Cape Gerbil
Gerbilliscus afra

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Gerbilliscus afra – (Gray, 1830)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Gerbilliscus – afra
Common Names: Cape Gerbil, Cape Gerbille (English), Kaapse Nagmuis (Afrikaans)
Synonyms: Tatera afra (Gray 1830), africanus, caffer, gilli, schlegelii (Dempster 2013)
Taxonomic Note: Although previously classified as Tatera afra (Skinner & Chimimba 2005), this species was recently recognised by Musser and Carleton (2005) as Gerbilliscus afra (Gray 1830). Aside from Gerbilliscus paeba (readily distinguished from G. afra by its smaller size, relatively long tail and hairy soles on its hindfeet), this is the only Gerbilliscus species present within its range (Monadjem et al. 2015), and no subspecies have been recognised (Dempster 2013). Treated by Musser and Carelton (2005) under Gerbilliscus afra (Gray, 1830).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Smith, C.1 & da Silva, J.M.2
Reviewer: Taylor, P.J.3
Contributors: Roxburgh, L.1 & Patel, T.1
Institutions:1Endangered Wildlife Trust,2South African National Biodiversity Institute,3University of the Free State
Previous Assessors and Reviewers: Palmer, G. & Cassola, F.
Assessment Rationale
Listed as Least Concern in view of its wide distribution, presumed large population, and because it is unlikely to be declining fast enough to qualify for listing in a more threatened category. Additionally, it adapts well to changing land use to the extent that it is considered an agricultural pest, which is being controlled with some success through the provision of nesting boxes and perches for Barn Owls (Tyto alba). This intervention should be encouraged as part of a holistic management technique.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No Change
Red List Index
Red List Index: No change
Recommended citation: Smith C & da Silva JM. 2025. A conservation assessment of Gerbilliscus afra. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
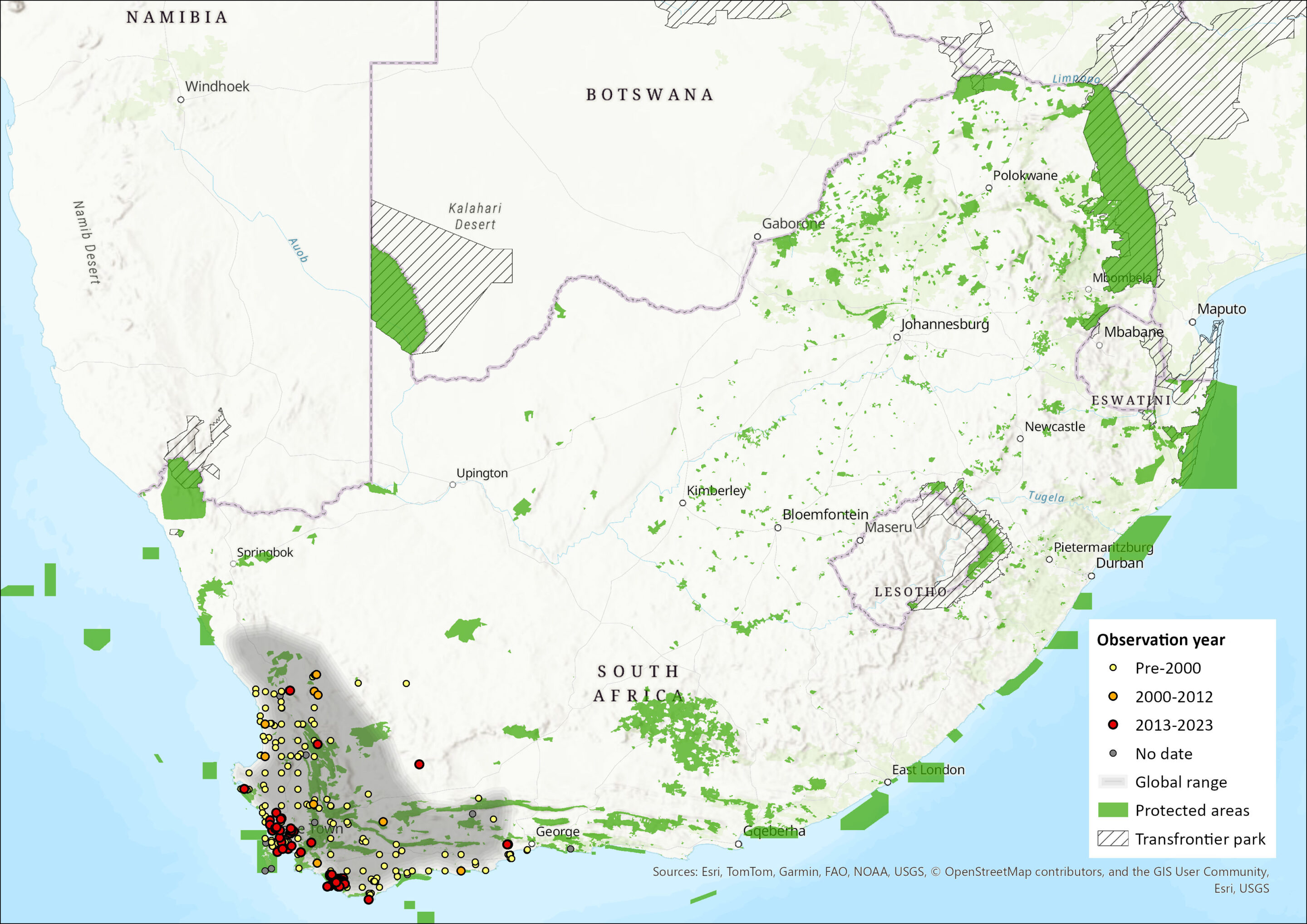
The Cape Gerbil is endemic to the southwestern region of South Africa, extending from Nieuwoudtville in the Northern Cape southwards to the Cape Peninsula of the Fynbos Biome of the Western Cape, eastwards to Herold’s Bay (near George, Western Cape) (Skinner & Chimimba 2005; Avery et al. 2005; Monadjem et al. 2015). Its distribution is somewhat fragmented across the low lying regions of the Western Cape Province. The estimated extent of occurrence (EOO) is 150,176 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Cape Gerbil (Gerbilliscus afra) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Western Cape | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
This species only occurs in the Western and Northern Cape provinces, South Africa. There is a possibility that it might be affected by the 1.5-4°C increase in temperature predicted under various climate change scenarios (Engelbrecht et al. 2024). This species creates burrows and might be able to avoid some temperature extremes by seeking refuge in the burrows. Rainfall is predicted to decrease in its range. These decreases will result in a more arid and drought-prone habitat which could affect food availability in the species distribution range.
Population Information
This species is locally abundant and common in suitable habitats (Dempster 2013). The population is stable within a widely fluctuating range depending on season and rainfall.
Population Information
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While this species has been investigated in a phylogenetic context (Colangelo et al. 2007), no population genetic assessment has been undertaken. It is abundant and seemingly connected across its range, and therefore is assumed to exist as a single metapopulation. Given no subpopulations are known to have gone extinct, it can be assumed that all known subpopulations (i.e., the metapopulation) remain. Consequently, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0. However, hidden structure might be revealed upon investigation as was found for G. leucogaster (McDonough et al. 2015). Given the limited information known about this species, such a study would be highly informative.
