Cape Fur Seal
Arctocephalus pusillus pusillus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Arctocephalus pusillus pusillus– (Schreber, 1775)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – OTARIIDAE – Arctocephalus – pusillus- pusillus
Common Names: Cape Fur Seal, Afro-Australian Fur Seal, Brown Fur Seal, South African Fur Seal (English), Kaapsepelsrob (Afrikaans), Lenyedi, Tau ya lewatle (Sepedi), Sili Ya Vhukuse (Venda), Inja Yolwandle Yesakapa (Xhosa), Imvu Yamanzi (Zulu)
Synonyms: Phoca pusilla Schreber, 1775
Taxonomic Note:
The Cape Fur Seal is congeneric with the Australian Fur Seal A. p. doriferus. The two subspecies are almost identical in both anatomy and behaviour (Warneke & Shaughnessy 1985, Kirkman & Arnould 2018). Repenning et al. (1971) separated them based on a single cranial character and separate geographic ranges. Very low genetic divergence indicates that they split relatively recently, with the Australian subspecies being established some 13 000 years ago (Lento et al. 1997; Wynen et al. 2001, Malan et al. 2022).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Seakamela, S.M.1 & Hofmeyr, G.J.G.2
Reviewer: Smith, C.3
Contributor: Nghimwatya, L. & Patel, T.
Institutions: 1Department of Forestry, Fisheries and the Environment, 2Port Elizabeth Museum at Bayworld, 3Endangered Wildlife Trust
Previous Assessors & Reviewers: Hofmeyr, G.J.G., Kirkman, S.P., Seakamela, S.M. & Pistorius, P.A.
Previous Contributors: Meyer, M., Oosthuizen, H., Lowry, L., Page-Nicholson, S. & Child, M.F.
Assessment Rationale
Due to its large population size, lack of major threats, and recently documented range expansions, the Cape Fur Seal of South Africa should remain classified as Least Concern. However, the subspecies is subject to legal harvesting across the order in Namibia, fisheries bycatch, deliberate killing at sea, possible illegal harvesting and the effects of novel diseases. Included in the latter is rabies, which was been recorded in fur seals for the first time in May 2024 (van Helden 2024). Of further concern is High Pathogenicity Avian Influenza (HPAI) which has resulted in elevated mortalities of sympatric species of seabirds (Abolnik et al. 2024). In the future, climate change may also pose threats to the species (Kovacs et al. 2012). It is therefore important that demographic changes, interactions with fisheries and possible infections are monitored.
Regional population effects:
Breeding colonies occur at numerous island and mainland sites along the east, south and west coasts of South Africa and the population is contiguous with the population in Namibia (Oosthuizen 1991; Kirkman et al. 2013, Makhado et al. 2024).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Seakamela SM & Hofmeyr GJG. 2025. A conservation assessment of Arctocephalus pusillus pusillus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
It is thought that historically the Cape Fur Seal population occurred on most, if not all, the coastal islands offshore of South Africa and Namibia. However, uncontrolled harvesting and habitat modification resulted in shifts in the distribution of the breeding population, with the bulk currently breeding at mainland sites while colonies at many former breeding islands remain extinct (Kirkman et al. 2007). Since the 1970s a range expansion has been recorded, with an increase in the number of breeding colonies from 23 to 40 (Kirkman et al. 2013). While the southeastern-most extent of the breeding range remains Algoa Bay in South Africa, the northwestern-most limit extended by 2009 from northern Namibia to a single Angolan colony at Ilha dos Tigres. Cape fur seals also occur at several non-breeding colonies or temporary haul-out sites throughout their range.
Within South Africa, the range of the breeding population is between Black Rocks in Algoa Bay, and Buchu Twins, south of the Orange River mouth. New breeding locations have been established within this range in recent years such as at Mitchell’s Bay, Cape Point and Cape Infanta (Seakamela 2025). In addition, some locations of historical breeding colonies such as Vondeling Island (Kirkman et al. 2013, Seakamela 2024) and Robberg Peninsula in Plettenberg Bay (Huisamen et al. 2012) have been re-colonised. While colonies were traditionally defined as sites with a consistent annual pup production of at least 100 pups Oosthuizen & David 1988) breeding took place at other sites. As of 2020, breeding took place at a total of 28 sites in South Africa, including those not meeting the traditional definition (Seakamela et al. 2024)
Cape Fur Seals are generally not migratory. However, small numbers follow the Sardine Run eastwards during winter (O’Donoghue et al. 2010). While this species breeds seasonally, haul-outs are occupied year-round (David 1987a). Feeding is generally restricted to the continental shelf area (David 1987b). Vagrants have therefore only been recorded twice, in Gabon (Thibault 1999) and on the Prince Edward Islands (Kerley 1983).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
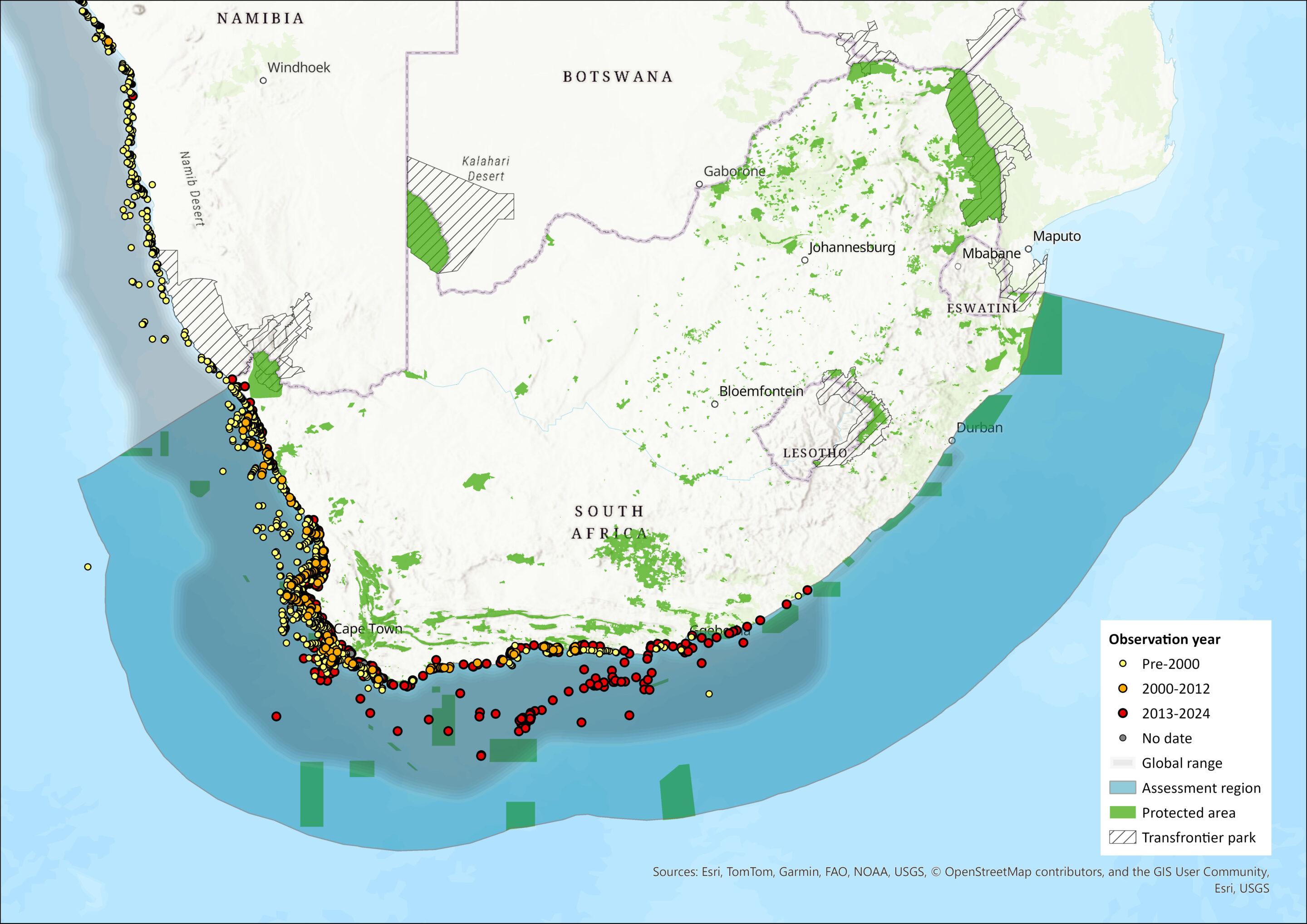
Map
Figure 1. Distribution records for Cape Fur Seal (Arctocephalus pusillus pusillus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence within southern Africa
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Australia | Extant | Native | – | – |
| Australia -> New South Wales | Extant | Native | – | – |
| Australia -> South Australia | Extant | Native | – | – |
| Australia -> Tasmania | Extant | Native | – | – |
| Australia -> Victoria | Extant | Native | – | – |
| Gabon | Extant | Vagrant | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Africa -> Northern Cape Province | Extant | Native | ||
| South Africa -> Eastern Cape Province | Extant | Native | – | – |
| South Africa -> Marion-Prince Edward Is. | Extant | Vagrant | – | – |
| South Africa -> Western Cape | Extant | Native | – | – |
| South Africa -> KwaZulu Natal Province | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: The breeding range of the Cape Fur Seal is primarily associated with the cold, nutrient-rich Benguela Large Marine Ecosystem (BCLME) in the southeast Atlantic Ocean, with less than two percent of the population inhabiting the warm, nutrient-poor Agulhas Current Large Marine Ecosystem (ACLME) in the southwest Indian Ocean (Kirkman et al. 2016). Productivity in the BCLME is largely the result of seven known wind-driven upwelling cells (Santana-Casiano et al. 2009). In contrast, productivity in the ACLME’s is driven by two persistent upwelling cells only (Lutjeharms 2006).
