Cape Spiny Mouse
Acomys subspinosus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Acomys subspinosus – (Waterhouse, 1838)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Acomys – subspinosus
Common Names: Cape Spiny Mouse (English), Kaapse stekelmuis (Afrikaans), Kap-Stachelmaus (German)
Synonyms: No Synonyms
Taxonomic Notes: This species appears to be basal in phylogenies of Acomys (Barome et al. 2001; Verheyen et al. 2011).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Russo, I.M.1 & da Silva, J.M.2
Reviewer: Kennerley, R.3
Institutions:1Cardiff University, 2South African National Biodiversity Institute, 3IUCN SSC Small Mammal Specialist Group
Previous Assessors: Palmer, G., Midgley, J., Pence, G. & Avery, M.
Previous Reviewers: Child, M.F.
Previous Contributors: Avenant, N., Baxter, R., MacFadyen, D., Monadjem, A., Taylor, P. & Wilson, B.
Assessment Rationale
The Cape Spiny Mouse is listed as Least Concern because it is widespread in the Western Cape Province of South Africa with an estimated extent of occurrence (EOO) of 171,474 km². As this species mainly exists in rocky habitat that is unlikely to be transformed, there are suspected to be no major threats that could cause widespread population decline. Additionally, there may be localised losses of habitat quality due to the spread of alien invasive species, inappropriate fire regimes (too frequent fires), and edge effects associated with agricultural and residential land-uses (for example, use of pesticides and predation from domestic pets). Although the species remains widespread and regularly encountered, proactive mitigation measures, including protected area expansion and habitat restoration, should be continued to counteract habitat loss.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Russo IM & da Silva JM. 2025. A conservation assessment of Acomys subspinosus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
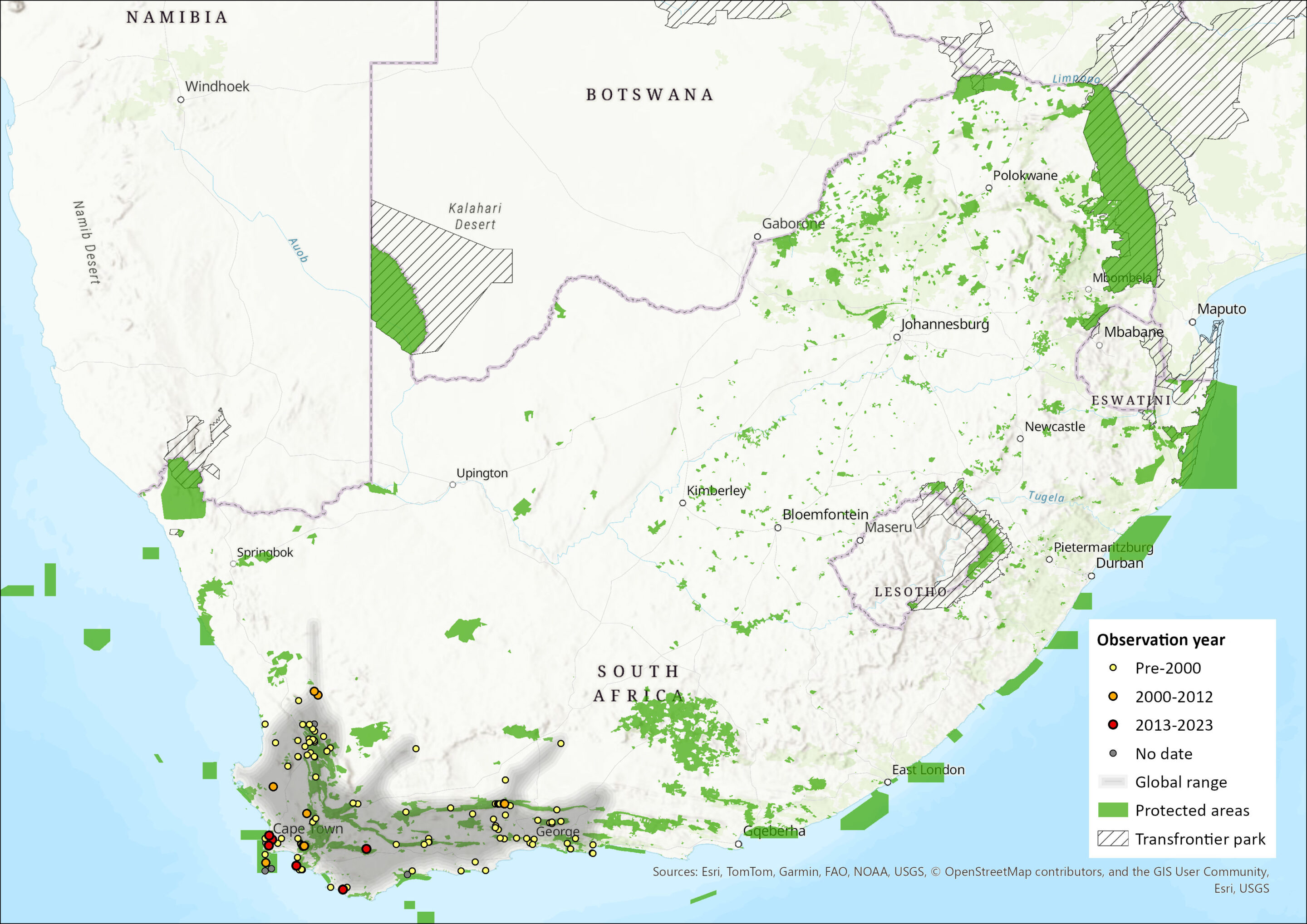
This species is a true fynbos endemic (Breytenbach 1982), occurring in the Western Cape Province (Avery et al. 2005) and extending marginally into the Eastern and Northern Cape Provinces (Avery and Avery 2011). Its range extends from Citrusdal in the west to Knysna in the east (Skinner and Chimimba 2005). It is generally associated with rocky habitats on mountain slopes, and thus can exist at high altitudes, but it also occurs in lowland fynbos habitats. No range