Cape Grysbok
Raphicerus melanotis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Raphicerus melanotis – (Thunberg, 1811)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Raphicerus – melanotis
Common Names: Cape Grysbok (English), Kaapse Grysbok (Afrikaans), Ingxungxu (Xhosa)
Synonyms: No Synonyms
Taxonomic Note:
No subspecies are recognised (Skinner and Chimimba 2005).
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: da Silva, J.M.1, de Villiers, M.S.2, Jansen van Vuuren, A.3, Landman, M.3, Swanepoel H.I.J.3 & Venter, J.A.3
Reviewer: Palmer, G.2
Institutions: 1South African National Biodiversity Institute, 2CapeNature, 3Nelson Mandela University
Previous Assessors: Palmer, G., Birss, C., Kerley, G., Feely, J., Peinke, D. & Castley, G.
Previous Reviewers: Child, M.F. & Mallon, D.P.
Contributors: Selier, J., Dorse, C., L. Jacobs, T. van der Westhuizen, K. Williams.
Assessment Rationale
The species is listed as Least Concern. Despite its restricted range, the Cape Grysbok is common, relatively adaptable, and there are no major threats that could cause range-wide declines. This species is well represented in protected areas and occurs on private farms where it can typically adapt to the predominant forms of land use provided there is sufficient suitable structured habitat remaining. However, estimates of population sizes are scarce and it is therefore difficult to extrapolate data from individual studies or locations to discern the status of the population nationally. Some studies are also relatively dated thus highlighting the need for more robust estimates of Cape Grysbok populations from sites throughout their distribution. The population trend is assumed to be generally stable in protected areas and on private land but decreasing in some other areas where human population densities are high. The effects of private conservation and wildlife ranching on this species should further be monitored and managed. There is also little information on the possible impacts of climate change, alien invasive vegetation and the expansion of certain agricultural industries in some areas (e.g., rooibos tea plantations and vineyards). However, climate change may make marginal habitats more suitable for agricultural expansion, putting pressure on remaining habitat patches where this species occurs, and thus represents an emerging threat. This, and the emerging threat of snaring, should be monitored for its impacts on Cape Grysbok.
Red List Index
Red List Index: No change
Recommended citation: da Silva JM, de Villiers MS, Jansen van Vuuren A, Landman M, Swanepoel HIJ & Venter JA. 2025. A conservation assessment of Raphicerus melanotis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
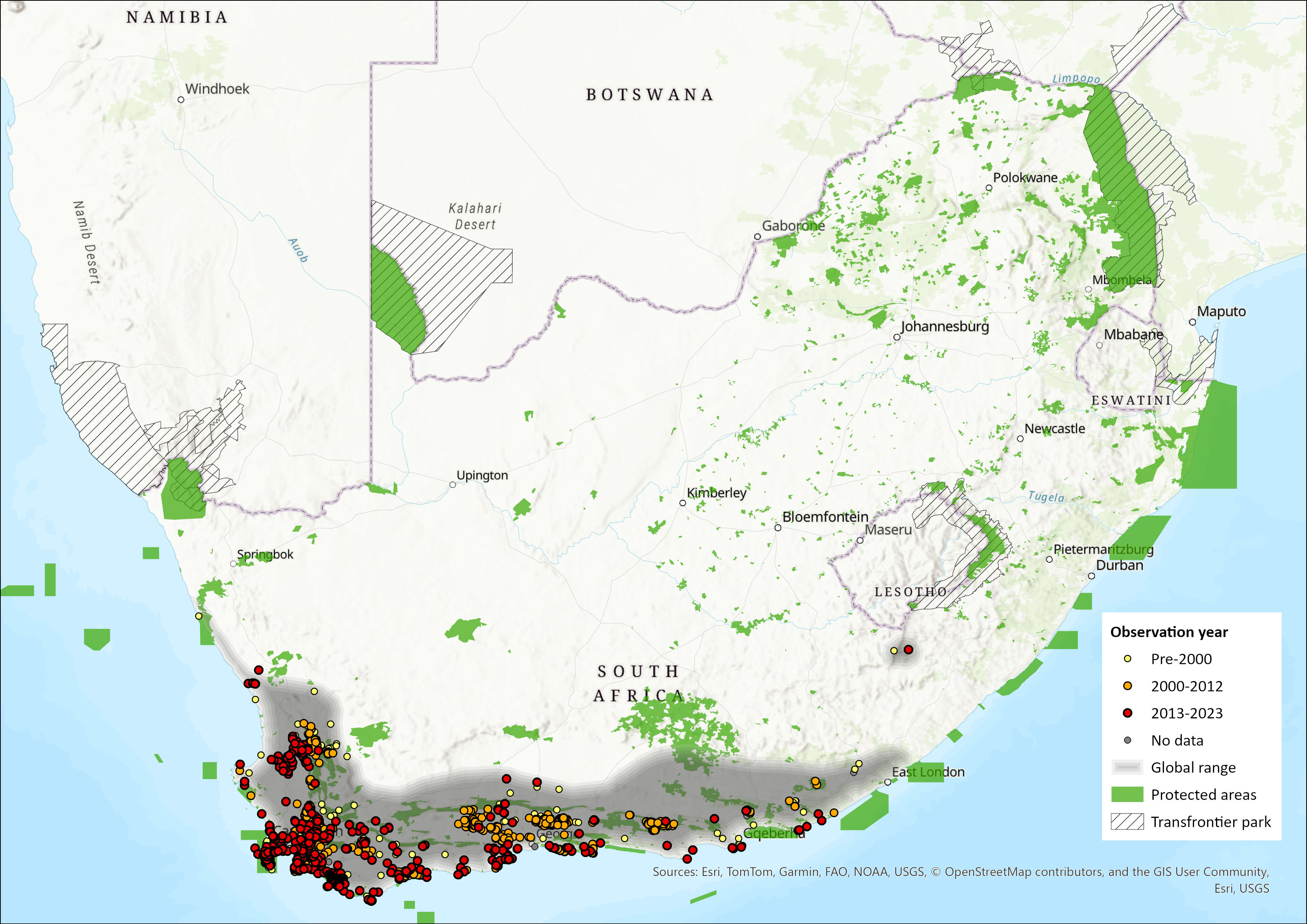
The Cape Grysbok is endemic to South Africa, and is largely confined to the Cape Floristic Region. It remains widespread and locally common within its historical range in the Western Cape and Eastern Cape provinces. It also marginally occurs in the Northern Cape. The most northern confirmed record and locality is van Rhynsdorp, Western Cape Province. In the Eastern Cape, little is known about its historical distribution (Skead 2007). Boshoff and Kerley (2013) provide two records for the Drakensberg/Lesotho but caution that they may be of material transported there through trade.
Cape Grysbok occur on at least six National Parks (Table Mountain, West Coast, Bontebok, Agulhas, Garden Route, Addo Elephant), 59 provincial natural reserves, several local authority nature reserves, and private land in the Western and Eastern Cape Provinces. There are no records for Cape Grysbok in Namaqua and Tankwa Karoo National Parks.