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 57. Indian Ocean – southwest | Extant | Native | – | – |
Climate change
Pinnipeds generally inhabit temperate or polar habitats, which are expected to be substantially impacted by a warming climate (Laidre et al. 2008, Simmonds & Isaac 2007, Kovacs et al. 2012, Gulland et al. 2022). This may have a number of impacts, including potentially influencing the availability of prey species. Some 98 % of the Cape Fur Seal population inhabit the Benguela Current system (Kirkman et al. 2013). In common with other eastern boundary systems, it is highly productive and supports a high biomass of top predators (Bakun et al. 2015). However, the system is not immune from the effects of anthropogenic climate change and has experienced increased an intensity in upwelling with concomitant increases in sea surface temperature offshore and decreases inshore (Santos et al. 2012). However, changes are not uniform, with local characteristics and anthropogenic influences influencing regional changes (Blamey et al. 2015). This has had an impact on the abundance of various fish species, including prey of seals, with populations declining or shifting their distribution southwards (van der Lingen 2006, Blamey et al. 2015). It is possible that these changes are partially responsible for the recent changes in the distribution of the Cape fur seal, with declines in southern Namibian and northern South African colonies, and increases elsewhere (Kirkman et al. 2013, Seakamela et al. 2024). Species that range widely and are generalist feeders are thought to be more resilient to climate change (Laidre et al. 2008, Kovacs et al. 2012). Cape fur seals share these characteristics (De Bruyn et al. 2003, Mecenero et al. 2006, Skern-Mauritzen et al. 2009, Connan et al. 2014, Botha et al. 2020, Botha et al. 2023). They also show an inclination to relocate to other breeding colonies when under stress (Kirkman et al. 2013). However, they are not immune to the effects of climatic aberrations, as shown by the mortality event in 1994 (Roux 1998). Genetic analysis suggests that Cape fur seals have been impacted by past climatic changes (Mathee et al. 2006, Malan et al. 2022).
Climate change may also be responsible for the direct mortality of vulnerable age classes in pinnipeds (Kovacs et al. 2012, Gulland et al. 2022). Cape Fur Seals pups are at risk on hot, windstill days when they suffer higher mortality (De Villiers & Roux 1992, Roux 1998). Therefore, an increase in temperatures ashore during the breeding season may limit the range of Cape Fur Seals in the future (Kovacs et al. 2012). Further, climate change may result in increased susceptibility to novel diseases (Harvell et al. 2002, Burek et al. 2008).
Population information
The size of the foraging population of Cape Fur Seals (individuals older than one year) in the early 1990’s was estimated at about 1.7 million individuals with some 75 % of pup production at the three major colonies at Cape Cross, the Wolf Bay-Atlas Bay-Long Island complex and Kleinzee (Butterworth et al. 1995, Kirkman et al. 2013). However, there have been substantial changes in distribution since then, including range expansion. Despite this, overall abundance appears to have remained relatively stable (Kirkman et al. 2007, Kirkman et al. 2013, Seakamela et al. 2024a). Concomitant with the establishment of new breeding colonies, especially in northern Namibia and southern Angola, there has been a decline in other colonies, especially the major colonies in the southern Namibia (Kirkman et al. 2013). Currently, approximately 40 breeding colonies are found throughout the range of the species (Kirkman et al. 2013). Pup counts from the 2017/2018 breeding season indicated that some 65% of pup production (298,772) took place in Namibia (Mwaala et al. 2024) and most of the remainder in South Africa (104,332) (Seakamela et al. 2024a).
Current abundance of the foraging population in South Africa is suggested to be 652,075 individuals based on pup production (Seakamela et al. 2024a). Numbers have not increased since the 1990’s (Butterworth et al. 1995, Kirkman et al. 2013, Seakamela et al. 2024a). Although new colonies have been established on the South African coast, with breeding now taking place at 28 sites, this increase is offset by a substantial reduction in pup production at the largest mainland colony, at Kleinsee (Seakamela et al. 2024a). Although this colony has declined by about 40 % from its peak abundance, it is still responsible for most pup production in South Africa (Kirkman et al. 2013, Seakamela et al. 2024a). The second largest colony is on Vondeling Island, despite it only being recolonised in the early 2000s.
Cape Fur Seals have experienced major mortality events in recent years. In 1994 poor environmental conditions resulted in poor pup recruitment and the death of adults in southern Namibia (Roux 1998). This was followed, in 2021 by a die-off of pups from colonies on the west coast of South Africa (Sgqolana 2021a, b, Bega 2021). However, these events seem to have had a negligible impact on the population numbers (Seakamela et al. 2024).
Generation length has been calculated at 9.1 years (Pacifici et al. 2013). Population change over three generations from 1972–2020 has been positive (Unpublished Data, DFFE).
Population Information
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: No
Current population trend: Stable
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: None
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: None
Probability of extinction in the wild within 100 years: None
Population genetics
Uncontrolled harvesting from the 17th century reduced Cape Fur Seal abundance to less than 100,000 individuals by the beginning of the 20th century (Shaughnessy and Butterworth 1981) possibly resulting in a demographic bottleneck. However, the population had recovered to a foraging population of more than 1,700,000 individuals by the early 1990s (Butterworth et al. 1995). Mitochondrial haplotype diversity of Cape Fur Seals remaining reasonably high (Matthee et al. 2006, Robbertse et al. 2025). However, nucleotide diversity is low. It is suggested that this was due to an ancient demographic bottleneck (Matthee et al. 2006). This was likely followed by expansion during the Last Glacial Maximum, starting some 18,000 years ago (Matthee et al. 2006, Malan et al. 2022). This resulted in the migration of individuals to Australia, founding what would become the congeneric Australian fur seal (Lento et al. 1997, Malan et al. 2022). Genetic analysis shows that contact between the two subspecies ceased some 13,000 years ago (Malan et al. 2022).
