
Cape Roof Bat
Laephotis capensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Laephotis capensis – (A. Smith, 1829)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Neoromicia – capensis
Common Names: Cape Roof Bat, Cape Bat, Cape House Bat, Cape Serotine Bat (previous name) (English), Kaapse Dakvlermuis (Afrikaans)
Synonyms: Eptesicus capensis (A. Smith, 1829); Pipistrellus capensis (A. Smith, 1829); Vespertilio capensis A. Smith, 1829
Taxonomic Note:
Laephotis capensis was previously classified as Eptesicus capensis. However, generic distinctiveness led to its reclassification in 1987 as N. capensis, alongside N. somalica, N. tenuipinnis and N. zuluenesis (Hill & Harrison 1987). Recent research, involving extensive genetic and morphological sampling, has now placed it in the genus Laephotis (Monadjem et al. 2020b). This species is highly variable morphologically and ecologically and may represents a complex of several similar species (Monadjem et al. 2020). Further taxonomic studies are necessary to confirm the status of populations currently allocated to L. capensis.
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Howard, A.1, Lötter, C.A.2, Balona, J.3, Richardson, E.J.4, & da Silva, J.M.5
Reviewer: de Villiers, M.6
Institutions: 1University of the Free State, 2Inkululeko Wildlife Services (Pty) Ltd, 3Gauteng and Northern Regions Bat Interest Group, 4Independent Consultant at Richardson & Peplow Environmental, 5South African National Biodiversity Institute, 6CapeNature
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., MacEwan, K., Cohen, L., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.,
Previous Contributors: Relton, C., Raimondo, D. & Child, M.F.
Assessment Rationale
Listed as Least Concern in view of its wide distribution (in the assessment region alone the EOO is 1,638,011 km2) and its presumed large population. It is locally common in a wide range of habitats including built-up areas and has been recorded from formally protected areas within the assessment region. Its use of buildings and other anthropogenic structures as roosts has possibly led to its numbers increasing in urbanized and other areas. This species is, however, very susceptible to mortality from wind turbines (Doty & Martin 2013; Aronson 2022) and while not yet in critical decline, needs closer monitoring.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Howard A, Lötter CA, Balona J, Richardson EJ & da Silva JM. 2025. A conservation assessment of Laephotis capensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is widespread and abundant over much of sub-Saharan Africa. It has been recorded from Guinea Bissau in the west, to Somalia, southern Sudan and Eritrea in the east, ranging south throughout most of southern Africa (ACR 2015). In the assessment region, the species is widespread and common, occurring in all provinces of South Africa and also in Lesotho and Eswatini. The type specimen is from Grahamstown (Monadjem et al. 2020). The estimated extent of occurrence is 1,638,011 km².
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