shifts have been documented. Its extent of occurrence (EOO) is estimated to be 171,474 km². Its area of occupancy (AOO) is estimated to be 74,165 km² (calculated using quarter degree square cells) using all fynbos vegetation types within the EOO (Mucina and Rutherford 2006). The current amount of untransformed fynbos will be lower than this estimate but will still be above the 2,000 km² threshold for criterion B2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 1000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Cape Spiny Mouse (Acomys subspinosus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | – |
| South Africa -> Eastern Cape Province | Extant | Native | – | – |
| South Africa -> Northern Cape Province | Extant | Native | – | – |
| South Africa -> Western Cape | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Currently, although around 76% of the Western Cape is still considered natural or near-natural, information from CapeNature indicates that there is a continuing decline in natural habitat from agricultural expansion, especially on lower- to mid-slope areas (below 1,000 m) caused by planted pastures and rooibos, wine and fruit cultivation, which may impact the species in the future as climate change makes higher elevation habitats more suitable for agriculture.
Population Information
This species is widespread but not abundant. It is never trapped in high numbers at a site (J. Midgley unpublished data; Fleming and Wilson 2002)) and it is never the dominant species in an area (Avery et al. 2005). Although direct population estimates and trends are not available for this species, we suspect a declining population based on ongoing habitat loss from agricultural expansion in the Western Cape Province (Pence 2014), which may be exacerbated by the current trend of planted pastures and rooibos, wine and fruit cultivation expanding onto lower- and mid-slopes below 1,000 m asl (Pence 2014). However, given that the core rocky habitats of the species are likely to be left untransformed and that habitat continues to be protected—between 2007 and 2014, 775 km² were added to the conservation estate of the Western Cape Province (Pence 2014)—the net effect on population trend is unknown and thus assumed to be stable. Provided the current protected area network persists this species should not decline. Further research should test this assumption at sites from across the province.