Cape Fur Seals are dispersed along some 3,000 km of coastline. Despite this, both mitochondrial and nuclear DNA analysis do not support sub-structuring (Matthee et al. 2006, Robbertse et al. 2025). This is suggested to be partly attributable to relatively low female philopatry (Oosthuizen 1991, Matthee et al. 2006). While the Lüderitz upwelling cell is considered a barrier between marine organisms occurring in the northern and southern Benguela systems (Lett et al. 2024), this has not hindered the movement of Cape Fur Seals (Skern-Mauritzen et al. 2009).
The effective population size (Nc) of Cape Fur Seals is estimated to lie between 65,208 and 195,623.
Habitats and ecology
The Cape Fur Seal is one of the largest of all the fur seals. It is also highly sexually dimorphic. Adult males are 2–2.3 m long and average 247 kg in weight. Adult females are 1.2–1.6 m long and weigh an average of 57 kg. At birth they weigh around 6 kg (Shaughnessy 1979). Females become sexually mature at 3–6 years. While males reach sexual maturity at the same age, they are only socially mature at 9–12 years (Wickens & York 1997). The annual pregnancy rate of mature females has been estimated at 71% (Wickens & York 1997). Gestation is known to last 51 weeks, including a three-month delay of implantation but longevity and adult mortality are unknown (Reijnders et al. 1993; Butterworth et al. 1995; Wickens & York 1997).
Cape Fur Seals are highly polygynous with breeding males defending territories where multiple females gather to pup. Breeding occurs from late October to the beginning of January, with adult males arriving at the colonies first. Females give birth 1.5–2 days after their arrival. The peak of pupping is in the first week of December, although there is some variation between colonies (David 1987a). Mothers attend to their pup for about 6-7 days before coming into oestrous, mating, and then departing on their first foraging trip (Rand 1955). From then until weaning at 10-12 months of age, the mothers alternate between regular foraging trips to sea and shore visits to nourish their pups with milk (David 1987a). The Cape Fur Seal is positively thigmotactic, a trait that they have in common with sea lions but not with other fur seals.
Cape Fur Seals spend the majority of their lives at sea, typically over the continental shelf and within 80 kilometres of the shore (David 1987). While they also inhabit the relatively warm waters of the Agulhas Current Large Marine Ecosystems (ACLME), some 98 % are found in the waters of the cooler and more productive Benguela Current Large Marine Ecosystem (BCLME). Cape Fur Seals are generalist foragers that take a wide variety of pelagic, demersal and benthic prey, including Cape Hake (Merluccius spp.), Horse Mackerel (Trachurus spp.), Pelagic Goby (Sufflogobius bibarbatus), Pilchards (Sardinops sp.), Anchovy (Engraulis capensis), Squid ( Loligo sp.), Rock Lobster (Jasus lalandi), shrimp, prawns, and amphipods. However, they prefer Pelagic Teleost Fish (David 1987a; de Bruyn et al. 2003; Mecenero et al. 2006; Huisamen et al. 2012, Connan et al. 2014). Although their habitat has been subject to ecosystem changes, their prey preferences have not altered, only the ratios consumed (Gumede 2023, Mwaala et al. 2024). Cape Fur Seals feed on a similar diet throughout their range, but with greater proportion of benthic fish in the ACLME in some years (Castley et al. 1991, Stewardson 2001, Huisamen et al. 2012, Connan et al. 2014). Predation on seabirds has been reported, including species of conservation concern such as Cape Gannets (Morus capensis) (Makhado et al. 2006), African Penguins (Spheniscus demersus) (Johnson et al. 2006), and Cape Cormorants (Phalocrocorax capensis) (Marks et al. 1997). Recently, Cape Fur Seals have also been recorded to kill Kelp Gulls (Larus dominicanus) (Gumede 2023).
Cape Fur Seals haul out to breed on land and their pups are largely restricted to terrestrial habitat (David 1987). Historically, they bred on offshore islands. However, colonies were established in isolated mainland areas in the 20th century, possibly due to disturbance caused by harvesting at adjacent islands (Rand 1972, Shaughnessy and Butterworth 1981). Mainland sites have become the largest colonies, perhaps because space is less restricted (David 1987). In recent years, with the redistribution of prey species, colonies have been established at sub-optimal localities such as on low-lying rocks (for example, Grotto Bay) which are exposed to strong surf and below steep cliffs (for example, Cape Point, Robberg Peninsula) both of which are unlikely to result in high pup mortality.
Ecosystem and cultural services: The Cape Fur Seal is the only pinniped regularly breeding on the South Africa coast. As a top predator they most likely have a critical role in prey regulation and the structure and functioning of the ecosystem. While the nature of this role is not completely known, due the complexity of the marine food web, modelling studies have provided some insight in this regard. Although Cape Fur Seals are frequently suggested to be significant competitors with commercial fisheries, modelling has suggested that predation by Cape Fur Seals on predatory fish (for example, hake) could possibly result in greater biomass of target fish available for fisheries, with potential for a net negative impact on the fishery if seal numbers were reduced (Punt & Butterworth 1995).
Pinnipeds are potential indicators of changes in ecosystem health or abundance of marine resources, especially because they are partially terrestrial and therefore more accessible to researchers (Sanders et al. 2025). Changes in numbers, distribution or other characteristics of marine higher predators, such as the Cape Fur Seal, could be symptomatic of changes occurring at lower trophic levels, such as may be caused by effects of overfishing or other environmental (e.g. climatic) changes (Kirkman et al. 2011).
The Cape Fur Seal also serves as prey for other predators at sea, including the Great White Shark (Carcharodon carcharius) (Martin et al. 2005) and Killer Whales (Orcinus orca) (Rand 1955). Ashore, their pups are preyed upon by Black-backed Jackals (Lupullela mesomelas), African Lions (Panthera leo) and Brown Hyaenas (Hyaena brunnea) (Oosthuizen et al. 1997; Wiesel 2010, Stander 2019).
The attractiveness of seal colonies on the southwest coast to Great White Sharks, and associated shark-seal predatory interactions, is important for the commercial success of the Great White Shark viewing ecotourism industry. In addition, Cape Fur Seal colonies provide a reliable attraction (unlike more elusive taxa targeted for tourist viewing such as some whales, dolphins and sharks) and are part of the viewing experience on offer by several ecotourism ventures. Some seal colonies on the mainland can also be viewed by land-based tourists, usually as part of a broader package (Kirkwood et al. 2003). Examples are at the Kleinsee colony which is situated in a mining lease and is one of the viewing experiences on offer during mine tours, and the Bird Island Reserve in Lambert’s Bay, where a seal colony is visible from a tourist hide beside the gantry.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
| 12.1. Marine Intertidal -> Marine Intertidal – Rocky Shoreline | – | Suitable | Yes |
| 13.1. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Sea Cliffs and Rocky Offshore Islands | – | Suitable | Yes |
Life History
Generation Length: 9
Age at Maturity: Female or unspecified: 4-5
Age at Maturity: Male: 3-4 (puberty), 9-10 (social maturity)
Size at Maturity (in cms): Female: 147
Size at Maturity (in cms): Male: 199
Longevity: 31 (based on specimens in captivity)
Average Reproductive Age: 9.4
Maximum Size (in cms): 241
Size at Birth (in cms): 70-80 cm
Gestation Time: ~8 months (from implantation)
Reproductive Periodicity: Annual
Average Annual Fecundity or Litter Size: 1
Natural Mortality: 0.2-0.3 (old estimate-needs revision)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Non-migratory, feeding mostly over the continental shelf
Congregatory: Yes, highly gregarious on land. Can be gregarious or solitary at sea.
