Cape Grey Mongoose
Herpestes pulverulentus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Herpestes pulverulentus – (Wagner, 1839)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – HERPESTIDAE – Herpestes – pulverulentus
Common Names: Cape Grey Mongoose, Small Grey Mongoose (English), Klein Grysmuishond, Kaapse Grysmuishond (Afrikaans)
Synonyms: Galerella pulverulenta (Wagner, 1839)
Taxonomic Note:
Sometimes included in the genus Galerella (Meester et al. 1986; Wozencraft 2005; Veron et al. 2022). Cavallini (2013) recognised three subspecies: Herpestes pulverulentus pulverulentus (present in most of the range), H. p. basuticus (present at higher altitudes, from the east of Lesotho to the west of KwaZulu-Natal), and H. p. ruddi (in the extreme northwestern section of the distribution range). Lynch (1981) noted that character gradients are present in individuals from contiguous areas.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Filonzi, J.1, Adams, E.C.2, da Silva, J.M.3 & Do Linh San, E.4
Reviewer: Power, R.J.5
Institutions: 1University of Fort Hare, 2Endangered Wildlife Trust, 3South African National Biodiversity Institute, 4Sol Plaatje University, 5North West Province’s Directorate of Biodiversity Management
Previous Assessors & Reviewers: Do Linh San, E., Mbatyoti, K.V., Palmer, G., Stuart, C., Stuart, M., Avenant, N. & Cavallini, P.
Previous Contributor: Relton, C.
Assessment Rationale
The Cape Grey Mongoose is listed as Least Concern because it is common and adaptable, with a catholic diet, there are no major threats, and it is present in several protected areas in its range. The distribution range appears not to have changed significantly since the last assessment.
Regional population effects: The range is continuous and dispersal between regions occurs as this species is not constrained by fences.
Red List Index
Red List Index: No change
Recommended citation: Filonzi J, Adams EC, da Silva JM & Do Linh San E. 2025. A conservation assessment of Herpestes pulverulentus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
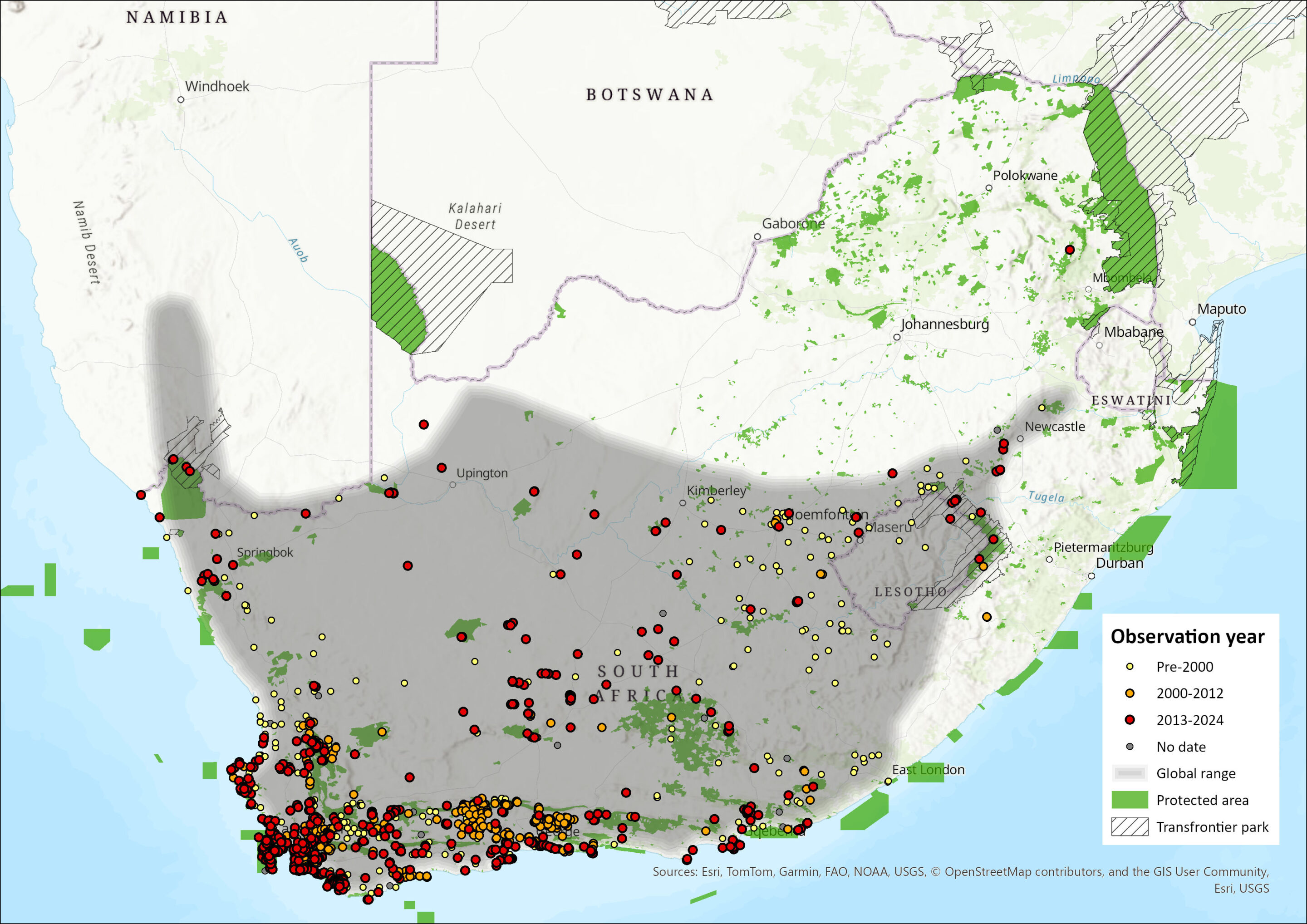
This species is endemic to southern Africa, ranging throughout the Western Cape, Northern Cape, Free State (Lynch 1983) and Eastern Cape provinces of South Africa, with a marginal intrusion into southern Namibia (Pallett & Thomson 2022), east to Lesotho and extreme western KwaZulu-Natal (Cavallini 2013). Persistence at the northern edge of distribution range the Northern Cape province has been confirmed in the Korannaberg mountain range stretching inside Tswalu Kalahari Reserve (Filonzi pers. obs. 2025). In 1990, an animal was trapped in Wakkerstroom, Mpumalanga, 200 km north of known records in KwaZulu-Natal and Lesotho (Bronner 1990). It is present from sea level around the Western Cape to 1,900 m asl in KwaZulu-Natal (Cavallini 2013). Although there is a slight overlap in the distribution range of Cape Grey Mongoose and that of the closely related Slender Mongoose (Herpestes sanguineus), it is speculated that the otherwise disjoint distribution ranges are a result of competitive exclusion between these species (Skinner & Chimimba 2005).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m