The range has not expanded either naturally or through the private sector. If anything it has contracted through the loss of scattered habitat fragments that have been structurally altered (become less dense), or have been totally transformed through the introduction and expansion of alien invasive vegetation (Kerley et al. 2010), increased densities of megaherbivores (Tambling et al. 2013) and the expansion of certain agricultural industries in some areas (for example, rooibos tea plantations and vineyards). This trend is likely to continue with the effects of climate change making such fragments amenable to alternative land uses. The area of occupancy (AOO) calculated for Cape Grysbok is 3,684 km². For more detailed discussion of the distribution range see East (1999) and Castley and Lloyd (2013).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Cape Grysbok (Raphicerus melanotis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The effects of climate change on Cape Grysbok in the following decades are predicted to be minimal. The change in suitable habitat for the species is predicted to range between –3% and + 4% for the time period 2011-2040, depending on the climate model and emissions scenario applied. Furthermore, the spatial overlap between current and future Cape Grysbok distribution is predicted to be high. Collectively, these results suggest a low climate change threat to the species in the short- to medium-term. However, climate change is expected increase the suitability of marginal habitats for cultivation (Hannah et al. 2013), and land transformation may occur as a result of the influence of climate change and corresponding economic changes on the livelihoods of farmers or rural communities (Curtis 2013 Pote et al. 2006). For example, farmers may transform virgin renosterveld into cropland or burn vegetation at unfavourable times in order to generate grazing for livestock, which threatens the biodiversity and functioning of these ecosystems (Curtis, 2013). Records of fire-related Cape Grysbok mortalities in the Western Cape suggest that the species might also be impacted by climate change related increases in fire frequency and intensity.
Population information
Cape Grysbok are normally solitary and cryptic in their behaviour and therefore seldom seen. They are particularly difficult to see in dense vegetation, which is exacerbated in fire-prone areas such as the fynbos of the Western Cape (Castley and Lloyd 2013). Estimates of population sizes are scarce and it is therefore difficult to extrapolate data from individual studies or locations to discern the status of the population nationally. Some studies are also dated and more recent data are needed. For example, Scott (1991) studied the distribution of small antelopes in De Hoop Nature Reserve between 1985 and 1987 and recorded densities of 0.21 animals / 100 km travelled for Cape Grysbok compared with 2.64 for Steenbok (Raphicerus campestris). Castley and Lloyd (2013) suggest that comparisons such as this may not be accurate estimates of Cape Grysbok populations given their dense habitat preferences. In the City of Cape Town area, Cape Grysbok can occur in high densities on some relatively small (less than 100 ha) isolated properties. For example, it is estimated that up to 30 animals occur on 17 hectares on the Milnerton Race Course section of the Table Bay Nature Reserve (C. Dorse pers. comm., 2024). At reserves where management limits predation from dogs and snaring, and in the absence of natural predators such as Caracal (Caracal caracal) and natural competitors such as Common Duiker (Sylvicapra grimmia), densities can become elevated and this can negatively impact highly threatened vegetation (C. Dorse pers. comm., 2024). In the 1990s and 2000s, there were signs of significant over-browsing at several sites including False Bay, Zandvlei, and the Cape Flats (University of the Western Cape) nature reserves. Caracal appear to have increased at these sites and are now far more frequently observed, and over-browsing is far less notable. At the University of the Western Cape, however, it appears that Cape Grysbok have completely disappeared (C. Dorse pers. comm., 2024). The causal factors for this recent decline need to be investigated. A recent study by Swanepoel et al. (2021) indicated that Cape Grysbok have similar levels of habitat use between De Hoop Nature Reserve and the surrounding agricultural landscape. All indications are however that Cape Grysbok occur “freely” in the landscape—on and off protected areas, on agricultural land, on game farms, and in vineyards.
