
Cape Horseshoe Bat
Rhinolophus capensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Rhinolophus capensis – Lichtenstein, 1823
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – capensis
Common Names: Cape Horseshoe Bat, Southern Africa Horseshoe Bat (English), Kaapse Saalneusvlermuis, Kaapse Hoefystervlermuis (Afrikaans)
Synonyms: No Synonyms
Taxonomic notes: Although records have been reported from Zambia and Malawi, these have been shown to be misidentified (Ansell 1986; Koopman 1993). Considering this species is confined to the Fynbos and Succulent Karoo Biomes, it is unlikely to occur anywhere else on the continent (Monadjem et al. 2010). Together with R. denti, R. simulator, and R. swinnyi, R. capensis makes up the capensis species-group (Csorba et al. 2003).
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.1, Lotter, C.2, Bastian, A3. & da Silva, J.M.4
Reviewer: Howard, A.1
Institutions: 1University of the Free State, 2Inkululeko Wildlife Services (Pty) Ltd, 3University of KwaZulu-Natal, 4South African National Biodiversity Institute
Previous Assessors and Reviewers: Jacobs D, Cohen L, Richards LR, Monadjem A, Schoeman C, MacEwan K, Sethusa T, Taylor PJ.
Previous Contributors: Raimondo, D. & Child, M.F.
Assessment Rationale
An endemic species to South Africa. While declining in parts of its range, the species is listed as Least Concern in view of its wide distribution, its known large population (there are many records of this species occurring in high numbers in coastal caves), and because it is unlikely to be declining fast enough to qualify for listing in a more threatened category. The species has an EOO of 440,027 km2.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Taylor P, Lötter C, Bastian A & da Silva JM. 2025. A conservation assessment of Rhinolophus capensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This South African endemic is mainly restricted to the coastal belt, typically 100-200 km wide (but possibly further inland), of the Northern Cape, Western Cape and the Eastern Cape provinces of South Africa, and occurs from just south of the border of Namibia in the west as far east along the coast as the vicinity of East London (Skinner and Chimimba 2005). It occurs mainly in the South West Cape biotic zone (BZ), but extends marginally into the Karoo, Highveld, Coastal