Cape Genet
Genetta tigrina

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Genetta tigrina – (Schreber, 1776)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – VIVERRIDAE – Genetta – tigrina
Common Names: Cape Genet, Cape Large-spotted Genet, South African Large-spotted Genet, Tigrine Genet (English), Grootkolmuskeljaatkat (Afrikaans), !Noreb (Damara, Nama), Insimba (Ndebele, Swati, Zulu), Thsipathoko (Sepedi), Thsipa, T’sipa, Tsipa e matheba a maholo (Sesotho), Tshipathokolo, Thokolo (Setswana), Tsimba (Shona, Venda), Msimba-mangovo, Nsimba (Tsonga), Inyhwagi (Xhosa)
Synonyms: Genetta methi Roberts 1948
Taxonomic Note:
This species was until recently considered conspecific with the Rusty-spotted Genet (G. maculata) (Coetzee 1977; Meester et al. 1986). It is here regarded as distinct based on evidence provided by Crawford-Cabral (1980–81), Crawford-Cabral and Pacheco (1992), Schlawe (1981), Gaubert (2003) and Gaubert et al. (2005b). See Wozencraft (2005) and Gaubert (2013) for further discussion. Through a naked eye Cape Genet differs from Rusty-spotted Genet by the absence of rusty spots and the presence of “black socks”, i.e. the coloration of the whole hindfoot and that of the posterior part of the upper arms and around forefoot pads is dark. The Cape Genet also possesses a middorsal crest (i.e. longer hairs along the spine) and longer tail hairs (4–4.5 cm versus 2–3 cm) than Rusty-spotted Genet, but these characteristics cannot always be identified during fleeting field encounters or even on pictures. Despite the presence of other morphological and some genetic differences, further investigations are needed in order to solve the taxonomic status of G. tigrina relative to G. maculata. [Note that the species name “maculata” is invalid; see details in Rusty-spotted Genet account.]
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Carvalho, F.1,2, Ehlers Smith, Y.3, Patel, T.4 & da Silva, J.M.5
Reviewer: Do Linh San, E.6
Previous Assessors and Reviewers: Widdows, C.D., Roberts, P.D., Maddock, A.H., Carvalho, F., Gaubert, P. & Do Linh San, E.
Previous Contributors: Hoffmann, M., Page-Nicholson, S. & Child, M.F.
Institutions: 1University of Fort Hare, 2CIBIO – University of Porto,3Ezemvelo KZN Wildlife, 4Endangered Wildlife Trust, 5South African National Biodiversity Institute, 6Sol Plaatje University
Assessment Rationale
The Cape Genet is listed as Least Concern because it has a relatively wide range in South Africa, appears to be common, occurs in human-modified habitats, does not face major threats, and is present in several protected areas across its distribution. Population size and trend estimates are not available for most areas, but it is thought that the population is currently stable across the entire range. However, we caution for the impacts that agriculture intensification and rise of road constructions might have in a near future.