Although no comprehensive subpopulation trend data are available, the metapopulation is suspected to be stable (for example, aerial counts suggest a stable subpopulation on Baviaanskloof Nature Reserve between 2008 and 2014; D. Peinke unpubl. data), although there are indications of localised declines. Anecdotal evidence suggests that numbers of Cape Grysbok and Bushpig (Potamochoerus larvatus) both declined in the Main Camp section of AENP as mega-herbivore numbers increased (Tambling et al. 2013, G. Castley unpubl. data).
Based on available habitat, and a requirement of between 6 and 456 ha per animal depending on the vegetation type, Cape Grysbok numbers could be up to 231,448 in the Cape Floristic Region, down from an estimated population of 322,977 in the pre-habitat transformation model (Kerley et al. 2003). This modelled estimate is almost an order of magnitude higher than earlier estimates (East 1999). Using the number of land parcels (2,438) on which Cape Grysbok are either present (720 land parcels) or persist (indicating that the subpopulation is persistent and breeding; 1,718 land parcels) in the Western Cape Province, at the calculated densities of 6 to 456 hectares per animal it is estimated that the 77,269 ha of protected area could sustain between 1,704 to 129,544 animals.
Population Information
Current population trend: Stable, but decreasing in some areas.
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown, but ranges from 1,000 to > 200,000.
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No. They are suspected to move between multiple land cover and land use types.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
The complete mitogenome for Cape and Sharpe’s Grysbok (R. sharpei) has been assembled and a comparison with the mitogenome of R. campestris (Steenbok) revealed that Cape and Sharpe’s Grysbok are not sister taxa (i.e., are not most closely related to each other; de Jager and Lorenzen 2023), despite having similar morphologies (Du Toit, 2005; Groves and Grubb 2011; Castley and Lloyd 2013).
DNA analysis of both museum and wild specimens has revealed two genetically distinct clades (possibly Ecologically Significant Units) within the Cape Grysbok, with individuals from the Eastern Cape forming a distinct group from the Western Cape (de Jager. Pers comm, June 2024). Additional genetic structure within the Western Cape is suggested by phylogenetic analysis, with the Cape Town area forming a distinct group (de Jager. Pers comm, June 2024); however more comprehensive sampling is needed to confirm this structure. The genetic diversity within the Eastern and Western subpopulations is high. While the contemporary effective population size (Ne) for these groups is still under investigation, they likely exceed the historical Ne (from ~11kya) of just over 10 000 individuals (de Jager, pers comm, June 2024), suggesting stable and healthy populations.