Forest Mosaic and Afromontane-Afroalpine BZs (Bernard 2013). We follow Herselman and Norton (1985) and Monadjem et al. (2020) in extending its range to just south of the Orange River on the Namibian border, but it may also occur in southern Namibia (Griffin 1999). As R. capensis is difficult to discern from R. acrotis (previously R. clivosus) and R. darlingi, records north of 32°S may need vetting (Bernard 2013). Similarly, one record from north-east Eastern Cape (Lynch 1989) needs vetting. Its current estimated extent of occurrence is over 440,027 km². Call recordings of this species suggest that this species occurs as far inland as Sanddrift and Victoria West in the Northern Cape, and Aberdeen and Molteno in the Eastern Cape (Inkululeko Wildlife Services unpubl. data).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 18 m asl (Mondajem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,833 m asl (Mondajem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
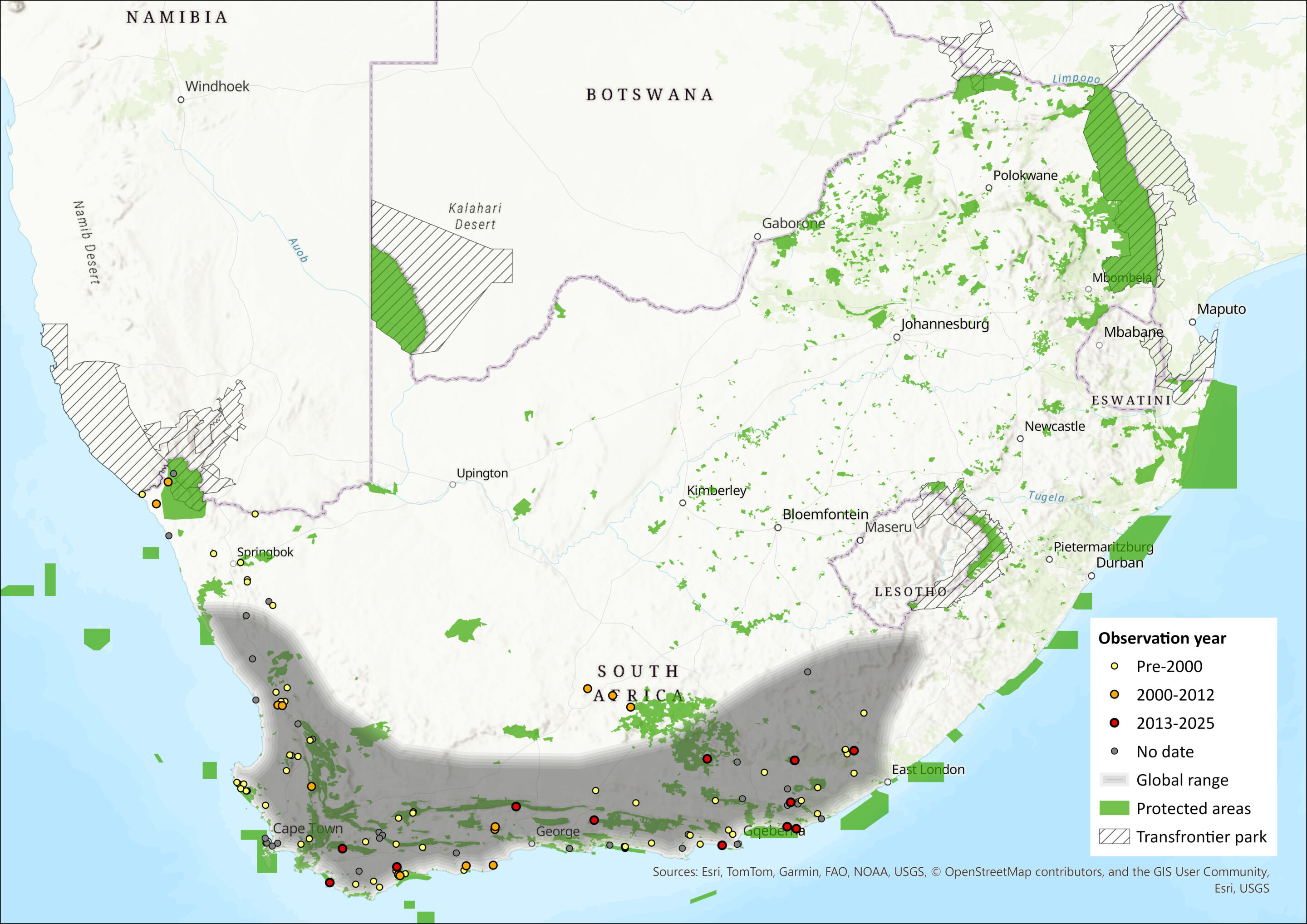 Figure 1. Distribution records for Cape Horseshoe Bat (Rhinolophus capensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Cape Horseshoe Bat (Rhinolophus capensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Namibia | Possibly Extant | Native | – | |
| South Africa | Extant | Native | – | – |
| South Africa -> Eastern Cape Province | Extant | Native | – | – |
| South Africa -> Northern Cape Province | Extant | Native | – | – |
| South Africa -> Western Cape | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Sherwin et al. (2012) identified seven risk factors for bats encompassing three broad aspects: 1) biogeography – small range size, high latitude or high altitude range and a range occupying a geographic area likely to become water stressed; 2) foraging niche – frugivory and species restricted to aerial hawking; and 3) dispersal ability – species with restricted dispersal behaviour. These identified risk factors suggest that R. capensis may be impacted by climate change more significantly than congeners which have larger distribution ranges and/or which occupy more temperate or mesic areas that are less prone to droughts (such as R. acrotis). Taylor et al. (2024) indeed reported an appreciable reduction of about 10-20% in the range of this species under future climate models.