Elevation Upper Limit (in metres above sea level): 1,900 m
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Cape Grey Mongoose (Herpestes pulverulentus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Lesotho | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No recent research or literature has focused on how climate change might affect this species in the future. However, Fynbos regions, in which this species is dominant, have been shown to experience an increase in mean monthly temperatures, as well as an increase in cumulative monthly rainfall over a 48-year period (Slingsby et al. 2017). This increase in temperature and rainfall affected the plant species diversity (Slingsby et al. 2017) and would in turn affect the small mammal communities on which the Cape Grey Mongoose depends. Otomys irroratus, a fynbos species and one of the main prey items of the Cape Grey Mongoose, has been modelled to experience a decline in their range as a result of climate change by 2050 (Taylor et al. 2016). Nama Karoo regions are also expected to experience changes, with an increase in rainfall observed over the past few decades (Harmse et al. 2020). This could facilitate the encroachment of the Savannah biome into the Nama Karoo (DEA 2016). With Cape Grey Mongoose observed in the Maloti-Drakensberg montane area recently, the area could be a refuge from the effects of climate change, as it has been observed to be for other mammals (Taylor et al. 2024).
Population information
The Cape Grey Mongoose is common within its wide range in the assessment region. In 1990, densities of up to 10 individuals / km² have been recorded in suitable habitat (Cavallini & Nel 1990a). As such, given this species’ large distribution range and assuming a minimum average density of at least 0.1 individual / km², it was estimated that there were well over 10,000 mature individuals in the assessment region. In GFRNR, a density of 0.3 individual / km² was recorded considering only collared animals, thus total density is supposedly much higher (Filonzi, 2022). Therefore, considering the minor changes of the distribution range and the adaptability of the species, the estimate of well over 10,000 mature individuals in the assessment region is supposedly still valid.
Population Information
Current population trend: Unknown, but probably stable based on lack of threats and wide habitat tolerance and extent of occurrence.
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: Unlikely due to the catholic diet and plentiful alternative food sources.
Continuing decline in number of subpopulations: Unlikely based on recent records and distribution range.
All individuals in one subpopulation: Unknown
Number of mature individuals in population: Probably > 10,000.
