Cape Ground Squirrel
Geosciurus inauris

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Geosciurus inauris – (Zimmermann, 1780)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – SCIURIDAE – Geosciurus – inauris
Common Names: Cape Ground Squirrel, Ground Squirrel, South African Ground Squirrel (English), Waaierstertgrondeekhoring (Afrikaans), Sehlora (Sepedi), Lesamane (Sesotho), Sekata-mosima, Sekate (Setswana), Sinkwe (siSwati), Unomatse (isiXhosa), Maxindlani (Xitsonga), Ingwejeje yaseKapa, Intshidane yaseKapa (isiZulu)
Synonyms: No Synonyms
Taxonomic Note: Previously Xerus inauris. Three genetic clades, corresponding to geographical restriction, have been identified (Herron et al. 2005), but the genetic differences are not sufficient to warrant the recognition of further taxa (Monadjem et al. 2015). No subspecies are recognised (Skinner & Chimimba 2005). Although doubts have been raised regarding the specific distinctness of X. inauris and X. princeps, molecular research supports each as a monophyletic lineage (Herron et al. 2005).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Vos, S.1
Reviewer: Waterman, J.1
Contributor: da Silva, J.M.2
Institutions: 1University of Manitoba,2South African National Biodiversity Institute
Previous Assessors and Reviewers: Waterman, J., Wilson, B. & Child, M.F.
Previous Contributors: Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Monadjem, A., Palmer, G. & Taylor, P.
Assessment Rationale
This species has a wide distribution across the assessment region, occurs in many protected areas – including Kgalagadi Transfrontier Park – and there are no major threats that could cause range-wide population declines. Habitat available for Ground Squirrels is stable if not expanding, as they can exist on overgrazed lands, and thus the expansion of livestock and wildlife ranching may benefit this species. Furthermore, it is a resilient species, adapting its social organisation, space use and breeding to climactic fluctuations, and thus will likely be able to adapt to climate change. No specific interventions are necessary at present.
The Damara Ground Squirrel (Xerus princeps) is Not Evaluated in this revision as, although it may marginally exist in South Africa (for example, at Augrabies National Park; J. Waterman pers. obs.), it is an extreme edge of range species and there is no evidence of a breeding population. It is therefore considered vagrant. However, it may become more prevalent within South Africa (inhabiting mountains, cliffs and gorges) as climate change increases arid conditions, and may thus require a reassessment in future.
Regional population effects: The species’ range is continuous throughout the arid areas of southern Africa and thus dispersal is possible across Namibia and Botswana. Populations in Namibia and Botswana are not expected to decline.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No Change
Red List Index
Red List Index: No change
Recommended citation: Vos S. 2025. A conservation assessment of Geosciurus inauris. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
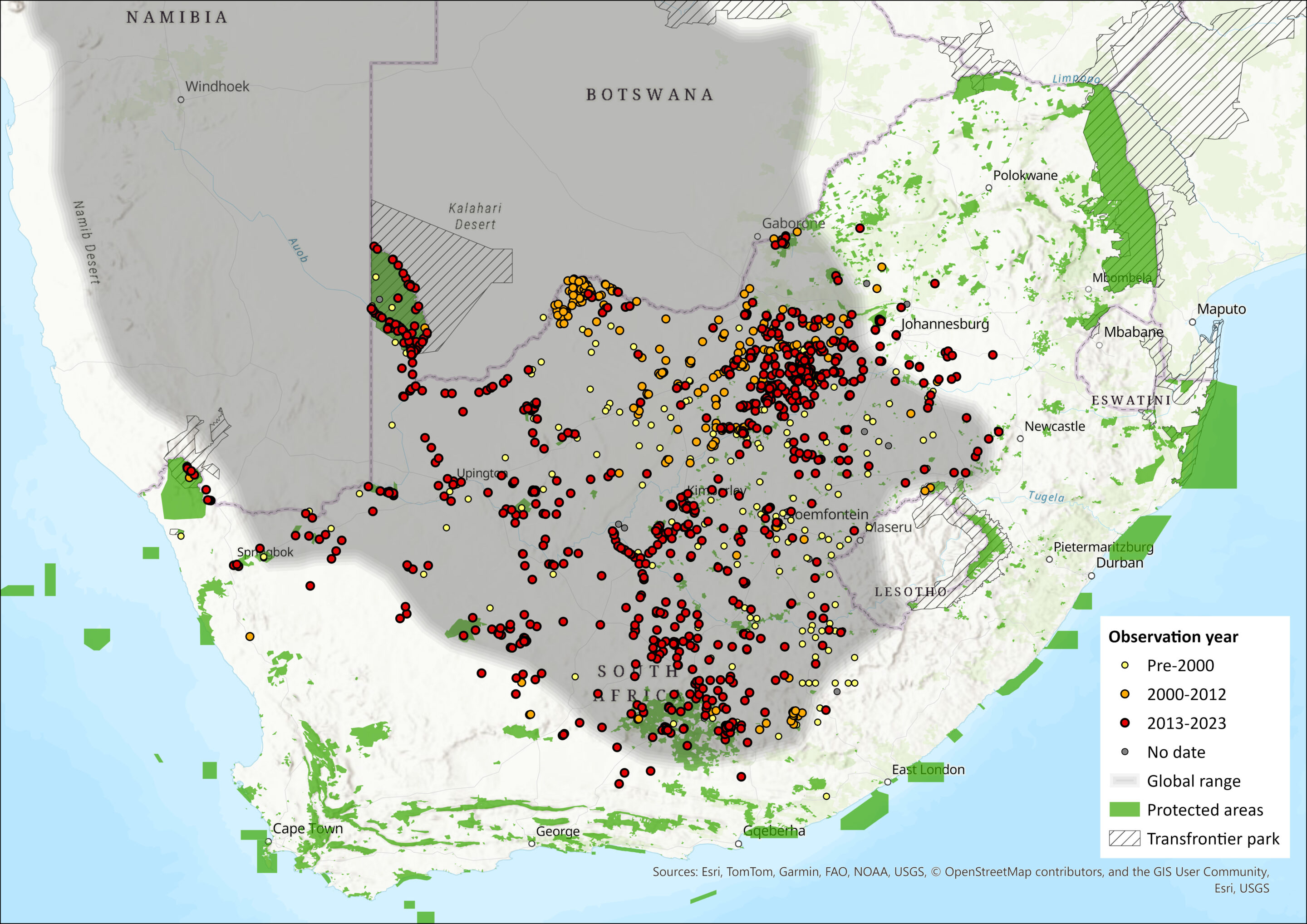