Systems
System: Terrestrial (breeding), Marine (foraging)
General Use and Trade Information
Cape Fur Seals were hunted heavily from the 17th to the 19th centuries, and abundance was reduced to low levels but have recovered under protective legislation first introduced in 1893 (Butterworth et al. 1995). Commercial harvesting took place in South Africa until this was suspended in 1990 (Hanks 1990; Wickens et al. 1991). The killing of seals for profit in South Africa is now prohibited in terms of the Marine Living Resources Act (MLRA 1998). However, Cape Fur Seals are still harvested commercially in Namibia and are also subject to trophy hunting (Campbell et al. 2011; Japp et al. 2012). Products of commercial seal harvesting include fur products from pelts, leather products from skin, the male genitalia for traditional aphrodisiacs, oil for medicinal uses and meat, bone meal and oil as additives to animal fodder, as well as the subsistence consumption of meat (Campbell et al. 2011). However, import/export data from CITES indicated that there has been a drastic drop in export of Cape Fur Seal products from Namibia (CITES 2024). Since the last assessment, an annual Total Annual Catch (TAC) of 60 000 was set for pups and 8 000 for bulls. This was reduced to 6 000 for bulls in 2023. The actual number of pups taken dropped from 24% of the TAC in 2016 to 4% of the TAC in 2023. For bulls, the harvest ranged between 53% and 91% of the TAC, with a marked drop in 2022 (62%) and 2023 (58%).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | – | – | – |
National Commercial Value: Yes (non-consumptive)
International Commercial Value: Yes (non-consumptive)
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: The take in Namibia has declined.
Threats
Cape Fur Seals face both direct threats, such as mortality from fisheries bycatch, entanglement in debris, and illegal killing, and various indirect threats such as trophic interactions with fisheries, shifts in prey abundance and distribution, climate change impacts, contamination by oil and other pollutants and novel diseases. Although good data are missing, direct threats are thought to kill relatively few animals. However, these mortalities could still result in localised demographic impacts (Shaughnessy 1980; Wickens et al. 1992). These impacts come primarily from the fisheries industry. The numbers of seals killed illegally by fishermen, especially where seals interfere with fishing operations (e.g. line or long-line fisheries) are unknown but are potentially higher than bycatch- or entanglement-related mortality levels (Wickens et al. 1992; David & Wickens 2003). There is however improved awareness of the illegality of the practice. There are also attempts to improve the reporting of animals killed at sea or those washing up on beaches with evidence of having been killed (Sanders et al. 2025). Bycatch in fisheries operations remains a challenge. While the quantity of seals caught in fisheries operations is not known, fisheries are mandated through permit conditions to report incidents. Although bycatch numbers have been estimated to be low (< 1% of population size), they could still result in localised demographic impacts (Shaughnessy 1980; Wickens et al. 1992), Entanglement incidents are primarily in debris originating in the fisheries industry (Curtis et al. 2021, Henry 2022, Forsberg 2023). Although there are disentanglement efforts in some parts of the species’ range, many entanglements likely lead to the death of affected individuals.
Indirect threats potentially pose a greater threat than direct impacts. Cape Fur Seals prey on several species that are also targeted by commercial or recreational fisheries (David 1987b; Wickens et al. 1992). The effects of these trophic interactions with fisheries on the seal population are difficult to quantify given the complexities of the marine food web and the fact that seals also prey on a range of other species that are not targeted by fisheries (David 1987b). However, these interactions are of concern considering increasing trends in fishing effort and declining catch rates (for example, Mann 2013). This may be exacerbated by shifts in prey distribution and abundance associated with ecosystem changes (e.g. related to climate change). As an example, there has been a recent south and eastward shift in the epicentre of anchovy and sardine biomass along the South African coast (Roy et al. 2007; Coetzee et al. 2008) which would have negatively impacted on availability of these prey items to some colonies.
Indirect impacts have also resulted in the greatest recorded mortalities of Cape Fur Seals. The so-called Benguela Nino event in 1994/1995, resulted from unfavourable environmental conditions with detrimental effects on prey populations and therefore feeding conditions. This led to the deaths of almost an entire cohort of pups and many adults in part of the range (Roux 1998). This is the largest mass die off recorded for any seal species (Harwood 2002). A lower magnitude die-off occurred over a wider area (Namibia and South Africa) between 2020 and 2021, killing over 10,000 individuals, mostly pups. In South Africa, laboratory test from 8 carcasses from the 2021 mass die-off indicated protein energy malnutrition as a cause of death (Seakamela et al. 2021). The uncertainty brought by climate change may result in further such events. Such events would also likely be exacerbated by fishing pressure, just as the 1994/1995 event in Namibia is believed to have been intensified by high levels of fishing at the time (Boyer & Hampton 2001). In recent years relatively high numbers of dead or malnourished stranded Cape Fur Seals have been observed on the west coast of South Africa, where fish stocks are known to be depleted (Department of Environmental Affairs unpubl. data). Acting in unison with climate change and fishing pressures are disease outbreaks.
Several morbillivirus epidemics have occurred in true seal (Phocid) populations in the northern hemisphere, and it is thought that the infections were transmitted to the seals by domestic dogs or terrestrial carnivores (Kennedy et al. 2000). Although no epidemic diseases are known to have infected Cape Fur Seals historically, there are now emerging health concerns in the population. In 2024, positive cases of rabies virus that is genetically similar to a canine strain found in Black-backed Jackals were detected in the Western Cape (Van Helden 2024, Department of Agriculture 2024). The virus is now endemic in the population, suggesting that it has been in the population for some time prior to detection. High Pathogenicity Avian Influenza (HPAI) is a potential future concern. This disease has resulted in the mortalities of numbers of sympatric species of seabirds (Abolnik et al. 2024). While impact on Cape Fur Seals have not yet been recorded, HPAI has resulted in mass mortalities of pinnipeds elsewhere (Alava et al. 2024, Campagna et al. 2024).
Other potential threats of climatic changes include predicted sea level rises and increased frequencies of extreme weather events (e.g. storms). These may threaten the viability of seal colonies at small, low-lying islands such as Black Rocks in Algoa Bay which are already vulnerable to catastrophic events (Stewardson 1999, Hofmeyr et al. 2011) and could result in localised depletions. A further threat is increased mortality, especially of young pups, associated with increased air temperatures, a decrease in cooling winds or an increase in the number of hot days per year. This threat may be most relevant to mainland colonies (e.g. de Villiers & Roux 1992).
Seal-targeted tourism has increased in popularity in recent years. Tourism activities include boat-based viewing, swim-with (snorkelling and scuba diving) and illegal feeding for selfies. Boat-based activities have been shown to elicit behavioural response from seals relatively more than other activities (Cate 2013). Two sites in South Africa have established dive-with or snorkelling operations, one in Plettenberg Bay and another in Hout Bay. In Hout Bay, the impacts of this industry were found to be minimal and are not expected to be adverse at the colony or population level (Heidi 2020). In Plettenberg Bay, presence of the boat and swimmers did elicit short-term behavioural responses such a decreased state of restfulness (Basson 2024). Illegal seal feeding tourism has also gained notoriety. This activity has been argued to have potentially negative impacts at the individual and population level. Some of the impacts include dependency on humans, habituation, aggression when food is unavailable, potential disease transfers and alteration to natural behavioural patterns (Orams 2002). This activity is mostly limited to Hout Bay and Kalk Bay (both in Cape Town); at the former, cases of aggression and bites to tourists and feeders have been reported. Furthermore, some of these animals are considered obese and have been diagnosed with blindness and other comorbidities.