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: Naturally fragmented.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
A phylogenetic investigation on the genus Acomys, identified only one lineage within Acomys subspinosus, confirming its range is restricted to South Africa (Aghova et al. 2019). To date this is the only study examining this species at a molecular level, however interest in the genus as a research organism for regenerative medicine has been suggested (see Gaire et al. 2021).
Since no population genetic study has been undertaken and the phylogenetic study found a single lineage, the species is thought to exist as a single metapopulation within the fynbos biome. Consequently, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining). However, genetic isolation may be taking place if habitat transformation has created substantial gaps, exceeding their dispersal capabilities.
Given that the species has never been known to be abundant and population numbers are thought to be declining, estimates of effective population size (Ne) are not possible, thereby preventing the calculation of the GBF’s Ne 500 genetic indicator.
It is highly recommended that a population genetic study be undertaken to better understand the population genetic structure and diversity within the species and to quantify the estimated indicator values.
Habitats and ecology
This species is often, but not only, associated with rocky areas on mountain slopes in fynbos vegetation and is almost entirely dependent on fynbos that has all its functional components. The species is widespread but poorly correlated with vegetation structure (Bond et al. 1980), which may be due to its specific habitat compositional requirements, such as rodent-pollinated plants and plants with nuts. This species is thus generally not found in modified habitats. It appears to favour more mature fynbos where it can find seeds, particularly from restios (to which it is partial), but also takes green plant material, insects, millipedes and snails (Stuart and Stuart 2007). It changes its diet from primarily insects in winter and spring to mainly seeds in summer and autumn, which parallels a foraging behaviour shift from seed consumption to seed burial (Rusch et al. 2014). However, isotope analysis suggests its diet is stable for most of the year besides summer (van den Heuvel and Midgley 2014). It is nocturnal and thus a prey species for owls (Avery et al. 2005) and sometimes nests in holes rather than cracks and crevices (Breytenbach 1982). It seems to be an opportunistic breeder (Fleming and Nicolson 2002) and to disappear after fire before slowly recovering (van Hensbergen et al. 1992). It is absent from fire-breaks so is probably disadvantaged by frequent fires.
Ecosystem and cultural services: This species is also a keystone species as it scatter-hoards (by burying) seeds (Midgley and Anderson 2005) and pollinates flowers (Letten and Midgley 2009; Turner et al. 2011). It may even be dependent on these resources. For instance, Fleming and Nicholson (2002) noted how breeding and population numbers depended on access to rodent-pollinated Protea humiflora. As up to 76% of seed caches contain just one seed, suggesting that scatter-hoarding may have evolved as an anti-pilfering strategy (Rusch et al. 2013), Cape Spiny Mice may help sustain landscape heterogeneity.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 6. Rocky areas (e.g. inland cliffs, mountain peaks) | – | Suitable | – |
Life History
| Generation Length | Justification | Data Quality |
| 1-2 | Inferred (see Pacifici et al. 2013) | good |
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Although no major or specific threats are suspected to be impacting this species, habitat loss and degradation are suspected to be causing local declines. Habitat loss from agricultural expansion and urbanisation is suspected to have severely affected individuals existing in fringe habitats on low-lying fynbos areas; of the 24% that is severely degraded or entirely devoid of natural vegetation. About 14% is attributed to intensive agricultural land uses (Pence 2014).
The fynbos habitat is impacted in parts by invasive alien plants (IAPs) which, despite control efforts, are expanding (van Wilgen et al. 2012). Since the Cape Spiny Mouse feeds primarily on the seeds of restios (Skinner and Chimimba 2005), replacement of natural vegetation by non-indigenous species may impact the species and effectively reduce its area of occupancy. Similarly, altered fire regimes caused by synergies between IAPs and climate change may be or become a threat if the fire return interval becomes so short that seeds preferred by the Cape Spiny Mouse cannot be produced.