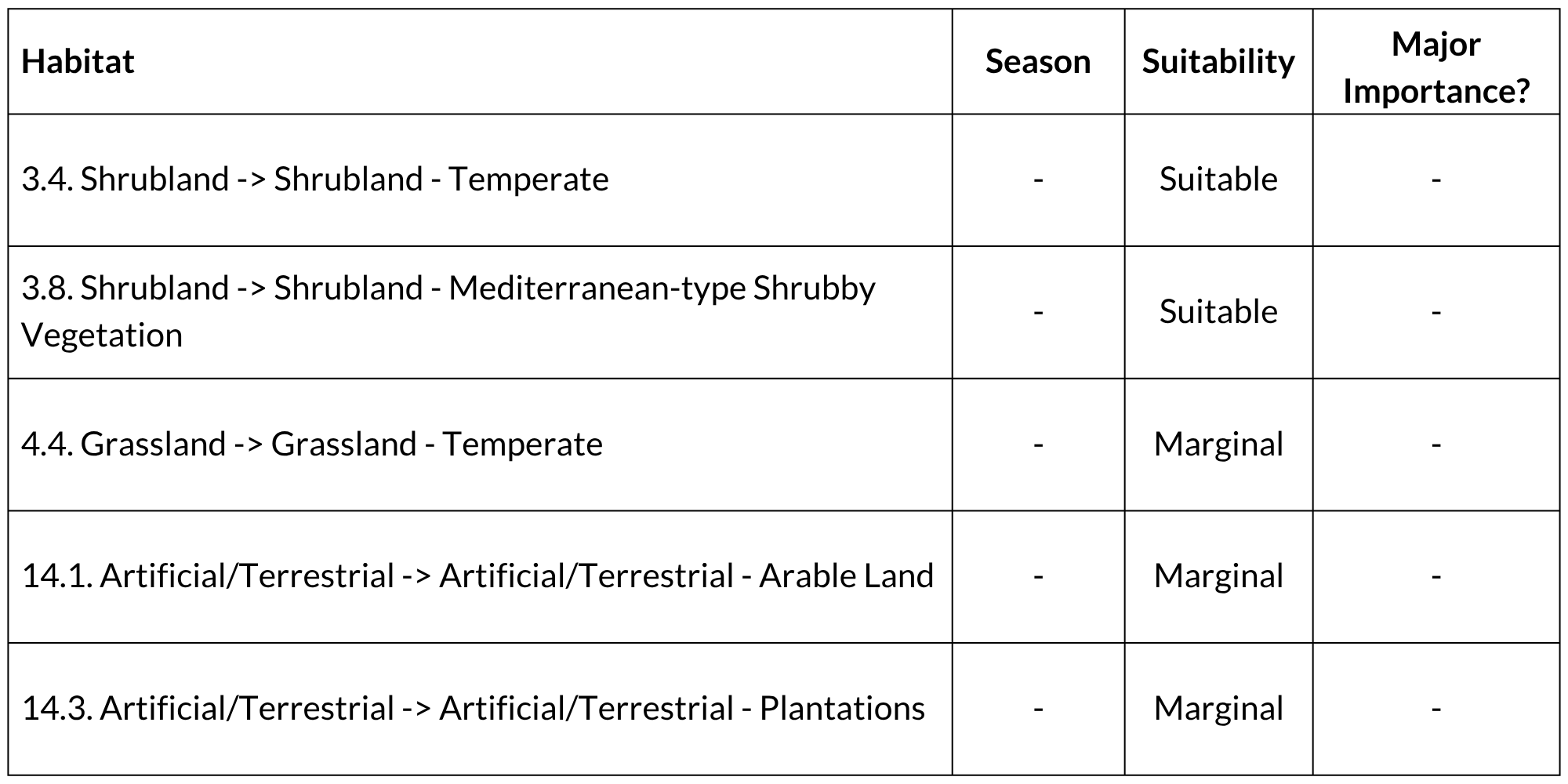
Habitats and ecology
The natural (historical) distribution of Cape Grysbok is primarily associated with the Fynbos Biome and extends into the Forest, Succulent Thicket and Succulent Karoo Biomes and marginally into the Nama-Karoo and Grassland Biomes. They are absent from the Desert and Savanna Biomes. They are locally common in thickets, shrublands and the fynbos habitats. Dense cover is an important habitat requirement. Their presence in the high-altitude grasslands of the north-eastern Cape is conditional on the proximity of forest fragments and bush clumps, although they may also use long grass for cover (Castley and Lloyd 2013). They also enter developed areas such as vineyards and agricultural areas (East 1999, Jansen van Vuuren et al. 2021, 2022, Swanepoel 2021), and have been blamed, along with the Common Duiker, for extensive damage to young shoots in tea plantations in the Cedarberg (C.T. Stuart and T. Stuart pers. comm. in Castley and Lloyd 2013). Cape Grysbok are generally regarded as selective browsers (Stynder 2009, Kigozi et al. 2008, Kerley et al. 2010, Jansen van Vuuren et al. 2022). While the inclusion of grass in the diet has also been reported (Jansen van Vuuren et al. 2022, Kerley et al. 2010, Manson 1974), its importance may fluctuate with environmental changes (Faith 2011). In the Overberg, crops such as Barley is used to supplement other natural vegetation in the diet (Jansen van Vuuren et al. 2022). This is an adaptable species and can survive in human-modified landscapes provided that vegetation with the required understorey cover remains (e.g., Jansen van Vuuren et al. 2022).
Ecosystem and cultural services: Cape Grysbok has been long suspected to be insignificant in seed dispersal in the coastal dunefields (Castley et al. 2001). However, Sigwela (2004) found that the species disperses the seeds of two plant species in thicket habitats, and a more recent dietary study in the Overberg revealed seeds of at least four different plant species in the dung of Cape Grysbok, similar to Steenbok (Jansen van Vuuren unpubl. data). They are also flagship species of the Cape Floristic Region.
IUCN Habitats Classification Scheme
Life History
Generation Length: Probably similar to Sharpe’s Grysbok, 4.8 years
Age at maturity: female or unspecified: Sexual maturity 18-24 months (earlier for females than males); social maturity later than sexual maturity
Age at Maturity: Male: Sexual maturity 18-24 months (earlier for females than males); social maturity later than sexual maturity
Size at Maturity (in cms): Female: 18-24 months
Size at Maturity (in cms): Male: 54 cm shoulder height
Longevity: 6-8 years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 7 months
Reproductive Periodicity: Up to 2 lambs per female per year
Average Annual Fecundity or Litter Size: 1 lamb per lambing event
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Males are territorial.
