Cape Porcupine
Hystrix africaeaustralis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Hystrix africaeaustralis – Peters, 1852
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – HYSTRICIDAE – Hystrix – africaeaustralis
Common Names: Cape Porcupine, Cape Crested Porcupine (English), South African Porcupine, Southern African Porcupine (English), Kaapse ystervark (Afrikaans), Ingungumbane, Inungu(IsiZulu, Swati), Noko (Sepedi, Sesotho, Setswana); Nungu (Xitsonga, Tshivenda)
Synonyms: Hystrix africae australis Peters, 1852; Hystrix capensis (Grill, 1860); Hystrix australis (Fitzinger, 1867) [nomen novum]; Hystrix africae-australis prittwitzi Müller, 1910; Hystrix stegmanni (Müller, 1910); Hystrix africae-australis zuluensis (Roberts, 1936); Hystrix africæaustralis africæaustralis
(Allen, 1939) (Mammal Diversity Database 2025)
Taxonomic Note: No subspecies are currently recognised (Monadjem et al. 2015).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1 & da Silva, J.M.2
Reviewer: Patel, T.3
Institutions: 1Durban Natural Science Museum,2South African National Biodiversity Institute,3Endangerrd Wildlife Trust
Previous Assessors and Reviewers: Bragg, C. & Child, M.F.
Previous Contributors: Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
The species remains Least Concern in view of its wide distribution within the assessment region, its occurrence in many habitats, including agricultural and urban environments, and its relatively high densities in some regions. Hunting and snaring for bushmeat and persecution for damage caused to fences and crops may result in local declines. Holistic management strategies, such as creating artificial passageways in game fences and establishing sustainable quill and meat trades, should be employed for this species, with positive effects for the wider socio-ecological community.
Regional population effects: Extensive and well connected throughout all range states. Rescue effects are possible. Females do not conceive while living in their natal groups and dispersal is a prerequisite for successful reproduction (van Aarde 1987a; van Aarde & van Wyk 1991). However, it is unknown what distances they travel when dispersing.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR & da Silva JM. 2025. A conservation assessment of Hystrix africaeaustralis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Cape Porcupines have a wide distribution in sub-Saharan Africa, avoiding the tropical forests of the Congo basin, and the driest parts of the Namib Desert (Monadjem et al. 2015). They occur from Kenya and southern Uganda in the north, through Tanzania, Rwanda, southeastern Democratic Republic of the Congo, extreme southwestern Congo, Angola, Zambia, Malawi and Mozambique, and then south throughout southern Africa (although they are absent from much of central Botswana).
