Cape Golden Mole
Chrysochloris asiatica

2025 Red list status
Regional Population Trend
Change compared
to 2016
No Change
Overview
Chrysochloris asiatica – (Linnaeus, 1758)
ANIMALIA – CHORDATA – MAMMALIA – AFROSORICIDA – CHRYSOCHLORIDAE – Chrysochloris – asiatica
Common Names: Cape Golden Mole (English), Kaapse Gouemol, Kaapse Kruipmol (Afrikaans), Kapgoldmull (German)
Synonyms: Talpa asiatica Linnaeus, 1758
Taxonomic Note:
Previously included Kilimatalpa as a subgenus (Bronner 1995), although Kilimatalpa is now recognised as a separate genus (Bronner et al. 2024).
Up to ten subspecies have previously been recognised, but size and colour varies clinally, hence validity of these taxa is dubious (Meester et al. 1986). The only known specimen of damarensis may have been incorrectly labelled as no Chrysochloris have subsequently been collected in Damaraland, Namibia (Meester 1974).
Red List Status: Least Concern
Assessment Information
Assessor: Mynhardt, S.1 & da Silva, J.2
Reviewer: Asher, R.J.3
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3University of Cambridge
Previous Assessor: Bronner, G.
Previous Reviewers: Asher, R.J. & Taylor, A.
Assessment Rationale
A widespread species that does not appear to be in decline. Although habitat alteration/degradation is ongoing in some parts of this species range, it has adapted well to mildly transformed habitats ranging from vineyards and pasturelands to city parks and gardens; habitat alteration is not, therefore, considered to be a major threat and it is therefore listed as Least Concern.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Mynhardt S & da Silva JM. 2025. A conservation assessment of Chrysochloris asiatica. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
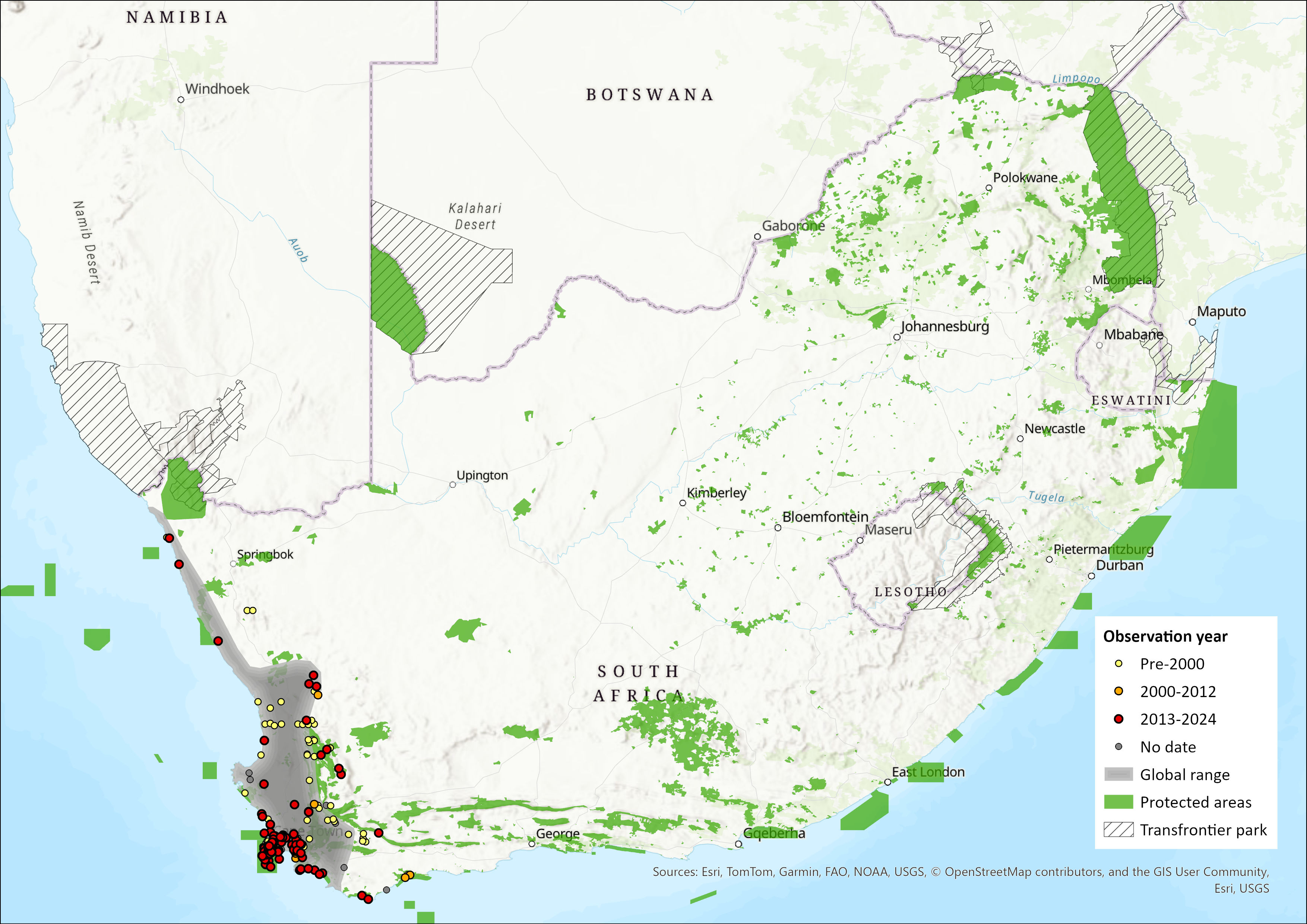
This species is endemic to South Africa. Ranges from the Cape Peninsula (Western Cape) south-eastwards across Cape Flats to Bredasdorp, Swellendam, possibly as far as Knysna; and northwards along the Atlantic coast to Port Nolloth in Northern Cape. Occurs inland to Ceres, Worcester, Sutherland and Stellenbosch in the south, and Calvinia, Garies and Kamieskroon in the north, but does not penetrate the arid Karoo (Taylor et al. 2018). One specimen (type of C. damarensis) recorded from Damaraland in Namibia is probably a misidentified specimen from Little Namaqualand (Meester et al. 1986).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Cape Golden Mole (Chrysochloris asiatica) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Eastern Cape Province | Extant | Native | – | Resident |
| South Africa -> Northern Cape Province | Extant | Native | – | Resident |
| South Africa -> Western Cape | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Although the potential impacts of climate change on this species have not been directly assessed, climate change is expected to impact most golden mole species. Since these subterranean animals are restricted to soft soils for burrowing, and have very low vagility, their ability to migrate or shift their distribution ranges in response to climate change is very limited. Climate change is likely to impact golden moles through changes in vegetation type, soil moisture and prey availability, and coastal species additionally through reduced available habitat in developed/developing areas, as rising sea levels reduce the area of undeveloped coastal dune habitats between the coast and its associated developments.
Population information