This species is restricted to southern Africa, south of the Cunene and Zambezi Rivers (Griffin & Coetzee 2008). It is distributed widely in Namibia, except where replaced by X. princeps in the north-west (Skinner & Chimimba 2005), and is absent from coastal regions. It is confined to the semi-desert Kalahari in Botswana. In South Africa, it occurs mainly in the arid interior, being widely distributed in the arid parts of North West (Power 2014), Free State, the Northern Cape (northern and north-eastern parts of the province) provinces, and southwards to the Graaff Reinet District in the Eastern Cape, which marks the most southerly limit of its distribution (Skinner & Chimimba 2005). There are isolated subpopulations in northern Limpopo Province (I. Gaigher & W. Collinson pers. obs. 2015). It also occurs in western Lesotho (Lynch 1994). The greater part of its range is within the Nama-Karoo and Succulent Karoo biomes in areas with a mean annual rainfall of 100–500 mm (Skinner & Chimimba 2005). The species’ range is continuous throughout the arid areas of southern Africa, except in areas of the western Namib (Skurski & Waterman 2005), which is reflected by mitochondrial DNA work that suggests all subpopulations are genetically connected and represent only a single species (Herron et al. 2005).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Cape Ground Squirrel (Geosciurus inauris) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The distribution of Cape Ground Squirrels spans an area that is projected to experience an average temperature rise of 2.5°C and decline in precipitation of <5 mm in the next 50 years, according to the IPCC (west Southern Africa, worst scenario SSP5-8.5, baseline 1850-1900). Cape Ground Squirrels are relatively ubiquitous throughout their distribution (Figure 1) and feed on abundant resource like annual grasses, seeds and bulbs (Herzig & Straschil 1978). The abundance of resources declines with a decrease in precipitation (Plessis 1999) but the decline in precipitation in this region is marginal and the species is very well adapted to arid and hot conditions. Cape Ground Squirrels are heterothermic, using both physiological and behavioural adaptations to regulate their body temperature. For instance, they concentrate their urine and only expel dry faeces (Haim et al. 1987; Van Heerden & Dauth 1987). They use their tails as a parasol to shield their bodies from excessive solar radiation and use their burrows for thermoregulation (Bennett et al. 1984; Fick et al. 2009; Scantlebury et al. 2012). These burrow systems maintain a relatively consistent temperature of around 14° C, shielding them from temperature extremes (Herzig & Straschil 1979). They have been found to reduce reproductive effort during years of extreme drought, but during these years, no changes in body condition or group sizes were found (Fenton & Waterman 2000). A current study does demonstrate that males delay dispersal in response to climatic conditions that are favourable for reproduction, suggesting they maximize reproductive success in good years to offset the bad years (Vos et al. unpublished). A study on the historic biogeography of the species indicates they have evolved in an arid corridor from the horn of Africa to the South African cape and that it was the colder regions in their distribution, rather than the hotter ones that caused fragmentation of the population (Herron & Waterman 2005). In addition, they have been found to respond to higher temperatures with a reduction in spine length and larger hind foot size (Warrington 2022), indicating that their generation time of approximately 1 year (Waterman 1996) is short enough to make the species adaptable to changing climatic conditions.
