Cape Mountain Zebra
Equus zebra ssp. zebra

2025 Red list status
Least Concern
Regional Population Trend
Unknown
change compared
to 2016
No Change
Overview
Equus zebra ssp. zebra – Linnaeus, 1758
ANIMALIA – CHORDATA – MAMMALIA – PERISSODACTYLA – EQUIDAE – Equus – zebra – zebra
Common Names: Cape Mountain Zebra (English), Bergkwagga, Kaapse Bergsebra (Afrikaans), Daou (Khoikhoi), Dou (San), iDauwa (Xhosa), Cebra Montanesa del Cabo (Spanish; Castilian), Zèbre de montagne du Cap (French)
Synonyms: No Synonyms
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: de Villiers, M.S.1, Kerley, G.2 & da Silva, J.M.3
Reviewers: Shultz, S.4 & Britnell, J.2
Institutions: 1CapeNature, 2Nelson Mandela University, 3South African National Biodiversity Institute, 4University of Manchester
Acknowledgements: Assessors and contributors of previous versions are thanked for providing the foundation for this assessment.
Previous Assessors & Reviewers: Hrabar, H., Birss, C., Peinke, D., King, S., Novellie, P. & Child, MF.
Previous Contributor: Dean Peinke
Assessment Rationale
The Cape Mountain Zebra is endemic to southern South Africa, and occurs in the Nama Karoo, Succulent Karoo, Grassland and Fynbos Biomes of the Western and Eastern Cape Provinces, extending marginally into the Northern Cape Province. Although extra-limital subpopulations exist, they are not included in this assessment. Reduced to fewer than 80 individuals in the 1950s, the current (2022/23) mature population size (excluding extralimital subpopulations) is between 2,852 to 3,889 individuals (using a 55% and 75% mature population structure, respectively). About half (51%) of the population (1461-1993 mature individuals) occurs on formally protected areas. Overall, the population increased steadily over a period of approximately three generations (1986-2014): average annual rate of subpopulation growth was 8.6% from 1985-1995; 9.6% from 1995-1998; 8.3% from 2002-2009; and 9.2% from 2009-2014. However, population growth slowed considerably between 2014 and 2023.
Major threats to Cape Mountain Zebra include a loss of genetic diversity through inbreeding and genetic drift, hybridisation with other equids including Hartmann’s Mountain Zebra (Equus zebra hartmannae) and Plains Zebra (Equus quagga), and a shortage of large areas of suitable habitat. Priority conservation interventions are (a) the establishment of a scientifically-based metapopulation management plan with the aim of enhancing genetic diversity amongst isolated subpopulations, (b) genetic testing for hybridization and management of at-risk subpopulations, and (c) an ongoing drive to secure suitable habitat within the natural distribution range through protected area expansion, including biodiversity stewardship agreements and (d) the growth of private subpopulations.
The population has been consistently increasing for over four decades, the minimum number of mature animals in the subpopulation is estimated to be 2852, and there have been more than 1000 mature individuals in formally protected areas alone for more than 10 years. Cape Mountain Zebra are therefore listed as Least Concern. This is a conservation success story, but further action and collaboration between stakeholders is required to ensure that it continues on this trajectory.
With high risks of inbreeding, genetic drift and a loss of heterozygosity due to extensive population fragmentation, the Cape Mountain Zebra remains a Conservation Dependent species which requires urgent metapopulation management and careful monitoring of subpopulation performance and heterozygosity. This applies especially to subpopulations in small areas with limited grassy habitat.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation:de Villiers MS, Kerley G & da Silva JM. 2025. A conservation assessment of Equus zebra ssp zebra. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
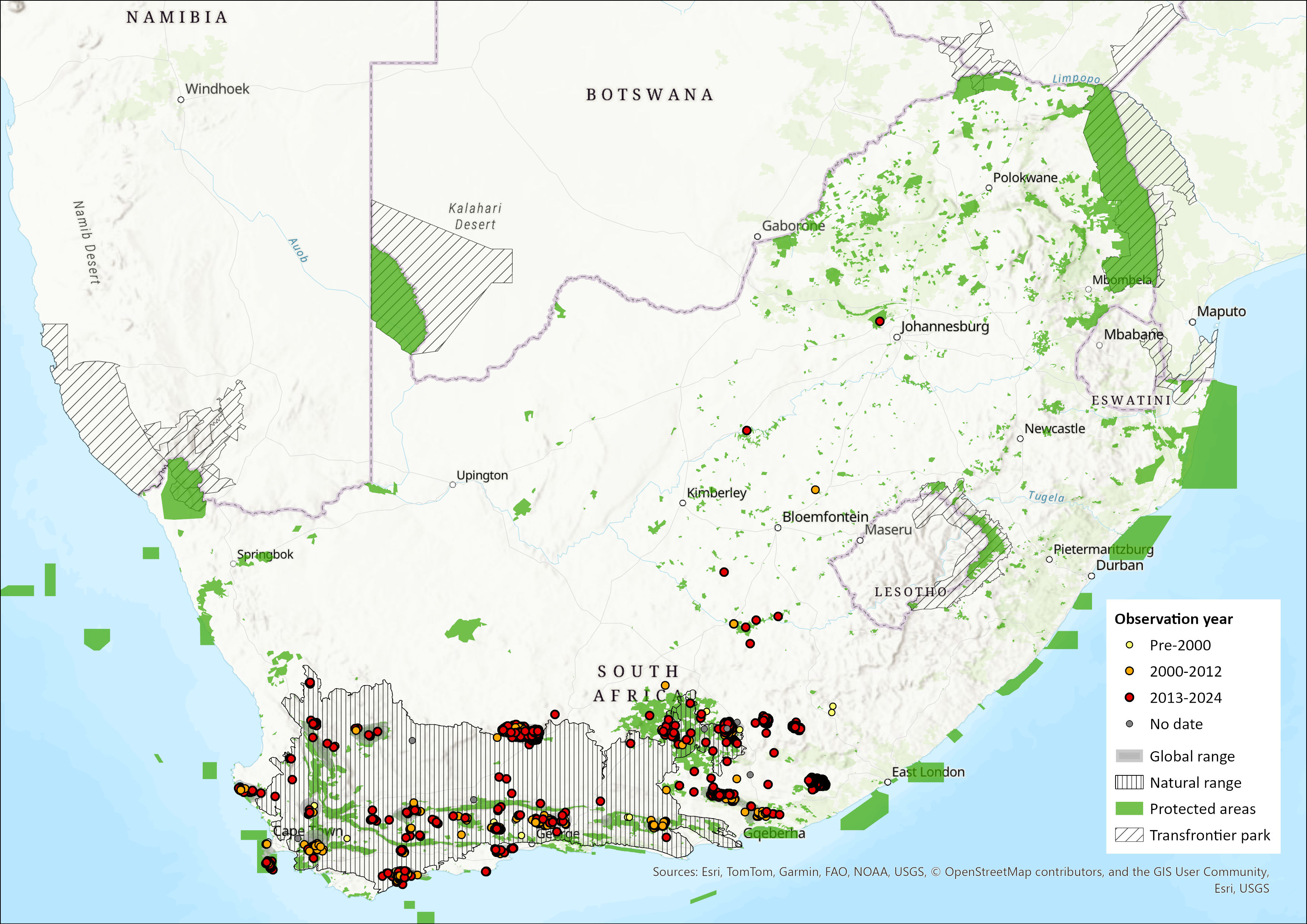
Geographic Range
The Cape Mountain Zebra is endemic to southern South Africa and occurs in the Nama Karoo, Succulent Karoo, Grassland and Fynbos Biomes (Skead 2007, 2011; Boshoff et al. 2015). Its historical distribution extended throughout the Great Escarpment range in the Cape, south of the Orange River, including the Cape Fold Belt Mountains (the southern parts of the current eastern Western Cape Province and the western mountains of the Eastern Cape) and the southern extent of the Northern Cape province (Figure 1). It is postulated that in historical times, Cape Mountain Zebra were separated from Hartmann’s Mountain Zebra (Equus zebra hartmannae) by the Knersvlakte, which separates the Kamiesberg in the north from the Roggeveldberge in the south (Novellie et al. 2002). There are no historical (pre-1920) records of Hartmann’s Mountain Zebra south of the Orange River (Skead 2011) and this subspecies now occurs mainly in Namibia.