Chrysochloris asiatica is common in most habitats, depending on soil friability and invertebrate prey resources. They are less common on rocky mountain slopes, but nevertheless have still colonized even some mountain plateaux. In prime habitat, inferred densities are up to 4 individuals / ha (G.N. Bronner unpubl. data).
Current population trend: Unknown
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
While phylogenetic assessments have been undertaken on golden moles that have incorporated this species (e.g., Asher et al. 2010, Bronner et al. 2024), limited sampling included within these studies has prevented more fine-scale examination of the population structure within the species.
A complete reference genome exists for the species (GCA_000296735.1), as well as a mitochondrial genome (NC_004920.1), which could facilitate a population genomic study as was conducted on A. hottentotus (Mynhardt et al. 2024). Subpopulation structure was revealed within A. hottentotus, which could suggest a similar outcome for C. asiatica.
While the effective population sizes (Ne) for this species’ populations have not been quantified, given that it is considered common in most habitats, it is likely to be genetically stable and healthy (e.g., Ne > 500). This should be verified through population genomic analyses.
Habitats and ecology
The Cape Golden Mole occurs within sandy soils in the Fynbos and Nama-Karoo biomes, and has been recorded from a wide variety of vegetation types, most commonly in Renosterveld, Fynbos and Strandveld Succulent Karoo. They are common in parks, gardens and cultivated lands, and are known to readily invade lawns and golf courses. This species also forages on beaches, presumably for amphipods and isopods associated with kelp wracks (Bennett and Spinks 1995, Bronner 2013). They coexist with Fynbos Golden Moles (Amblysomus corriae) in the Stellenbosch region, but comparatively prefer drier, sandier soils (Broom 1907).
Cape Golden Moles are predominantly active at night or during the late afternoons, particularly after rainfall events (Skinner and Chimimba 2005). As a solitary and occasionally aggressive (to conspecifics) species, only females raising young coexist (Skinner and Chimimba 2005). Breeding is restricted to the wet winter season of the Western Cape, with females producing between one and three young (Skinner and Chimimba 2005).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | Resident | Suitable | Yes |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | Resident | Suitable | Yes |
| 3.4. Shrubland -> Shrubland – Temperate | Resident | Marginal | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | Resident | Suitable | Yes |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | Resident | Suitable | Yes |
| 3.7. Shrubland -> Shrubland – Subtropical/Tropical High Altitude | Resident | Marginal | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | Resident | Suitable | Yes |
| 4.4. Grassland -> Grassland – Temperate | Resident | Suitable | No |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | Resident | Marginal | – |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | Resident | Suitable | No |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | Resident | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | Resident | Suitable | No |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | Resident | Suitable | No |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: 9-12
Size at Maturity (in cms): Male: 10-12
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): 5
Gestation Time: (Not specified)
Reproductive Periodicity: Recorded births (June to August) suggest that reproduction peaks in the wet winter months, at least in the south-western Cape.