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: No. Cape Grey Mongooses have a broad habitat tolerance and can exist in agricultural and rural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
To date no population genetic study has been undertaken on this species; however, it would be greatly informative to understand the evolutionary links, and hence validity, associated with the three proposed subspecies. It is possible that where the species is contiguous in range, genetic isolation by distance might be observed. Given that it is considered fairly common throughout its range in the assessment region, the species may be considered a single population of adequate effective population size to maintain adaptive potential (i.e., Ne > 500 based on an estimate size of 10,000 mature individuals; assuming a conversion ratio between 0.1 and 0.3).
Habitats and ecology
Cape Grey Mongooses have a wide habitat tolerance, although they are essentially found in Karoo and karroid bushveld and sclerophyllous scrub (Cavallini 2013), as well as Fynbos, thicket and even afromontane scrub (Stuart & Stuart 2015). They are often associated with refuge areas, such as dense bushes and rocky outcrops, and avoid open fields with short vegetation, where they are vulnerable to predation by mesocarnivores (Caracal Caracal caracal, Black-backed Jackal Lupulella mesomelas, etc.; E. Do Linh San & M.J. Somers unpubl. data) and large birds of prey (Cavallini & Nel 1990a). They appear to avoid moist and wet high forests (Crawford et al. 1983). They are essentially terrestrial, but readily climb bushes and tree trunks, which they occasionally use to defecate (latrines) or rest during the day. In the Great Fish River Nature Reserve (GFRNR), Eastern Cape, some radio-tracked individuals used riverine forests as nocturnal shelter – sleeping in hollow Cape Bushwillow (Combretum caffrum) trees – and as foraging grounds during the day (E. Do Linh San & M.J. Somers unpubl. data). Cape Grey Mongooses otherwise use dense bushes or rock crevices to sleep during the night. They are not fossorial but may occasionally use burrows dug by other species. They are sometimes seen close to human settlements, foraging in gardens and on food scraps, and sleeping in attics (E. Do Linh San pers. obs. 2006; Cavallini 2013; Bernard et al. 2024).
The Cape Grey Mongoose is essentially a diurnal species, with timing of onset and end of activity generally correlated with sunrise and sunset (de Satgé et al. 2017). Longer activity periods and higher activity levels are recorded in summer, while activity is more restricted in winter (E. Do Linh San & M.J. Somers unpubl. data). Cape Grey Mongooses are also less active under rainy and cold weather conditions, which likely reduce prey activity and therefore availability. They have a catholic diet, though their primary food is generally small mammals –Rhabdomys spp., Otomys spp. and Grammomys spp. have been recorded as the most common prey species – and insects (du Toit 1980; Stuart 1981, 1991; MacDonald & Nel 1986; Cavallini & Nel 1990b; Avenant & Nel 1997; Stuart & Stuart 1998; Mbatyoti 2010), and they readily kill snakes (Branch & Hanekom 1987). They have also been reported feeding on larger mammals such as Cape Porcupine (Hystrix africaeaustralis) and hares (Lepus spp.) (Cavallini 1992; Stuart & Stuart 1998), although this probably mostly takes place through scavenging, as directly observed with hares (E. Do Linh San pers. obs. 2007). This species does readily scavenge.
The Cape Grey Mongoose is regarded as a solitary species, but groups of two, three and up to five individuals have been observed (Stuart 1981, 1991). Groups often comprise one adult female and her offspring, sometimes with the presence of a second adult.
Male home range size in the West Coast National Park varied from 52 to 92 ha (Cavallini & Nel 1990a). Female home range size is likely smaller, as suggested by the case of one radio-tracked female who ranged over 31 ha. Juveniles may, however, move over much larger areas (Cavallini & Nel 1990a). In the GFRNR, the average home range size of males (83 ha) was significantly larger than that of females (40 ha) (Filonzi 2022). This difference in size is at least partly linked to sexual dimorphism, as trapped males were on average 20% heavier than females (Filonzi 2022). No seasonal difference in home range size was recorded (Filonzi 2022). Home range overlap has been observed in both males and females (Cavallini & Nel 1990a; Stuart 1991: Filonzi 2022). As reported in the Slender Mongoose (Waser et al. 1994), males exhibit some degree of tolerance or sociality, with two or three males of different sizes (and likely ages) ranging over a common area, although rarely sharing the same nocturnal resting site (Cavallini & Nel 1990a; E. Do Linh San & M.J. Somers unpubl. data).
The breeding season probably runs from June to December, as lactating females have been observed in August and from November to February (Stuart 1981; Lynch 1983). Litter size is 1–3 pups; it is unknown whether females can have two litters per breeding season. Pups are often reared in rock crevices, hollow trees, wood piles or farm outbuildings (Stuart 1981). Life expectancy in captivity is more than 11 years (Weigl 2005).