Population Information
This species is common throughout its range (Bernard 2013) and is relatively well represented in museums (Monadjem et al. 2020). Skinner and Chimimba (2005) state that they are abundant in the Western Cape and the Eastern Cape, where there are many records from coastal caves. It can be found in colonies consisting of thousands of individuals (Herselman and Norton 1985, Taylor 2000, Skinner and Chimimba 2005). For example, there are an estimated 19,000 individuals in De Hoop Guano Cave (McDonald et al. 1990a), but future monitoring and updated estimates should be investigated since the last published information is now over 30 years old.
Current population trend: Stable
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: 19,000 individuals have been recorded in the De Hoop Guano Cave (McDonald et al. 1990a).
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Rhinolophus capensis has been investigated in a phylogenetic context and is represented as a single lineage across its range (Stoffberg 2008; Taylor et al. 2018; Taylor et al. 2024). While a fine scale population genetic study has not been undertaken, this, along with its ability to disperse between colonies, suggests the species exists as a single inbreeding metapopulation. However, climatic projections show a greatly reduced and bisected distribution for the species, which could result in isolated subpopulations to the west and south of South Africa.
Based on evidence that single colonies can be comprised of several thousand animals, it is likely that Ne for the species (population) exceeds 500.
Habitats and ecology
This bat has been recorded from a range of habitats but is generally closely associated with the Fynbos and Succulent Karoo Biomes (Monadjem et al. 2020). Populations roost in suitable coastal and sea caves, and have been recorded from dark lofts, and disused mines (Taylor 2000, Csorba et al. 2003) but apparently avoids houses (Bernard 2013). They often share caves with R. acrotis and Miniopterus natalensis (Herselman and Norton 1985, Stoffberg 2008). They forage predominantly in the canopy of trees (McDonald et al. 1990b), or in orchards surrounding wetlands and over the wetlands themselves (Sirami et al. 2013). They are clutter foragers, feeding primarily on Coleoptera and Lepidoptera (Jacobs et al. 2007, Monadjem et al. 2020). Small-scale migrations of 10 km have been recorded (Taylor 2000). Sometimes hibernates in winter but torpor not as deep as R. clivsous (R.T.F Bernard pers. obs.).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | Yes |
| 3.5. Shrubland – Subtropical/tropical dry | Suitable | Yes | |
| 3.6. Shrubland – Subtropical/tropical moist | Suitable | Yes | |
| 4.4. Grassland – Temperate | Suitable | Yes | |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | Yes |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Small-scale migrations of 10 km have been recorded (Taylor 2000).
Congregatory: The largest known colony (in De Hoop Guano Cave) was reported to comprise up to 19,000 individuals (McDonald et al. 1990a).
Systems
System: Terrestrial
General Use and Trade Information
This species is not traded.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
It may be declining in parts of its range due to disturbance of cave roosts (often by recreational and tourism activities), and the conversion of suitable foraging habitat to agricultural use. Implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021) poses a serious threat to established colonies of this species in old mine tunnels. The rapidly growing number of renewable energy developments in the Northern, Western, and Eastern Cape provinces could also reduce the foraging habitat of this species, but due to the clutter foraging habits of this species, it is regarded as having a “Low” risk of fatality from wind turbines (MacEwan et al. 2020).
Conservation
The species is recorded from more than 10 protected areas including: West Coast National Park; De Hoop Nature Reserve; Garden Route National Park; Langeberg Nature Reserve; Addo Elephant National Park; Great Fish Nature Reserve; Kologha Forest Reserve and Kubusi Indigenous State Forest.