Within the assessment region, they occur widely across all provinces, as well as Eswatini and Lesotho. For example, Power (2014) recorded them as common throughout the North West Province, occurring in every vegetation type but showing some local preference for riparian and rocky habitats. In the Drakensberg Midlands, relative occupancy of ten terrestrial mammal species was highest for Cape Porcupine (Ramesh & Downs 2015). They may be locally absent from some areas where there is high hunting pressure (Skinner & Chimimba 2005).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m asl (Skinner and Chimimba 2005)
Elevation Upper Limit (in metres above sea level): 2000 m asl (Skinner and Chimimba 2005)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
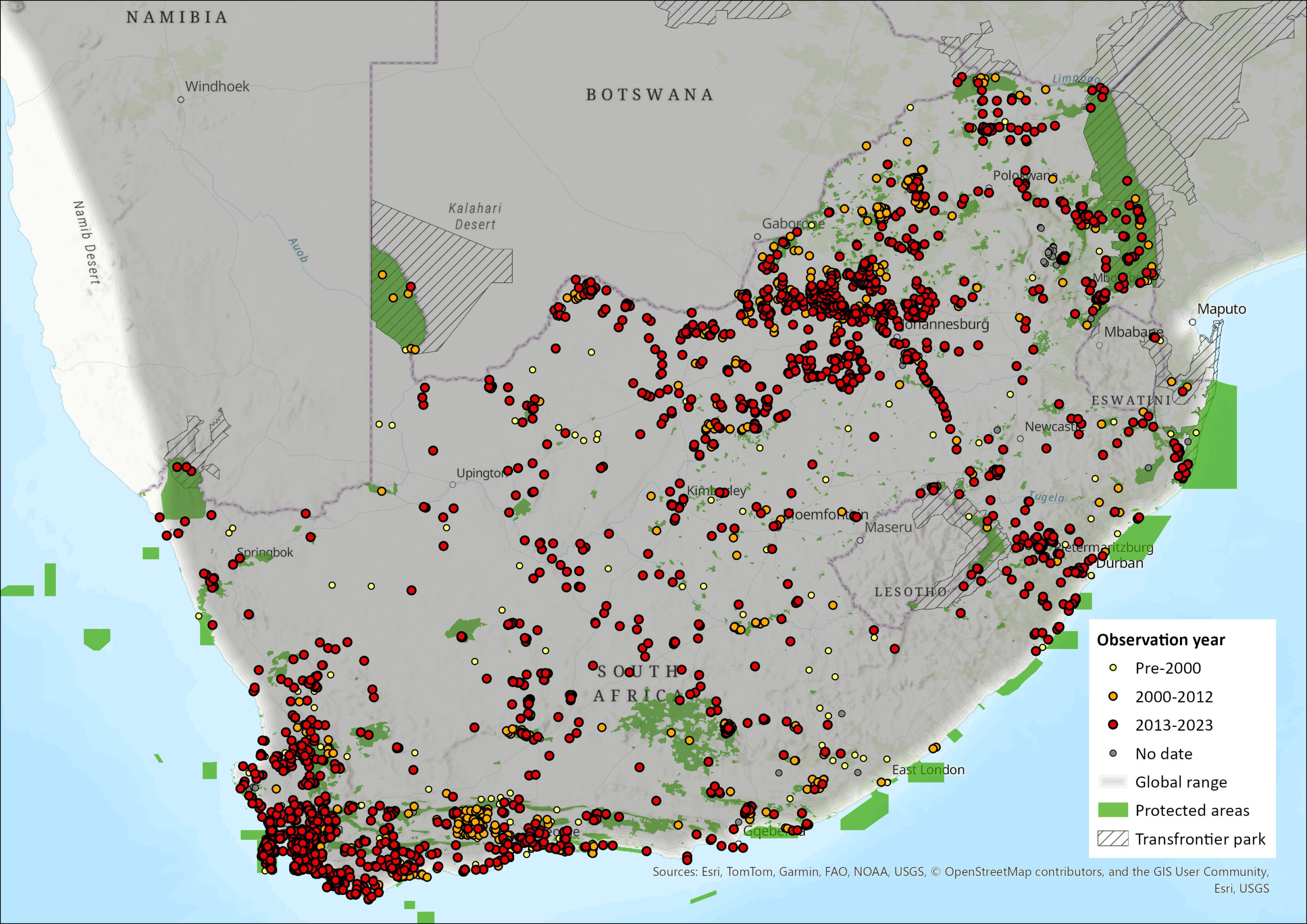
Map
Figure 1. Distribution records for Cape Porcupine (Hystrix africaeaustralis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
Cape Porcupines are widely distributed and inhabit a diverse range of habitats, generally exhibiting a broad tolerance to both daily and seasonal environmental fluctuations (Skinner et al. 1984; Barthelmess 2006). Despite this general adaptability, hey possess a relatively narrow thermoneutral zone between 24 °C and 28 °C (Haim et al. 1990a). Cooler ambient temperature adapted individuals exhibited signs of hyperthermia when ambient temperatures exceeded 35 °C (Haim et al. 1990a). Their physiological responses, including digestion rates, appear to be influenced by the climatic conditions of their respective regions. Populations adapted to lower ambient temperatures (proxy for temperate, winter-rainfall environments), tend to consume more water and food yet display lower digestion rates compared to those exposed to higher ambient temperatures (typical of tropical, summer-rainfall regions) (Haim et al. 1990a). This suggests that the Cape Porcupine can adapt to seasonal climatic changes by adjusting its metabolic rate, in addition to its thermal conductance (Haim et al. 1992). Haim et al. (1990a) postulated that low metabolic rates and high thermal conductance are key adaptations in this large burrowing rodent to inhabiting arid and semi-arid environments. However, the challenges associated with increasingly unpredictable environmental conditions driven by climate change may, in time, present a notable threat to this iconic African rodent. Southern Africa has been notably affected by cyclic El Niño Southern Oscillation (ENSO) events, resulting in a reduction of over 25% in average precipitation since 1960 (Sardans et al. 2024). This prolonged decline in rainfall is expected to contribute to the expansion of arid environments, which may adversely affect the long-term persistence of certain taxa inhabiting these regions, including the Cape Porcupine.