Ecosystem and cultural services: Like other small carnivores, the Cape Grey Mongoose may predate on pest species, such as rodents and insects (e.g. Cavallini & Nel 1990b; Mbatyoti 2010). This small carnivore is also an important scavenger within the Cape Flats Dune Strandveld vegetation subtype (Spies et al. 2018; Finaughty et al. 2024). The species scavenging behaviour has been important in elucidating a forensic application when examining decomposition of bodies (Spies et al. 2018; Finaughty et al. 2024).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: More than 11 years in captivity
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: June to December
Average Annual Fecundity or Litter Size: 1–3 pups per litter
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is mostly not known to be harvested or traded in any form. It may rarely be found in herbalist shops (Simelane & Kerley 1998), however there are no recent records of it.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
No |
– |
– |
– |
National Commercial Value: No
International Commercial Value: No
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species. Like most small carnivores the Cape Grey Mongoose is occasionally victim of road traffic collisions. On farmland it might also be poisoned accidentally when it feeds on carcasses set out for damage-causing predators such as Black-backed Jackals or Caracals. Conflicts with farmers over poultry are rarely cited and the species is usually tolerated (Nieman et al. 2020).
Conservation
This species occurs in numerous protected areas in its range, including the West Coast, Namaqua, Mokala, Mountain Zebra, and Greater Addo Elephant national parks, as well as the Great Fish River Nature Reserve. No specific conservation interventions are necessary at this stage.
Recommendations for land managers and practitioners:
- No specific management interventions have been identified.
Research priorities:
- Monitoring subpopulations to detect trends across various land-uses.
- General studies on the biology (notably reproductive aspects) and ecology of this species in different habitat types, i.e Grasslands, and afromontane habitats in the north and north-east of its range
- Re-assessing genetic structure in the species population
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
Bibliography
Avenant NL, Nel JAJ. 1997. Prey use by four syntopic carnivores in a strandveld ecosystem. South African Journal of Wildlife Research 27: 86–93.
Bernard, A., Fritz, H., Dufour, A. B., Venter, J. A., and Guerbois, C. 2024. A local ecological knowledge-based assessment of anthropodependence for large mammals in anthropogenic landscapes. Biological Conservation, 290, 110450.
Branch WR, Hanekom N. 1987. The herpetofauna of the Tsitsikamma Coastal and Forest National Parks. Koedoe 30: 49–60.
Bronner, G.N. 1990. New distribution records for four mammal species, with notes on their taxonomy and ecology. Koedoe 33: 1 –7.
Cavallini P. 1992. Herpestes pulverulentus. Mammalian Species 409: 1–4.
Cavallini, P. 2013. Herpestes pulverulentus Cape Grey Mongoose (Small Grey Mongoose). In: J.S. Kingdon and M. Hoffmann (eds), The Mammals of Africa. 5. Carnivores, Pangolins, Rhinos and Equids, pp. 311–314. Bloomsbury, London, UK.
Cavallini, P. and Nel, J.A.J. 1990a. Ranging behaviour of the Cape grey mongoose Galerella pulverulenta in a coastal area. Journal of Zoology (London) 222: 353-362.
Cavallini, P. and Nel, J.A.J. 1990b. The feeding ecology of the Cape grey mongoose, Galerella pulverulenta (Wagner, 1839) in a coastal area. African Journal of Ecology 28: 123-130.
Crawford PB, Crawford SAH, Crawford RJM. 1983. Some observations on Cape grey mongooses Herpestes pulverulentus in the Tsitsikamma National Parks. South African Journal of Wildlife Research 13: 35–40.
DEA (Department of Environmental Affairs). 2016. Long Term Adaptation Scenarios for South Africa (ed. Munzhedzi, S. M., Khavhagali, V. P., Midgley, G. M., de Abreu, P., Scorgie, S., Braun, M., Abdul, Z., Pegram, G., Baleta, H. and Seddon-Daines, D.). Long-Term Adaptation Scenearios Flagship Research Programme. Pretoria.
de Satgé, J., Teichman, K. and Cristescu, B. 2017. Competition and coexistence in a small carnivore guild. Oecologia, 184, 873-884.
du Toit, C.F. 1980. The yellow mongoose Cynictis penicillata and other small carnivores in the Mountain Zebra National Park. Koedoe 23: 179–184.
Filonzi, J. 2022. The spatial ecology of the small grey mongoose (Galerella pulverulenta) in the Great Fish River Reserve, South Africa. MSc dissertation, University of Bologna, Bologna, Italy.