Regional population effects: We suspect that there is dispersal across the distribution range, although possible barriers to the movements of this species at a landscape scale may exist.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Carvalho F, Ehlers Smith Y, Patel T & da Silva JM. 2025. A conservation assessment of Genetta tigrina. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
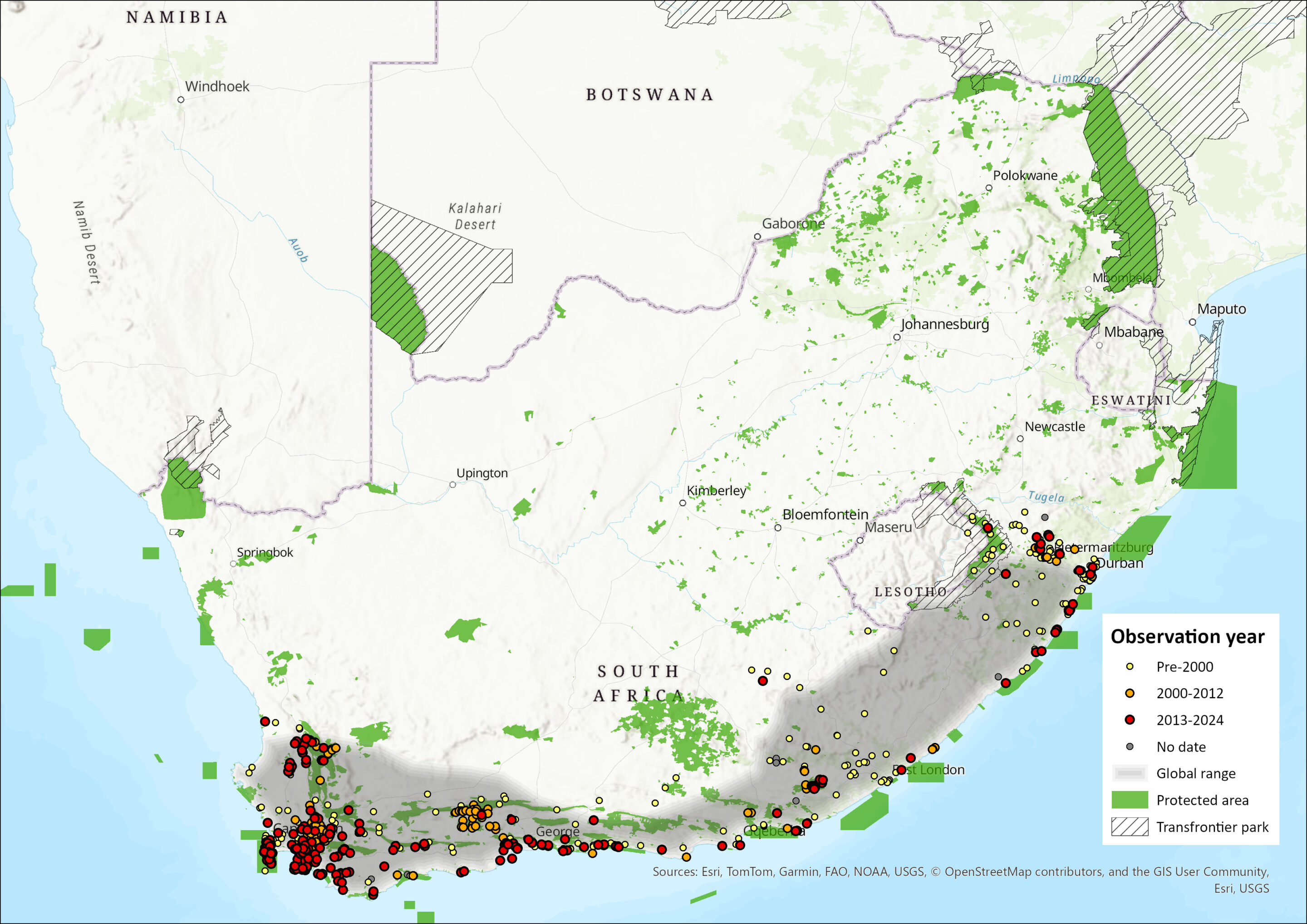
The Cape Genet is the only carnivore species endemic to the assessment region. Morphometric analysis and coat pattern variation indicate that it is solely distributed in higher rainfall areas – and particularly in coastal forests – from the Western Cape to southern KwaZulu-Natal, south of 32°S, and to the neighbouring Lesotho border (Schreiber et al. 1989; Bronner et al. 2003; Gaubert 2003; Gaubert et al. 2005a, 2005b; Gaubert 2013). Two subspecific populations of G. tigrina may occur; G. t. tigrina, ranging from southern Western Cape Province to south KwaZulu-Natal and G. t. methi from the Pondoland area, southern KwaZulu-Natal (Roberts 1951), although morphometric and genetic evidence did not support such differentiation (Gaubert et al. 2005a). Historic distributions of G. tigrina elsewhere mentioned in Lack (1977), Wemmer (1977) and Skinner and Smithers (1990) now reclassify to other species, with the Kenyan population for example reclassifying to G. maculata. The Cape Genet is considered sympatric with at least two other genet species (G. maculata and G. genetta) and this may result in a hybridisation zone in these areas where ranges overlap. Genetta tigrina naturally but rarely crosses with G. maculata in a restricted hybridisation zone within KwaZulu-Natal. Genetta genetta has hybrid compatibility with G. tigrina in captivity and genetic evidence showed a case of hybridisation between the species in the wild (Gaubert et al. 2005a). Accordingly, in areas where G. tigrina is sympatric with G. genetta, such as in some areas of the Eastern Cape (Boshoff et al. 1994; E. Do Linh San pers. obs. 2005–2015), further investigation of any hybridisation is needed.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Cape Genet (Genetta tigrina) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Lesotho | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
So far, no specific research on how climate change will affect this species has been carried out. However, genets are prone to high thermoregulatory challenges. In a recent telemetry activity study in a semi-arid Albany Thicket landscape (Eastern Cape, South Africa), Cape Genets showed a pronounce decrease in activity during the dry-cold season (March-September) and stopped totally below 0ºC (Carvalho et al. 2024b). Similarly, during hot days (> 35 º C) due to their nocturnal nature they avoid moving during those days, although some activity might happen near resting sites in dense vegetation areas like the riverine forests where they prevail (Carvalho