Population Information
It is a fairly common species across the assessment region, with relatively high densities. For example, in the semi-arid landscapes around Nieuwoudtville, Northern Cape Province, burrow entrances were found to be a good predictor of the number of porcupines it contained, where occupied burrow density was estimated to be 2.6 burrows / km2 and density on the study farm was estimated to be 8 individuals / km2 (Bragg et al. 2005). This high density in a semi-arid area was proposed by Bragg et al. (2005) to be a result of the high food availability in the region (high geophyte density). However, within more arid regions, porcupine numbers can be as low as 0.8 individuals / km2 such as in the Kalahari (Bragg, unpubl. data). Thus, we infer that there are over 10,000 mature individuals within the assessment region. However, hunting pressure and persecution (by farmers and farm labourers because it is a considered an agricultural pest, for bushmeat by rural communities, and for harvesting of the quills for use in the decor sector) may account for local extinctions in some areas, and current density estimates from throughout its range should be ascertained to determine population size more accurately.
Cape Porcupines live in extended family groups, consisting of a breeding male, breeding female and offspring of consecutive years (van Aarde 1987b). Female porcupine offspring do not conceive while in their natal groups and thus dispersal is a prerequisite for reproduction (van Aarde 1987b). This has important implications for the regulation of population size (van Aarde 1987a), because, if there are insufficient resources available in the environment, mature offspring cannot disperse from their natal group and thus cannot reproduce (van Aarde 1987a, 1987b). In captivity, the gestation period is c. 93 days, average litter size is 1–3, and young are usually suckled for 100 days or more (van Aarde 1985a, 1985b). Offspring attain sexual maturity between 1–2 years of age, during which time annual survival is relatively high for a rodent (van Aarde 1987a, 1987b, 1987c). The inter-litter interval in captivity is approximately one year (van Aarde 1985b).
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Yes. Locally, due to hunting pressure and persecution. Agricultural land expansions and forest fragmentation are growing threats.
Number of mature individuals in population: >10,000
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: In the absence of population genetic studies, presumed to represent one metapopulation (see below).
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
Severely fragmented: No. Can exist in human modified habitats.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unknown, but unlikely given the population size and its wide distribution
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unknown, but unlikely
Probability of extinction in the wild within 100 years: Unknown
Population Genetics
No population genetic studies have been conducted on this species; however, it is believed to exist as a single metapopulation given that its range is extensive and well connected. Moreover, it is abundant, often occurring in high densities, suggesting its effective population size should exceed the 500 threshold; however, this should be confirmed.
Habitats and ecology
Porcupines of the genus Hystrix are the largest African rodents with a mass of up to 20 kg (Monadjem et al. 2015). They typically rest during the day in rock crevices, small caves, or burrows, the latter either dug by Aardvark (Orycteropus afer) or by the porcupines themselves. Shelters often contain an accumulation of bones carried in by the porcupines themselves, as they practice osteophagy as a means of dietary supplementation (Skinner & Chimimba 2005). They are found in most of the types of vegetation encountered in southern Africa (including the coastal areas of the Namib Desert), from sea level to 2,000 m asl (Skinner & Chimimba 2005). They are generally absent from forest and are only found here marginally. In Nylsvley Nature Reserve, Limpopo Province, they showed a preference for Burkea over Acacia savannah due to higher concentrations of food in the former (de Villiers et al. 1994; de Villiers & van Aarde 1994). In the Bokkeveld Plateau, in the Northern Cape, they showed seasonal changes in preference for habitats based on the habitat’s substrate, seasonal food availability and refuge capacity. They can also exist in human-modified areas, such as croplands and suburban gardens.
Porcupines feed predominantly on roots, geophytes and tubers, which are dug up from under the ground using their strong incisors. Cape Porcupines not only consume plants selectively (Bragg 2003; de Villiers & van Aarde 1994) and sometimes en masse (Bragg 2003), but they are also able to consume (without any apparent side-effects) geophyte species that are known to be toxic for livestock. They also feed on fallen fruits and gnaw bones. Due to the combination of their diet and digging abilities, porcupines can become agricultural pests in farming areas (Monadjem et al. 2015). They do not appear to scavenge (Shaw et al. 2015). They are nocturnal, territorial and solitary foragers, although they can occasionally be found foraging in groups of two to three animals. They are monogamous and live in groups comprising either an adult pair, an adult pair and their offspring from consecutive litters, or an adult male and young of the year (Skinner & Chimimba 2005). They are long-lived and have a slow reproductive rate.