Finaughty, D.A., French, G.M., Adams, K.S., Spies, M.J. and Gibbon, V.E. 2024. Quantifying forensically relevant vertebrate scavenging: a pilot study to develop a methodological framework using cape grey mongoose (Galerella pulverulenta) as an illustrative model. Forensic Sciences Research, owae069.
Harmse, C. J., Du Toit, J. C., Swanepoel, A., and Gerber, H. J. (2020). Trend analysis of long-term rainfall data in the Upper Karoo of South Africa. Transactions of the Royal Society of South Africa, 76(1), 1-12.
Langley CH. 1986. Cape grey mongoose attacks grysbok lambs. African Wildlife 40: 203.
Lynch CD. 1981. The status of the Cape grey mongoose, Herpestes pulverulentus Wagner, 1839 (Mammalia: Viverridae). Navorsinge van die Nasionale Museum, Bloemfontein 4: 121–168.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1–218.
MacDonald JT, Nel JAJ. 1986. Comparative diets of sympatric carnivores. South African Journal of Wildlife Research 16: 115–121.
Mbatyoti OA. 2010. The diet of the Cape grey mongoose Galerella pulverulenta in the Albany Thicket Biome (South Africa). B.Sc. Honours Thesis. University of Fort Hare.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Nieman, W. A., Wilkinson, A., & Leslie, A. J. (2020). Farmer attitudes and regional risk modelling of human–wildlife conflict on farmlands bordering the Boland Mountain Complex, South Africa. African Journal of Wildlife Research, 50(1), 36-54.
Pallett, J. and Thomson G 2022. A conservation assessment of Small Grey Mongoose Herpestes pulverulentus. In: NCE, LCMAN, MEFT (eds) 2022. Conservation Status and Red List of the Terrestrial Carnivores of Namibia. Pp 141-142. MEFT, LCMAN & NCE, Windhoek, Namibia
Simelane, T. S., & Kerley, G. I. H. (1998). Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research, 28(4), 121-126.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Spies, M.J., Gibbon, V.E. and Finaughty, D.A. 2018. Forensic taphonomy: vertebrate scavenging in the temperate southwestern Cape, South Africa. Forensic science international, 290, 62–69.
Slingsby, J. A., Merow, C., Aiello-Lammens, M., Allsopp, N., Hall, S., Kilroy Mollmann, H., Turner, R., Wilson, A. M., and Silander Jr, J. A. (2017). Intensifying postfire weather and biological invasion drive species loss in a Mediterranean-type biodiversity hotspot. Proceedings of the National Academy of Sciences, 114(18), 4697-4702.
Stuart, C. T. (1981). Notes on the mammalian carnivores of the Cape Province, South Africa. Department of Nature and Environmental Conservation.
Stuart C. 1991. Aspects of the biology of the small grey mongoose Galerella pulverulenta. Mustelid & Viverrid Conservation 4: 1–4.
Stuart, C. and Stuart, T. 1998. Notes on the diet of four species of viverrid in a limited area of southern Namaqualand, Northern Cape, South Africa. Small Carnivore Conservation 19: 9–10.
Stuart, C. and Stuart, M. 2015. Stuarts’ Field Guide to Mammals of Southern Africa including Angola, Zambia & Malawi. Struik Nature, Penguin Random House, Cape Town, South Africa.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Taylor, P. J., Kearney, T. C., Clark, V. R., Howard, A., Mdluli, M. V., Markotter, W., Geldenhys, M., Richards, L. R., Rakotoarivelo, A. R., Watson, J., Balona, J. and Monadjem, A. (2024). Southern Africa’s Great Escarpment as an amphitheater of climate‐driven diversification and a buffer against future climate change in bats. Global Change Biology, 30(6), e17344.
Taylor, P. J., Nengovhela, A., Linden, J., and Baxter, R. M. (2016). Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia, 80(4), 359-375.
Veron, G., Patou, M.-L. and Jennings, A.P. 2022. Systematics and evolution of the mongooses (Herpestidae, Carnivora). In: E. Do Linh San, J.J. Sato, J.L. Belant and M.J. Somers (eds), Small Carnivores: Evolution, Ecology, Behaviour and Conservation, pp. 61–78 Wiley-Blackwell, Oxford, UK.
Waser PM, Keane B, Creel SR, Elliot LF, Minchella DJ. 1994. Possible male coalitions in a solitary mongoose. Animal Behaviour 47: 289–294.
Weigl, R. 2005. Longevity of Mammals in Captivity; from the Living Collections of the World.
Wozencraft, W.C. 2005. Order Carnivora. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World: A Taxonomic and Geographic Reference. Third Edition, pp. 532–628. Johns Hopkins University Press, Baltimore.