Although once widely distributed throughout the mountainous regions of the former Cape Province, over-hunting and agricultural expansion reduced the Cape Mountain Zebra population to fewer than 80 individuals by the 1950s (Millar 1970a, b). Only three remnant subpopulations in the former Natural Distribution Range survived, these being in the Mountain Zebra National Park, Kammanassie Nature Reserve and Gamkaberg Nature Reserve (Smith et al. 2008). Subpopulations were subsequently introduced or reintroduced to a number of protected areas. In the Western Cape, these included Karoo, Addo Elephant, Bontebok, Tankwa Karoo and Camdeboo national parks, and De Hoop and Anysberg nature reserves. Cape Mountain Zebra were also moved onto Commando Drift and Tsolwana nature reserves in the Eastern Cape, and Baviaanskloof Wilderness Area (Hrabar and Kerley 2015)
Based on data collected between 2021 and 2023, the current distribution is on 108 fenced and isolated subpopulations in or close to the historical range (Scientific Authority of South Africa 2024). There are also four subpopulations in the Free State and North West provinces but these are significantly extralimital (Boshoff and Kerley 2013) and are excluded from this assessment. The Commando Drift and Tsolwana Nature Reserve subpopulations might be extralimital, as there are no historical records of the species east of the Great Fish River (Skead 2007); these subpopulations are also excluded here. Novellie et al. (2002) regarded the West Coast National Park as being within the historical range but there is some doubt about this given that it is on the coast and 70 km from the closest historically-recorded subpopulation in Piketburg (Skead 2011); for the purposes of this assessment, this subpopulation is excluded. The total area currently potentially available to the Cape Mountain Zebra is estimated to be greater than 935,191 ha and capable of supporting a much larger population than currently exists (Scientific Authority of South Africa 2024). There is thus no need to introduce this subspecies outside its natural range for conservation purposes.
The area of occupancy (AOO) has increased from a total AOO of 8,566 km² in 2016 to 19 293 km² in 2022/2023. There has been significant expansion of protected areas since 2016 (e.g., the purchase of 5,669 ha to be incorporated into Gamkaberg Nature Reserve, and the addition of 397 ha to Karoo National Park) and growth of the private sector contribution (e.g., a contribution of 52,100 ha by Sanbona Nature & Wilderness Reserve, and the creation and expansion of the Mountain Zebra-Camdeboo Protected Environment). However, Cape Mountain Zebra habitat is likely to remain severely fragmented due to game fencing and habitat transformation. Translocations between subpopulations as part of a metapopulation plan potentially reduce the impacts (such as loss of genetic diversity) of fragmentation, but these do not take place consistently enough (Hrabar and Kerley 2015). Thus, further reintroductions to formally and privately protected areas should be facilitated by a metapopulation management plan and should follow the International Union for the Conservation of Nature (IUCN) guidelines on translocations (IUCN/SSC 2013).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Cape Mountain Zebra (Equus zebra ssp. zebra) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Eastern Cape Province | Extant | Native | – | Resident |
| South Africa -> Free State | Extant | Introduced | – | Resident |
| South Africa -> Northern Cape Province | Extant | Introduced | – | Resident |
| South Africa -> Western Cape | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The climate of the Greater Cape Floristic Region (representing core habitat for the Cape Mountain Zebra) has warmed over the last century, and further warming as well as moderate drying are predicted (Altwegg et al. 2014). The impacts of climate change will vary between and within biomes (Department of Environmental Affairs 2015). Overall, the distribution of the Grassland Biome is predicted to shrink. Temperatures are likely to increase, with increased fire intensity. Climate change impacts on rainfall are equivocal, but rainfall variability is predicted to increase. Increased temperatures and CO2 concentrations are likely to result in the invasion of woody plants into grassland, with a concomitant decrease in habitat suitability for Cape Mountain Zebra. The Fynbos Biome is predicted to experience a decrease in rainfall frequency and an increase in rainfall intensity, as well as increased summer rainfall in the east (Department of Environmental Affairs 2015). Altered fire regimes will impact biodiversity patterns and processes, especially in lowland areas, but might favour Cape Mountain Zebra by promoting grasses over woody plants in places. However, this might be overshadowed by the impact of increasing pressure on water resources available to the subspecies. The Nama Karoo, between the Succulent Karoo Biome in the west and the mesic Savanna Biome in the west, is expected to be at risk from high temperatures exceeding physiological comfort thresholds for livestock. Elevated temperatures, combined with certain rainfall thresholds, could result in increased pests and pathogens. A higher frequency of extreme rainfall events is also a risk. A preferred anthropogenic adaption is a switch from agriculture to ecotourism and wildlife management, which could result in greater habitat availability for Cape Mountain Zebra. The Succulent Karoo biome is expected to remain stable over time, with the Desert Biome expanding into areas previously occupied by the Nama-karoo Biome. However, reduced rainfall and increased drought frequency might result in decreased forage quality and quantity and thus a deterioration in habitat suitability, especially for the Gamkaberg subpopulation (Department of Environmental Affairs 2015).
Overall, however, it is expected that the distribution of Cape Mountain Zebra will decrease due to climate change. The species distribution model for the period 2014-2070 predicts a change in Cape Mountain Zebra habitat of between -3% and -20%, and for the period 2071-2100, a change of between -23% and +15% (Cowan 2025).
Population Information
The Cape Mountain Zebra is the smallest living zebra, and differs from Hartmann’s Mountain Zebra by its smaller size, slightly thicker black stripes, minor striping variations on the rump, and a shorter mane which doesn’t extend onto the forehead beyond the ears (Novellie et al. 2002). Typical breeding herds consist of small harems comprising an adult stallion and one to three (maximum five) mares and their dependent foals. Non-breeding groups consist primarily of bachelor stallions but sometimes include juvenile females (Penzhorn 2013), and dispersal from natal herds is a mechanism of inbreeding avoidance. The average breeding group size ranges from 3.4–3.8 individuals (Klingel 1968, Penzhorn 1984, Smith et al.2008). Bachelor group size has been estimated at 2.5±1 (Lloyd and Rasa 1989). Breeding herds are long-term, stable social structures and tenure by herd stallions and breeding mares is often life-long (Rasa and Lloyd 1994). On De Hoop, dominance in mares correlates with fitness (Lloyd and Rasa 1989). Generation length has been calculated as 16 years. Gestation period is approximately 12 months and mares can produce a single foal every 25 months (range 12-69 months; birth rate of 0.337-0.46 foals per female per year for females >30 months) (Smith et al. 2008). Penzhorn and Lloyd (1987) recorded age at first foaling as 38-105 months, although a two-year old female produced a foal at Sanbona Nature & Wilderness Reserve (L. Eichenberger pers. comm.) Females >21 years old can still reproduce (Penzhorn and Lloyd 1987). Individuals can live up to ca 26 years. On De Hoop Nature Reserve, a predator-limited environment, survivorship during the first year of life was found to be 82.9% and 77.5% for male and female foals, respectively. Seventy six percent of surviving male foals and 84% of females survived to maturity (Smith et al. 2008). Infanticide by mares was inferred based on indirect evidence in the 1980s (e.g. Lloyd and Rasa 1989) but evidence for this behaviour has only recently been documented and suggested to occur after stallion turnovers (Britnell et al. 2021).