Population Information
This is a common species wherever it occurs. In Namibia, it exists at densities of c. 400 individuals / km2 and, in S.A. Lombard Nature reserve, North West Province, the density is higher (Manjerovic et al. 2022). The population is considered stable based on no net decline in habitat.
Local declines may be expected in areas where Black-backed Jackal (Lupullela mesomelas) numbers are high due to the removal of apex predators. For example, at three sites in the Northern Cape, there was a negative relationship between Ground Squirrel and Black-backed Jackal abundance (Bagniewska & Kamler 2014).
Population Information
Current population trend: Stable, inferred from stable habitat and high densities.
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No. Their habitat is connected, and they can utilise agricultural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
A phylogenetic study of the species indicates at least three distinct clades of Cape Ground Squirrel exist. One clade in the inlands of South Africa, one living in the northern parts of Namibia and one in between but with a distribution overlapping that of the northern clade (Herron et al. 2005). However, due to sampling limitations, the study can’t exclude the existence of additional clades. Based on this information, we can say that at least three genetically distinct clusters (possible Evolutionarily Significant Units [ESUs]) exist within the species, with one occurring in South Africa. Based on this information, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining). Unless subpopulations are known to have gone extinct, this value should not change regardless of the number of subpopulations (additional genetic clusters) identified within the assessment region.
While no census (Nc) or effective population size (Ne) data exists for the species, density estimates in some areas suggest that across the assessment region there is well over 10,000 individuals. Even though the 10 000 value is not an actual census count, the quantity can still be used as a proxy for quantifying the GBF’s headline genetic indicator – proportion of populations with an Ne > 500. Based on the assumption that the species is likely to occur as a single metapopulation, the Ne of the species is ~1000-3000 individuals, based on a Ne/Nc conversion ratio of 0.1-0.3. Having the 1 population above Ne 500, translates to an indicator score of 1.0 (1/1 population with Ne > 500).
Habitats and ecology
They have a preference for open terrain with little bush cover and a substrate suitable for burrowing, occurring on open calcareous ground on the fringes of dry pans, watercourses and floodplains, on open overgrazed ground, and in open grassland or karroid areas, providing the substrate is suitable (Skinner & Chimimba 2005). They avoid extremely loose sandy areas for making their burrows but are found burrowing near the dunes of the Kalahari Desert in stable zones. They are locally common along the edges of salt pans or at old kraal sites and overgrazed patches, and there are isolated colonies occurring in the northern bushveld regions (Power 2014).
