisance of the fact that such large, focussed, initiatives are already in progress. Examples include the Canis–Caracal Programme (University of the Free State and NMB); the national Predation Management Information Centre (UFS and PMF); Neil Viljoen Predation Management (assisted by the PMF); Cape Leopard Trust; Endangered Wildlife Trust, Carnivore Conservation Programme; Cheetah Conservation Fund (CCF); Cat Conservation Trust; and an increasing number of studies by the universities of Cape Town, Free State, KwaZulu-Natal, Pretoria, Stellenbosch, NMMU, and others.
At this stage, and with the knowledge that circumstances may differ markedly between areas/farms, the following general recommendations can be made to land managers (based on the research studies produced so far on the topic):
- Healthy natural prey populations, and temporal planning of farm management around the time of year when these densities are high or low, can significantly reduce the risk of losses; this includes when and where the stock lamb, and where they forage during which time of the year.
- Be aware of the Caracal’s behaviour, such as habitat preferences, the peak times when Caracals’ energy needs are highest (for example, when females lactate/have young), their social structure, and the important role that territorial individuals can play in limiting the time that non-territorial cats spend on the property (following Avenant 1993, it has been proposed that non-territorial cats are more prone to take the easy and “not-natural” prey; an idea currently further under research).
- Search for and apply methods (e.g. poison collars) that only take out the culprit (in other words, strive towards a situation where territorial cats do no or relatively little damage and where they can assist in the management process by excluding non-territorial animals); be aware that blanket control methods may only offer short-term relief on the property, and will most probably lead to escalated problems/costs for the owner, surrounding owners and the ecosystem in future.
- Search for and apply the best husbandry methods that suit the situation and management style.
- Use a combination of methods (and the steps indicated here), and change/rotate them to prevent habituation.
- Be aware that success requires dedication and continuous hard work, and that there is no single method that will provide a solution on its own.
- Know that relief from your current situation will most probably not happen overnight and that the situation will gradually change; persevere with your intended management plan.
- Strive to get as large an area (most properties) as possible to follow the same holistic management principles.
- Be aware that areas that have both Caracal and Black-backed Jackal present are in for a larger challenge.
- Invite researchers and conservation officials to be involved with research on your farm, and in such way find out what works best for your specific situation.
Nature conservation agencies should strive to work together with land managers, but also support the PMF in such a way where all or most of the Caracal-killed records, as well as the stock- and wildlife-loss records, are sent to the Predation Management Information Centre at the University of the Free State (see https://pmfsa.co.za/), and in that way assist to get an overall picture of what happens where, what is the resulting effect of management efforts in that area, and also to assess the impact of predator control on this species’ populations.
Research priorities: As an important damage-causing species in large parts (> 75%) of southern Africa, more research is needed on farm management methods and tools that allow for the protection of livestock rather than the management of mesopredators themselves, how the predators react to different management strategies, and how to mitigate conflicts with human livelihoods, especially in the livestock and game farming industries. Additionally, we can benefit from a regional focus on the status and ecology of the species. More generally, research is needed on:
- Population size and trends (this can be promoted by citizen science, especially in areas where densities are low or where very specific research questions are asked).
- Impact of livestock and game farming on Caracal population size and breeding ecology, compared with undisturbed large natural areas (du Plessis et al. 2015).
- Impact of human–predator conflict management strategies (both lethal and non-lethal) on Caracal numbers, reproduction, social structure, habitat selection and dispersal.
- Landscape scale trophic cascades resulting from the localised removal of Caracals (as seen in Australia).
- Landscape genetics and determination of source/sink areas.
- At a national scale, the number of Caracals killed during predator-control operations.
- Spatial ecology of the species with size of home range in relation to prey density and human activity.
- Effects of sympatric apex predators on the population size/density, survival and behaviour of Caracals (and are these effects density dependent?).
- Predatory impact of the Caracal on game ranches.
- Whether there are “hotspots” of Caracal predation, and if yes, the characteristics of those “hotspots”.
- Whether increased densities of natural prey buffer livestock losses caused by Caracals.
- Non-lethal control methods for the Caracal (i.e. scent avoidance, bio-fencing, etc.).
- Evidence for range expansion/contraction.
- Development of an App to record livestock depredation and mortality of Caracal on farmlands (aimed at livestock and game farmers in particular).
- Investigate the impact of anthropogenic threats (e.g., vehicle collisions, environmental pollutants, disease and genetic isolation) on the health and survival of populations of Caracals exposed to rapid urbanisation.