Cape Mountain Zebras were once widespread and numerous within their historical range (Skead 2011). Hunting, persecution and habitat loss to agriculture in the 20th and 21st centuries reduced them to just 80 individuals in three relict subpopulations in the 1950s (Bigalke 1952; Millar 1970a, b). The founders of these subpopulations were 19, five and six individuals respectively on Mountain Zebra National Park (MZNP), Kammanassie Nature Reserve and Gamka (now Gamkaberg) Nature Reserve (Millar 1970a, b; Lloyd 1984). The total population therefore bottlenecked at around 30 individuals at three locations. There has since been a significant population recovery and the current population, based on surveys conducted between 2021 and 2023, is estimated at 5,185 individuals. (This excludes the extralimital subpopulations listed above; there are a further 590 individuals located significantly outside of the Natural Distribution Range.) It should, however, be noted that there was limited data available for private properties in the Eastern Cape at the time of this assessment. Demographic data from nine subpopulations from the Western Cape (in systems with minimal mortality/few predators) revealed a mature population size of ca 67% based on average numbers of mature individuals in both breeding and stallion herds (C. Birss unpubl. data). To compensate for variation between areas, we use a mature population structure of 55–75%, which translates the current total of 5185 to between 2,852-3,889 mature individuals. The two largest subpopulations are on the Mountain Zebra and Karoo national parks, which together support 37% of the global population. The population now comprises 108 isolated and mostly small subpopulations, with an average size of 204 individuals (range 2-1223) on protected areas and 23 on private land in the Western Cape (range 4-71). The contribution of private land to Cape Mountain Zebra conservation has increased steadily over the years from 1985-2015 (Hrabar and Kerley 2015). In 2022-2023, the population had grown to 5,185 individuals, 2,657 of which were on formally protected areas.
Estimating annual growth rate trends is challenging because most subpopulations are actively managed (maintained at sustainable stocking levels) and animals are often removed or augmented, which masks true growth rate. Also, growth rates vary between subpopulations and habitats. However, the available information (Novellie et al. 1996; Novellie et al. 2002; Hrabar and Kerley 2013, 2015; Scientific Authority of South Africa 2024) indicates that the population grew steadily from 1985 to 2015, with an annual average growth rate of between 8 and 10%. Since 2015, however, the rate of increase has declined considerably. This in part reflects the confinement of some populations to grass-poor habitats (Lea et al. 2016), which are situated at the edges of the Cape Mountain Zebra’s ‘historical climatic niche (Britnell et al. 2024). Such ecological marginalization (Britnell et al. 2023) is reflected in declines in fecundity (foal: mare ratio), density and population growth (Britnell et al. 2024).
An increase in available habitat has contributed to the growth of the population over time. Privately-owned land played a crucial role in preventing the extinction of the Cape Mountain Zebra, when the last few animals in the Cradock area were saved from extinction by local farmers in the 1930s (Skead 2011). This subpopulation was formally protected in 1937 by the proclamation of the MZNP, which was expanded in 1964 to incorporate Cape Mountain Zebra subpopulations occurring on neighbouring private farms (Penzhorn 1975). The subsequent increase of the MZNP subpopulation enabled the translocation of individuals to 25 other protected areas during the 1980s and early 1990s, a number of which were private game ranches (Novellie et al. 2002). The expansion of MZNP and Karoo National Park allowed the growth of the two largest subpopulations. Similarly, Eastern Cape Parks and Tourism Agency (and its predecessors) removed 235 Cape Mountain Zebra from Commando Drift and Tsolwana nature reserves (166 from Command Drift and 69 from Tsolwana). Twenty-nine of these animals were translocated to the Baviaanskloof Nature Reserve and a further 206 were sold to the private sector. In the Western Cape, Anysberg and Gamkaberg nature reserves have both expanded in area, and stewardship agreements have assisted the establishment of Cape Mountain Zebra on some private properties. For example, in 2016, 28 zebra from De Hoop Nature Reserve were translocated to Sanbona Nature & Wilderness Reserve. Currently, Cape Mountain Zebra have limited dispersal opportunities due to landscape fragmentation and anthropogenic barriers such as fences and roads. The translocation of animals out of established subpopulations not only reduces density-dependent feedback in these subpopulations but creates subpopulations in new areas, each with the potential to increase, while at the same time securing additional habitat. This approach has greatly improved the conservation status of the Cape Mountain Zebra.
Key life history parameters differ according to habitat type and Winker et al. (2016) developed three population scenarios based on different sets of survival and fecundity input parameters. Based on these scenarios and using actual population data from 1985 to 2015, forward projections were made for nine subpopulations of Cape Mountain Zebra. These subpopulations showed considerable variation in growth rates but growth was highest for reserves at the eastern edge of the Natural Distribution Range and lowest for Gamkaberg Nature Reserve. Population growth performance was generally better in the Nama Karoo Biome and the interface between the Nama Karoo and Grassland Biomes, than in the Fynbos Biome (Winker 2016).The two largest populations (MZNP and Karoo NP) were predicted to reach their upper limits by 2020 (Winker et al. 2016), indicating the need for further reserve expansion to maintain population growth. In fact, actual population figures in 2020 for these reserves were closer to the lower than the upper confidence intervals of the predictions and thus somewhat lower than the estimated limit (MZNP, 876 actual compared to 1200 limit, and Karoo NP 773 actual compared to 1000 limit) (data supplied by SANParks, average of 2019 and 2021 counts used for Karoo NP). This can be at least partly attributed to off-takes in the intervening years, and the impact of lion predation (both these national parks support lion populations). The 2020 population on De Hoop Nature Reserve and neighbouring Overberg Test Range was 72 (de Villiers 2021), lower than the predicted lower confidence interval (81) and far lower than Winker et al.’s (2016) estimated carrying capacity (170). This was partly due to the translocation of 28 Cape Mountain Zebra from the area to Sanbona Nature & Wilderness Reserve in 2016, however competition with other large herbivores likely also plays a role (de Villiers and Shaw 2019). Of particular concern is the small, relict population of genetically distinct Cape Mountain Zebra at Gamkaberg Nature Reserve, which contains one-third of the genetic diversity of the taxon. The predicted upper limit for this population was 40 and currently, this population totals only 30 individuals (CapeNature, unpublished data). The inclusion of additional suitable habitat into Gamkaberg Nature Reserve to allow for population growth is thus critical. To this end, three properties near Gamkaberg, totalling 5669 ha, were purchased in recent years, with the ultimate aim of linking Gamkaberg to Outeniqua Nature Reserve. The incorporation of several additional properties into Gamkaberg is currently under consideration.
Population Information
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: 1223
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
The designation of Hartmann’s Mountain Zebra and the Cape Mountain Zebra as separate Evolutionary Significant Units (ESUs) has been debated. These subspecies are not reciprocally monophyletic and hence do not fit the definition of an ESU according to Moritz (1994); however, they do demonstrate highly restricted gene flow. Accordingly, these subspecies were considered as separate conservation management units in the 2016 Red Data Book, and are treated as such in this revision.
As a result of historical anthropogenic isolation of small subpopulations, the Cape Mountain Zebra has low genetic diversity represented in three relict, genetically distinct stocks: Cradock, Gamkaberg and Kammanassie (Moodley and Harley 2005), with 95% of the population belonging to Cradock stock. Until 2016, De Hoop Nature Reserve and neighbouring Overberg Test Range were the only sites with a mixture of stocks (Cradock and Kammanassie) and had the highest genetic diversity (Moodley and Harley 2005).
While some genetic structure exists, the diversity within and between localities is extremely low (Moodley and Harley 2005; Smith et al. 2007; Kotze et al. 2019). Some isolated subpopulations have lost multiple private alleles and show increased susceptibility to diseases such as equine sarcoids (Sasidharan, 2006; Sasidharan et al. 2011), highlighting the risk of inbreeding depression. Indeed, estimates of effective population size (Ne) for animals in the Mountain Zebra National Park (MZNP)– the largest subpopulation– are critically low (Ne = 1.7-15.1) due to very high levels of inbreeding. Despite population estimates of over 1000 mature individuals at this location, the majority of zebras are therefore essentially genetically identical. The same is expected within the other, smaller subpopulations. Continued isolation will result in further loss of genetic variation through genetic drift. Given that the Cape Mountain Zebra population is highly fragmented with little potential for natural gene flow, and that the subspecies is recognized as a single management unit, translocations and breeding between sites are strongly encouraged and should primarily occur between subpopulations derived from different genetic stocks.
There have been four actions towards this aim:
- In 2016, 28 zebra were translocated from De Hoop to Sanbona Nature & Wilderness Reserve in the Little Karoo under a custodianship agreement with CapeNature. This subpopulation was supplemented over the years and, unlike the relatively stable DHNR-OTR subpopulation, grew considerably and totalled 73 individuals in 2023 (Eichenberger 2024).