Because the species is presumed to be locally abundant numbering in excess of 10,000 individuals, it can be inferred that this would translate into it having an effective population size (Ne) exceeding 1000 (assuming a 0.1 conversion ratio of Ne to census size). While the 10,000 value is not an actual census count, the quantity can still be used as a proxy for quantifying the proportion of populations with an Ne > 500 indicator, which is valued at 1.0 (1/1 population with Ne > 500).
Habitats and ecology
The Cape Gerbil occurs within sandy soils or sandy alluvium (Skinner & Chimimba 2005), and appears to prefer the edge of scrubland regions and Karoo habitats. It is well adapted to cultivated lands (Dempster 2013), often thriving in agricultural areas during favourable environmental conditions. It is known to exhibit population explosions, and thus can become a pest in some areas, especially grain lands. It is considered a pest particularly for wheat farmers in the Western Cape (Grain SA). It also occurs in vineyards where it does not reach particularly high densities. Its presence here may yet be found to be beneficial, possibly assisting with drainage and nutrient cycling.
This species has been documented feeding on grass, roots, seeds and bulbs (Roberts 1951; Dempster 2013). They utilise sandy soils to excavate extensive burrows, comprising resting and breeding chambers (Skinner & Chimimba 2005). Breeding usually commences after the winter rainfall in the Western Cape (Measroch 1954), when they produce between three and five young (Dempster et al. 1992).
Ecosystem and cultural services: It may yet be found that their presence in vineyards is in fact beneficial from an agricultural point of view. They are also a tremendous food source for numerous predators from Barn Owls to snakes and Weasels (Poecilogale albinucha).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 8.2. Desert -> Desert – Temperate | – | Marginal | – |
Life History
| Generation Length | Justification | Data Quality |
| 1-2 | Inferred (see Pacifici et al. 2013) | good |
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
General Use and Trade Information
General notes regarding trade and use of this species: (Not specified)
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
No major threats have been identified for this species. However, it is persecuted as an agricultural pest – millions can be found on wheat fields in the West Coast area of the Western Cape (Potter 2004). Farmers often use poison to control the population, which has knock-on effects for the ecosystem (Makundi & Massawe 2011).
Current habitat trend: Stable
Conservation
This species is present in many protected areas within the assessment region, for example De Hoop and Riverlands Nature Reserves and Agulhas and West Coast National Parks. There are no specific interventions necessary, but biocontrol of population explosions through the provisioning of nest boxes and hunting perches for Barn Owls is an effective part of a holistic ecosystem management strategy. Barn Owls respond to prey explosions by increasing reproduction (Potter 2004; Makundi & Massawe 2011). This management intervention also saves farmers the cost of buying poison and the mortality of non-target species including domestic stock. For example, in the grain-growing areas of the Western Cape Province, Cape Gerbil numbers increase exponentially in good rain years, and this nocturnal pest can cause considerable damage to farmland and crops in the area.