- Determine the success of genetic rescue and increased protection to reduce anthropogenic risks for the spatially and genetically isolated Cape Peninsula Caracal population.
The following research projects are currently ongoing:
- Canis–Caracal Programme (CCP), run by the African Large Predator Research Unit, UFS: aims at finding solutions to reduce the widespread impact of predation on the livestock industry (national). Contact details: Prof. H.O. de Waal, Department of Animal, Wildlife and Grassland Sciences and African Large Predator Research Unit (ALPRU), PO Box 339, Internal Box 70, University of the Free State, Bloemfontein, 9300, South Africa. Email: dewaalho@ufs.ac.za.
- Predation Management Information Centre (PMIC): collating and analysing reliable information on predation and predation management methods, which will be made available continuously to a management information system (MIS). A team of dedicated staff members handles calls and enquiries. Experts in the team are available to provide advice to farmers. The centre is also responsible for the management of information and resources. Aim: to generate information that can be used to reduce the widespread impact of predation on the livestock industry. A collaborative initiative between the UFS and the PMF. Contact details: Email: PredationMC@ufs.ac.za. Telephone: 051 401 2210 (on week days from 08:00–16:00).
- Cape Leopard Trust (CLT): regularly provide advice and assistance to farmers when they experience predation by Caracals. As part of the collaboration, Snare Free initiative (https://snarefree.co.za), the NGO also helps respond to Caracals (and other wildlife) caught in snares. In case you discover a live wild animal caught in a snare in the Western Cape, call the Snare Free hotline for assistance on 076 127 8485. Contact details: Helen Turnbull, CEO. Email: contact@capeleopard.org.za; website: http://www.capeleopard.org.za.
- Urban Caracal Project: participants in this project include the University of Cape Town, Panthera, and South African National Parks, and private landowners in Cape Town. This project aims to 1) establish baseline information – distribution, population size, health status of individuals – about the Caracal population in the Cape Peninsula; 2) evaluate the effects of urbanisation on the behaviour, movement patterns, diet and genetic health of Caracals; and 3) assess the threats to survival for Caracals. Contact details: Dr Laurel Serieys, Project Founder. Email: Lserieys@panthera.org; Dr Gabriella Leighton, Project Coordinator. Email: gabi.leighton1@gmail.com; website: www.urbancaracal.org.
- Cat Conservation Trust: involved in ex-situ breeding, awareness and research programmes on four wild cat species, including the Caracal, in the Eastern Cape Karoo in South Africa. Contact details: Richard and Marion Holmes. Email: info@karoocats.org;
- Landmark Foundation’s Leopard & Predator Project: based in the Eastern Cape, and advocate non-lethal control measures of species such as Caracal. Contact details: Dr Bool Smuts, Director. Email: bool@landmarkfoundation.org.za.
Encouraged citizen actions:
- Report sightings on virtual museum/social media platforms (for example, iNaturalist MammalMAP, and www.UrbanCaracal.org), especially outside protected areas. Caracal sightings are very rare, so the use of camera traps by citizen scientists is encouraged as more data can accrue this way than through direct observations.
- For the farmers and hunters controlling the species, it is crucial that they report all the dead animals (trapped, shot or poisoned), as well as their livestock losses due to the species, with photographs and GPS coordinates, to the national Predation Management Information Centre (PMIC; email: PredationMC@ufs.ac.za).
- Livestock farmers can get on board by encouraging scientific research on their properties.
- Livestock farmers can actively monitor and record the effectiveness of the management methods they are implementing. Of more value would be if these management methods are designed, implemented and monitored on a sound scientific basis (through liaison with the scientific community) and if the results are shared and published in peer-reviewed literature as a way to promote accuracy, reliability and application.
Bibliography
Avenant NL, Steenkamp E, de Waal HO. 2009. Reviewing a case study on the effects of different management options to reduce predation on small livestock in the Karoo. Southern African Wildlife Management Association Symposium. Cape Town, South Africa.
Avenant NL, de Waal HO, Combrinck W. 2006. The Canis–Caracal Programme: a holistic approach. National Workshop on the Holistic Management of Human–Wildlife Conflict in South Africa. Ganzekraal Conference Centre, Western Cape.