They are diurnal, promiscuous and highly social, with males forming ephemeral all-male groups that vary in composition regulary and are essentially independent of female groups except during breeding. Males in a Kalahair population form social hierarchies but female groups do not (Wilson 1996). However, no male dominance hierarchies have been found in another population living in a high resource site, demonstrating intraspecific variation in the social structure of this species (Manjerovic et al. 2022). Individual vigilance during foraging decreases with group size (Edwards & Waterman 2011). Breeding can occur year-round, and females are capable of having up to three litters of one to two young annually (Waterman 1996), where orphans are often adopted by related females in the same group (J. Waterman unpubl. data). Males have a high reproductive skew, with 65% of the males failing to produce offspring (Manjerovic et al. 2022). Female reproductive suppression exists in groups with more than one adult female; however, females can split off to start a new matriline resulting in group fission (Waterman 2002). Although it has been postulated that their parasol-like tails are used to decrease body temperature whilst foraging during the day, new data suggest that the tail simply provides comfort during the heat of the day and that they retreat into burrows to dissipate a heat load and remain active diurnally (Fick et al. 2009). To aid thermoregulation, they adapt burrow emergence and immergence times according to the time of year, further demonstrating that they use both physiological and behavioral adaptations to maintain their body temperature (Scantlebury 2012). Unlike the Damara Ground Squirrel (X. princeps), this species has no arboreal tendencies and is purely ground-living (Skinner & Chimimba 2005). They are predominantly herbivorous (leaves, grass stems, seeds, bulbs, roots and plant stems), being able to digest cellulose, but do eat insects (Skinner & Chimimba 2005).
The ‘landscape of fear’ for this species has been quantified in Augrabies Falls National Park, Northern Cape Province (van der Merwe & Brown 2008): among three colonies, only 3–22% of the space resulted in low foraging costs while 31–92% of the sampled areas represented very high foraging costs. Overall, they are a resilient and dynamic species, being able to adapt their home range size and reproductive output in response to drought conditions (Waterman & Fenton 2000) and thus will likely be able to adapt to climate change in the future.
Ecosystem and cultural services: Many other species (for example, Suricates Suricata suricata and Yellow Mongooses Cynictis penicillata) use the burrows of Cape Ground Squirrels for refuge, suggesting they are ecological engineers (Waterman & Roth 2007). Furthermore, in central South Africa and the Namib desert grasslands, controlled, replicated studies revealed increased diversity and abundance of small mammals and invertebrates in burrow areas and a higher abundance of plants (Ewacha et al. 2016). Thus, the burrowing and foraging behaviour of this species creates habitat for plant and animal communities. However, Power (2014) found no evidence of enhanced small mammal diversity in relation to Ground Squirrel burrow clusters in three landscapes.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Marginal | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Suitable | – |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | – | Unknown | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: 10 months (Waterman 1996)
Age at Maturity: Male: 8 months (Waterman 1996)
Size at Maturity (in grams): Female: 568 (Herzig-Straschil 1978)
Size at Maturity (in grams): Male: 596 (Herzig-Straschil 1978)
Longevity: 11 years (Warrington et al. 2022)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 48 days (Waterman 1996)
Reproductive Periodicity: Year-round breeding (Herzig-Straschil 1978; Waterman 1996)
Average Annual Fecundity or Litter Size: Litter sizes of 1-3, average 1.9 (Herzig-Straschil 1978)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Females remain in their natal environment and males disperse over larger distances (Waterman 1995). Female home ranges average 4 hectares and, after dispersal, male home range average 12.5 hectares (Waterman 1995). Males move throughout their home range sleeping in empty burrow clusters, therefore, after dispersal males spent considerably more time in locomotion compared to females and males that delay dispersal to remain in their natal environment (Waterman 1995).
Congregatory: Yes. females live in matrilineal family group (Waterman 1995). Males disperse and live in ephemeral fission-fusion all-male bands but may delay dispersal to remain in their natal family group (Waterman 1995).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence for significant utilisation of this species.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major identified threats that could cause population declines. It is considered to be an agricultural pest in some parts of its range (Griffin & Coetzee 2008) but has not been reported as such within the assessment region (Power 2014). There are localised complaints about damage to gravel and farm roads as a result of burrow formations (B. Wilson unpubl. data). They tend to prefer Schmidtia kalaharensis (J. Waterman unpubl. data), a grass that is characteristic of degraded lands, such as where there is overgrazing. Thus, livestock and wildlife ranching, contrary to what was reported in the previous assessment (Friedmann & Daly 2004), are not threats but are probably beneficial to this species.
Habitat trend: Stable. Occurs on modified and agricultural lands (including road verges) and is commensal with humans. It does not occur in long grass, so overgrazed areas are suitable. It can thus occur on overstocked ranchlands, and will possibly benefit with the expansion of wildlife ranching across South Africa. Furthermore, as this species is adapted to arid conditions, drier conditions are not expected to constrain its distribution (Herron et al. 2005).