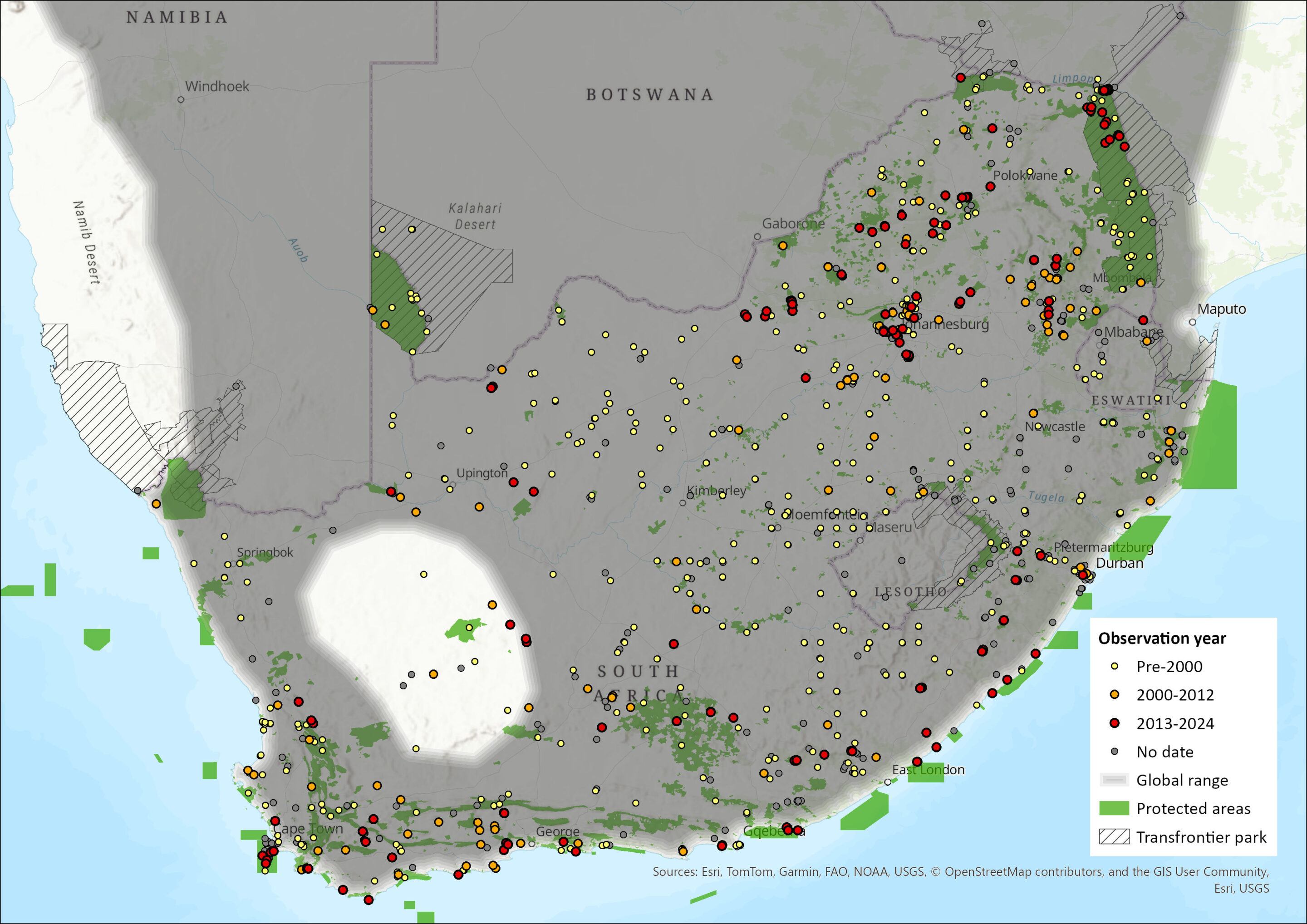 Figure 1. Distribution records for Cape Roof Bat (Laephotis capensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Cape Roof Bat (Laephotis capensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence within southern Africa
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No formal study on the direct effects of climate change in this species has yet been conducted. However, due to the increasing temperatures across most of the species’ distribution (Mbokodo et al. 2020), the roosting preferences and foraging areas are predicted to shift. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa and assessment region (Adams & Hayes, 2021; Mbokodo et al. 2020).
Population Information
In general, this is a locally common species (ACR 2015) and is well represented in museums with over 800 specimens examined in Monadjem et al. (2020). Earlier accounts of this species indicated it roosts singly or in groups of two or three individuals, which appears to be the case when it rests in natural roosts. However, it is now often found roosting in buildings in either medium mixed groups or large groups of females comprising maternity colonies. For example, in Zimbabwe large colonies of at least 100 individuals were observed roosting in houses (see Monadjem et al. 2020). Its use of buildings and other anthropogenic structures as roosts has possibly led to its numbers increasing in urbanised and other areas. However, in Renewable Energy Development Zones and other areas where there are clusters of operational wind farms, the cumulative impact of these may result in population declines especially in the absence of effective fatality mitigation for this species.
Current population trend: Stable, with localised increases and decreases within certain areas
Continuing decline in mature individuals? No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Although common and widespread throughout the assessment region, L. capensis forms at least two distinct clades in South Africa that are genetically differentiated from other L. capensis in Africa (Taylor et al. 2022). Additional genetic clusters might be revealed through a more fine–scale investigation of the population genetic structure and diversity of the species. While no population sizes are known, the species is estimated to be abundant and common and hence it is assumed that the effective population sizes for each subpopulation exceed the 500 threshold.
Habitats and ecology
It appears to tolerate a wide range of environmental conditions from arid semi-desert areas to montane grasslands (at altitudes as high as 1,600 m asl in the Drakensberg, KwaZulu-Natal; Taylor 1998), forests (dry and moist), bushveld, Acacia woodland, savannahs (dry and moist), and Mediterranean shrubland (Skinner & Chimimba 2005; Monadjem et al. 2020). However, it is possibly less abundant in low-lying, hot savannahs in the far east of southern Africa (Monadjem et al. 2020). It roosts under the bark of trees and similar vegetation, at the base of aloe leaves, between cracks in walls and under the roofs of houses, including thatched, corrugated iron and tiled (Lynch 1983; Monadjem 1998; ACR 2015). The species is recorded from all bioregions in the assessment region.