Avenant NL, du Plessis J, Senoge J. 2014. Mammals of the Mohale Dam catchment. Specialist report for Contract 1273: Biological Resources Monitoring within Phase 1 of the LHWP Catchments 2013-14. Report no. AEC/14/12 submitted by Anchor Environmental Consultants to the Lesotho Highlands Development Authority.
Avenant NL, du Plessis JJ. 2012. Letšeng Expansion Project: Project Kholo. Mammal Specialist Report. Environmental Resources Management (Southern Africa) Pty Ltd.
Avenant NL. 1992a. Neem rooikat in diens. Custos 21: 35–37.
Avenant NL. 1992b. Stock thief or ally? Custos 21: 39–41.
Avenant NL. 1993. The caracal Felis caracal Schreber, 1776, as predator in the West Coast National Park. M.Sc. Thesis. University of Stellenbosch.
Avenant NL. 1996. Die rooikat: gehaat of geliefd? Culna 51: 18–20.
Avenant NL. 2004. Conserving Mountain Biodiversity in Southern Lesotho: Mammal report. United Nations Development Plan, Lesotho.
Avenant NL. 2007. Rooikat & rooijakkals: is daar hoop vir kleinveeboerdery in Suid-Afrika? Culna 62: 3–5.
Avenant NL. 2012. Rooikat en jakkalsprobleme: die rooies, die groenes, die blindes en die opportuniste. Culna 67: 30–32.
Avenant NL. Die rooikat: aktiwiteite en sosiale gedrag. Culna 52: 23–24.
Avenant, N. L. and Nel, J. A. J. 1998. Home-range use, activity, and density of caracal in relation to prey density. African Journal of Ecology 36: 347-359.
Avenant, N.L. 2011. The potential utility of rodents and other small mammals as indicators of ecosystem “integrity” of South African grasslands. Wildlife Research 38: 626–639.
Avenant, N.L. and du Plessis, J.J. 2008. Sustainable small stock farming and ecosystem conservation in southern Africa: a role for small mammals? Mammalia 72: 258-263.
Avgan B, Henschel P, Ghoddousi A. 2016. Caracal caracal. The IUCN Red List of Threatened Species 2014.
Badenhorst CG. 2014. The economic cost of large stock predation in the North West Province of South Africa. M.Sc. Thesis. University of the Free State.
Barnes B. 1991. Giants Castle: A Personal History. Privately published, Pietermaritzburg, South Africa.
Bekker SJ. 1994. Die invloed van sosio-ekologiese veranderlikes op die aard en omvang van die probleemdierkwessie in suid-oos Kaapland. M.Sc. Dissertation. University of Stellenbosch.
Bergman DL, de Waal HO, Avenant NL, Bodenchuk MJ, Marlow MC, Nolte DL. 2013. The need to address black-backed jackal and caracal predation in South Africa. In: Armstrong JB, Gallagher GR. (ed.), Wildlife Damage Management Conference., pp. 86–94. Lincoln, USA.
Blaum, N., Tietjien, B., and Rossmanith, E. 2009. Impact of livestock husbandry on small- and medium-sized carnivores in Kalahari Savannah Rangelands. Journal of Wildlife Management 73: 60-67.
Bothma J du P, Le Riche EAN. 1994. Range use of an adult male caracal in the southern Kalahari. Koedoe 37: 105–108.
Bothma J du P, Suich H, Spencely A. 2009. Extensive wildlife production on private land in South Africa. In: Suich H, Child B, Spenceley A. (ed.), Evolution and Innovation in Wildlife Conservation: Parks and Game Ranches to Transfrontier Conservation Areas, pp. 147–162. Earthscan, London, UK.
Braczkowski, A., Watson, L., Coulson, D., Lucas J., Peiser, B. and Rossi, M. 2012. The diet of caracal, Caracal caracal in two areas of the southern Cape, South Africa, as determined y scat analysis. South African Journal of Widllfie Research 42(2): 111-116.
Brand, D. J. 1989. Die verspreiding von rooikatte en bobbejane in Kaapland, en die skade wat hulle in die landbou hier berokken – Rooikatte. Die beheer von rooikatte (Felis caracal) en bobbejane (Papio ursinus) in Kaapland met behulp van meganiese metodes, Universiteit van Stellenbosch.
Davies R. 1997. Caracal Felis caracal. In: Mills G, Hes L. (ed.), The Complete Book of Southern African Mammals, pp. 186–187. Struik Winchester, Cape Town, South Africa.