et al. 2014, 2024b). Additionally, Carvalho and colleagues (2024b), also found that Cape Genets found away from the coast, reduce their occupancy and abundance significantly in areas where they occur in sympatry with Small-spotted Genets (Genetta genetta). In those areas they show abundances 3-4 times lower compared to Small-spotted Genets and are constrained to wider and dense Combretum riverine forests (Carvalho et al. 2024a, b). In this semi-arid Albany Thicket landscapes those riverine forests are the only refuges that allow the species to persist away from the mild coast weather (Carvalho et al. 2024b). In areas where extreme temperature might affect their occupancy, maintaining buffering areas like riverine forests might be crucial for their persistence across landscapes (Carvalho et al. 2024b).
Population Information
There is no precise information on their abundance, but they are not uncommon. In areas of sympatry with G. genetta, trapping success seems to indicate that G. tigrina may be 3–4 times less abundant than the former species (F. Carvalho & E. Do Linh San pers. obs. 2005–2019, Carvalho et al. 2024b).
Population Information
Current population trend: Unknown, but probably stable due to a lack of severe threats.
Continuing decline in mature individuals: Unknown, but probably not.
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: It is not currently possible to determine whether there are clearly delineated subpopulations, and if so, how many.
Severely fragmented: Not currently, since Cape Genets can exist in multiple habitats (including in rural areas) as long as there is dense vegetation cover.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While Genetta tigrina has been studied in a broad phylogenetic context (Gaubert et al. 2005, Ferguson et al. 2018), no fine scale population genetic study has been undertaken within the assessment region.
While population structure is unknown, it is expected this species is able to travel considerable distances (at least one individual per generation), and therefore it is expected to exist as a single metapopulation. Based on this assumption, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining).
While precise population sizes (e.g. census [Nc]) or effective population size (Ne) data do not exist, the perception that the population is stable and of least concern, suggests that there could well be 10,000 or more individuals in the assessment region. Even though the 10,000 value is not an actual census count, this quantity can still be used as a proxy for quantifying the GBF’s headline genetic indicator – proportion of populations with an Ne > 500. Based on the assumption that the species is likely to occur as a single metapopulation, the Ne of the species is roughly estimated at ~1,000-3,000 individuals, based on a Ne/Nc conversion ratio of 0.1-0.3. Having the 1 population above Ne 500, translates to an indicator score of 1.0 (1/1 population with Ne > 500).