Ngcobo et al. (2019) examined porcupine home ranges across three land-use types—agroecosystems, peri-urban, and suburban environments—and reported an overall mean estimated home range of 39.37 ± 6.33 hectares (n = 9), with individual ranges spanning from 13.19 to 67.19 hectares. Porcupines located in agroecosystems exhibited the lowest estimated home range sizes; approximately half that of urban associated individuals (Ngcobo et al. 2019).
Ecosystem and cultural services: Cape Porcupines are ecosystem engineers. Foraging diggings or pits obstruct the flow of resources, trapping windborne organic matter and fine soil particles such as silt and clay, which would normally be captured by shrubs and their hummocks. The scale of the engineering effects caused by Cape Porcupines in the Nieuwoudtville region in the Northern Cape is on a par with, or at an even greater scale than, many other ecosystem engineer species’ impacts reported in the literature. The fact that Cape Porcupines can dig up to 0.87 m3 of soil / hectare over a year, which is equivalent to 5,859 tonnes / year and that their disturbance can cover up to 510,391 m2 demonstrates the scale and intensity of their disturbance patterns (C. Bragg unpubl. data). Cape Porcupine disturbance created distinct soil property changes in chemicals and moisture, and probably also texture, and therefore creates a mosaic of modified, unmodified and regenerating patches that provide habitats of differing resource availability and physical characteristics (C. Bragg unpubl. data). Additional disturbance impacts of Cape Porcupines take the form of the widespread distribution of their multi-entrance burrows (c. 60 on the 4,000 ha study area; Bragg et al. 2005). Cape Porcupines are also important patch creators, such as shown by Bragg (2003) through their maintenance of the Endangered Sparaxis pillansii geophytic species in the landscape of the Nieuwoudtville region, through their regular diggings and foraging activities. In addition, Kraai et al. (2022) demonstrated that porcupine-induced mortality of dominant tree species, such as Spirostachys africana, Vachellia nilotica, and V. robusta, in mesic savannas may promote structural heterogeneity in woody vegetation. This ecological effect could benefit landowners and reserve managers, particularly in areas where woody plant encroachment poses a management challenge (Kraai et al. 2022).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Suitable | – |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | – | Unknown | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
Life History
Generation Length: 7 years
Age at Maturity: Female or unspecified: 8 – 18 months (van Aarde 1987)
Age at Maturity: Male: 9 – 18 months (Van Aarde 1985)
Size at Maturity (in cms): Female: Mean total length = 51.6 ± 7.32 cm (Van Aarde 1987)
Size at Maturity (in cms): Male: Mean total length = 51.2 ± 4.73 cm (Van Aarde 1987)
Longevity: can live up to 10 years in the wild, but can attain 20 years in captivity
Average Reproductive Age: Typically 13 months, but ranges from 8 to 18 months (Van Aarde 1985, 1987)
Maximum Size (in cms): Female maximum total length = 81.39 cm; Male maximum total length = 75.6 cm (Van Aarde 1987)
Size at Birth: Young weigh 300 – 400 g at birth (Van Aarde 1985)
Gestation Time: Gestation lasts 93 – 94 days (Van Aarde 1985)
Reproductive Periodicity: Capable of breeding throughout the year but will typically conceive only once with most births occurring between August and March (late winter to early autumn) (Van Aarde 1985).
Average Annual Fecundity or Litter Size: Females give birth to 1 – 3 porcupettes (Van Aarde 1985)
Natural Mortality: This species is subject to predation by a range of medium- to large-sized carnivores, including but not limited to honey badgers, leopards and lions (Happold, 2013; Arbon 2019; Peterhans 2019)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Estimated home range sizes vary between 13.19 to 67.19 ha (Ngcobo et al. 2019a), with individuals in natural environments expanding their home range size in winter (Corbet and van Aarde 1996). Radio-tracked individuals from agroecosystems appear to maintain the same home range size irrespective of seasonal changes. Their dispersal patterns and long-term movement patterns remain largely unstudied.