- In 2021, CapeNature translocated an at-risk dispersing mare (Cradock-Kammanassie stock) from De Hoop Nature Reserve to Bredasdorp to Bontebok National Park.
- A genetic rescue project involving all three stocks was initiated in 2021. Under a custodianship agreement between CapeNature and Sanbona, three Gamkaberg stallions were translocated to a dedicated camp on Sanbona, to which seven mares of Cradock or Cradock-Kammanassie stock were later added. So far, the project has produced six foals, including the only Cape Mountain Zebra in existence which are a mixture of Cradock-Gamkaberg and Cradock-Gamkaberg-Kammanassie stock. The long-term aim is to integrate offspring from this project into other Cape Mountain Zebra subpopulations, thereby helping to restore the genetic integrity of the population.
- In 2024, a second genetic rescue project was initiated when a Gamkaberg stallion and two Kammanassie mares (all at-risk, dispersing individuals) were translocated to Zebraskop, a property purchased by WWF-SA and managed by CapeNature.
Further genetic profiling of individuals is strongly recommended to ensure maximal genetic diversity is achieved with every interbreeding effort to prevent further inbreeding.
Habitats and ecology
Palaeozoological evidence suggests that Mountain Zebra (Equus zebra) occupied open grassland and low-lying habitats during the late Pleistocene era, and that climatic changes during the Holocene resulted in vegetation changes which favoured browsers (Faith 2012). The absence of Cape Mountain Zebra from most Holocene sites in the Cape Floristic Region suggests that the historic distribution of this taxon represents a recent range expansion made possible by active human management of vegetation through fire to stimulate grassland production for livestock (Faith 2012). There is abundant evidence that access to grassy habitat is associated with subpopulation growth in Cape Mountain Zebra (Grobler 1983; Winkler and Owen-Smith 1995; Watson et al. 2005; Watson and Chadwick 2007; Kraaij and Novellie 2010; Smith et al. 2011; Weal et al. 2015; Lea et al. 2016; Lea et al. 2017 but see Britnell et al 2024b; Oliver 2019; Britnell et al. 2024). The north-eastern part of the Cape Floristic Region, with its summer rainfall and escarpment grasslands, is ideal for these bulk grazers. The southwest, with winter-rainfall and fynbos and succulent Karoo habitat, is not as suitable (Boshoff et al. 2002; Kerley et al. 2003; Lea et al. 2016; Britnell et al. 2024) and subpopulations in this region are regarded as “ecological refugees” persisting in sub-optimal habitat (Lea et al. 2016; Britnell et al. 2024) and isolated from one another by habitat loss and fragmentation and dispersal barriers such as fences.
Cape Mountain Zebras are bulk grazers which select for leafy, tufted and highly palatable grasses such as Themeda triandra, Cymbopogon pospischilii, Sporobolus fimbriatus and Panicum stapfianum. They are fairly adaptable, moving seasonally between upland and lowland habitats and increasingly utilizing browse as the quality and quantity of grass declines in winter (Penzhorn 1982; Novellie et al. 1988; Penzhorn and Novellie 1991). They do not graze as closely to the ground as many antelope species found in the same habitat (Grobler 1983). As hindgut fermenters, they require large volumes of forage (Novellie 1990).
This has important implications for the two genetically significant subpopulations at Kammanassie and Gamkaberg nature reserves. Studies on these reserves and on Bontebok National Park have found that the subspecies prefers recently burnt grass-rich areas (Watson et al. 2005, 2011; Watson and Chadwick 2007). Between 1974 and 2002, subpopulation growth at Gamka Nature Reserve improved after grass growth was stimulated by fire (Watson et al. 2005). In 2016 and 2017, the Gamkaberg subpopulation temporarily exceeded the estimated limit of 40 (Winker et al. 2016) after a veld fire (CapeNature, unpublished data), however, in 2022, the subpopulation comprised only 30 individuals. Several management options for this genetically important subpopulation were proposed in 2005: (a) translocation to another protected area; (b) acquisition of adjacent land; (c) burning preferred habitat at unnaturally short intervals; (d) forming a conservancy with adjacent landowners; and (e) leasing cultivated land for pasture (Watson et al. 2005). Of these, (a) and (b) have been implemented thus far. Trend data for the Kammanassie subpopulation are limited because of the rugged terrain and limited road network, but aerial surveys covering the reserve and neighbouring private land recorded 37 and 56 animals in 2018 and 2023 respectively. Most of the animals were located off the nature reserve on private land, and their distribution was influenced by a combination of habitat type, waterpoint distribution and topography (none located above 1200 masl (de Villiers 2024).Rainfall on the western side of the survey area has decreased since 2013 (CapeNature unpublished data) and this, together with over-abstraction, has resulted in many of the springs on the mountain drying up – 64% of the 55 natural springs that are monitored every year have dried up completely (CapeNature 2020). The impacts of this on the Kammanassie Cape Mountain Zebra has to some extent been ameliorated by management interventions, such as the installation of artificial watering points on the reserve and improved accessibility to waterpoints on nearby private properties (de Villiers 2024).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | Resident | Suitable | Yes |
Life History
| Generation Length | Justification | Data Quality |
| 16 | Pacific 2013 | medium |
| Age at maturity: female or unspecified |
| 38-105 months (but one record of 24 months) |
Age at Maturity: Male: 4-4.5 years (but can be as low as 3.5 years)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: Up to 26 years
Average Reproductive Age: e Hoop: 7 and 9 years for breeding stallions and mares, respectively. Mountain Zebra National Park: Median age 67 months (range 38-105 months).
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
| Gestation Time |
| 1 Years |
| Reproductive Periodicity |
| 25 months |
Average Annual Fecundity or Litter Size: 1 Birth rate of 0.337-0.46 foals per female per year for females >30 months
| Natural Mortality |
| 26% |
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Not a Migrant (but seasonal altitudinal movements)
Congregatory: Congregatory (year-round)
Systems
System: Terrestrial
General Use and Trade Information
Cape Mountain Zebra are utilised for trophy hunting, sport hunting, photographic tourism, live sales and the captive wildlife industry. Unlike several other wild ungulate species maintained in the private sector, this subspecies has not been subjected to artificial selection for economically or aesthetically desirable characteristics.
In 2016, Cape Mountain Zebra was transferred from CITES Appendix I to Appendix II. Trophy and sport hunting occur on private properties in the Eastern Cape and Western Cape, with permits issued if the criteria relating to monitoring and provision of data are met. Between 2017 and 2022, 58 wild-sourced trophies were exported from South Africa (CITES 2024). According to the Department of Forestry, Fisheries and the Environment’s Professional Hunter’s Register, about 257 animals were hunted by international clients between 2010 and 2021, however this figure is likely an overestimate resulting from confusion between Plains Zebra and Cape Mountain Zebra hunts (Scientific Authority of South Africa 2024). In the Western Cape, permits were issued to hunt a total of 51 Cape Mountain Zebra from 2018-2023. The demand for trophy hunting is limited due to regulatory constraints around the import of trophies into the United States under the USA Endangered Species Act of 1973. The market value of the subspecies initially increased sharply after transfer to Appendix II (from R20,000 to R65,000 per individual) (Scientific Authority of South Africa 2024). However, the current live sale value is estimated at less than R5000 per animal (Scientific Authority of South Africa 2024). The lack of financial incentive could risk disinvestment of some private landowners in conservation of the subspecies (Scientific Authority of South Africa 2024). Currently, no hunting of Cape Mountain Zebra takes place on any national or provincial protected areas
The main trade is in live animals. On protected areas, translocations are used as a tool to manage subpopulation sizes and/or to improve genetic diversity. Formally protected areas also sell to private landowners (102 out of 112 animals translocated from formally protected areas between 2009 and 2015 were moved to private land), but do not reintroduce animals from the private sector or captive-bred facilities (Hrabar and Kerley 2015). Most trade in live animals is, however, between private landowners. For example, at least 254 Cape Mountain Zebra were translocated within the private sector between 2009 and 2015 (Hrabar and Kerley 2015).