Habitats and ecology
Cape Genets are found in woodlands and other densely vegetated habitats such as Fynbos and Albany Thicket in Western and Eastern Cape. However, coastal forests are possibly the species’ preferred habitat across its distribution range (Stuart 1981; Gaubert 2013). They have also often been reported as occurring in riparian habitats, especially when crossing open agricultural land (Roberts et al. 2007; Gaubert 2013). These habitats may therefore act as important dispersal corridors. Cape Genets have also been recorded within close proximity of permanent water reservoirs (Rowe-Rowe 1992), open grasslands and exotic scrublands (Western Cape) during foraging (Maddock & Perrin 1993; Gaubert 2013), but not too far from areas with vegetation cover, also due to the predation risk during those incursions. They have also been found using pine plantations and urban areas in KwaZulu-Natal (Rowe-Rowe 1992; Widdows 2016). They are good climbers, thus are able to select resting sites in elevated places such as hollow trees (Stuart 1981), rocky crevices and roofs in barns, outbuildings and other farmhouses. Nevertheless, in areas where the shortage of elevated roost sites (for example, coastal shrubland areas) is evident, they rest below dense thickets and under tree roots (Roberts 2006). So far, only two studies looked at the spatial ecology of this species. In Vernon Crooks Nature Reserve, Maddock (1988) found an average home range size of 22 ha (range 6–33 ha), but these values are based on only three Genets radio-tracked during short periods (range 1–5 months). Within the urban landscape of Kloof (KZN), Widdows (2016) recorded an average 95% kernel density home range size of 32 ha (range 17–48 ha). However, these values are based on the GPS tracking of only two individuals over a period of 2–3 months.
Within the Indian Ocean Coastal Belt forest, the species was the third most photographed mammal. Given that the species is considered an arboreal associated species much time was spent on the ground, where photographs using camera traps were captured. The species had an overall site occupancy of 0.72 (72%) across 250 camera trap sites surveyed and was found within habitat patches nestled in anthropogenic land use types such as urban and farmland (Ehlers Smith et al. 2018). In these habitats an increased Functional Height Diversity of the vegetation profile positively influenced the species occupancy, suggesting that they have a preference for heterogenous structures within their habitat (Ehlers Smith et al. 2017). In coastal areas G. tigrina outcompetes G. genetta and dominates, whereas in more interior transitions areas where both species exists like in the Eastern Cape Albany Thicket landscapes, G. tigrina only persists in wider Combretum riverine forests (Carvalho et al. 2024a, b, Unpublish data). In an Albany Thicket landscape (Eastern Cape), they prevail only in wider and dense Combretum riverine forests, as it has been recently shown in a year-round radiotracking study of 6 individuals (3 males and 3 females), they leave the riverine forest frequently to hunt in the bushclump Karroid Thickets, but within 50 meters form the riverine corridors (F. Carvalho, unpublished data). They clearly avoided open areas such as grasslands and bare soil areas likely due to the high predation risk in these treeless areas (F. Carvalho, unpublished data).
Cape Genets have an opportunistic generalist diet, and Roberts et al. (2007) found in a study in the Eastern Cape that the main bulk is comprised of small mammals (Dendromus sp., Otomys sp., Graphiurus sp. and Mastomys sp.) and arthropods (Coleoptera and Orthoptera). Other important items were birds, although they were eaten in lower numbers than expected from a semi-arboreal predator; the peak of consumption was recorded in winter and spring during the nesting period (Roberts et al. 2007). A huge amount of grass has also been found and it is believed to aid digestion (Roberts et al. 2007; Gaubert 2013). Maddock (1988) in southern KwaZulu-Natal also found a large proportion of small mammals and other vertebrates, including birds, as well as numerous fruits. In other areas, rodents, birds (including Columbidae), insects, spiders and pill millipedes, freshwater crabs and earthworms have been reported (Stuart 1981). They also feed on human leftovers and pet food in some urban backyards, and scavenge on fish in some beach areas (Roberts et al. 2007).