Average Annual Fecundity or Litter Size: Litter size 1-3 (Bronner 2013)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
General notes regarding trade and use of this species: This species is not known to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Natural habitats of this species have undoubtedly been dramatically altered by urbanisation, coastal tourism developments, mining for alluvial diamonds and agriculture during the past 350 years, but this is not considered a threat as it coexists (indeed even thrives) in close association with humans. The species is persecuted by gardeners and greenkeepers and regularly caught by domestic dogs and cats. Its distribution nonetheless remains fairly widespread given its cryptic nature.
Conservation
Although the species is formally protected in many national and provincial conservation areas, management is not consistent across parks and provinces. Research to clarify status of described forms/subspecies is needed, as distinct geographical ranges do not all enjoy high levels of habitat protection. The range of C. asiatica overlaps with the Western Cape fold mountains. Populations attributed to C. asiatica, for example near Modderfontein (USNM 342427) and Kamiesberg (MCZ 39606) in Northern Cape, to Port Elizabeth (BMNH 1921.1.3.2) and Knysna (Oslo 17462, 17464) in or near Eastern Cape, are approximately 1000km apart on either side of major geographic barriers and lack direct gene flow. Such populations may also be distinct from populations around Cape Town, Stellenbosch and their suburbs. Therefore, multiple, distinct evolutionary lineages are likely present.
Recommendations for land managers and practitioners:
- None, this species adapts well to transformed habitats providing that transformation is not too severe.
Research priorities:
- Research to clarify the status of described forms/subspecies is needed, as some of these may be distinct taxa given that their range largely corresponds with the Western Cape Fold Mountains, which may serve as barriers to gene flow between some populations.
- Surveys needed to determine subpopulation size, trend and distribution, particularly within urban and agricultural areas.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Deposit any dead specimens found in a state or provincial museum, together with information on the date and site where found.
- Create native vegetation gardens.
Bibliography
Afrotheria Specialist Group. 2014. Specialist Group website. Available at: http://www.afrotheria.net/index.php.
Bennett, N.C. and Spinks, A.C. 1995. Thermoregulation and metabolism in the Cape golden mole (Insectivora: Chrysochloris asiatica). Journal of Zoology (London) 76: 957–971.
Bronner, G.N. 1995. Systematic revision of the golden mole genera Amblysomus, Chlorotalpa and Calcochloris (Insectivora: Chrysochloromorpha; Chrysochloridae). Ph.D. Thesis, University of Natal.
Bronner, G.N. 2013. Chrysochloris asiatica. In: J. Kingdon, D. Happold, T. Butynski, M. Hoffmann, M. Happold and J. Kalina (eds), Mammals of Africa, Volume I: Introductory Chapters and Afrotheria , pp. 242-244. Bloomsbury Publishing, London.
Bronner, G.N. and Jenkins, P.D. 2005. Order Afrosoricida. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World, pp. 70-81. The Johns Hopkins University Press, Baltimore, MD, USA.
Bronner, G.N., Mynhardt, S., Bennett, N.C., Cohen, L., Crumpton, N., Hofreiter, M., Arnold, P. and Asher, R.J., 2024. Phylogenetic history of golden moles and tenrecs (Mammalia: Afrotheria). Zoological Journal of the
Linnean Society, 201(1), pp.184-213.
Broom, R. 1907. A contribution to the knowledge of the Cape golden moles. Transactions of the South African Philosophical Society 18: 283–311.
IUCN. 2015. The IUCN Red List of Threatened Species. Version 2015.2. Available at: www.iucnredlist.org . (Accessed: 23 June 2015).
Meester, J. 1974. Family Chrysochloridae. Smithsonian Institution Press, Washington, DC, USA.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Pence, G.Q.K. 2014. Western Cape Biodiversity Framework 2014 status update: Critical Biodiversity Areas of the Western Cape. CapeNature, Cape Town.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Third edition. Cambridge University Press, United Kingdom, Cambridge.
Taylor WA, Mynhardt S, Maree S (2018). Family Chrysochloridae. In: Handbook of the Mammals of the World – Volume 8 Insectivores, Sloths and Colugos. DE Wilson & RA Mittermeier (eds). Lynx Edicions. ISBN-13: 978-84-16728-08-4.