It is an insectivorous, clutter-edge forager (Monadjem et al. 2020), with a diet that is known to vary seasonally and geographically. At Sengwa, Zimbabwe, a study found that Coleoptera and Trichoptera made up the majority of the diet of L. capensis, with Lepidoptera, Hemiptera and Diptera making up a lesser proportion (Fenton 1985). However, moth consumption by this syntonic species has been shown to increase six-fold under artificial lights (Minnaar et al. 2015), which may place unprecedented pressure on eared-moth populations (which are less effective at evading syntonic species under artificial light conditions) and increase interspecific competition with allotonic bat species for this prey type, given the global increase in light pollution.
Ecosystem and cultural services: As this species is one of the most widespread and common insectivorous bat species in the assessment region, it may play an especially important role in controlling insect populations such as agricultural pest species (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus decrease the need for pesticides. It is also an important prey species for owls due to its abundance and clutter-edge foraging style (slow flight at the edge of vegetation) (ACR 2015).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Marginal | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Marginal | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Suitable | – |
Life History
Generation Length: Three years (Richardson pers comm/Bats KZN Rehab data, unpub.)
Age at Maturity: Female or unspecified: 12 months
Size at Maturity (in cms): Female: Forearm mean = 3.45 cm ± 0.217 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Forearm mean = 3.28 cm ±0.195 cm (Monadjem et al. 2020)
Longevity: 7-8 years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: Females are mono-oestrus and give birth in the warm, wet season (Monadjem et al. 2020).
Average Annual Fecundity or Litter Size: 1-4 pups, usually 1 (Monadjem et al. 2020)
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: Natural roosts tend to comprise 1-3 individuals. In buildings, larger groups of up to 100 (male and female) individuals may occur (Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
A recent study by Tarango et al. (2025) listed this species as imported into the U.S.A so online e-commerce platforms should be monitored for illegal trade in bat taxidermy and specimens from the assessment region.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Of all bat fatalities reported by Aronson (2022) from data that were collated from 25 operational wind farms in South Africa for the period 2011-2020, L. capensis was the second most prevalent (19% of all recorded fatalities). This threat should be monitored for its impacts on local populations. The potential cumulative impact of multiple wind farms in certain Renewable Energy Development Zones (such as the Cookhouse and Komsberg REDZ) is especially concerning, given that multiple wind farms in these areas have exceeded their bat fatality threshold for several years (SABAA unpubl. data).
Conservation
It is a widespread and adaptable species and occurs in numerous protected areas across the assessment region. However, taxonomic research may reveal several cryptic species, which will require a re-examination of regional threats and possible interventions.
To mitigate mortalities of this species from turbine collisions on wind farms, development of wind farms must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species in buildings and elsewhere, and also around large tree clumps and water supplies (for buffer recommendations see MacEwan et al. 2020 or later). Turbine-fatalities of this species must be reduced below fatality threshold values (as calculated according to MacEwan et al. 2018 or later) with interventions such as: i) ultrasound to deter bats (Weaver et al. 2020); ii) blanket curtailment of turbines at low wind speeds during seasons and hours of the night when this species is most active (Berthinussen et al. 2010; Arnett et al. 2011; Hayes et al. 2019); iii) acoustic smart curtailment of turbines in response to real-time bat activity (https://www.wildlifeacoustics.com/smart-system); and/or optimized smart curtailment of turbines during predicted periods of high bat activity based statistical modelling (https://west-inc.com/wp-content/uploads/2024/04/Optimized-Smart-Curtailment-SOQ_FINAL.pdf). As high bat fatalities at wind farms cannot be easily offset (Aronson et al. 2018; Mark Botha pers. comm.), avoidance and minimisation of bat fatalities is critical.
Recommendations for land managers and practitioners:
- Development of wind farms must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species.
- Turbine-fatalities of this species must be reduced with interventions such as ultrasound to deter bats and curtailing turbines during low wind speeds.
- Data sharing by wind farm managers into a national database is needed to be able to calculate cumulative impacts and thereafter implement collaborative mitigation and management efforts.
- Better post-construction compliance monitoring at wind farms.
Research priorities:
- A meta-analysis of all recorded L. capensis fatalities at wind farms in South Africa to date, to assess the cumulative impact of this industry on this species, and to identify any spatial, temporal, or turbine-specific trends, which may assist in devising effective fatality mitigation for future implementation.
- Research to assess the efficacy of different mitigation methods to reduce bat mortality at wind farms.