The Cape Genet is a nocturnal species, with a peak of the activity occurring during the first hours of the night after leaving the day-time resting sites (Maddock & Perrin 1993; E. Do Linh San and M.J. Somers unpubl. data). Individuals are solitary, but they may form pairs during the breeding season. Intraspecific communication is mainly done through olfaction, notably with faeces deposited at conspicuous latrines (Rautenbach 1982; Maddock 1988; Widdows 2016). These are strategically located at the animals’ home range boundaries, such as at the junctions of routes, in large tree branches and high rocks, where they are less likely to be washed away and the scent might be more effectively spread (Roberts 2006; Gaubert 2013). Cape Genets also communicate by using their perineal glands, especially during the breeding season, when they rub their glands against large tree trunks, rocks or shrubs. Rusty-spotted Genets (G. maculata) are capable of producing vocalisations in several situations such as during courtship, cub rearing and territorial defence (Wemmer 1977), and it is likely to be valid for Cape Genets too. [N.B.: Wemmer’s (1977) study was based on observations of captive G. tigrina collected in Tanzania, but following the change of specific nomenclature these animals now correspond to G. maculata.]
Ecosystem and cultural services: The Cape Genet is expected to play an important role in ecosystems by preying on rodents and other small mammals, and also promoting seed dispersal due to its omnivorous diet. Moreover, this species might have the potential to become a successful example of pacific coexistence between humans and wildlife in the context of the ongoing growth of urban landscapes. Locally, genets are also of cultural importance, as detailed in the next section.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.6. Grassland -> Grassland – Subtropical/Tropical Seasonally Wet/Flooded | – | Marginal | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Suitable | – |
Life History
Generation length: 4 years (E. Do Linh San pers. comm. 2015)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Genet hides and tails are sometimes used in traditional Zulu clothing items. In local traditional medicine pieces of genet skin are used as stick-fight charms, or to adorn hats, and parts of the body are used to treat ailments of eyes. Some people eat the flesh (Cunningham & Zondi 1991).
Different genet species are kept as exotic pets. While this is especially common in the USA, it does not seem to be popular in the assessment area. It is likely that a large majority of pet genets originate from captive breeding programmes rather than from the wild. The number and proportion of Cape Genets kept as pets both globally and in the assessment region is unknown.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | – | No | – |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 3. Medicine – human & veterinary | – | true | – | – |
| 10. Wearing apparel, accessories | true | true | – | – |
| 12. Handicrafts, jewellery, etc. | – | true | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
We suspect that there are currently no known major threats to this species. Cape Genets occasionally kill domestic poultry in both rural and peri-urban areas and are regarded as problem animals by those that incur losses. They are therefore occasionally killed by farmers in retaliation for predation on small domestic stock and poultry, which could influence numbers in some areas (Stuart 1990).
The extent of road mortality on Cape Genet populations within the assessment region is unknown, although individuals of this species have been recorded in the Endangered Wildlife Trust’s road collision database (W. Collinson unpubl. data). In both urban and rural areas Cape Genets are also known to be killed by feral and domestic cats and dogs, and are sometimes deliberately poisoned or killed indirectly by the consumption of poisoned rodents. Within urban areas attacks by domestic dogs and collisions with motor vehicles are clearly common threats to Genets. Over a 12-month period between 2012 and 2013, 19 Genets were killed by collisions with vehicles and 27 killed by domestic dogs within Durban and surrounding municipalities (Widdows 2016).
However, it is believed that these sources of mortality, in addition to the non-consumptive use of this species, do not currently threaten local populations.
Conservation
Despite the abovementioned threats, Cape Genets occur commonly outside protected areas. They are also present in several protected areas throughout their range, such as Table Mountain, Garden Route, and Addo Elephant national parks (www.sanparks.org), Great Fish River Complex (E. Do Linh San pers. obs. 2010) and Dwesa-Cwebe Wildlife Reserve (Roberts et al. 2007). In southern KwaZulu-Natal the species is present within Umtamvuna, Mbumbazi, Mpenjati, Skyline, Oribi Gorge and Vernon Crookes Nature Reserves. Animals from Hluhluwe-Imfolozi Game Reserve are Central African Large-spotted Genets G. maculata (P. Gaubert pers. obs. 2014).