A quantitative study of owls as bio-control agents was recently conducted on a West Coast wheat farm with a network of over 80 Barn Owl nest boxes (Potter 2004). The size of the owl population resident on this property varied from year to year, from about 15–40 pairs. Cape Gerbils made up about 90% of the diet of breeding owls and, collectively, the owls ‘harvested’ about 35,000 gerbils per year (Potter 2004). Overall, using owls to control gerbil damage was twice as effective as using poison, and much less expensive in the long-term. An optimal array of owl nest boxes in this area requires about one nest box per 25 ha of cropland, with each box spaced at least 500 m from its nearest neighbour. While the capital outlay required to set up such an array of boxes can be more or less the same as the cost of poison needed to control gerbils on an average farm in an average year, should scrap material be used, as in the case on the West Coast, it can be considerably less. Maintenance costs for an existing nest box scheme are probably less than 5% of the cost of another application of poison (in addition to the hidden financial costs of environmental damage caused by repeatedly dosing the farm with toxins). An added advantage of not using poison is that numerous other predators, such as snakes, mongooses, genets and weasels, are also maintained. As most are resident, they keep the pressure on throughout the year, making the chances of the rodent numbers getting beyond the capabilities of the Barn Owls less likely.
Recommendations for land managers and practitioners:
- Barn Owls are being used successfully to control this species through the provision of nesting boxes and hunting perches. This should be encouraged as a holistic management intervention.
Research priorities:
- Taxonomic resolution of the Gerbilliscus genus.
- Population size, distribution and trend estimates.
- Diet, reproduction and general biology.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Encourage the biocontrol of this species by installing Barn Owl nest boxes and hunting perches in areas affected by population explosions causing agricultural damage
Bibliography
Avery DM, Avery G, Palmer NG. 2005. Micromammalian distribution and abundance in the Western Cape Province, South Africa, as evidenced by Barn owls Tyto alba (Scopoli). Journal of Natural History 39:2047–2071.
Colangelo, P., Granjon, L., Taylor, P.J. & Corti, M. 2007. Evolutionary systematics in African gerbilline rodents of the genus Gerbilliscus: Inference from mitochondrial genes, Molecular Phylogenetics and Evolution, 42(3), 797-806. https://doi.org/10.1016/j.ympev.2006.10.001
Dempster ER. 2013. Gerbilliscus afra Cape Gerbil. Pages 270271 in Happold DCD, editor. Mammals of Africa. Volume III: Rodents, Hares and Rabbits. Bloomsbury Publishing, London, UK.
Dempster ER, Perrin MR, Nuttall RJ. 1992. Postnatal development of three sympatric small mammal species of southern Africa. Zeitschrift für Säugetierkunde 57:103–111.
Engelbrecht, FA., Steinkopf, J., Padavatan, J. & Midgley, GF. 2024. Projections of future climate change in southern Africa and the potential for regional tipping points. In: von Maltitz, G.P., et al. Sustainability of Southern African Ecosystems under Global Change. Ecological Studies, vol 248. Springer, Cham. https://doi.org/10.1007/978-3-031-10948-5_7.
IUCN. 2016. The IUCN Red List of Threatened Species. Version 2016-2. Available at: www.iucnredlist.org. (Accessed: 04 September 2016).
Makundi RH, Massawe AW. 2011. Ecologically based rodent management in Africa: potential and challenges. Wildlife Research 38:588–595.
McDonough, M.M., Šumbera, R., Mazoch, V., Ferguson, A.W., Phillips, C.D. and Bryja, J., 2015. Multilocus phylogeography of a widespread savanna–woodland‐adapted rodent reveals the influence of Pleistocene geomorphology and climate change in Africa’s Zambezi region. Molecular Ecology, 24(20), pp.5248-5266.
Measroch V. 1954. Growth and reproduction in the females of two species of gerbil, Tatera brantsi (A. Smith) and Tatera afra (Gray). Proceedings of the Zoological Society of London 124:631–658.
Monadjem A, Taylor PJ, Denys C, Cotterill FP. 2015. Rodents of Sub-Saharan Africa: a biogeographic and taxonomic synthesis. Walter de Gruyter GmbH, Berlin, Germany.
Musser, G.G. and Carleton, M.D. 2005. Superfamily Muroidea. In: D.E. Wilson and D.A. Reeder (eds), Mammal Species of the World: a geographic and taxonomic reference, pp. 894-1531. The John Hopkins University Press, Baltimore, USA.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Potter L. 2004. Raptors for rodent control: Is the Barn Owl a viable control agent for pest rodents on South African farmlands? M.Sc. Thesis. University of Cape Town, Cape Town, South Africa.
Rathbun, G.B. (subeditor). 2005. Macroscelidea. In: J.D. Skinner and C.T. Chimimba (eds), The Mammals of the Southern African Subregion, 3rd edition, pp. 22-34. Cambridge University Press, Cambridge, UK.
Roberts A. 1951. The Mammals of South Africa. News Agency, Cape Town, South Africa.
Skinner JD, Chimimba CT. 2005. The Mammals of the Southern African Subregion. Third edition. Cambridge University Press, Cambridge, UK.