Deacon F. 2010. Aspekte rakende die ruimtelike ekologie van die rooijakkals (Canis mesomelas) as probleemdier in die Suid-Vrystaat. M.Sc. Dissertation. University of the Free State.
Drouilly, M., Clark, A., & O’Riain, M. J. (2018a). Multi-species occupancy modelling of mammal and ground bird communities in rangeland in the Karoo: A case for dryland systems globally. Biological Conservation, 224, 16-25.
Drouilly, M., Nattrass, N., & O’riain, M. J. (2018b). Dietary niche relationships among predators on farmland and a protected area. The Journal of Wildlife Management, 82(3), 507-518.
Drouilly, M., & O’Riain, M. J. (2019a). Wildlife winners and losers of extensive small-livestock farming: a case study in the South African Karoo. Biodiversity and Conservation, 28, 1493-1511.
Drouilly, M., Nattrass, N., & O’riain, M. J. (2019b). Global positioning system location clusters vs. scats: comparing dietary estimates to determine mesopredator diet in a conflict framework. Journal of Zoology, 310(2), 83-94.
Drouilly, M., Tafani, M., Nattrass, N., & O’Riain, J. (2019c). Spatial, temporal and attitudinal dimensions of conflict between predators and small-livestock farmers in the Central Karoo. African Journal of Range & Forage Science, 35(3-4), 245-255.
Drouilly, M., Kelly, C., Cristescu, B., Teichman, K. J., & O’riain, M. J. (2020). Investigating the hidden costs of livestock guarding dogs: a case study in Namaqualand, South Africa. Journal of Vertebrate Biology, 69(3), 20033-1.
Drouilly, M., Nattrass, N., & O’Riain, M. J. (2021). Beauty or beast? Farmers’ dualistic views and the influence of aesthetic appreciation on tolerance towards black-backed jackal and caracal. PLoS One, 16(3), e0248977.
Drouilly, M., Nattrass, N., & O’Riain, M. J. (2023). Small-livestock farmers’ perceived effectiveness of predation control methods and the correlates of reported illegal poison use in the South African Karoo. Ambio, 52(10), 1635-1649.
Ferreira NA. 1988. Sekere aspekte van die ekologie en die beheer van die rooikat (Felis caracal) in die Oranje-Vrystaat. Unpublished report, Orange Free State Provincial Administration,irectorate Environmental and Nature Conservation, Bloemfontein, South Africa.
Fuller, A., Mitchell, D., Maloney, S. K., Hetem, R. S., Fonsêca, V. F., Meyer, L. C., … & Snelling, E. P. (2021). How dryland mammals will respond to climate change: the effects of body size, heat load and a lack of food and water. Journal of Experimental Biology, 224(Suppl_1), jeb238113.
Greaves N, Clement R. 1993. When Lion Could Fly: And Other Tales From Africa. Lutterworth Press, Cambridge, UK.
Grobler, J. H. 1981. Feeding behaviour of the caracal Felis caracal Schreber, 1776, in the Mountain Zebra National Park. South African Journal of Zoology 16: 259-262.
Gunter Q. 2008. A critical evaluation of historical data on two damage causing predators, Canis mesomelas and Caracal caracal. M.Sc. Thesis. University of the Free State.
Inskip C, Zimmermann A. 2009. Human-felid conflict: a review of patterns and priorities worldwide. Oryx 43:18. doi:10.1017/S003060530899030X
Janczewski DN, Modi WS, Stephens JC, O’Brien SJ. 1995. Molecular evolution of mitochondrial 12S RNA and cytochrome b sequences in the Pantherine lineage of Felidae. Molecular Biology and Evolution 12: 690–707.
Jansen C. 2016. Diet of key predators responsible for livestock conflict in Namaqualand, South Africa. M.Sc. Dissertation. University of Stellenbosch.
Johnson, W. E. and O’Brien, S. J. 1997. Phylogenetic reconstruction of the Felidae using 16S rRNA and NADH-5 mitochondrial genes. Journal of Molecular Evolution 44: S98-S116.
Johnson, W.E., Eizirik, E., Pecon-Slattery, J., Murphy, W.J., Antunes, A., Teeling, E. and O’Brien, S.J. 2006. The late Miocene radiation of modern Felidae: A genetic assessment. Science 311: 73-77.
Kerley, G.I.H., Wilson, S.L. & Balfour, D. (Eds) 2018. Livestock Predation and its Management in South Africa: A Scientific Assessment. Centre for African Conservation Ecology, Nelson Mandela University, Port Elizabeth.