Conservation of the species should focus primarily on (i) solving the taxonomic status of the Cape Genet relative to the Rusty-spotted Genet, (ii) assessing levels of gene flow with co-occurring species, notably at its range limits (East and North) and (iii) deciphering population structure across its range. Beyond that, it would be important to evaluate whether the protected areas will suffice to sustain stable Cape Genet populations and assess the degree of connectivity among them. Indeed, due to the abovementioned increase in logging and deforestation activities and the expected rise of road construction and land use changes in South Africa (Collinson et al. 2015), the assessment of their effects on landscape functional connectivity would be crucial to ensure the long-term persistence of Cape Genets, as has been evaluated for other genet species elsewhere (Carvalho et al. 2016). Conservationists should encourage landowners to implement interventions that would reduce predation on Cape Genets, such as collar devices or sterilising pets in rural areas (see Recommendations below), as such interventions have been shown to significantly reduce wildlife mortality (Nelson et al. 2005; Claver et al. 2007).
Recommendations for land managers and practitioners:
Due to the often reported Cape Genet casualties either by feral and domestic cats and dogs (Widdows 2016), it is important to control the number of feral cats and dogs in both urban and rural areas, and encourage dog and cat owners to put collar-mounted bells, sonic bleepers or “pounce protectors” on their pets (Nelson et al. 2005; Calver et al. 2007). To reduce collisions with vehicles, mitigation measures such as road fencing and improvement of habitat near road crossing structures (for example, underpasses) should be implemented whenever possible (Collinson et al. 2015).
Research priorities:
- Taxonomic status of G. tigrina versus G. maculata.
- Competition and hybridisation patterns with sympatric species of genets (G. genetta and G. maculata).
- Population estimates, demographic parameters and possible barriers to dispersal across the species’ distribution range.
- Home range and habitat use of Cape Genets in a variety of landscapes (protected areas, agricultural areas and urban landscapes).
- The distribution for G. tigrina vs. G. maculata and G. genetta needs to be mapped properly to avoid the inadvertent introduction of one species into the range of the other – which is often a post rehabilitation issue. The large spots present in the G. tigrina and G. maculata are hard to distinguished for unexperienced people, which is even more evident with black and white pictures that most camera trap models take during the night.
- Determine genetic health and diversity of both rural and urban populations.
- Testing of rabies in Cape Genets and their potential role as vectors of the disease.
Encouraged citizen actions:
- Report sightings of any genet species on virtual museum/social platforms (for example, iNaturalist and MammalMAP), especially outside protected areas, as well as to Emmanuel Do Linh San (emmanuel.dolinhsan@gmail.com). GPS locations and photographs would be of great assistance.
- In addition, report Cape Genet sightings within KwaZulu-Natal to Craig Widdows (Craigw@hotmail.co.za).
- Put mammal and bird friendly devices (bells, sonic bleepers or “pounce protectors”) on pets, particularly dogs.
Bibliography
Carvalho, F., Ziko, B.A., Mrubata, Z., Xhobani, M., Plaatjie, A., Do Linh San, E. 2024a. Multi-scale habitat modelling of latrine site selection by sympatric carnivores in an Albany Thicket landscape, South Africa. Landscape Ecology, 39:1-9.
Carvalho, F., Galantinho, A., Sommers, M.J., Do Linh San, E. 2024b. Influence of season, sex, and interspecific interactions on the diel activity patterns of two sympatric African small carnivores. Scientific Reports, 14:29701.
Coetzee, C.G. 1977. Order Carnivora. Part 8. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-42. Smithsonian Institution Press, Washington, DC, USA.
Crawford-Cabral, J. 1980. The classification of the genets (Carnivora, Viverridae, genus Genetta). Boletim da Sociedade Portuguesa de Ciências Naturais 20: 97-114.