Congregatory: Usually solitary
Systems
System: Terrestrial
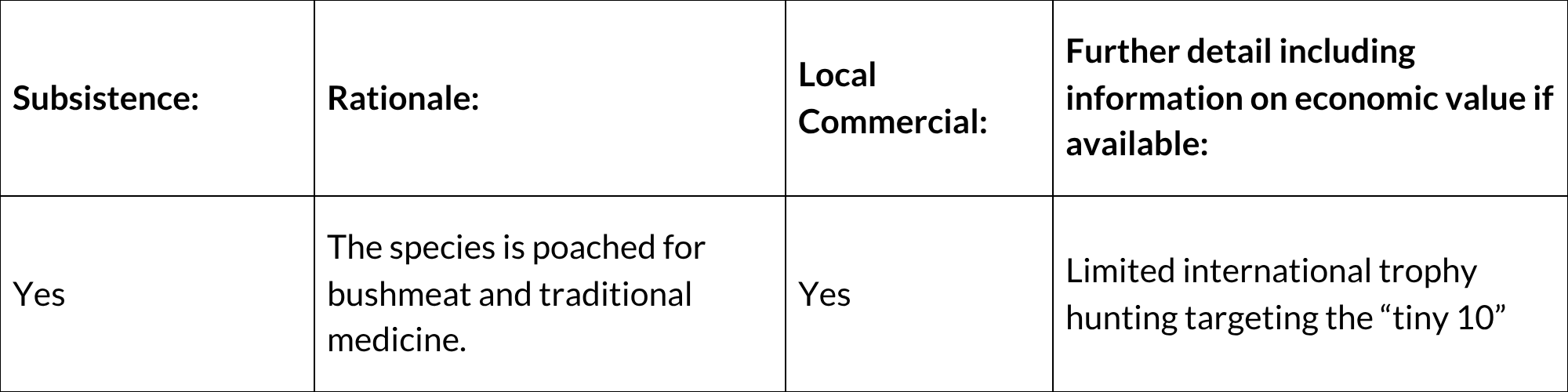
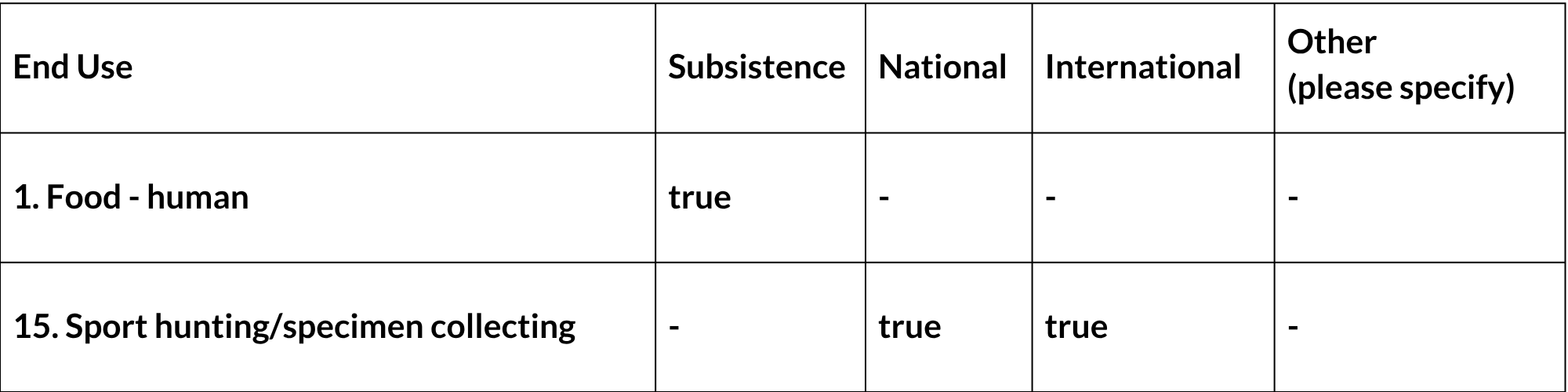
General Use and Trade Information
This species is poached as bushmeat, as it is vulnerable to being caught in snares. In the Boland region of the Western Cape, Cape Grysbok is the third–most preferred target, and the third-most frequently caught animal, in bushmeat poaching using wire snares, after Common Duiker and Cape Porcupine (Hystrix africaeaustralis) (Nieman et al. 2019). This might affect local subpopulations but is not expected to cause widespread population decline. This seems to be an emerging threat in the Western Cape at least and should be monitored. There is also limited international trophy hunting from hunters targeting the “tiny 10”. Over a six-year period (2018-2023), CapeNature received permit applications for the sport hunting of 43 Cape Grysbok, two applications to hunt animals because of crop damage and five applications to capture or cull animals for conservation purposes (CapeNature, unpubl. data). Hunting records are kept in the Eastern Cape but are not currently available. Cape Grysbok parts have also been recorded from traditional herbalist shops but at relatively low frequencies (number of items for sale) and rates of occurrence (number of outlets where items are for sale) (Simelane and Kerley 1998); there was not a high incidence of Cape Grysbok in 17 sampled townships and informal settlements in the Boland region (Nieman et al. 2019). CapeNature aims to manage off-takes through permits and requests that landowners provide evidence of the persistence of their subpopulations and registers of hunting history. Captive breeding is discouraged by CapeNature and the Eastern Cape Parks and Tourism Agency, and local translocations from within the ecotypic range are preferred. Landowners are required to supply evidence of the status of their population before permits to capture and translocate are supplied. The receiving property is also evaluated for suitability of habitat.