Illegal translocations and poaching occur on a limited scale. Some poaching for bushmeat has occurred in at least one subpopulation (Camdeboo National Park). Cases of Cape Mountain Zebra being hunted and sold or exported as Hartmann’s Mountain Zebra have also been reported. The CITES trade database holds records of six confiscated trophies from 1988 to 2022 (CITES 2024).
The effects of harvesting (for example, on behaviour, heterozygosity and fitness) are not monitored. The national management system is informal: there is no set structure to measure management activities against a larger adaptive management framework. In some cases, local management plans are available but there is no approved national plan that is aimed at managing the genetic integrity of the Cape Mountain Zebra.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Localised bushmeat consumption | Yes | Translocations/sales occur in most protected areas as part of management plans. |
National Commercial Value: Yes
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: (Not specified)
Threats
The primary threat to Cape Mountain Zebra is loss of genetic diversity. The original cause of this was the historical reduction of the population to about 30 individuals divided between three isolated and genetically distinct subpopulations. Approximately two-thirds of the genetic variation of the subspecies is now contained in two small subpopulations, representing the Gamkaberg and Kammanassie genetic stocks. Both have been described as refugee subpopulations, being characterized by relatively low population growth rates at least in part due to suboptimal habitat (Lea et al. 2016; Britnell et al, 2024). Loss of genetic diversity is not restricted to these two relict subpopulations; a decrease in over a period of 15 years was demonstrated for the Kammanassie Nature Reserve, De Hoop Nature Reserve and Mountain Zebra National Park subpopulations, with the largest reduction of heterozygosity (23%) at De Hoop (Kotzé et al. 2019). Further loss of diversity could reduce evolutionary potential and result in fitness-related issues (Kotzé et al. 2019).
The problems associated with small subpopulation size are exacerbated by the potentially reinforcing effects of poor offtake management practices. Offtakes not linked to a metapopulation management plan can retard recruitment and subpopulation growth rate by skewing sex ratios and disrupting social structures (Milner et al. 2007). When animals are sold and captured for translocation it is common practice to capture and translocate family groups and to ignore bachelor groups. This is particularly true when only small groups are sold or relocated. This practice can lead to an accumulation of males in the donor population which in turn can impact on the growth rate of these populations if not properly managed. Surplus males are required for the establishment of new herds with dispersing females, and a 1:1 sex ratio is therefore recommended for all removals. Loss of genetic diversity is further exacerbated by the social structure of the Cape Mountain Zebra, where a fraction of the males can dominate herds for an extended period of time, thereby reducing the effective population size further.
Problems associated with the fragmentation of the population into mostly small subpopulations are largely due to a lack of integrated, cross boundary, management action. Although a Biodiversity Management Plan for the subspecies has been published, achieving its objectives is constrained by poor alignment of regulations between South African regulatory authorities and limited capacity and resources for implementation of regulations (Novellie et al. 2017). Development of a metapopulation management plan which incorporates resource mobilization strategies is essential, and such a plan should be adopted into provincial and national conservation policies.
A second threat to the subspecies is hybridization with other equid species. Cape Mountain Zebra can hybridize with domestic equids, although the offspring are likely to be sterile. In 2024, for example, a zebra-donkey hybrid was found outside Baviaanskloof Wilderness Area (Dean Peinke, pers. comm.). A more serious threat is hybridization with other types of zebra. In 2015, it was estimated that 62% of the total population had been/was at risk of hybridization (Hrabar and Kerley 2015).
Hybridization between Hartmann’s Mountain Zebra (Equus zebra hartmannae) and Cape Mountain Zebra is a considerable risk. Hybrids are difficult to detect phenotypically and are reproductively viable so decreased genetic integrity can thus spread within a subpopulation. Although it is illegal to keep the two subspecies together, cases of hybridization do still occur. One Hartmann’s-Cape Mountain Zebra hybrid subpopulation was confirmed in the Eastern Cape through genetic testing (Hrabar and Kerley, 2015). Although all stallions were subsequently culled and replaced with Cape Mountain Zebra stallions, individuals from this site were used to establish at least two other subpopulations which may thus have contained hybrids. Genetic testing as a pre-requisite for translocations is thus critical. Since phenotypic assessments do not provide reliable results, SANBI’s Genetic Services Unit has developed genetic markers to test for hybrids. Some provinces have policies in place to prevent extralimital establishment and hybridization of this subspecies, e.g. KwaZulu-Natal and North-West provinces. Testing for hybrids is a requirement for translocations into and within the Western Cape. Despite this, hybridization is a major threat to the Anysberg Nature Reserve subpopulation. In 2016 a section of the boundary fence was destroyed in a fire, resulting in contact between res nullius Hartmann’s Mountain Zebra and the reserve’s Cape Mountain Zebra population; it is suspected that this has resulted in the birth of at least one hybrid foal.
Mountain Zebra rarely occur in sympatry with Plains Zebra as the former are adapted for life on rugged terrain due to their harder and faster growing hooves, thereby making them less suitable for habitation of soft flat plains (Skinner and Chimimba 2005). Initially, hybridization with Plains Zebra was not of great concern as fertile hybrids were thought to be unlikely due to the difference in chromosome numbers between the two species (Ryder et al. 1978, Cordingley et al. 2009). Plains Zebra were therefore introduced into four formally protected areas, including the MZNP in 1999 and Karoo NP in 1998 (the two largest CMZ populations), and into about 10 private populations. More recent evidence, however, shows that differences in chromosome number do not constitute a barrier to exchange of genes between equid species (Jónsson et al. 2014). In 2014, two Plains-Cape Mountain Zebra hybrids in MZNP were confirmed through genetic testing (Dalton et al. 2017). The fertility of these hybrids is not clear and further research into this threat is needed. All Plains Zebra have since been removed from MZNP. On MZNP, more than half of the Cape Mountain Zebra have been tested and found to be genetically pure.
Infrastructure such as roads and fences are barriers to movement and this can result in accidental mortalities when individuals attempt to disperse or to undertake seasonal migrations between high-lying and low-lying areas. Animals on public roads are at risk from vehicle collisions, and there have been cases of Cape Mountain Zebra dying as a result of being accidentally trapped in farm camps without water, and becoming entangled in fencing. Translocations to assist dispersal also carry risks and can result in mortalities.
A third threat is disease. Outbreaks of equine sarcoidosis were recorded at Bontebok National Park and Gariep Dam Nature Reserve (Sasidharan 2006; Marais et al 2007) and the occurrence of bovine papillomavirus-induced tumours in these subpopulations indicates a general immune system breakdown which might be related to inbreeding (Sasidharan et al. 2011). Although equine sarcoidosis is not fatal, animals with visible lesions are thought to act as a source of infection. Cape Mountain Zebra can also be asymptomatic carriers of African Horse Sickness (AHS), a disease which can be spread to domestic equids via midges or biting flies. This disease is not a threat to the subspecies but has certain management implications. Under the Animal Diseases Act of 1984, restrictions (set out by the Department of Agriculture in 2003) are in place for the movement of individuals, especially into the AHS-controlled areas of the Western Cape. Cape Mountain Zebra which are translocated from the AHS infected zone to the protection/surveillance zone must undergo a three-week quarantine and this has cost and logistical implications for metapopulation management. At Gamkaberg Nature Reserve and Sanbona Nature & Wilderness Reserve and, there have been a few cases of mortalities linked to dental abnormalities. These could be linked to deleterious alleles resulting from inbreeding, to inadequate diet, or simply old age.
A poorly understood but emerging threat is that of reintroduced large predators. This includes Lion (Panthera leo) (Addo Elephant, Mountain Zebra and Karoo national parks and Sanbona Nature & Wilderness Reserve) and Cheetah (Acinonyx jubatus) (MZNP and some private reserves). Data indicate that Cape Mountain Zebra were preferred prey for Lion in Karoo National Park (C. Tambling unpub. data), and anecdotal evidence suggests that Cheetah suppressed population growth in at least one privately-owned population. In 2022, there was an experimental introduction of fourteen Cape Mountain Zebra into the northern predator camp on Sanbona Nature & Wilderness Reserve and one zebra was soon killed by lions. A further two zebra were killed when lions broke into the southern, predator-free camp (Eichenberger 2023). Further monitoring and research are needed to assess the extent and implications of this threat on Sanbona and elsewhere.