Kok, O.B. 1996. Dieetsamestelling van enkele karnivoorsoorte in die Vrystaat, Suid-Afrika. South African Journal of Science 92:393-398. [In Afrikaans, English summary]
Kyriazis C.C., Serieys L.E.K., Bishop J.M., Drouilly M., Viljoen S., Wayne R.K. & Lohmueller, K.E. The influence of gene flow on population viability in an isolated urban caracal population. Mol Ecol. 2024;1–43
Leighton, G.R.M., Bishop, J.M., O’Riain, M.J., Broadfield, J., Merondun, J., Avery, G., Avery, D. M. & Serieys, L.E.K. (2020) An integrated dietary assessment increases feeding event detection in an urban carnivore. Urb Ecosyst. doi:10.1007/s11252-020-00946-y
Leighton G. R. M., Bishop J. M., Merondun J., Winterton, D. J., O’Riain, M. J. & Serieys, L.E.K. (2022a) Hiding in plain sight: risk mitigation by a cryptic carnivore foraging at the urban edge. Anim Conserv 1–15. doi: 10.1111/acv.12732
Leighton G.R.M., Bishop J.M., Camarero, P., Mateo, R. O’Riain, M. J. & Serieys, L.E.K. (2022b) Poisoned chalice: Use of transformed landscapes associated with increased persistent organic pollutant concentrations and potential immune effects for an adaptable carnivore. Sci Total Environ. doi.org/10.1016/j.scitotenv.2022.153581
Leighton, G.R.M., Froneman, W., Serieys, L.E.K. & Bishop, J.M. (2023) Trophic downgrading of an adaptable carnivore in an urbanising landscape. Sci. Rep. doi.org/10.1038/s41598-023-48868-x
Leighton, G.R.M., Froneman, W., Serieys, L.E.K. & Bishop, J.M. (2024) Sustained use of marine subsidies promotes niche expansion in a wild felid Sci Total Environ. doi.org/10.1016/j.scitotenv.2024.169912
Lloyd PH, Millar JCG. 1983. A questionnaire survey (1969-1974) of some of the larger mammals of the Cape Province. Bontebok 3: 1-49.
Marker, L. L. and Dickman, A. J. 2005. Notes on the spatial ecology of caracals (Felis caracal), with particular reference to Namibian farmlands. African Journal of Ecology 43: 73-76.
Martins QE. 2010. The ecology of the leopard Panthera pardus in the Cederberg Mountains. Ph.D Thesis. University of Bristol, Bristol, UK.
McManus JS, Dickman AJ, Gaynor D, Smuts BH, Macdonald DW. 2015. Dead or alive? Comparing costs and benefits of lethal and non-lethal human–wildlife conflict mitigation on livestock farms. Oryx 49: 687-695.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Melville H. 2004. Behavioural ecology of the caracal in the Kgalagadi Transfrontier Park, and its impact on adjacent small stock production units. M.Sc. Dissertation. University of Pretoria.
Melville HIAS, Bothma J du P, Mills MGL. 2004. Prey selection by caracal in the Kgalagadi Transfrontier Park. South African Journal of Wildlife Research 34: 67–75.
Moffett R. 2010. SeSotho Plant and Animal Names, and Plants Used by the Basotho. Sun Press, Bloemfontein, South Africa.
Moolman LC. 1984. ‘n Vergelyking van die voedingsgewoontes van die rooikat Felis caracal binne en buite die Bergkwagga Nasionale Park. Koedoe (Suppl.) 27: 122–129.
Moolman LC. 1986. Aspekte van die ekologie en gedrag van die rooikat Felis caracal Schreber, 1776 in die Bergkwagga Nasionale Park en op die omliggende plase. M.Sc. Dissertation. University of Pretoria.
Müller, L., Briers‐Louw, W. D., Amin, R., Lochner, C. S., & Leslie, A. J. (2022). Carnivore coexistence facilitated by spatial and dietary partitioning and fine‐scale behavioural avoidance in a semi‐arid ecosystem. Journal of Zoology, 317(2), 114-128.
Nattrass, N., Conradie, B., Stephens, J., & Drouilly, M. (2020a). Culling recolonizing mesopredators increases livestock losses: Evidence from the South African Karoo. Ambio, 49, 1222-1231.