National Commercial Value: Yes
International Commercial Value: Yes
Is there harvest from captive/cultivated sources of this species? Unknown
Harvest Trend Comments: (Not specified)
Threats
The increase in human settlements (or urban development), agriculture, and alien invasive species have reduced available habitat (Skowno et al. 2019, CapeNature 2023). Localised declines occur due to habitat transformation and loss of dense vegetation. For example, Cape Grysbok ranges have seen local declines in numbers from areas such as the AENP where escalating numbers of elephants have opened up or destroyed thicket habitats (Castley and Lloyd 2013, Tambling et al. 2013). Additionally, some game farms are over stocked and under managed resulting in the opening up of areas of dense vegetation, thus reducing habitat for Cape Grysbok. Even where numbers are not excessive, extra-limital browsers can compete for forage and space (Spear and Chown 2009, Spear et al. 2011). Increasing development is associated with an increase in fencing and road networks. These barriers to dispersal can also be a direct cause of mortality. Of 134 mortalities recorded by CapeNature, at least 49 were the result of collisions with vehicles and these occurred across 16 magisterial districts across the width of the Western Cape Province (CapeNature, unpublished data 2024). Two of the mortalities were the result of predation by dogs, and eight Grysbok might have died during fires (CapeNature, unpublished data 2024).
Cape Grysbok are illegally hunted with dogs, which may lead to local subpopulation declines, and this is sometimes related to illegal betting and possibly organized crime (Donnie Malherbe, pers. comm.). They are also both accidentally and deliberately caught with snares for bush meat. In 2022, there were two incidents near Worcester involving the species, one of poaching and one of smuggling of a live, wild-caught animal (C. Claassen, pers. comm.). In the Boland region of the Western Cape, Cape Grysbok is the third-most preferred target, and the third-most frequently caught animal, in bushmeat poaching using wire snares, after Common Duiker and Cape Porcupine (Nieman et al. 2019). This might affect local subpopulations in certain areas. The threat of snaring in the Boland region was confirmed by Cape Leopard Trust patrols, which recorded 671 snares. Seven carcasses with meat removed were found in or near snares and included two Cape Porcupine, a Common Duiker, a Bushpig, a Cape Grysbok, a Helmeted Guineafowl (Numida meleagris), and a Common Genet (Genetta genetta) (Kendon et al. 2022). Snaring behaviour in the Boland was influenced by poverty, a perceived lack of regulation and punishment, interpersonal development, and factors such as proximity to major residential areas and roads, as well as proximity to protected areas (Nieman et al. 2019). Other areas in the Western Cape where illegal hunting may have occurred are the George District and Dana Bay (CapeNature, unpublished data). Between 2021 and 2022, Cape Leopard Trust conducted 309 interviews with farmers and conservation officials across the Western Cape. Three respondents confirmed that a Cape Grysbok had been found in a snare on their land at some time (Riversdale- Albertinia, Calitzdorp, and Oudtshoorn areas). This indicates a more widespread threat of snaring for Grysbok. (van Tonder in prep.). Localised unsustainable offtakes of trophy males for hunting may lead to population structure disruptions and localised declines.
Additionally, the habitat available to this species is declining in area and quality. Agriculture and urbanisation has reduced mainly lowland habitat and likely habitat quality [but there has not been a severe decrease in habitat quality]. For example, Pence (2014) calculated that between 2006 and 2011, 536 km² of land was converted to agriculture in the Western Cape Province (107 km² per year, which equates to 0.08% of the surface area of the province per year). Similarly, approximately 2026 km2 of fynbos was cleared between 1990 and 2014 (84.4 km2 per year, Skowno et al. 2019). Urban areas have expanded by 27% and 9% between 1990 and 2014 in the Western and Eastern Cape provinces, respectively (GeoTerraImage 2022), which we infer to mean increasing mortality from poaching, snaring and dog hunting. Even though there may not be any empirical evidence for specific impacts of extra-limital species on Cape Grysbok, several publications have highlighted the detrimental impacts of introducing extra-limital and/or exotic species that compete for resources with an endemic species (Castley et al. 2001, Spear and Chown 2009, Spear et al. 2011). Observational data from AENP further indicate that the increase in numbers of larger herbivores is associated with declines in the more cryptic species (for example, Grysbok and Bushpig). Finally, we suspect that habitat loss from agricultural expansion may become likely as climate change makes marginal habitats more suitable for cultivation. For example, climate change is projected to increase the suitability of upslope habitats for viticulture, increasing the footprint of winelands by 14% by 2050 (Hannah et al. 2013).