The growing focus on oceans economy could introduce further challenges and may exacerbate existing ones. Emerging changes in land-use patterns may impact the breeding habitat of the species. The growth of oceans economy may result in more residential and commercial property developments and operations closer to breeding colonies. This may cause breeding females to desert their preferred habitat for less optimal habitats. In the assessment region, Marine Spatial Planning programmes have been introduced to better manage potential threats of a growing oceans economy. Further out to sea, foraging grounds will be more susceptible to oil spills due to the increase in ship traffic and prevailing adverse weather which is known to cause marine incidents. Like other fur seals, Cape Fur Seals are vulnerable to oil spills which can reduce the insulation properties of their fur, be ingested in toxic concentrations or affect populations of prey (Kirkwood & Goldsworthy 2013).
Conservation
Although Cape Fur Seals have been protected in South Africa since 1893, they were still subject to government-run or government-authorised commercial harvests until 1990 (Wickens et al. 1991; Butterworth et al. 1995). Various legislations have been passed over time to meet the changing management requirement. The Seabirds and Seals Protection Act (SBSPA; Act no. 46 of 1973), regulated sealing methods and provided broad protection for seals in South Africa and the then South West Africa. The Commercial killing of seals for profit are now prohibited in terms of the Marine Living Resources Act (Act no. 18 of 1998; Policy on the management of seals, seabirds and shorebirds (Government Gazette No. 30534, 2007). To better manage human interactions with seals (for example. culling, feeding, captivity), the Threatened or Protected Species Regulations (Government Gazzette No. 40876 of 2017) were gazetted in terms of the National Environmental Management: Biodiversity Act (10/2004), also known as NEM:BA. Concurrently and in terms of NEM:BA, A number of seal colonies occur within or adjacent to marine protected areas (MPAs) or reserves. These include:
- Bird Island, Lambert’s Bay (Bird Island Reserve, CapeNature).
- Black Rocks (Bird Island MPA, SANParks) Cape Point (Table Mountain National Park MPA, SANParks)
- Duikerklip (Table Mountain National Park MPA, SANParks)
- Quoin Rock and Geyser Rock (Dyer Island Nature Reserve Complex, CapeNature)
- Robberg Peninsula (Robberg Nature Reserve and Marine Protected Area, CapeNature)
- Sammy’s Bay (Namaqua National Park, SANParks)
- Strandfontein Point (Namaqua National Park, SANParks)
- Vondeling Island (Vondeling Island Reserve, CapeNature)
Besides Vondeling Island, seals also haul out on other islands adjacent to the West Coast National Park around which MPAs have been declared (e.g. Jutten Island, SANParks). Three mainland breeding colonies in the Northern Cape including the South Africa’s largest Cape Fur Seal breeding colony near Kleinsee, occur in mining lease areas where human access is restricted, affording these colonies a level of protection. Cape Fur Seals would further benefit from more stringent regulation of reporting of bycatch and improvement of fishing gear design. An example of the impact of such interventions are the mitigation measures implemented in New Zealand’s hoki trawl fishery, which reduced Hooker’s Sea Lion (Phocarctos hookeri) bycatch by almost 90%. The population has since showed signs of recovery (Porritt & Goodman 2005). Ongoing monitoring and research within the assessment region should be maintained.
Recommendations for land managers and practitioners:
- Continuation of regular population censuses (aerial pup surveys).
- Monitor changes in key demographic parameters, especially those that may be related to climatic or other changes.
- Regional (transboundary) cooperation with regard to research and monitoring, including standardisation of research methods.
- Regulation of seal ecotourism activities.
- Ensuring protection of mainland seal colonies situated in current mining lease areas where human access has been restricted, in the event that these areas become accessible to the public.
- Preventing commercial operations (including mining and development) from encroaching into seal breeding territories.
- Prevention of direct mortality due to fisheries operations.
- Initiate national disease surveillance and monitoring initiatives.
Research priorities:
The following research topics are currently being investigated:
- Demography, diet, foraging behaviour and fisheries interactions of Cape Fur Seals in Algoa Bay (Port Elizabeth Museum, Nelson Mandela University and Department of Forestry, Fisheries and the Environment, DFFE).
- Seal-seabirds interactions at Vondeling Island (DFFE/CapeNature, ongoing).
- Diet of the Cape Fur Seal at key colonies on the West and South Coasts (DFFE, long-term monitoring effort).
- Cape Fur Seal pup trends from comprehensive aerial surveys (DFFE, long term monitoring effort).
- Identifying and determining the size of home ranges and core feeding areas for Cape Fur Seals (DFFE).
- Monitoring of marine debris entangling the Cape Fur Seal (DFFE/Two Oceans Aquarium-A long-term monitoring effort).
- Assessing the effects of “swim-with-seal” tourism activities on a seal colony (Nelson Mandela University/DFE).
- Cape Fur Seal health and disease surveillance (SeaSearch, DFFE, Stellenbosch University, Port Elizabeth Museum and others)
- Investigation of population parameters required for extending pup counts to total population size (including e.g. mark-recapture, pup growth, initial pup survival at Kleinsee breeding colony (DFFE).
The following research topics should be investigated:
- Updated assessments of extent of trophic interactions between seals and fishing, including development of population and bioenergetics models to determine consumption rates, and spatial models of foraging effort distribution.
- Updated assessments of operational interactions between seals and fisheries to provide estimates of effects on the population and revised recommendations for mitigation.
- Assessment of effects of seal ecotourism activities on seal colonies.
- Assessments of effects of variability in the environment and prey availability on the population, through long-term monitoring of key parameters or indices, e.g. diet, foraging trip duration, foraging distributions, breeding phenology, abortion rates, pup birth mass and growth, early survival rates, at key locations.
Encouraged citizen actions:
- Citizens contribute to science and conservation management by reporting stranded, sick, injured or entangled seals to appropriate authorities. There has been an increase in reporting on virtual platforms, such as iNaturalist.
Bibliography
Abolnik, C., Roberts, L.C., Strydom, C., Snyman, A. And Roberts, D.G. 2024. Outbreaks of H5N1 High Pathogenicity Avian Influenza in South Africa were caused by two distinct sub-genotypes of clade 2.3.4.4b viruses. Viruses 16: 896.
Alava, J.J., Tirapé, A., Denkinger, J., Calle, P, Rosero, P., Salazar, S., Fair, P.A. and Raverty, S. 2024. Endangered Galápagos sea lions and fur seals under the seige of lethal avian flu: a cautionary note on emerging infectious viruses in endemic pinnipeds of the Galápagos Islands. Frontiers in Veterinary Science 11: 1457035
Bakun, A., Black, B.A., Bograd, S.J., Garcia-Reyes, M., Miller, A.J., Rykaczewski, R.R. and Sydeman, W.J., 2015. Anticipated effects of climate change on coastal upwelling ecosystems. Current Climate Change Reports, 1, pp.85-93.