Crawford-Cabral, J. and Pacheco, A.P. 1992. Are the Large-spotted and the Rusty-spotted genets separate species (Carnivora, Viverridae, genus Genetta)? Garcia de Orta, Série de Zoologia, Lisboa 16: 7-17.
Cunningham, A.B. and Zondi, A.S. 1991. Use of animal parts for the commercial trade in traditional medicines. Institute of Natural Resources, University of Natal, Pietermaritzburg, South Africa.
Ehlers Smith, Y.C., Ehlers Smith, D.A., Ramesh, T. et al. Forest habitats in a mixed urban-agriculture mosaic landscape: patterns of mammal occupancy. Landscape Ecol 33, 59–76 (2018). https://doi.org/10.1007/s10980-017-0580-1
Ehlers Smith, Y.C., Ehlers Smith, D.A., Ramesh, T. et al. The importance of microhabitat structure in maintaining forest mammal diversity in a mixed land-use mosaic. Biodivers Conserv 26, 2361–2382 (2017). https://doi.org/10.1007/s10531-017-1360-6
Ferguson, A.W., Roble, H.R. and McDonough, M.M. 2019. Noteworthy record of the Ethiopian genet, Genetta abyssinica,(Carnivora, Viverridae) from Djibouti informs its phylogenetic position within Genetta. Mammalia, 83(2), pp.180-189.
Gaubert, P. 2003. Description of a new species of genet (Carnivora; Viverridae; genus Genetta) and taxonomic revision of forest forms related to the Large-spotted Genet complex. Mammalia 67: 85-108.
Gaubert, P. 2003a. Systématique et phylogénie du genre Genetta et des énigmatiques “genet-like taxa” Prionodon, Poiana et Osbornictis (Carnivora, Viverridae): caractérisation de la sous-famille des Viverrinae et étude des patrons de diversification au sein du continent africain. Ph.D. Thesis, Muséum National d’Histoire Naturelle.
Gaubert, P. 2013. Genetta tigrina Cape Genet. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 247-249. Bloomsbury, London, UK.
Gaubert, P., Taylor, P.J. and Veron, G. 2005a. Integrative taxonomy and phylogenetic systematics of the genets (Carnivora, Viverridae, genus Genetta): a new classification of the most speciose carnivoran genus in Africa. In: B.A. Huber, B.J. Sinclair and K.-H. Lampe (eds), African Biodiversity: Molecules, Organisms, Ecosystems, pp. 371-383. Springer, New York, USA.
Gaubert, P., Taylor, P.J., Fernandes, C.A., Bruford, M.W. & Veron, G. 2005b. Patterns of cryptic hybridization revealed using an integrative approach: a case study on genets (Carnivora, Viverridae, Genetta spp.) from the southern African subregion.
IUCN. 2015. The IUCN Red List of Threatened Species. Version 2015-4. Available at: www.iucnredlist.org. (Accessed: 19 November 2015).
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Plaatjie, A., Carvalho, F., Marikhele, R., Madikiza, Z.J.K. Do Linh San, E. 2024. Cape genet (Genetta tigrina) diet in the Albany Thicket biome, South Africa. African Zoology, 59:111–117.
Schlawe, L. 1981. Material, Fundorte, Text- und Bildquellen als Grundlagen für eine Artenliste zur Revision der Gattung Genetta G. Cuvier, 1816. Zoologische Abhandlungen (Dresden) 37: 85-182.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Stuart, C.T. 1990. The conservation status of mustelids and viverrids in Southern Africa. Small Carnivore Conservation 3: 16.
Widdows, C., Roberts, P.D., Maddock, A.H., Carvalho, F., Gaubert, P., Do Linh San, E. 2016. A conservation assessment of Genetta genetta. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Wozencraft, W.C. 2005. Order Carnivora. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World: A Taxonomic and Geographic Reference. Third Edition, pp. 532-628. Johns Hopkins University Press, Baltimore.