Conservation
Cape Grysbok are conserved in protected areas throughout their natural distribution range in the Western and Eastern Cape Provinces. Regulated harvesting through conservation legislation aims to ensure that off-takes are sustainable. In addition, it occurs widely in local authority and forestry reserves and on private land. In the Overberg region, Cape Grysbok are heavily dependent on pockets of natural vegetation but can utilise transformed habitats (Jansen van Vuuren et al, 2022; Jansen van Vuuren 2021; Swanepoel 2021). Research is being initiated by CapeNature and the Cape Leopard Trust to investigate the impacts of bush meat poaching in natural areas where Cape Grysbok occurs in close proximity to highly populated urban areas. While no direct conservation interventions are necessary at present, several interventions will benefit this and other species in the region:
- Continue with private land conservation initiatives including biodiversity servitude and stewardship agreements to protect patches of remaining habitat, thus enabling better connectivity across the landscape for this species and enabling range shifts in adaptation to climate change.
- Allow habitats to recover and interspecific competition to decrease by reducing stocking rate, especially of exotic and extralimital species. Awareness and training programmes should be provided to landowners in key habitat areas. Provincial conservation authorities should also systematically monitor trophy hunting and translocation activities to gauge the sustainability of the practice.
- Increased enforcement of laws protecting wildlife should be used to discourage illegal poaching. Linked to this is the substitution of sport hunting with dogs with alternative recreational opportunities.
Recommendations for land managers and practitioners:
- Regulate translocation to avoid genetic contamination of eco-typical variation within Cape Grysbok and hybridisation with Sharpe’s Grysbok (R. sharpei).
- Protected area managers and private landowners should ensure their properties are stocked at an ecological level and composition that will sustain the habitat structure preferred by Cape Grysbok.
- Regulate and monitor the impact of trophy hunting.
- Monitor the population status and habitat quality and identify causes of negative trends.
Research priorities:
- Impact of bush meat poaching in areas adjacent to highly-populated urban areas.
- Genetic variation (eco-typical variation) across bioregional gradients related to connectivity and gene-flow to ensure resilient sub-populations (climate change).
- A population census needs to be initiated to identify population status and trends as well as identifying causes of any negative trends observed.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Create conservancies to increase/secure habitat for the species.
- Install permeable fences to allow connectivity across landscapes.
Bibliography
Avery G. 1990. Avian fauna, palaeoenvironments and palaeoecology in the Late Quaternary of the Western and Southern Cape, South Africa. University of Cape Town.
Badgley, C., Barry, J.C., Morgan, M.E., Nelson, S.V., Behrensmeyer, A.K., Cerling, T.E. and Pilbeam, D., 2008. Ecological changes in Miocene mammalian record show impact of prolonged climatic forcing. Proceedings of the National Academy of Sciences, 105(34), pp.12145-12149.
Boone, R.B., 2019. Weather and climate impacts on browsing and grazing ungulates. The ecology of browsing and grazing II, pp.197-213.
Boshoff AF, Kerley GIH. 2013. Historical incidence of the larger mammals in the Free State Province (South Africa) and Lesotho. Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth.
Castley JG, Boshoff AF, Kerley GIH. 2001. Compromising South Africa’s natural biodiversity: inappropriate herbivore introductions. South African Journal of Science 96: 365–378.
Castley, G. and Lloyd, P. 2013. Raphicerus melanotis. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa, Bloomsbury Publishing, London, UK.
Curtis, O. E. (2013). Management of Critically Endangered renosterveld fragments in the Overberg, South Africa. Department of biological sciences, University of Cape Town. Doctor of Philosophy.
Castley, J.G., Bruton, J.S., Kerley, G.I.H. and McLachlan, A., 2001. The importance of seed dispersal in the Alexandria Coastal Dunefield, South Africa. Journal of Coastal Conservation, 7, pp.57-70.
Department of Environmental Affairs. 2015. Climate Change Adaptation Plans for South African Biomes (ed. Kharika, J.R.M., Mkhize, N.C.S., Munyai, T., Khavhagali, V.P., Davis, C., Dziba, D., Scholes, R., van Garderen, E., von Maltitz, G., Le Maitre, D., Archibald, S., Lotter, D., van Deventer, H., Midgely, G. and Hoffman, T). Pretoria.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Faith J.T. 2012. Late Quaternary dietary shifts of the Cape grysbok (Raphicerus melanotis) in southern Africa. Quaternary Research 75: 159–165.
GeoTerraImage 2022. South African National Land Cover Change Assessment (1990-2014). Report to the Department of Environmental Affairs, South Africa.