Basson, R. 2024. Behavioural responses of Cape fur seals (Arctocephalus pusillus) to swim-with-seal tourism activities in the Robberg Marine Protected Area in South Africa. MSc dissertation. Cape Peninsula University of Technology, Cape Town. South Africa
Bega, S. 2021. Thousands of thin Cape fur seals die on West Coast beaches. Mail & Guardian – Environment. https://mg.co.za/environment/2021-11-01-thousands-of-thin-cape-fur-seals-die-on-west-coast-beaches/. Accessed 21 Jul 2023
Botha JA, Kirkman SP, Arnould JPY, Lombard AT, Hofmeyr GJG, Meÿer MA, Kotze PGH, Pistorius PA. 2020. Geographic variation in at-sea movements, habitat use and diving behaviour of female Cape fur seals. Marine Ecology Progress Series 649: 201–218.
Botha, JA, Truman, CN, Kirkman, SP, Arnould, JPY, Lombard, AT, Connan, MA, Hofmeyr, GJG, Seakamela, SM Pistorius, PA. 2023. Geographic, temporal, and individual-based differences in the trophic ecology of female Cape fur seals. Ecology and Evolution 13: e9790.
Boyer DC, Hampton I. 2001. An overview of the living marine resources of Namibia. South African Journal of Marine Science 23: 5–35.
Burek, KA, Gulland, FM and O’Hara, TM. 2008. Effects of climate change on Arctic marine mammal health. Ecological Applications, 18(sp2), pp. S126-S134.
Butterworth, D.S., Punt, A.E. and Wickens, P.A. 1995. The effects of future consumption by the Cape fur seal on catches and catch rates of the Cape hakes. 3. Modelling the dynamics of the Cape fur seal Arctocephalus pusillus pusillus. South African Journal of Marine Science 16: 161-183.
Campagna, C, Uhart, M, Falabella, V, Campagna, J, Zavattieri, V, Vanstreels, RE and Lewis, MN. 2024. Catastrophic mortality of southern elephant seals caused by H5N1 avian influenza. Marine Mammal Science, 40(1).
Campbell R, Knowles T, O’Connor S. 2011. The economics of seal hunting and seal watching in Namibia. A report for Humane Society International, World Society for the Protection of Animals, Bont Voor Dieren (NL) and Respect for Animals (UK). Economists at Large, Melbourne, Australia.
Castley, JG, Cockcroft, VG and Kerley, GIH. 1991. A note on the stomach contents of fur seals Arctocephalus pusillus pusillus beached on the south-east coast of South Africa. South African Journal of Marine Science, 11(1), pp.573-577.
Cate, JR. 2013. The effects of tourism on the behaviour of the New Zealand fur seal (Arctocephalus forsteri). PhD thesis, University of Canterbury, New Zealand.
CITES Trade Database. 2024. Compiled by UNEP-WCMC for the CITES Secretariat. Available at: trade.cites.org. Accessed 7/29/2024.
Coetzee, J.C., Van der Lingen, C.D., Hutchings, L. and Fairweather, T.P. 2008. Has the fishery contributed to a major shift in the distribution of South African sardine? ICES Journal of Marine Science: Journal du Conseil 65(9): 1676-1688.
Connan, M, Hofmeyr, GJG, Smale, MJ and Pistorius, PA. 2014. Trophic investigations of Cape fur seals at the easternmost extreme of their distribution. African Journal of Marine Science, 36(3), pp.331-344.
Curtis, S., Elwen, S.H., Dreyer, N. and Gridley, T. 2021. Entanglement of Cape fur seals (Arctocephalus pusillus pusillus) at colonies in central Namibia. Marine Pollution Bulletin, 171, p.112759.
David, J. and Wickens, P. 2003. Management of Cape fur seals and fisheries in South Africa. In: N.J. Gales, M.A. Hindell and R. Kirkwood (eds), Marine mammals: fisheries, tourism and management issues, pp. 116-135. CSIRO, Collingwood, Australia.
David, J.H.M. 1987a. Diet of the South African (Cape) fur seal (1974-1 985) and an assessment of competition with fisheries in southern Africa. South African Journal of Marine Science 5: 693-713.
David, J.H.M. 1987b. South African fur seal, Arctocephalus pusillus pusillus. In: J. P. Croxall and R. L. Gentry (eds), Status, biology, and ecology of fur seals, pp. 73-77. NOAA Techn. Rep. NMFS 51.
de Bruyn, P.J.N., Bester, M.N., Mecenero, S., Kirkman, S.P., Roux, J.-P. and Klages, N.T.W. 2003. Temporal variation of cephalopods in the diet of Cape fur seals in Namibia. South African Journal of Wildlife Research 33: 85-96.
de Villiers, D. J., and Roux, J.-P. 1992. Mortality of newborn pups of the South African fur seal Arctocephalus pusillus pusillus in Namibia. South African Journal of Marine Science 12: 881-889.
Department of Agriculture. 2024. Rabies in Cape fur seals: outbreak update report, 31 October 2024. https://www.nda.gov.za/images/outbreaks/Rabies/2024/rabies-in-seals_dalrrd-report_oct2024_final.pdf
Forsberg, S.T.W., 2023. Disentangling entanglement in Cape fur seals for better management of plastic pollution impacts.
Gulland, FM, Baker, JD, Howe, M, LaBrecque, E Leach, L., Moore, SE, Reeves, RR and Thomas, PO. 2022. A review of climate change effects on marine mammals in United States waters: Past predictions, observed impacts, current research and conservation imperatives. Climate Change Ecology, 3, p.100054.
Gumede, NC. 2023. Variability in the diet of Cape fur seals and their interaction with fisheries off the South African coast, 2010-2019. MSc dissertation, University of Cape Town, South Africa
Hanks J. 1990. Report to the subcommittee of the Sea Fisheries Advisory Committee appointed at the request of the Minister of Environment Affairs and of Water Affairs, to advise the Minister on scientific aspects of sealing. Southern African Nature Foundation.
Harvell, CD, Mitchell, CE, Ward, JR, Altizer, S, Dobson, AP, Ostfeld, RS and Samuel, MD. 2002. Climate warming and disease risks for terrestrial and marine biota. Science, 296(5576), pp.2158-2162.
Harwood, J. 2002. Mass die-offs. In: Perrin WF, Wursig B, Thewisse JGM (ed.), Encyclopaedia of Marine Mammals, pp. 724–726. Academic Press, New York, USA.
Heidi, T. 2020. Assessing the sustainability of seal tourism at Duiker Island, Hout Bay. MSc Dissertation, University of Cape Town, South Africa.
Henry, M.R., 2022. Entanglement of Cape Fur Seals (Arctocephalus pusillus pusillus) in South Africa.
Huisamen J, Kirkman SP, Watson LH, Cockcroft VG, Jewell R, Pistorius PA. 2012. Diet of the Cape fur seal Arctocephalus pusillus pusillus in Plettenberg Bay, South Africa, and the implications for fisheries. African Journal of Marine Science 34: 431–441.
Huisamen J, Kirkman SP, Watson LH, Cockcroft VG, Pistorius PA. 2011. Re-colonisation of the Robberg Peninsula (Plettenberg Bay, South Africa) by Cape fur seal. African Journal of Marine Science 33: 453–462.
Japp, D.W., Purves, M.G. and Wilkinson, S. 2012. Benguela Current Large Marine Ecosystem: State of the Stocks Review. Report No. 2 (2012). Benguela Current Commission. Capricorn Fisheries Monitoring, Cape Town, South Africa.
Johnson, RL, Venter A, Bester MN, Oosthuizen WH. 2006. Seabird predation by white shark, and Cape fur seal, at Dyer Island. 36: 23–32.
Kennedy, S, Kuiken, T, Jepson, PD, Deaville, R, Forsyth, M, Barrett, T, Van de Bildt, MW, Osterhaus, AD, Eybatov, T, Duck, C and Kydyrmanov, A. 2000. Mass die-off of Caspian seals caused by canine distemper virus. Emerging infectious diseases, 6(6), p.637.