Congregatory: Known to forage in small family groups, with monogamous male-female pairs occupying the same home range (Corbet and van Aarde 1996).
Systems
System: Terrestrial
General Use and Trade Information
They may be extensively hunted for bushmeat, even within formally protected areas (for example, Hayward 2009). They are also used in traditional medicine (e.g. the use and international trade in bezoars; Heinrich et al. 2020) and the quills are used as ornaments. The quill trade is suspected to have a negligible impact on the population as a whole (for example, Power 2014) but might have local impacts on subpopulations (Chevallier & Ashton 2006). They are also hunted as trophies in some regions.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
– |
– |
– |
National Commercial Value: No
International Commercial Value: Viewed in some countries as a valuable source of meat, with their quills used for trinkets and other decorative items.
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? None reported
Harvest Trend Comments: The harvesting of porcupines from the wild remains largely undocumented; as such, harvesting trends and likely impacts on local population numbers are difficult to ascertain.
Threats
Bushmeat hunting and persecution may be causing local decline or even extinctions in some areas. Such local depletion should be monitored and documented.
They are persecuted primarily for digging holes beneath fences, which, aside from the cost of repair, may allow predators into a farm or wildlife ranch with subsequent damage to livestock or game (Rust et al. 2015). Given the expansion of intensive wildlife ranching and the breeding of rare game species, which require increased fencing of properties, this threat could be increasing. They are also persecuted for damaging crops in agricultural areas (for example, Power 2014), especially where root crops are grown, and for ring-barking trees (which exposes the tree’s heartwood and increases susceptibility to fungal infections). Correspondingly, Cape Porcupine occupancy was negatively correlated with human abundance, which could be related to persecution, and livestock activity in the Drakensberg Midlands, KwaZulu-Natal Province (Ramesh & Downs 2015). Conversely, porcupine occupancy along the Coastal Belt of southern KwaZulu-Natal, was positively influenced by huma abundance (Ehlers-Smith et al. 2017), suggesting that scale-dependent regional effects must be considered in species occupancy modelling studies. Further studies show that due to their opportunistic and often destructive foraging behaviour, Cape Porcupines are generally perceived negatively by suburban homeowners and farmers in cropland areas (Ngcobo et al. 2019b). This perception has contributed to increasing human–porcupine conflict in anthropogenically-transformed regions, a trend that is likely to intensify with the ongoing expansion of suburban developments and agricultural landscapes (Ngcobo et al. 2019b). In the Eastern Cape, anecdotal reports suggest Cape Porcupines are being severely depleted by farmers and local communities to the extent that some subpopulations are locally extinct. Cape Porcupines are also often killed on roads (Collinson et al. 2015) and drowning incidents in residential pools are becoming more common (Durban Natural Science Museum records).
Conservation
They occur in many protected areas within the assessment region. Based on current understanding, no direct interventions may be deemed necessary at present. However, elsewhere it has been recommended that, on account of increased persecution and exploitation, porcupine species ought to be a listed on CITES Appendix II to effect monitoring of resident populations and the regulation of any likely international trade (Gomez 2021). The same may be true for the Cape Porcupine, as there are currently no reliable population estimates available to determine the population size within the assessment region. Consequently, the impact of localised hunting on porcupine populations over recent years remains undocumented and potentially unsustainable and warrants scientific investigation.
In general, this species would benefit from encouraging landowners and local communities to adopt holistic management techniques, such as burying water pipes below ground to avoid porcupine damage and setting up porcupine-permeable fences. For example, research from Namibia has shown that swing gates may be an effective alternate passageway for burrowing animals. Schumann et al. (2006) and Rust et al. (2015) showed that the installation of swing gates decreased the number of holes created for the duration of the studies. Similarly, the use of discarded car tyres installed into wildlife-proof fences has been demonstrated to be a cost-efficient and effective way to reduce damage and facilitate dispersal, where Cape Porcupines and Black-backed Jackals (Lupulella mesomelas) used the tyres most frequently (Weise et al. 2014). Setting up electrified fencing around crops of high value would also provide a deterrent to porcupines and minimise agricultural losses.