Current habitat trend: Although there is a continuing decline in fynbos habitat within the Western Cape Province (Pence 2014), primarily through agricultural expansion, especially vineyards (for example, Fairbanks et al. 2004), it is uncertain how this impacts Cape Spiny Mouse subpopulations considering the species primarily inhabits rocky areas and mountain slopes. As 76% of the province remains natural or near-natural and the protected area network continues to expand (Pence 2014), we suspect that habitat loss from agricultural expansion will only impact the species if higher-elevation areas are increasingly converted to crops, which may become likely as climate change makes such areas suitable. For example, climate change is projected to increase the suitability of upslope habitats for viticulture, increasing the footprint of winelands by 14% by 2050 (Hannah et al. 2013). Such trends in transformation of mid- and upperslopes should be monitored.
Conservation
The Cape Spiny Mouse has been recorded from virtually all protected areas in the Western Cape, including all eight components of the Cape Floral Region Protected Areas World Heritage Sites, which includes the Baviaanskloof in the Eastern Cape.
No specific interventions are necessary at this stage. However, protected area expansion and biodiversity stewardship schemes, especially to counteract the potential for vineyards to transform the mountain slopes where this species occurs, are encouraged. Progress is being made in protected area expansion in the Western Cape, especially in Critical Biodiversity Areas (CBAs; Pence 2014). Stewardship on private lands may be particularly promising. For example, the Biodiversity Stewardship Programme has added over 490 km² to the Western Cape’s formal protected area network since its launch in 2003, by entering into biodiversity agreements with private landowners (Maree et al. 2015). Such agreements should be enhanced through best practice management techniques for both viticulture and biodiversity, a new field dubbed vinecology, which is actively implemented in South Africa (reviewed in Viers et al. 2013).
Ongoing clearing of IAPs and restoration of fynbos habitats will also likely benefit this species by creating habitat with indigenous restio species and thus food resources.
Recommendations for land managers and practitioners:
- It is critical that the fight to eradicate IAPs from our protected areas continues and that every effort be made to maintain the “natural” fire regime within the fynbos. Landowners should be incentivised to employ the Working for Water Programme (Department of Environmental Affairs) to restore habitats.
Research priorities:
- Rate of future habitat loss in the Western Cape, especially in higher altitude areas due to climate change and viticultural expansion.
- Estimating population size through density estimates and total natural habitat available. This would enable a threshold of habitat loss to be calculated below which the population is expected to be fewer than 10,000 mature individuals.
- Effectiveness of IAP removal in increasing Cape Spiny Mouse and small mammal occurrence and abundance.
- Effectiveness of implementing vinecology management in the survival of Cape Spiny Mouse and other small mammals should be monitored and evaluated.
- Research into how the Cape Spiny Mouse responds to fire frequency and the extent of dependence on nut-fruited plants and mammal-pollinated plants.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP). As this species is easily distinguished by its spines, geo-referenced observations, especially from outside protected areas, will help to map distribution and habitat preference.
Bibliography
Aghová, T., Palupčíková, K., Šumbera, R., Frynta, D., Lavrenchenko, L.A., Meheretu, Y., Sádlová, J., Votýpka, J., Mbau, J.S., Modrý, D. and Bryja, J., 2019. Multiple radiations of spiny mice (Rodentia: Acomys) in dry open habitats of Afro-Arabia: evidence from a multi-locus phylogeny. BMC Evolutionary Biology, 19, pp.1-22.
Avery, D.M. and Avery, G. 2011. Micromammals in the Northern Cape Province of South Africa, past and present. African Natural History 7: 9-39.
Avery, D.M., Avery, G. and Palmer, N.G. 2005. Micromammalian distribution and abundance in the Western Cape Province, South Africa, as evidenced by Barn owls Tyto alba (Scopoli). Journal of Natural History 39: 2047–2071.
Barome P-O, Volobouev V, Monnerot M, Mfune JK, Chitaukali W, Gautun J-C, Denys C. 2001. Phylogeny of Acomys spinosissimus (Rodentia, Muridae) from north Malawi and Tanzania: evidence from morphological and molecular analysis. Biological Journal of the Linnean Society 73: 321-340.