Conservation
Past conservation measures, including strict regulations on trade (CITES), regulation of hunting and regulation of translocations, have effectively mitigated the major historical threats responsible for the critical losses in the 19th and first half of the 20th century. Conservation actions should now be guided by the Cape Mountain Zebra BMP-S (Birss et al. 2016) implementation framework, which is based on the following prioritized objectives:
- Maintenance of genetic diversity in the meta-population,
- Monitoring and research to inform adaptive management,
- Consistent and uniform implement legislation, regulations, policies and guidelines, and
- Effective communication, collaboration and coordination between stakeholders and the public.
Conservation measures to address the threat of loss of genetic diversity are now critical. The population is fragmented, with mostly small subpopulations. Genetic diversity is low due to the small number of founder individuals and lack of dispersal opportunities. Genetic diversity is represented in three distinct stocks. 95% of individuals are of Cradock stock while the remainder, belonging to the Gamkaberg and Kammanassie stocks, are located in suboptimal habitat which limits growth. The genetic rescue project at Sanbona Nature & Wilderness Reserve, and the planned genetic rescue project at Zebraskop near Gamkaberg Nature Reserve, are a first step towards integrating these alleles into the metapopulation. Over the years, there have been many assisted dispersals and range expansions by means of translocations. In the Western Cape, biological samples of all translocated individuals are submitted to the national Biobank. This database of genetic material would be crucial to determine, monitor, and/or manage genetic heterogeneity within the metapopulation. However, translocations are not guided by a metapopulation strategy which informs the number, sex, and frequency of additions/removals required in order to prevent any further loss of genetic variation. There are also no formal translocation guidelines in place, or a centralized record of translocations and their outcomes.
Protected Area expansion is key to population growth. Britnell et al. (2024) show that grass-rich habitat is most suitable for the subspecies, resulting in the highest fecundity, densities, and subpopulation growth. Such habitats are mostly found in the east and north-east of the Natural Distribution Range. This region should therefore serve as a focal area for protected area expansion. One such initiative is the planned expansion and connection of the Mountain Zebra National Park and Camdeboo National Park (Davis 2024). This expanded area has the potential to support a Cape Mountain Zebra subpopulation of about 10 000 individuals, which would exceed the estimated mean + 95% CI of the Minimum Viable Population size for mammals (Trail et al. 2007). This would clearly provide a very high level of confidence in the persistence of the Cape Mountain Zebra.
Population growth, particularly of the genetically significant but small Gamkaberg and Kammanassie subpopulations, is essential but is dependent on the extent and suitability of available habitat. Additional land near Gamkaberg Nature Reserve has been secured but corridors to allow the core Gamkaberg subpopulation to access this have not yet been created. There have been few opportunities for the expansion of Kammanassie Nature Reserve through land purchase, but stewardship plays an important role, with two Contract Nature Reserves adjacent to the nature reserve providing refuges for dispersing individuals. Another conservation mechanism which is being implemented in the Kammanassie area is that of non-binding custodianship agreements, whereby private landowners commit to implement best practices to protect dispersing Cape Mountain Zebra on their properties.
The private sector plays an important role in habitat expansion. Almost half of all subpopulations are located on private land. This is, however, hampered by the lack of financial incentives to introduce and manage subspecies. In the private sector, conservation was reportedly the most common motivation behind acquiring Cape Mountain Zebra, while hunting was the least common reason (Hrabar and Kerley 2015). Most private owners agreed with regulating the possession, translocation and hunting of Cape Mountain Zebra through a permit system (as long as the process is efficient), but around 50% of owners did not agree that the subspecies should be restricted to the natural distribution range. Such considerations should be taken into account in designing an incentive system.
Cape Mountain Zebra is vulnerable to hybridization with other equids, including Hartmann’s Mountain Zebra, Plains Zebra, horses and donkeys. The urgent need to eliminate the threat of hybridisation with Plains Zebra has been recognised by SANParks and plans are in place to remove all remaining Plains Zebra from areas with Cape Mountain Zebra (Hrabar and Kerley 2015). Furthermore, all individuals captured for translocation from affected SANParks subpopulations are subject to genetic testing and are kept in holding camps until confirmed as pure. Hybrid individuals are euthanised to prevent further genetic contamination. As the genetic integrity of Cape Mountain Zebra depends on their relative abundance to Plains Zebra in a subpopulation (sensu Piett et al. 2015), it is important to sustain large subpopulations of Cape Mountain Zebra. Managing the hybrid threat with from both Hartmann’s and Plains Zebra relies on active participation in the Biodiversity Management Plan. Incentives should be developed to encourage private landowners to participate.
Recommendations for land managers and practitioners:
- Determine the minimum viable population size through a population viability analysis.
- Develop a metapopulation management strategy which addresses genetic rescue (mixing of stocks) and genetic bolstering (exchanges between subpopulations originating from the same stock), and management actions required to ensure the viability of subpopulations of various sizes (e.g., the number, sex, and frequency of additions/removals required in order to prevent any further loss of genetic variation).
- Develop translocation guidelines. The population simulation model for Cape Mountain Zebra (Winker 2016) is one tool that can be used to evaluate alternative management strategies. Genetic testing for hybridization prior to movement of animals should be a pre-requisite and any hybrid individuals should be euthanised. Pathogen testing should also be a pre-requisite, e.g. testing for the tick-borne disease, equine piroplasmosis, which has been found in clinically healthy Cape Mountain Zebra (Smith et al. 2019). Prior to translocations or introductions, habitat suitability should be assessed based on indices that have been developed (e.g. Novellie and Winkler 1993), and post-translocation monitoring of performance and habitat use should inform refinement of the index over a wide range of habitats. The guidelines should be informed by research investigation the possible long-term effects of translocation on the animals involved and how these effects can be mitigated. They should include recommendations for essential post-release monitoring.
- Reduce the vulnerability of the Gamkaberg and Kammanassie subpopulations through habitat expansion (land purchase, stewardship, and informal custodianship agreements), including the creation of functional corridors that will allow the core subpopulations to access additional habitat. Expansion can include reclaimed agricultural lands that have been converted to grasslands for livestock.
- Reduce the vulnerability of small populations, including the Gamkaberg and Kammanassie subpopulations, through habitat management, e.g. implementation of integrated burn thresholds aimed at maintaining landscape diversity which includes areas of grassy fynbos, and employment of ecological stocking rates to reduce grazing competition with other game species.
Captive breeding and ex situ management are not necessary.
Research priorities:
- Investigate the impacts of reintroduced large predators on Cape Mountain Zebra subpopulations, and develop and implement appropriate management interventions to mitigate such impacts.
- Determine effective subpopulation size and minimum viable population size.
- Build on previous research into habitat suitability for Cape Mountain Zebra. Current evidence suggests that poor habitat suitability is a major factor limiting reproduction and population growth in key relict (and genetically distinct) populations, and habitat suitability is strongly linked to fecundity and reproductive success and thus effective population sizes, hybridisation, genetic erosion and inbreeding risks (Jake Britnell, pers. comm., 2025).
- Investigate the potential for expansion of the population within the available habitat based on an improved understanding of habitat suitability and niche marginality, with particular focus on the relict Gamkaberg and Kammanassie subpopulations (Britnell et al. 2024).
- Investigate the possible long-term effects of translocation on Cape Mountain Zebra.
- Assess the effectiveness of using integrated fire thresholds aimed at maintaining diversity inclusive of grassy habitats and subpopulation performance.
- Evaluate the severity of genetic threats, for example, the extent of hybridisation with Hartmann’s Mountain Zebra and Plains Zebra and the extent and consequences of inbreeding, including an improved understanding of how it relates to the sarcoid virus and other diseases.