Even though porcupines probably play a major role in maintaining the heterogeneity of landscapes and facilitating the persistence of patches of endangered geophyte species in vegetation types of conservation priority (C. Bragg unpubl. data), the Cape Porcupine continues to be actively eradicated from agricultural landscapes in South Africa. Clearly their value in the ecosystem has been understated and their conservation value needs to be recognised.
Recommendations for land managers and practitioners:
- Porcupine-friendly fences must be encouraged in farm and ranch lands to reduce damage.
- Managers of high-priority vegetation types, such as Renosterveld, which contain high geophyte diversity, should be aware of the ecological value of porcupine herbivory and disturbance as a driver of diversity.
- The trade in Porcupine quills, both locally and internationally, must be properly documented and managed more sustainably and ethically.
- Promote “green corridors” in urban environments to limit the threat of roadkill and persecution by domestic animals.
Research priorities:
- Population genetic studies are urgently needed to establish the effective population size (Ne) and determine genetic diversity of local populations.
- Investigations into the extent and impact of local harvesting, either for bushmeat or the quill industry, are needed to determine the impact on the regional population.
- The frequency of snaring, inadvertent capture in game-fencing and electrocution, as well as roadkill incidents should be systematically documented to support scientific understanding and inform conservation strategies.
- Documenting the survival rates of juveniles, in natural and transformed habitats, is essential for accurately assessing recruitment within populations.
Encouraged citizen actions:
- Report sightings (inclusive of spoor and dropped quills) on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Do not buy quill décor items unless the product is shown to be from sustainably harvested sources.
Bibliography
Arbon, K. 2019. Predation of porcupine Hystrix africaeaustralis in the den by honey badger Mellivora capensis. Biodiversity Observations, 10, pp.10-4.
Bragg CJ, Donaldson JD, Ryan PG. 2005. Density of Cape porcupines in a semi-arid environment and their impact on soil turnover and related ecosystem processes. Journal of Arid Environments 61: 261–275.
Bragg CJ. 2003. Porcupine ecology in Nieuwoudtville, a hotspot of geophyte diversity. M.Sc. Thesis. University of Cape Town.
Chevallier N, Ashton B. 2006. A report on the porcupine quill trade in South Africa. IFAW (International Fund for Animal Welfare).
Collinson, W.J., Reilly, B.K., Parker, D.M., Bernard, R.T. and Davies-Mostert, H.T., 2015. An inventory of vertebrate roadkill in the greater Mapungubwe Transfrontier conservation area, South Africa. African Journal of Wildlife Research, 45(3), pp.301-311.
de Villiers MS, van Aarde RJ, Dott HM. 1994. Habitat utilization by the Cape porcupine Hystrix africaeaustralis in a savanna ecosystem. Journal of Zoology 232: 539–549.
de Villiers MS, van Aarde RJ. 1994. Aspects of habitat disturbance by Cape porcupines in a savanna ecosystem. South African Journal of Zoology 29: 217–220.
Ehlers Smith, Y.C., Ehlers Smith, D.A., Ramesh, T. and Downs, C.T. 2018. Forest habitats in a mixed urban-agriculture mosaic landscape: patterns of mammal occupancy. Landscape Ecology, 33(1), pp.59-76.
Gomez, L. 2021. The illegal hunting and exploitation of porcupines for meat and medicine in Indonesia. Nature Conservation, 43, pp.109-122.
Haim, A., Van Aarde, R.J. and Skinner, J.D. 1990a. Metabolism and thermoregulation in the Cape porcupine, Hystrix africaeaustralis. Physiological Zoology, 63(4), pp.795-802.
Haim, A., Van Aarde, R.J. and Skinner, J.D. 1990b. Metabolic rates, food consumption and thermoregulation in seasonal acclimatization of the Cape porcupine Hystrix africaeaustralis. Oecologia, 83(2), pp.197-200.
Haim, A., Van Aarde, R.J. and Skinner, J.D. 1992. Urinary characteristics of the Cape porcupine Hystrix africaeaustralis: effects of photoperiod and temperature. Journal of Basic and Clinical Physiology and Pharmacology, 3(2), pp.165-175.
Hayward MW, White RM, Mabandla KM, Bukeye P. 2005. Mammalian fauna of indigenous forest in the Transkei region of South Africa: an overdue survey. South African Journal of Wildlife Research 35: 117-124.
Hayward MW. 2009. Bushmeat hunting in Dwesa and Cwebe Nature Reserves, Eastern Cape, South Africa. South African Journal of Wildlife Research 39: 70-84.