Kerley, G.I.H. 1983. Record for the Cape fur seal Arctocephalus pusillus pusillus for subantarctic Marion Island. South African Journal of Zoology 18: 139-140.
Kirkman SP, Oosthuizen WH, Meÿer MA, Seakamela SM, Underhill LG. 2011. Prioritising range-wide scientific monitoring of the Cape fur seal in southern Africa. African Journal of Marine Science 33(495–509).
Kirkman, S.P., Oosthuizen, W.H., Meyer, M.A., Kotze, P.G.H, Roux, J.-P. and Underhill, L.G. 2007. Making sense of censuses and dealing with missing data: trends in pup counts of Cape fur seal Arctocephalus pusillus pusillus for the period 1972-2004. South African Journal of Marine Science 29: 161-176.
Kirkman, S.P., Yemane, D., Oosthuizen, W.H., Meÿer, M.A., Kotze, P.G.H., Skrypzeck, H., Vaz Velho, F. and Underhill, L.G. 2013. Spatio-temporal shifts of the dynamic Cape fur seal population in southern Africa, based on aerial censuses (1972-2009). Marine Mammal Science 29: 497-524.
Kirkman, S. P., & Arnould, J. P. 2018. Cape and Australian fur seals: Arctocephalus pusillus pusillus and A. p. doriferus. In Würsig B, Perrin W.F., Thewissen J.G.M. (Eds) Encyclopedia of Marine Mammals (pp. 158-161). Academic Press.
Kirkwood, R. and Goldsworthy, S.D. 2013. Fur Seals and Sea Lions. CSIRO Publishing, Collingwood, Victoria, Australia.
Kovacs, KM, Aguilar, A, Aurioles, D, Burkanov, V, Campagna, C, Gales, N, Gelatt, T, Goldsworthy, SD, Goodman, SJ, Hofmeyr, GJG, Härkönen, T, Lowry, L, Lydersen, C, Schipper, J, Sipilä, T, Southwell, C, Stuart, S, Thompson, D and Trillmich, F. 2012, Global threats to pinnipeds. Marine Mammal Science, 28: 414-436. https://doi.org/10.1111/j.1748-7692.2011.00479.x
Laidre, KL, Stirling, I, Lowry, LF, Wiig, Ø, Heide-Jørgensen, MP and Ferguson, SH. 2008. Quantifying the sensitivity of Arctic marine mammals to climate‐induced habitat change. Ecological applications, 18(sp2), pp. S97-S125.
Lento, G.M., Haddon. M., Chambers, G.K. and Baker, C.S. 1997. Genetic variation of southern hemisphere fur seals (Arctocephalus spp.): Investigation of population structure and species identity. Journal of Heredity 88(3): 202-208.
Lutjeharms, JRE. 2006. Three decades of research on the greater Agulhas Current. Ocean Science, 3(1), pp.129-147.
Makhado, A.B., Crawford, R.J. and Underhill, L.G. 2006. Impact of predation by Cape fur seals Arctocephalus pusillus pusillus on Cape gannets Morus capensis at Malgas Island, Western Cape, South Africa. African Journal of Marine Science, 28(3-4), pp.681-687.
Makhado A.B, Amaro A, Crawford R.J.M, Gotlieb T, Morais M, Mwaala D.N, Nghimwatya L, Seakamela S.M, Tom D and Witteveen. 2024. Atlas of marine turtles, seabirds, and seals in the Benguela Current and adjacent regions. Population sizes and trends, conservation status, and Important Bird and Biodiversity Areas for breeding. Benguela Current Convention, Cape Town South Africa.
Malan, A., Von Der Heyden, S., Herron, S., Arnould, J. P. Y., Kirkwood, R., & Matthee, C. A. 2022. Palaeoclimatic changes resulted in range expansion and subsequent divergence in brown fur seals, Arctocephalus pusillus. Biology Letters, 18(8), 20220285.
Mann, B.Q. 2013. Southern African Marine Linefish Species Profiles. Special Publication No. 9. Southern African Association for Marine Biological Research Oceanographic Research Institute, Durban.
Marks, M. M., Brooke, R. K., & Gildenhuys, A. M. 1997. Cape fur seal Arctocephalus pusillus predation on Cape cormorants Phalacrocorax capensis and other birds at Dyer Island, South Africa.
Martin, A.R., Hammerschlag, N., Collier, R.S. and Fallows, C. 2005. Predatory behaviour of white sharks (Carcharodon carcharias) at Seal Island, South Africa. Journal of the Marine Biological Association of the UK 85: 1121-1135.
Matthee, C.A., Fourie, F., Oosthuizen, W.H., Meÿer, M.A. and Tolley, K.A. 2006. Mitochondrial DNA sequence data of the Cape fur seal (Arctocephalus pusillus pusillus) suggest that population numbers may be affected by climatic shifts. Marine Biology 148: 899-905.
Mecenero, S., Kirkman, S.P. and Roux, J.-P. 2006. A dynamic fish consumption model for lactating Cape fur seals Arctocephalus pusillus pusillus based on scat analyses. Journal of Marine Science 63: 1551–1566.
Mwaala, DN, Wilhelm, MR, Kirkman, SP and Roux, JP. 2023. Geographical and seasonal patterns in the diet of Cape fur seals Arctocephalus pusillus pusillus in Namibia, based on extensive scat analyses. African Journal of Marine Science, 45(4), pp.285-294.
Oosthuizen, W.H. 1991. General movements of South African (Cape) fur seals Arctocephalus pusillus pusillus from analysis of recoveries of tagged animals. South African Journal of Marine Science 11: 21-29.
Oosthuizen, W.H., Meyer, M.A., David, J.H.M., Summers, N.M., Kotze, P.G.H., Swanson, S.W. and Shaughnessy, P.D. 1997. Variation in jackal numbers at the Van ReenenBay seal colony with comment on the likely importance of jackals as predators. African Journal of Wildlife Research 27: 26-29.
O’Donoghue, S.H., Whittington, P.A., Dyer, B.M. and Peddemors, V. 2010. Abundance and distribution of avian and marine mammal predators of sardine observed during the 2005 KwaZulu-Natal sardine run survey. African Journal of Marine Science 32: 361-374.
Orams, M.B. 2002. Feeding wildlife as a tourism attraction: a review of issues and impacts. Tourism Management, 23(3), pp.281-293.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Porritt J, Goodman J. 2005. Fishing for Good. Forum for the Future, London, UK.
Punt, A.E. and Butterworth, D.S. 1995. The effects of future consumption by the Cape fur seal on catches and catch rates of the Cape hakes. 4. Modelling the biological interaction between Cape fur seals Arctocephalus pusillus pusillus and the Cape hakes Merluccius capensis and M. paradoxus. South African Journal of Marine Science 16: 255-285.
Rand, R.W. 1955. Reproduction in the female Cape fur seal, Arctocephalus pusillus (Schreber). Proceedings of the Zoological Society, London 124: 717-740.
Reijnders, P., Brasseur, S., van der Toorn, J., van der Wolf, P., Boyd, I., Harwood, J., Lavigne, D. and Lowry, L. 1993. Seals, fur seals, sea lions, and walrus. Status survey and conservation action plan. IUCN Seal Specialist Group.
Repenning, C.A., Peterson, R.S. and Hubbs, C.L. 1971. Contributions to the systematics of the southern fur seals, with particular reference to the Juan Fernández and Guadalupe species. In: W.H. Burt (ed.), Antarctic Pinnipedia, pp. 1-34. Antarctic Research Series 18, American Geophysical Union, New York, USA.