Conservation
The species is common in many protected areas across the assessment region, including Kgalagadi Transfrontier Park. Additionally, the expansion of wildlife ranches across the arid parts of its range may be expanding habitat for this species as it occurs in areas that have been overgrazed or where there are high densities of grazers (J. Waterman unpubl. data). No specific conservation interventions are necessary at present.
Recommendations for land managers and practitioners: None
Research priorities: An ongoing research project (since 2002) is being conducted in S. A. Lombard Nature Reserve, North West Province, by Prof. Jane Waterman. Research areas include parasitology (Hillegass et al. 2008, 2010), and its energetic costs (Scantlebury et al. 2007); physiology (Scantlebury et al. 2012), including the effects of hormone physiology and resource availability on reproduction (Jackson et al. 2007; Pettitt et al. 2008; Pettitt & Waterman 2011); gender-bias in immunology (Manjerovic & Waterman 2012); effectiveness of chemical immobilization and anaesthesia (Joubert et al. 2011); and behavioural ecology, including interactions with predators and group dynamics (Belton et al. 2007; Unck et al. 2009).
Research priorities regarding the conservation assessment are as follows:
- The abundance and distribution of the species given various grazing intensities of both livestock and wildlife ranching.
- The efficacy of this species as an ecological engineer in enhancing small mammal abundance, invertebrate abundance and plant diversity.
Encouraged citizen actions:
- Report sightings of this species, especially outside protected areas, on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Bagniewska JM, Kamler JF. 2014. Do black-backed jackals affect numbers of smaller carnivores and prey? African Journal of Ecologyvvvv 52: 546–567.
Belton LE, Ball N, Waterman JM, Bateman PW. 2007. Do Cape ground squirrels (Xerus inauris) discriminate between olfactory cues in the faeces of predators versus non-predators? African Zoology 42: 135–138.
Bennett AF, Huey RB, John-Alder H, Nagy KA. 1984. The parasol tail and thermoregulatory behavior of the Cape ground squirrel Xerus inauris. Physiological Zoology 57: 57–62.
Edwards S, Waterman JM. 2011. Vigilance and grouping in the southern African ground squirrel (Xerus inauris). African Journal of Ecology 49: 286–291.
Ewacha MV, Kaapehi C, Waterman JM, Roth JD. 2016. Cape ground squirrels as ecosystem engineers: modifying habitat for plants, small mammals and beetles in Namib Desert grasslands. African Journal of Ecology 54: 68–75.
Waterman, F. & Fenton. 2000. The effect of drought on the social structure and use of space in Cape ground squirrels, Xerus inauris. Ecoscience 7 (2): 131-136
Fick LG, Kucio TA, Fuller A, Matthee A, Mitchell D. 2009. The relative roles of the parasol-like tail and burrow shuttling in thermoregulation of free-ranging Cape ground squirrels, Xerus inauris. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 152: 334–340.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Griffin M, Coetzee N. 2008. Xerus inauris.
Haim, A., Skinner, J. D., & Robinson, T. J. 1987. Bioenergetics, thermoregulation and urine analysis of squirrels of the genus Xerus from an arid environment. South African Journal of Zoology, 22(1), 45–49.
Van Heerden, J. V., & Dauth, J. (1987). Aspects of adaptation to an arid environment in free-living ground squirrels Xerus inauris. Journal of Arid Environments, 13(1), 83–89.
Herron MD, Waterman JM, Parkinson CL. 2005. Phylogeny and historical biogeography of African ground squirrels: the role of climate change in the evolution of Xerus. Molecular Ecology 14: 2773–2788.
Herzig-Straschil. 1978. On the biology of Xerus inauris (Zimmerman, 1780). Saugertierkunde, 43, 262–278.
Herzig-Straschil. 1979. Xerus inauris (Rodentia, Sciuridea)—An inhabitant of the arid regions of southern Africa. Folia Zoologica 28: 119-124.
Hillegass MA, Waterman JM, Roth JD. 2008. The influence of sex and sociality on parasite loads in an African ground squirrel. Behavioral Ecology 19: 1006–1011.
Hillegass MA, Waterman JM, Roth JD. 2010. Parasite removal increases reproductive success in a social African ground squirrel. Behavioral Ecology 21: 696–700.