Heinrich, S., Toomes, A. and Gomez, L. 2020. Valuable stones: The trade in porcupine bezoars. Global Ecology and Conservation, 24, p.e01204.
Hystrix africaeaustralis (ASM Mammal Diversity Database #1001283) fetched October 16, 2025. Mammal Diversity Database. 2025. https://mammaldiversity.org/taxon/1001283
Kraai, U.M., Tsvuura, Z., Tjelele, T.J., Mkhize, N.R. and Kraai, M. 2022. Utilisation of woody plants by the cape porcupine in Mesic savannas in South Africa. Zoological Studies, 61, p.e40.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of sub-Saharan Africa: a biogeographic and taxonomic synthesis. De Gruyter, Berlin, Germany.
Ngcobo, S.P., Wilson, A.L. and Downs, C.T. 2019a. Home ranges of Cape porcupines on farmlands, peri-urban and suburban areas in KwaZulu-Natal, South Africa. Mammalian Biology, 96(1), pp.102-109.
Ngcobo, S.P., Wilson, A.L. and Downs, C.T. 2019. Habitat selection of Cape porcupines in a farmland-suburban context in KwaZulu-Natal, South Africa. Mammalian Biology, 98(1), pp.111-118.
Peterhans, J.C.K., Celesia, G.G. and Gnoske, T.P. 2019. Lion-porcupine interactions in Africa, including impacts on lion predatory behavior. Journal of East African Natural History, 108(1), pp.1-15.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Ramesh T, Downs CT. 2015b. Impact of land use on occupancy and abundance of terrestrial mammals in the Drakensberg Midlands, South Africa. Journal for Nature Conservation 23: 9-18.
Ramesh T, Kalle R, Rosenlund H, Downs CT. 2016. Native habitat and protected area size matters: Preserving mammalian assemblages in the Maputaland Conservation Unit of South Africa. Forest Ecology and Management 360: 20-29.
Rust NA, Nghikembua MT, Kasser JJW, Marker LL. 2015. Environmental factors affect swing gates as a barrier to large carnivores entering game farms. African Journal of Ecology 53: 339-345.
Sardans, J., Miralles, A., Tariq, A., Zeng, F., Wang, R. and Peñuelas, J. 2024. Growing aridity poses threats to global land surface. Communications Earth & Environment, 5(1), p.776.
Schumann M, Schumann B, Dickman A, Watson LH, Marker L. 2006. Assessing the use of swing gates in game fences as a potential non-lethal predator exclusion technique. South African Journal of Wildlife Research 36: 173-181.
Shaw JM, Merwe R van der, Merwe E van der, Ryan PG. 2015. Winter scavenging rates under power lines in the Karoo, South Africa. African Journal of Wildlife Research 45: 122–126.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skinner, J.D., Van Aarde, R.J. and Van Jaarsveld, A.S. 1984. Adaptations in three species of large mammals (Antidorcas marsupialis, Hystrix africaeaustralis, Hyaena brunnea) to arid environments. South African Journal of Zoology, 19(2), pp.82-86.
Weise FJ, Wessels Q, Munro S, Solberg M. 2014. Using artificial passageways to facilitate the movement of wildlife on Namibian farmland. South African Journal of Wildlife Research 44: 161-166.
van Aarde RJ, van Wyk V. 1991. Reproductive inhibition in the Cape porcupine, Hystrix africaeaustralis. Journal of Reproduction and Fertility 92: 13–19.
van Aarde RJ. 1985a. Age determination of Cape porcupines, Hystrix africaeaustralis. South African Journal of Zoology 20: 232–236.
van Aarde RJ. 1985b. Reproduction in captive female Cape porcupines (Hystrix africaeaustralis). Journal of Reproduction and Fertility 75: 577–582.
van Aarde RJ. 1987a. Pre-and postnatal growth of the Cape porcupine Hystrix africaeaustralis. Journal of Zoology 211: 25–33.
van Aarde RJ. 1987b. Reproduction in the Cape porcupine Hystrix africaeaustralis: An ecological perspective. South African Journal of Science 83: 605–607.
van Aarde RJ. 1987c. Demography of a Cape porcupine, Hystrix africaeaustralis, population. Journal of Zoology 213: 205–212.