The South Africa Department of Mineral Resources and Energy should engage with the South African Bat Assessment Association with regards to future closure of old mine tunnels in the country, so as to minimise the impact of this on cavity-roosting bat species such as R. acrotis. Renewable energy developments must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (for buffer recommendations see MacEwan et al. 2020 or later). Monitoring of population trends in response to the threat of mine closure or expansion, and renewable energy development, is recommended. Bats of the genus Rhinolophus are generally susceptible to indirect poisoning through the use of insecticides and thus there is a need to evaluate the impact of this threat on populations and to investigate alternative methods of insect control (D. Kock pers. comm. 2004).The species would benefit from further protected area establishment once key roost sites have been identified; and artificial wetlands in agricultural landscapes should be managed for biodiversity by conserving patches of native vegetation around the waterbodies (Sirami et al. 2013). Further studies are needed into the distribution of this bat it may occur in southern Namibia.
Recommendations for land managers and practitioners:
- The South Africa Department of Mineral Resources and Energy should engage with the South African Bat Assessment Association with regards to future closure of old mine tunnels in the country, so as to minimise the impact of this on cavity-roosting bat species such as R. acrotis.
- Renewable energy development must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (see MacEwan et al. 2020).
- The species would benefit from further protected area establishment, and by conserving patches of native vegetation around the waterbodies.
- Reduce pesticide use in agricultural landscapes.
Encouraged citizen actions:
- Minimise disturbance at caves when visiting.
- Citizens can report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Ansell, W.F.H. 1986. Some Chiroptera from south-central Africa. Mammalia 50: 507-520.
Bernard, R.T.F. 2013. Rhinolophus capensis. In: Happold, M. and Happold, D.C.D. (eds), Mammals of Africa Volume IV: Hedgehogs, Shrews and Bats, pp. 314–316. Bloomsbury Publishing, London.
Csorba, G.P., Ujhelyi, P. and Thomas, N. 2003. Horseshoe Bats of the World. Alana Books, Shropshire, England.
Griffin, M. 1999. Checklist and provisional national conservation status of Amphibians, Reptiles and Mammals known or expected to occur in Namibia. Biodiversity Inventory, Ministry of Environment and Tourism, Windhoek.
Herselman, J. C. and Norton, P. M. 1985. The distribution and status of bats (Mammalia: Chiroptera) in the Cape Province. Annals of the Cape Provincial Museums 16: 74-126.
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-2. Available at: www.iucnredlist.org. (Accessed: 14 September 2017).
Jacobs, D.S., Barclay, R.M., Walker, M.H. 2007. The allometry of echolocation call frequencies of insectivorous bats: why do some species deviate from the pattern? Oecologia 152: 583–594.
Koopman, K.F. 1993. Order Chiroptera. In: D.E. Wilson and D.M. Reeder (eds), Mammal species of the world: a taxonomic and geographic reference, pp. 137–241. Smithsonian Institution Press, Washington, D. C., USA.
Lynch, C.D. 1989. The mammals of the north-eastern Cape Province. Navorsinge van die Nasionale Museum Bloemfontein 25: 1-116.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association. South Africa.
McDonald, J.T., Rautenbach, I.L. and Nel, J.A.J. 1990. Foraging ecology of bats observed at De Hoop Provincial Nature Reserve, southern Cape Province. South African Journal of Wildlife Research 20: 133–145.
McDonald, J. T., Rautenbach, I. L. and Nel, J. A. J. 1990. Roosting requirements 582 Bats of Southern and Central Africa and behaviour of five bat species at De Hoop Guano Cave, southern Cape Province of South Africa. S. Afr. J. Wildl. Res. 20: 157–161.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Sirami, C., Jacobs, D.S. and Cumming, G.S. 2013. Artificial wetlands and surrounding habitats provide important foraging habitat for bats in agricultural landscapes in the Western Cape, South Africa. Biological Conservation 164: 30–38.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stoffberg, S. 2008. Rhinolophus capensis (Chiroptera: Rhinolophidae) 810: 1-4.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., Kearney, T.C., Clark, V.R., Howard, A., Mdluli, M.V., Markotter, W., Geldenhuys, M., Richards, L.R., Rakotoarivelo, A.R., Watson, J., Balona, J., Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate‐driven diversification and a buffer against future climate change in bats. Global Change Biology, 30(6), p.e17344.