Jackson TP, Waterman JM, Bennett NC. 2007. Pituitary luteinizing hormone responses to single doses of exogenous GnRH in female social Cape ground squirrels exhibiting low reproductive skew. Journal of Zoology 273: 8–13.
Joubert KE, Serfontein T, Scantlebury M, Manjerovic M-B, Bateman PW, Bennett NC, Waterman JM. 2011. Determination of an optimal dose of medetomidine-ketamine-buprenorphine for anaesthesia in the Cape ground squirrel (Xerus inauris). Journal of the South African Veterinary Association 82: 94–96.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Manjerovic, M. B., Hoffman, E. A., Parkinson, C. L., & Waterman, J. M. 2022. Intraspecific variation in male mating strategies in an African ground squirrel (Xerus inauris). Ecology and Evolution, 12(8).
Manjerovic MB, Waterman JM. 2012. Immunological sex differences in socially promiscuous African ground squirrels. PloS One 7.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
Pettitt BA, Waterman JM, Wheaton CJ. 2008. Assessing the effects of resource availability and parity on reproduction in female Cape ground squirrels: resources do not matter. Journal of Zoology 276: 291–298.
Pettitt BA, Waterman JM. 2011. Reproductive delay in the female Cape ground squirrel (Xerus inauris). Journal of Mammalogy 92: 378–386.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic
Du Plessis, W. P. 1999. Linear regression relationships between NDVI, vegetation and rainfall in Etosha National Park, Namibia. In Journal of Arid Environments (Vol. 42, pp. 235–260).
Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Scantlebury M, Danek-Gontard M, Bateman PW, Bennett NC, Manjerovic M-B, Joubert KE, Waterman JM. 2012. Seasonal patterns of body temperature daily rhythms in group-living Cape ground squirrels Xerus inauris. PloS One 7.
Scantlebury M, Waterman JM, Hillegass M, Speakman JR, Bennett NC. 2007. Energetic costs of parasitism in the Cape ground squirrel Xerus inauris. Proceedings of the Royal Society of London B: Biological Sciences 274: 2169–2177.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skurski DA, Waterman JM. 2005. Xerus inauris. Mammalian Species 781: 1–4.
Unck CE, Waterman JM, Verburgt L, Bateman PW. 2009. Quantity versus quality: how does level of predation threat affect Cape ground squirrel vigilance? Animal Behaviour 78: 625–632.
Warrington, M. H., & Waterman, J. 2023. Temperature-associated morphological changes in an African arid-zone ground squirrel. Journal of Mammalogy, 104(2), 410–420.
Warrington, M. H., Beaulieu, S., Vos, S., Jellicoe, R., Bennett, N. C., & Waterman, J. M. 2022. Personalities are not associated with different reproductive tactics in male Cape ground squirrels, Xerus inauris. Animal Behaviour, 193, 63–74.
Warrington, M. H., Beaulieu, S., Jellicoe, R., Vos, S., Bennett, N. C., & Waterman, J. M. 2024. Lovers, not fighters: Docility influences reproductive fitness, but not survival, in male Cape ground squirrels, Xerus inauris. Behavioral Ecology and Sociobiology, 78(1), 6.
Waterman JM, Fenton MB. 2000. The effect of drought on the social structure and use of space in Cape ground squirrels, Xerus inauris. Écoscience 7: 131–136.
Waterman, J. M. 2002. Delayed maturity, group fission and the limits of group size in female Cape ground squirrels (Sciuridae: Xerus inauris). Journal of Zoology, 256(1), 113–120.
Waterman, J. M. (1995). The Social Organization of the Cape Ground Squirrel (Xerus inauris; Rodentia: Sciuridae). Ethology, 101(2), 130–147.
Waterman. (1996). Reproductive biology of a tropical, non-hibernating ground squirrel. Journal of Mammalogy 77(1): 134-146.
Waterman JM, Roth JD. 2007. Interspecific associations of Cape ground squirrels with two mongoose species: benefit or cost? Behavioral Ecology and Sociobiology 61: 1675–1683.
Wilson B. 1996. The social life of ground squirrels (Xerus inauris). McGregor Miscellany 6: 6.
van der Merwe M, Brown JS. 2008. Mapping the Landscape of Fear of the Cape Ground Squirrel (Xerus inauris). Journal of Mammalogy 89: 1162–1169.