Guo, D., et al. (2017). “Impact of the Future Changing Climate on the Southern Africa Biomes, and the Importance of Geology.” Journal of Geoscience and Environment Protection 05(07): 1-9. Hannah, L., Roehrdanz, P.R., Ikegami, M., Shepard, A.V., Shaw, M.R., Tabor, G., Lu, Z., Marquet, P.A. and Hijmans, R.J. 2013. Climate change, wine, and conservation. Proceedings of the National Academy of Sciences of the United States of America 110: 6907-6912.
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-2. Available at: www.iucnredlist.org. (Accessed: 14 September 2017).
Jansen van Vuuren, A., 2021. The feeding ecology and habitat selection of small antelopes in the Overberg Renosterveld, Western Cape, MSc Dissertation, Department of Conservation Management, Faculty of Science, Nelson Mandela University, George Campus, Madiba Drive, George, South Africa.
Jansen van Vuuren A., Fritz H,, Venter J.A. 2022. Five small antelope species diets indicate different levels of anthrodependence in the Overberg Renosterveld, South Africa. African Journal of Ecology 60: 917-925.
Kerley G.I.H, Landman M., de Beer S. 2010. How do small browsers respond to resource changes? Dietary response of the Cape grysbok to clearing alien Acacias. Functional Ecology 24: 70–675.
Kerley, G. I. H., Pressey, R. L., Cowling, R. M. C., Boshoff, A. F. and Sims-Castley, R. 2003. Options for the conservation of large and medium-sized mammals in the Cape Floristic Region, South Africa. Biological Conservation 112: 169-190.
Kigozi F, Kerley G.I.H, Lessing J.S. 2008. The diet of Cape grysbok (Raphicerus melanotis) in Algoa Dune Strandveld, Port Elizabeth, South Africa. South African Journal of Wildlife Research 38: 79–81.
Manson J. 1974. Aspects of the biology and behaviour of the Cape Grysbok, Raphicerus melanotis Thunberg. University of Stellenbosch.
Novellie P.A., Randall R.M., Knight M.H. 1994. Restoring the diversity of ungulate species in the southern national parks: current state of progress and relevant background information. South African National Parks.
Pence, G.Q.K. 2014. Western Cape Biodiversity Framework 2014 status update: Critical Biodiversity Areas of the Western Cape. CapeNature, Cape Town.
Pote, J.M., Shackleton, C.M., Cocks, M.L., & Lubke, R.A. 2006. Fuelwood harvesting and selection in Valley Thicket, South Africa. Journal of Arid Environments, 67, 270-287.
Powell, M.B., Mills, A.J., & Marais, C. 2007. Carbon sequestration and restoration: challenges and opportunities in subtropical thicket.
Schmiedel, U., & Oldeland, J. 2018. Vegetation responses to seasonal weather conditions and decreasing grazing pressure in the arid Succulent Karoo of South Africa§. African Journal of Range & Forage Science, 35, 303 – 310.
Scott, H. A. 1991. Factors affecting the distribution of small antelope on the De Hoop Nature Reserve, Southern Cape. Bontebok 7: 7-15.
Simelane T.S., Kerley G.I.H. 1998. Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research 28: 121–126.
Sigwela, A.M. 2004. Animal seed interactions in the thicket biome: consequences of faunal replacements and land use for seed dynamics. PhD Dissertation, University of Port Elizabeth.
Skead, C.J. 2011. Historical incidence of the larger land mammals in the broader Western and Northern Cape provinces.In: Boshoff A.F., Kerley G.I.H, Lloyd P. (ed.), Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth.
Skead, C.J., Boshoff, A.F., Kerley, G.I.H., and Lloyd, P.H. 2007. Historical incidence of the larger land mammals in the broader Eastern Cape. Nelson Mandela Metropolitan University, Port Elizabeth.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skowno, A.L., Raimondo, D.C., Poole, C.J., Fizzotti, B. & Slingsby, J.A. (eds.). 2019. South African National Biodiversity Assessment 2018 Technical Report Volume 1: Terrestrial Realm. South African National Biodiversity Institute, Pretoria.
Spear D., Chown S.L. 2009. The extent and impacts of ungulate translocations: South Africa in a global context. Biological Conservation 142: 353–363.
Spear D., McGeoch M.A., Foxcroft L.C., Bezuidenhout H. 2011. Alien species in South Africa’s national parks. Koedoe 53(1-4).
Stynder D.D. 2009. The diets of ungulates from the hominid fossil-bearing site of Elandsfontein, Western Cape, South Africa. Quaternary Research 71: 62–70.
Swanepoel, H.I.J. 2021. The impact of transformed landscapes on the resource selection and occupancy of specialized browsing species in a lowland fynbos and renosterveld ecosystem. MSc Dissertation, Department of Conservation Management, Faculty of Science, Nelson Mandela University, George Campus, Madiba Drive, George, South Africa.
Tambling C.J., Minnie L., Adendorff J., Kerley G.I.H. 2013. Elephants facilitate impact of large predators on small ungulate prey species. Basic and Applied Ecology 14: 694–701.