Bibliography
Altwegg, R., West, A., Gillson, L. & Midgely, G.F. 2014. Impacts of climate change in the Greater Cape Floristic Region. In Allsopp, N., Colville, J.F. and Verboom, G.A. (eds): Fynbos: Ecology, Evolution, and Conservation of a Megadiverse Region (Oxford, 2014; online edn, Oxford Academic, 23 Oct. 2014), https://doi.org/10.1093/acprof:oso/9780199679584.003.0013, accessed 8 May 2024.
Bigalke, R. 1952. Early history of the Cape mountain zebra (Equus zebra zebra, Linn.). African Wildlife 6: 143–153.
Birss, C., Cowell, C., Hayward, N., Peinke, D., Hrabar, H.H. and Kotze, A. Biodiversity Management Plan
for the Cape Mountain Zebra in South Africa. Jointly developed by CapeNature, South African National
Parks, Eastern Cape Parks and Tourism Agency, National Zoological Gardens, Department of
Environmental Affairs, Northern Cape Department of Environment and Nature Conservation, Eastern
Cape Department of Economic Development, Environmental Affairs and Tourism and Free State
Department of Economic, Small business, Tourism and Environmental Affairs. Version 1.0.
Boshoff, A.F., Kerley, G.I.H. and Cowling, R.M. 2002. Estimated spatial requirements of the medium- to large-sized mammals, according to broad habitat units, in the Cape Floristic Region, South Africa. African Journal of Range & Forage Science, 19(1).
Boshoff, A.F. & Kerley, G.I.H. 2013. Historical incidence of the larger mammals in the Free State Province (South Africa) and Lesotho. Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth, South Africa.
Boshoff, A.F. Landman, M. and Kerley, G.I.H. 2015. Filling the gaps on the maps: historical distribution patterns of some larger mammals in part of southern Africa. Transactions of the Royal Society of South Africa. University of South Africa Press, Pretoria.
Britnell, J., Kerley, G.I.H., Antwis, R. and Shultz, S. 2024. A grazer’s niche edge is associated with increasing diet diversity and poor population performance. Ecology Letters 27: e14357.
Britnell, J.A., Vorster, L. and Shultz, S. 2021. Evidence of infanticide in the Cape mountain zebra. Behaviour 158: 1043-1055.
Britnell, J. A., Zhu, Y., Kerley, G. I. H., & Shultz, S. 2023. Ecological marginalization is widespread and increases extinction risk in mammals. Proceedings of the National Academy of Sciences, 120(3), e2205315120.
Britnell, J. A., Palme, R., Kerley, G. I., Jackson, J., & Shultz, S. 2024. Previous assessments of faecal glucocorticoid metabolites in Cape mountain zebra (Equus zebra zebra) were flawed. Functional Ecology, 38(9), 1862-1874.
CapeNature 2020. Swartberg Complex World Heritage Site & Nature Reserves: Protected Area Management Plan 2020-2030. Internal Report, CapeNature. Cape Town.
CITES 2024. CITIES Trade Database.
Cordingley JE, Sundaresan SR, Fischhoff IR, Shapiro B, Ruskey J, Rubenstein DI. 2009. Is the endangered Grevy’s zebra threatened by hybridization? Animal Conservation 12:505–513.
Cowan, OS. 2025. Modelling the change in potential species distribution of Cape Mountain Zebra (Equus zebra zebra) under predicted climate change scenarios. Technical Report, Endangered Wildlife Trust.
Department of Environmental Affairs. 2015. Climate Change Adaptation Plans for South African Biomes (ed. Kharika, J.R.M., Mkhize, N.C.S., Munyai, T., Khavhagali, V.P., Davis, C., Dziba, D., Scholes, R., van Garderen, E., von Maltitz, G., Le Maitre, D., Archibald, S., Lotter, D., van Deventer, H., Midgely, G. and Hoffman, T). Pretoria.
Dalton, D.L., Zimmermann, D., Mnisi, C., Taplin, M., Novellie, P., Hrabar, H. & Kotze, A. 2017. Hiding in plain sight: evidence of hybridization between Cape mountain zebra (Equus zebra zebra) and plains zebra (Equus quagga burchelli). African Journal of Wildlife Research 47(1): 59-64.
Davis, K. 2024. The Camdeboo-Mountain Zebra mega-reserve as an opportunity to protect viable populations of medium- to large-sized mammals. MSc thesis, Nelson Mandela University.
de Villiers, M.S. and Shaw. K. 2020. CapeNature Scientific Report to the Overberg Review Committee: Recommendations for Game Management on the Greater De Hoop Conservation Area in 2020. Unpublished report, CapeNature, Cape Town.
de Villiers, M.S. 2021. Annual biodiversity monitoring report: Cape mountain zebra, 2021-2022. Unpublished report, CapeNature, Cape Town.
de Villiers, M.S. 2024. Kammanassie aerial game survey, October 2023. Unpublished report, CapeNature, Cape Town.
Eichenberger, L. 2023. Annual Report – Cape Mountain Zebra.Unpublished report, Sanbona Nature and Wilderness Reserve NPC.
Eichenberger, L. 2024. Annual Report – Cape Mountain Zebra.Unpublished report, Sanbona Nature and Wilderness Reserve NPC.
Faith, J.T. 2012. Palaeozoological insights into management options for a threatened mammal: southern Africa’s Cape mountain zebra (Equus zebra zebra). Diversity and Distributions 18: 438–447.
Grobler, J. H. 1983. Feeding habits of the Cape mountain zebra Equus zebra zebra Linn. 1758. Koedoe 26: 159 – 168.
Henning, W., Novellie, P., Selier, J., Birss, C. and Hraber, H. 2016. Final report. Population trends and management strategy tools for Cape Mountain Zebra. Technical report commissioned by the Scientific Authority of South Africa.
Hrabar, H. and Kerley, G. I. H. 2013. Conservation goals for the Cape mountain zebra Equus zebra zebra—security in numbers? Oryx 47: 403–409.
Hrabar, H. & Kerley, G.I.H. 2015. Cape Mountain Zebra 2014/2015 Status Report. Centre for African Conservation Ecology Report 63, 32 pp.
IUCN. 2019. The IUCN Red List of Threatened Species. Version 2019-1. Available at: www.iucnredlist.org. (Accessed: 21 March 2019).
Jónsson H et al. 2014. Speciation with gene flow in equids despite extensive chromosomal plasticity. Proceedings of the National Academy of Sciences 111:18655–18660.
Kerley, G.I.H., Pressey, R.L., Cowling, R.M., Boshoff, A.F. and Sims-Castley, R. 2003. Options for the conservation of large and medium-sized mammals in the Cape Floristic Region hotspot, South Africa. Biological Conservation 112: 169 – 190.
Klingel H. 1968. Soziale Organisation und Verhaltensweisen von Hartmann-und Bergzebras (Equus zebra hartmannae und E. z.zebra). Mammalian Biology 25:76–88.
Komers PE, Curman GP. 2000. The effect of demographic characteristics on the success of ungulate re-introductions. Biological Conservation 93:187–193.
Kraaij, Tineke & Novellie, Peter. (2010). Habitat selection by large herbivores in relation to fire at the Bontebok National Park (1974-2009): The effects of management changes. African Journal of Range & Forage Science – AFR J RANGE FOR SCI. 27. 21-27. 10.2989/10220111003703450.
Kotzé A, Smith RM, Moodley Y, Luikart G, Birss C & van Wyk AM, et al. (2019) Lessons for conservation management: Monitoring temporal changes in genetic diversity of Cape Mountain Zebra (Equus zebra zebra). PLoS ONE 14(7): e0220331. https://doi.org/10.1371/journal.pone.0220331. Lea J, Kerley G, Hrabar H, Barry T & Shultz S (2016). Recognition and management of ecological refugees: A case study of the Cape Mountain Zebra. Biological Conservation 203: 207-215.
Lea, J.M.D., Walker, S.L., Kerley, G.I.H., Jackson, J., Matevich, S.C. and Shultz, S. 2017. Non-invasive physiological markers demonstrate link between habitat quality, adult sex ratio and poor population growth rate in a vulnerable species, the Cape mountain zebra. Functional Ecology 2017: 1-13.
Lloyd, P. 1984. The Cape Mountain Zebra 1984. African Wildlife 38(4): 144-149.