Nattrass, N., & O’Riain, M. J. (2020b). Contested natures: conflict over caracals and cats in Cape Town, South Africa. Journal of Urban Ecology, 6(1), juaa019.
Newbery CH. 1995. Mammal Checklist of the Provincial Nature Reserves. North West Parks Board, Mafikeng, South Africa.
Norton PM, Lawson AB. 1985. Radio tracking of leopards and caracals in the Stellenbosch area, Cape Province. South African Journal of Wildlife Research 15: 17–24.
Nowell, K. and Jackson, P. 1996. Wild Cats. Status Survey and Conservation Action Plan. IUCN/SSC Cat Specialist Group, Gland, Switzerland and Cambridge, UK.
Palmer R, Fairall N. 1988. Caracal and African wild cat diet in the Karoo National Park and the implications thereof for hyrax. South African Journal of Wildlife Research 18: 30-34.
Parker, K.H., Bishop J.M., Serieys, L.E.K., Mateo, R., Camarero, P. & Leighton, G.R.M. 2023 A heavy burden: Metal exposure across the land-ocean continuum in an adaptable carnivore. Environ. Pollut. doi.org/10.1016/j.envpol.2023.121585
Pohl, C.F. 2015. The diet of caracal (Caracal caracal) in the Southern Free State. Department of Zoology and Entomology, University of the Free State.
Potgieter GC, Marker LL, Avenant NL, Kerley GIH. 2013. Why Namibian farmers are satisfied with the performance of their livestock guarding dogs. Human Dimensions of Wildlife 18: 403–415.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Predation Management Forum (PMF). 2016. Predation Management Manual. Cape Town, South Africa.
Predation Management Forum (PMF). Website. Available at: http://www.pmfsa.co.za/.
Pringle JA & Pringle VL. 1979. Observations on the lynx Felis caracal in the Bedford district. African Zoology 14: 1–4.
Pringle JA. 1977. The distribution of mammals in Natal. Part 2. Carnivora. Annals of the Natal Museum 23: 93-115.
Ray, J.C., Hunter, L. and Zigouris, J. 2005. Setting conservation and research priorities for larger African carnivores. Wildlife Conservation Society, New York, USA.
Rowe-Rowe DT. 1978. The small carnivores of Natal. Lammergeyer 25: 1–48.
Rowe-Rowe, D.T. 1992. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Schepers A. 2016. The economic impact of predation in the wildlife ranching industry in Limpopo, South Africa. M.Sc. Thesis. University of the Free State.
Serieys LEK, Bishop J, Okes N, et al (2019) Widespread anticoagulant poison exposure in predators in a rapidly growing South African city. Sci Total Environ 666:581–590. doi: 10.1016/j.scitotenv.2019.02.122
Serieys L.E.K., Bishop J.M., Rogan M.S., Smith, J.A., Suraci, J.P., O’Riain, M.J. & Wilmers, C.C. (2023) Anthropogenic activities and age class mediate carnivore habitat selection in a human-dominated landscape. iScience 26:1–27. doi: 10.1016/j.isci.2023.107050
Serieys, L.E.K., Leighton, G.R.M., Merondun, J., Bishop, JM. 2024. Denning and maternal behavior of caracals (Caracal caracal). Mammalian Biology (in press)
Skead, C.J. 1980. Historical mammal incidence in the Cape Province: Vol. 1. The Western and Northern Cape. Department of Nature and Environmental Conservation, Cape Tpwn.
Skinner JD. 1979. Feeding behavior in Caracal Felis caracal. Journal of Zoology 189: 523–525.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smith, Y.C.E., Smith, D.A.E., Ramesh, T. and Downs, C.T., 2019. Novel predators and anthropogenic disturbance influence spatio-temporal distribution of forest antelope species. Behavioural processes, 159, pp.9-22.
Smithers, R.H.N. 1975. Family Felidae. In: J. Meester and H.W. Setzer (eds). The mammals of Africa: an identification manual. Smithsonian Institution, Washington D.C. Part 8.1. pp. 1-10.
Stadler H. 2006. Historical perspectives on the development of problem animal management in the Cape Province. In: Daly B, Davies-Mostert W, Evans S, Friedmann Y, King N, Snow T & Stadler H, (ed.), Workshop on Holistic Management of Human–Wildlife Conflict in the Agricultural Sector 94 of South Africa, pp. 11–16. Johannesburg, South Africa.
Strauss AJ. 2009. The impact of predation on a sheep enterprise in the Free State Province. M.Sc. Thesis. University of the Free State.