- Wind farm carcasses of this species could be used to obtain greater insight into the biogeography, genetics, reproductive biology, ecology (e.g. diet), and ecosystem services of this species throughout southern Africa.
- Taxonomic resolution is required to disentangle this possible species complex.
- Subpopulation sizes and trends at important roosts and determining population estimates.
- Physiological effects of agrochemicals.
Encouraged citizen actions:
- Reduce unnecessary disturbance of roosts and encourage safe co-habitation between bats and people.
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP). Since this species cannot be reliably identified based on easily observed external morphological features, calls may be more useful if these can be recorded with an ultrasonic bat detector.
- If bats are excluded from buildings, use bat-friendly methods (timing of removals relative to breeding season, use of “one-way valves” to prevent trapping, no fumigation, etc).
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Adams, R.A. and Hayes, M.A., 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Arnett EB, Huso MM, Schirmacher MR, Hayes JP. 2011. Altering turbine speed reduces bat mortality at wind-energy facilities. Frontiers in Ecology and the Environment 9: 209–214.
Aronson, J. 2022. Current state of knowledge of wind energy impacts on bats in South Africa. Acta Chiropterologica 24: 221-238.
Aronson, J., Sowler, S. and MacEwan, K. 2018. Mitigation guidance for bats at wind energy facilities in South Africa. Edition 2. South African Bat Assessment Association, South Africa.
Baerwald EF, Edworthy J, Holder M, Barclay RM. 2009. A large-scale mitigation experiment to reduce bat fatalities at wind energy facilities. Journal of Wildlife Management 73: 1077–1081.
Berthinussen, A., Richardson, O.C. and Altringham, J.D. 2010. Bat Conservation: Global Evidence for the Effects of Interventions. Synopses of Conservation Evidence. Pelagic Publishing, Exeter.
Boyles JG, Cryan PM, McCracken GF, Kunz TH. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Fenton MB. 1985. The feeding behaviour of insectivorous bats: echolocation, foraging strategies, and resource partitioning. Transvaal Museum Bulletin 21: 5–19.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S., 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Hayes, M.A., Hooton, L.A., Gilland, K.L., Grandgent, C., Smith, R.L., Lindsay, S.R., Collins, J.D., Schumacher, S.M., Rabie, P.A., Gruver, J.C., and Goodrich-Mahoney, J. 2019. A smart curtailment approach for reducing bat fatalities and curtailment time at wind energy facilities. Ecological Applications, 0(0), 2019, e01881.
Hill JE, Harrison DL. 1987. The baculum in the Vespertilioninae (Chiroptera: Vespertilionidae) with a systematic review, a synopsis of Pipistrellus and Eptesicus, and the descriptions of a new genus and subgenus. Bulletin of the British Museum (Natural History). Zoology 52: 225–305.
Kunz TH, Braun de Torrez E, Bauer D, Lobova T, Fleming TH. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
MacEwan, K., Aronson, J., Richardson, K., Taylor, P., Coverdale, B., Jacobs, D., Leeuwner, L., Marais, W. and Richards, L. 2018. South African Bat Fatality Threshold Guidelines. Edition 2. South African Bat Assessment Association, South Africa.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association, South Africa.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N., 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Minnaar C, Boyles JG, Minnaar IA, Sole CL, McKechnie AE. 2015. Stacking the odds: light pollution may shift the balance in an ancient predator–prey arms race. Journal of Applied Ecology 52: 522–531.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.
Monadjem A, Demos T., Dalton D., Webala P., Musila S., Kerbis Peterhans J., Patterson B. 2020b: 25. A revision of Pipistrelle-like bats (Mammalia: Chiroptera: Vespertilionidae) in East Africa with the description of new genera and species, Zoological Journal of the Linnean Society.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N., 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Rautenbach IL. 1982. Mammals of the Transvaal. No. 1, Ecoplan Monograph, Pretoria, South Africa.
Schoeman MC, Jacobs DS. 2008. The relative influence of competition and prey defenses on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS One 3: e3715.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Tarango, M., Kolby, J.E., Goodman, O.L., Anderson, C.J., Tinsman, J., Kirkey, J., Liew, A., Jones, M., Firth, C. and Reaser, J.K., 2025. Going batty: US bat imports raise concerns for species conservation and human health. One Health, 20, p.100999
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Weaver, S.P., Hein, C.D., Simpson, T.R., Evans, J.W., and Castro-Arellano, I. Ultrasonic acoustic deterrents significantly reduce bat fatalities at wind turbines. Global Ecology and Conservation 24: e01099