Lloyd, P.H. and Rasa O.A.E. 1989. Status, reproductive success and fitness in Cape mountain zebra (Equus zebra zebra). Behavioral Ecology and Sociobiology 25: 411–420.
Marais, H.and, Nel, P.and, Bertschinger, H.,and Schoeman, J. and Zimmerman, D. 2007. Prevalence and body distribution of sarcoids in South African Cape mountain zebra (Equus zebra zebra). Journal of the South African Veterinary Association. 78. 145-8. 10.4102/jsava.v78i3.306.
Millar, J. C. G. 1970a. Census of Cape mountain zebras: Part I. Afr. Wildlife 24: 17 – 25.
Millar, J. C. G. 1970b. Census of Cape mountain zebras: Part II. Afr. Wildlife 24: 105 – 114.
Milner JM, Nilsen EB, Andreassen HP. 2007. Demographic side effects of selective hunting in ungulates and carnivores. Conservation Biology 21:36–47.
Moodley, Y. and Harley, E.H. 2005. Population structuring in mountain zebras (Equus zebra): The molecular consequences of divergent demographic histories. Conservation Genetics 6: 953-968.
Moritz, C. 1994, Defining ‘Evolutionary Significant Units’ for conservation. Trends in Ecology & Evolution, 9(10): 373-375.
Novellie P. 1990. Habitat use by indigenous grazing ungulates in relation to sward structure and veld condition. Journal of the Grassland Society of Southern Africa 7:16–23.
Novellie, P., Birss, C., Cowell, C., Kerley, G.I.H., Peinke, D., Pfab, M., Selier, J. & Zimmermann, D. 2017. Adaptive governance of Cape mountain zebra, can it work? African Journal of Wildlife Research 47(2): 79-91.
Novellie PA, Fourie LJ, Kok OB, van der Westhuizen MC. 1988. Factors affecting the seasonal movements of Cape mountain zebras in the Mountain Zebra National Park. South African Journal of Zoology 23:13–19
Novellie, P., Lindeque, M. Lindeque, P., Lloyd, P. and Koen, J. 2002. Status and Action Plan for the mountain zebra (Equus zebra). In: P. D. Moehlman (ed.), Equids: Zebras, Asses and Horses. A Status Survey and Conservation Action Plan, pp. 28-42. IUCN, Gland, Switzerland.
Novellie, P. A., Millar, P. S. and Lloyd, P. H. 1996. The use of VORTEX simulations models in a long-term programme of re-introduction of an endangered large mammal, the Cape mountain zebra (Equus zebra zebra). Acta Oecologica 17: 657-671.
Novellie, P.A. & Winkler, A. 1993. A simple index of habitat suitability for Cape mountain zebras. Koedoe 36(2): 53-59.
Olivier J (2020) Ecology and habitat suitability of Cape Mountain Zebra (Equus zebra zebra) in the Western Cape, South Africa. M.Sc. Thesis, University of Stellenbosch, South Africa.
Penzhorn, B.L. 1975. Behaviour and population ecology of the Cape mountain zebra Equus zebra zebra Linn., 1758 in the Mountain Zebra National Park. Ph.D. thesis, University of Pretoria, Pretoria, South Africa.
Penzhorn, B.L 1982. Home range sizes of Cape Mountain Zebras Equus zebra zebra in the Mountain Zebra National Park. Koedoe 25: 103-108.
Penzhorn, B.L. 1984. A long-term study of social organisation and behaviour of Cape mountain zebras. Zeitschift für Tierpysychologle 64: 97-146.
Penzhorn B. 2013. Equus zebra Mountain Zebra. Pages 438–443 in Kingdon J, Hoffmann M, editors. Mammals of Africa. Volume V: Carnivores, Pangolins, Equids and Rhinoceroses. Bloomsbury Publishing, London, UK.
Penzhorn, B. L., Lloyd, P. H. 1987. Comparisons of reproductive parameters of two Cape mountain zebra populations. J. Reprod. Fert. Suppl. 35: 661 – 663.
Penzhorn, B.L. and Novellie, P.A. 1991. Some behavioural traits of Cape mountain zebras (Equus zebra zebra) and their implications for the management of a small conservation area. Applied Animal Behavioural Science 29: 293-299.
Piett S, Hager HA, Gerrard C. 2015. Characteristics for evaluating the conservation value of species hybrids. Biodiversity and Conservation 24:1931–1955.
Rasa, O.A.E. and Lloyd, P.H. 1994. Incest Avoidance and Attainment of Dominance by Females in Cape mountain zebra (Equus zebra zebra). Behaviour 128 (3 – 4).
Ryder OA, Epel NC, Benirschke K. 1978. Chromosome banding studies of the Equidae. Cytogenetic and Genome Research 20:323–350.
Sasidharan, S.P., 2006. Sarcoid tumours in Cape mountain zebra (Equus zebra zebra) populations in South Africa: a review of associated epidemiology, virology and genetics. Transactions of the Royal Society of South Africa 61(1), 11-18.
Sasidharan SP, Ludwig A, Harper C, Moodley Y, Bertshinger HJ & Guthrie AJ (2011) Comparative genetics of sarcoid tumour-affected and non-affected Mountain Zebra (Equus zebra) populations. South African Journal of Wildlife Research 41(1): 36 – 49.
Scientific Authority of South Africa. 2024. Non-detriment finding for Equus zebra zebra (Cape mountain zebra). Unpublished draft document, 19 February 2024.
Skead, C.J. 2007. Historical Incidence of the Larger Land Mammals in the broader Eastern Cape, Second Edition (eds. Boshoff, A.F., Kerley, G.I.H. and Lloyd, P.H.). Port Elizabeth: Centre for African Conservation Ecology, Nelson Mandela Metropolitan University. ISBN 1-920176-08-X
Skead, C.J. 2011. Historical Incidence of the Larger Land Mammals in the broader Western and Northern Cape, Second Edition (eds. Boshoff, A.F., Kerley, G.I.H. and Lloyd, P.H.). Port Elizabeth: Centre for African Conservation Ecology, Nelson Mandela Metropolitan University. ISBN 1-920176-08-X
Skinner, J.D. and Chimimba, C.T. (revisers) 2005. The Mammals of the Southern African Subregion. Third Edition. Cambridge University Press, Cape Town. ISBN 978-0521-84418-5.
Smith, R.K., Marais A., Chadwick, P., Lloyd, P.H. and Hill, R.A. 2008. Monitoring and management of the endangered Cape mountain zebra Equus zebra zebra in the Western Cape, South Africa. African Journal of Ecology 46: 207–213.
Smith, R.K., Ryan, E., Morley, E. and Hill, R.A. 2011. Resolving management conflicts: could agricultural land provide the answer for an endangered species in a habitat classified as a World Heritage Site? Environmental Conservation 38(3): 325-333.
Traill, L. W. Corey Bradshaw, J.A., and Brook, B. W. 2007. Minimum viable population size: A meta-analysis of 30 years of published estimates. Biol. Cons. 139:159–166.
Watson, L.H. and Chadwick, P. 2007. Management of Cape mountain zebra in the Kammanassie nature reserve, South Africa. South African Journal of Wildlife Research 37: 31–39.
Watson, L.H., Odendaal, H.E., Barry, T.J. and Pietersen, J. 2005. Population viability of Cape mountain zebra in Gamka Mountain Nature Reserve, South Africa: the influence of habitat and fire. Biological Conservation 122: 173–180.
Weel, S., Watson, L.H., Weel, J., Venter, J.A. & Reeves, B. 2015. Cape mountain zebra in the Baviaanskloof Nature Reserve, South Africa: resource use reveals limitations to zebra performance in a dystrophic mountainous ecosystem. African Journal of Ecology 53(4): 428-438.
Winker, H., Novellie, P., Selier, J., Birss, C. and Hrabar, H. 2016. Population trends and management strategy tools for Cape Mountain Zebra. Technical Report commissioned by the Scientific Authority of South Africa.
Winkler, A. and Owen-Smith, N. 1995. Habitat utilisation by Cape mountain zebras in the mountain Zebra National Park, South Africa. Koedoe, 38(1): 83 – 93.