Stuart CT. 1986. The incidence of surplus killing by Panthera pardus and Felis caracal in Cape Province, South Africa. Mammalia 50: 556–558.
Stuart, C. T. 1982. Aspects of the biology of the caracal (Felis caracal) in the Cape Province, South Africa. M.S. Thesis, University of Natal.
Stuart, C. and Stuart, T. 2013. Caracal caracal. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa, Academic Press, Amsterdam, The Netherlands.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1:1-58.
Sunquist, M. and Sunquist, F. 2002. Wild Cats of the World. University of Chicago Press.
Tambling, C.T, Avenant, N.L., Drouilly, M. & Melville, H. 2018. The Role Of Mesopredators In Ecosystems:
Potential Effects Of Managing Their Populations On Ecosystem Processes And Biodiversity. In: Livestock predation and its management in South Africa: a scientific assessment (Eds Kerley, G.I.H., Wilson, S.L. & Balfour, D.). Centre for African Conservation Ecology, Nelson Mandela University, Port Elizabeth, 214-236.
Teichman KJ, Cristescu B, O’Riain J, Hodges KE. 2015. Caracal diet and home range on farmlands in the Succulent Karoo.
Teichman, K. J., Cristescu, B., Crevier, L., O’Riain, M. J., & Hodges, K. E. (2023). Movement Choices of Persecuted Caracals on Farmlands in South Africa. Rangeland Ecology & Management, 88, 77-84.
Tensen, L., Drouilly, M., & van Vuuren, B. J. (2018). Genetic structure and diversity within lethally managed populations of two mesopredators in South Africa. Journal of Mammalogy, 99(6), 1411-1421.
Tensen, L., Drouilly, M., & Jansen van Vuuren, B. (2019). Insights into the genetic population structure of black-backed jackal and caracal in South Africa. African Journal of Wildlife Research, 49(1), 84-88.
Thorn, M., Green, M., Dalerum, F., Bateman, P.W. and Scott, D.M. 2012. What drives human-carnivore conflict in the North West Province of South Africa? Biological Conservation 150: 23-32.
Thorn, M., Green, M., Keith, M., Marnewick, K., Bateman, P.W., Cameron, E.Z. and Scott, D.M. 2011a. Large-scale distribution patterns of carnivores in northern South Africa: implications for conservation and monitoring. Oryx 45(4): 579-586.
Van Heezik, Y. M. and Seddon, P. J. 1998. Range size and habitat use of an adult male caracal in northern Saudi Arabia. Journal of Arid Environments 40: 109-112.
de Waal HO. 2009. Recent advances in co-ordinated predator management in South Africa. Merino SA Focus 2009(44–46).
du Plessis J. 2014. Rooijakkals- en rooikat-bestuur en die rol van predatore in ‘n ekosisteem. Culna 69: 11–13.
du Plessis JJ, Avenant NL, de Waal HO. 2015. Quality and quantity of the scientific information available on black-backed jackals and caracals: contributing to human–predator conflict management? African Journal of Wildlife Research 45: 138–157.
du Plessis JJ. 2013. Towards the development of a sustainable management strategy for Canis mesomelas and Caracal caracal on rangeland. Ph.D. Thesis. University of the Free State.
van Niekerk HN. 2010. The cost of predation on small livestock in South Africa by medium-sized predators. M.Sc. Dissertation. University of the Free State.
van Tonder, J. (2024) Investigating threats to leopards (Panthera pardus) and attitudes towards leopards across the Western Cape Province of South Africa. MSc thesis. Stellenbosch University
Veals, A. M., Burnett, A. D., Morandini, M., Drouilly, M., & Koprowski, J. L. (2020). Caracal caracal (Carnivora: Felidae). Mammalian Species, 52(993), 71-85.
Viljoen S, Justin O’Riain M, Penzhorn BL, Drouilly, M., Serieys, L.E.K., Cristescu, B., Teichman, K.J. & Bishop, J.M. (2020) Molecular detection of tick-borne pathogens in caracals (Caracal caracal) living in human-modified landscapes of South Africa. Parasit Vectors 13: doi: 10.1186/s13071-020-04075-5
Woodgate, Z. A., Drouilly, M., Nattrass, N., & O’Riain, M. J. (2023). Co-occurrence of black-backed jackal and caracal in the Karoo, South Africa. Journal of Arid Environments, 219, 105067.