Bond W, Ferguson M, Forsyth G. 1980. Small mammals and habitat structure along altitudinal gradients in the southern Cape mountains. South African Journal Zoology 15: 34-43.
Breytenbach, G.J. 1982. Small mammal responses to environmental gradients in the Groot Swartberg of the Southern Cape. University of Pretoria.
Fleming PA, Nicolson SW. 2002. Opportunistic breeding in the Cape spiny mouse (Acomys subspinosus). African Zoology 37(101-105).
Gaire, J., Varholick, J.A., Rana, S., Sunshine, M.D., Doré, S., Barbazuk, W.B., Fuller, D.D., Maden, M. and Simmons, C.S., 2021. Spiny mouse (Acomys): an emerging research organism for regenerative medicine with applications beyond the skin. npj Regenerative Medicine, 6(1), p.1.
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-2. Available at: www.iucnredlist.org. (Accessed: 14 September 2017).
Letten AD, Midgley JJ. 2009. Rodent pollination in the Cape legume Liparia parva. Austral Ecology 34: 233-236.
Maree KS, Pence GQK, Purnell K. 2015. Western Cape Protected Area Expansion Strategy: 2015 – 2020. Unpublished report. Produced by CapeNature. Cape Town, South Africa.
Midgley JJ, Anderson B. 2005. Scatterhoarding in Mediterranean Shrublands of the SW Cape, South Africa. In: Forget PM, Lambert JE, Hulme PE, Vander Wall SB (ed.), Seed Fate: Predation, Dispersal, and Seedling Establishment, Wallingford, UK.
Mucina, L. and Rutherford, M.C. 2006. The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria, South Africa.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pence, G.Q.K. 2014. Western Cape Biodiversity Framework 2014 status update: Critical Biodiversity Areas of the Western Cape. CapeNature, Cape Town.
Rusch UD, Midgley JJ, Anderson B. 2013. Competing seed consumers drive the evolution of scatter-hoarding: Why rodents do not put all their seeds in one larder. African Zoology 48: 152-158.
Rusch UD, Midgley JJ, Anderson B. 2014. Seasonal fluctuations in rodent seed caching and consumption behaviour in fynbos shrublands: Implications for fire management. South African Journal of Botany 93: 217-221.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stuart C, Stuart T. 2001. Field Guide to Mammals of Southern Africa. Struik Publishers, Cape Town, South Africa.
Turner RC, Midgley JJ, Johnson SD. 2011. Evidence for rodent pollination in Erica hanekomii (Ericaceae). Botanical Journal of the Linnean Society 166: 163-170.
Van Hensbergen HJ, Botha SA, Forsyth GG, Le Maitre DC. 1992. Do small mammals govern vegetation recovery after fire in Fynbos? In: van Wilgen BW, Richardson DM, Kruger FJ, van Hensbergen HJ (ed.), Fire in South African Mountain Fynbos, pp. 182-202. Springer, Berlin, Germany.
Verheyen W, Hulselmans J, Wendelen W, Leirs H, Corti M. 2011. Contribution to the systematics and zoogeography of the East-African Acomys. Zootaxa 3059: 1-35.
Viers, J.H., Williams, J.N., Nicholas, K.A., Barbosa, O., Kotzé, I., Spence, L., Webb, L.B., Merenlender, A. and Reynolds, M. 2013. Vinecology: pairing wine with nature. Conservation Letters 6: 287-299.
van Wilgen, B.W., Forsyth, G.G., Le Maitre, D.C., Wannenburgh, A., Kotzé, J.D.F., van den Berg, E. and Henderson, L. 2012. An assessment of the effectiveness of a large, national-scale invasive alien plant control strategy in South Africa. Biological Conservation 148: 28-38.
van den Heuvel IM, Midgley JJ. 2014. Towards an isotope ecology of Cape Fynbos small mammals. African Zoology 49: 195-202.