Robbertse, M, Hofmeyr, GJG, de Bruyn, PJN, Dalton, D and Mwale, M. 2025. Mitochondrial DNA diversity and phylogeographic patterns among South African Cape fur seals, Arctocephalus pusillus pusillus. Aquatic Conservation: Marine and Freshwater Ecosystems 35: e70210.
Roux, J.-P. 1998. The impact of environmental variability on the seal population. Namibia Brief 20: 138-140.
Roy, C., Van Der Lingen, C.D., Coetzee, J.C. and Lutjeharms, J.R.E. 2007. Abrupt environmental shift associated with changes in the distribution of Cape anchovy Engraulis encrasicolus spawners in the southern Benguela. African Journal of Marine Science 29: 309-319.
Sanders, YLB, Connan, MA, Jacquemet, S, van der Vyver, JSF and Hofmeyr, GJG. 2025. Spatial and temporal patterns of Cape fur seal strandings along the south-eastern South African coast suggest anthropogenic and environmental interactions. Marine Biology 172: e119
Santos, F., Gomez-Gesteira, M., Decastro, M. and Alvarez, I. 2012. Differences in coastal and oceanic SST trends due to the strengthening of coastal upwelling along the Benguela current system. Continental Shelf Research, 34, pp.79-86.
Santana-Casiano, JM, González-Dávila, M and Ucha, IR. 2009. Carbon dioxide fluxes in the Benguela upwelling system during winter and spring: A comparison between 2005 and 2006. Deep Sea Research Part II: Topical Studies in Oceanography, 56(8-10), pp.533-541.
Seakamela, SM. 2024. The effect and conservation implications of the re-establishment of a Cape fur seal Arctocephalus pusillus pusillus breeding colony at Vondeling Island, Saldanha Bay. MSc Dissertation. Department of Biological Sciences, University of Cape Town. South Africa
Seakamela S.M, McCue S.A, Mëyer M.A., Kotze P.G.H. 2025. South African Cape fur seal colonies and their status, 1971–2020. In: Makhado A.B, Amaro A, Crawford R.J.M, Gotlieb T, Morais M, Mwaala D.N, Nghimwatya L, Seakamela S.M, Tom D and Witteveen (eds), Atlas of marine turtles, seabirds, and seals in the Benguela Current and adjacent regions. Population sizes and trends, conservation status, and Important Bird and Biodiversity Areas for breeding. Cape Town: Benguela Current Convention.
Shaughnessy PD, Butterworth DS. 1981. Historical trends in the population size of the Cape fur seal (Arctocephalus pusillus). In: Chapman JA, Pursle D (eds), The Worldwide Furbearer Conference Proceedings. Falls Church, Virginia: Donnelly and Sons. pp 1305–1327.
Shaughnessy P.D. 1979. Cape (South African) Fur Seal. Mammals in the Seas, Vol. II: Pinniped species summaries and report on sirenians, pp. 37-40. Food and Agricultural Organization, Rome, Italy.
Shaughnessy, P.D. 1980. Entanglement of Cape fur seals with man-made objects. Marine Pollution Bulletin 11: 332-336.
Simmonds, MP and Isaac, SJ, 2007. The impacts of climate change on marine mammals: early signs of significant problems. Oryx, 41(1), pp.19-26.
Skern-Mauritzen, M., Kirkman, S.P., Olsen, E., Bjørge, A., Drapeau, L., Meÿer, M.A., Roux, J.P., Swanson, S. and Oosthuizen, W.H. 2009. Do inter-colony differences in Cape fur seal foraging behaviour reflect large-scale changes in the northern Benguela ecosystem? African Journal of Marine Science, 31(3), pp.399-408.
Sgqolana, T. 2021a. Hundreds of malnourished Cape fur seals wash up dead on South Africa’s shores. Daily Maverick. https://www.dailymaverick.co.za/article/2021-10-25-hundreds-of-malnourished-cape-fur-seals-wash-up-dead-on-south-africas-shores/. Accessed 21 Jul 2023
Sgqolana, T. 2021b. Malnourished and dead seals continue to wash up on South Africa’s coastal beaches. Daily Maverick. https://www.dailymaverick.co.za/article/2021-11-11-malnourished-and-dead-seals-continue-to-wash-up-on-west-coast-beaches/. Accessed 21 Jul 2023
Stander, P. E. 2019. Lions (Panthera leo) specialising on a marine diet in the Skeleton Coast Park, Namibia. Namibian Journal of Environment, 3, A-10.
Stewardson, C.L. 1999. The impact of the fur seal industry on the distribution and abundance of Cape fur seals Arctocephalus pusillus pusillus on the Eastern Cape Coast of South Africa. Transactions of the Royal Society of South Africa 54: 217-246.
Stewardson, CL. 2001. Biology and conservation of the Cape (South African) fur seal Arctocephalus pusillus pusillus from the Eastern Cape Coast of South Africa. PhD Thesis. The Australian National University
Stewardson, C.L., Bester, M.N. and Oosthuizen, W.H. 1998. Reproduction in the male Cape fur seal Arctocephalus pusillus pusillus: age at puberty and annual cycle of the testis. Journal of Zoology, 246(1), pp.63-74.
Thibault, M. 1999. Sighting of a South African fur seal on a beach in south-western Gabon. American Journal of Ecology 37: 119–120.
van der Lingen CD, Shannon LJ, Cury P, Kreiner A, Moloney CL, Roux J-P, Vaz-Velho F. 2006. Resource and ecosystem variability, including regime shifts, in the Benguela Current System. In: Shannon V, Hempel G, Malanotte-Rizzoli P, Moloney CL, Woods J (eds) Benguela: Predicting a Large Marine Ecosystem. Elsevier, Amsterdam, pp 147–185
Van Helden, L. 2024. Rabies in the Western Cape: seals, dogs and jackals. Western Cape Government Epidemiology Report. 16 (5) 1-3.
Warneke, R.M. and Shaughnessy, P.D. 1985. Arctocephalus pusillus, the South African and Australian fur seal: taxonomy, evolution, biogeography and life history. In: J.K. Ling and M.M. Bryden. (eds), Studies of Sea Mammals in South Latitudes, pp. 53-77. South Australian Museum, Adelaide, Australia.
Wickens, P. A., David, J. H. M., Shelton, P. A. and Field, J. G. 1991. Trends in harvest and pup numbers of the South African fur seal: Implications for management. South African Journal of Marine Science 11: 307-326.
Wickens, P. A., Japp, D. W., Shelton, P. A., Kriel, F., Goosen, P. C., Rose, B., Augustyn, C. J., Bross, C. A. R., Penney, A. J. and Krohn, R. G. 1992. Seals and fisheries in South Africa – competition and conflict. In: A.I.L. Payne, K.H. Brink, K.H. Mann, and R. Hilborn (eds.). Benguela Trophic Functioning. South African Journal of Marine Science 12: 773-789.
Wickens, P. and York, A.E. 1997. Comparative population dynamics of fur seals. Marine Mammal Science 13(2): 241-292.
Wiesel, I. 2010. Killing of Cape fur seal (Arctocephalus pusillus pusillus) pups by brown hyenas (Parahyaena brunnea) at mainland breeding colonies along the coastal Namib Desert. Acta Ethologica 13: 93-100.
Wynen, L.P., Goldsworthy, S.D., Insley, S.J., Adams, M., Bickham, J.W., Francis, J., Gallo, J.P., Hoelzel, A.R., Majluf, P., White, R.W.G. and Slade, R. 2001. Phylogenetic relationships within the eared seals (Otariidae: Carnivora): Implications for the historical biogeography of the family. Molecular Phylogenetics and Evolution 21(2): 270-284.