Cape Fox
Vulpes chama

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Vulpes chama – (A. Smith, 1833)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – CANIDAE – Vulpes – chama
Common Names: Cape Fox, Silver Fox, Silver Jackal (English), Draaijakkals, Silwerjakkals, Silwervos (Afrikaans), Mophémé (Sesotho), Lesiê, Losiê (Tswana)
Synonyms: No Synonyms
Taxonomic Note: None
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Power, R.J.1 & da Silva, J.M.2
Reviewer: Do Linh San, E.3
Contributor: Nicholson, S.4
Institutions: 1North West Provincial Government, 2South African National Biodiversity Institute, 3Sol Plaatjie University, 4Endangered Wildlife Trust
Previous Assessors and Reviewers: Kamler, J.F., Palmer, G., Cowell, C., Mills, M., Stuart, C., Stuart, M. & Do Linh San, E.
Previous Contributor: Hoffmann, M.
Assessment Rationale
The Cape Fox is listed as Least Concern as it is widespread within the assessment region and has expanded its range over recent decades. It is generally common to fairly abundant across much of its range, although problem animal control activities, especially indiscriminate poisoning and trapping, have resulted in local subpopulation reductions in some areas. Local subpopulations may also be low or even absent in areas where Black-backed Jackals (Lupulella mesomelas) are abundant, due to interspecific competition, including intraguild predation. This may represent an emerging threat if poor management or land-use change increases Black-backed Jackal numbers. Population size and trend estimates are not available for most areas, but it is thought that the population is currently stable across the entire range. Interventions include the establishment of conservancies and holistic ecosystem management of damage-causing animals.
Regional population effects: We suspect that there is dispersal across regional borders as the range is continuous across southern Africa and the species is not usually constrained by fences. There is a broad front of its national distribution with Namibia and Botswana, and they freely cross the Nossob into Botswana in the Kgalagadi Transfrontier Park.
Red List Index
Red List Index: No change
Recommended citation: Power RJ & da Silva JM. 2025. A conservation assessment of Vulpes chama. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
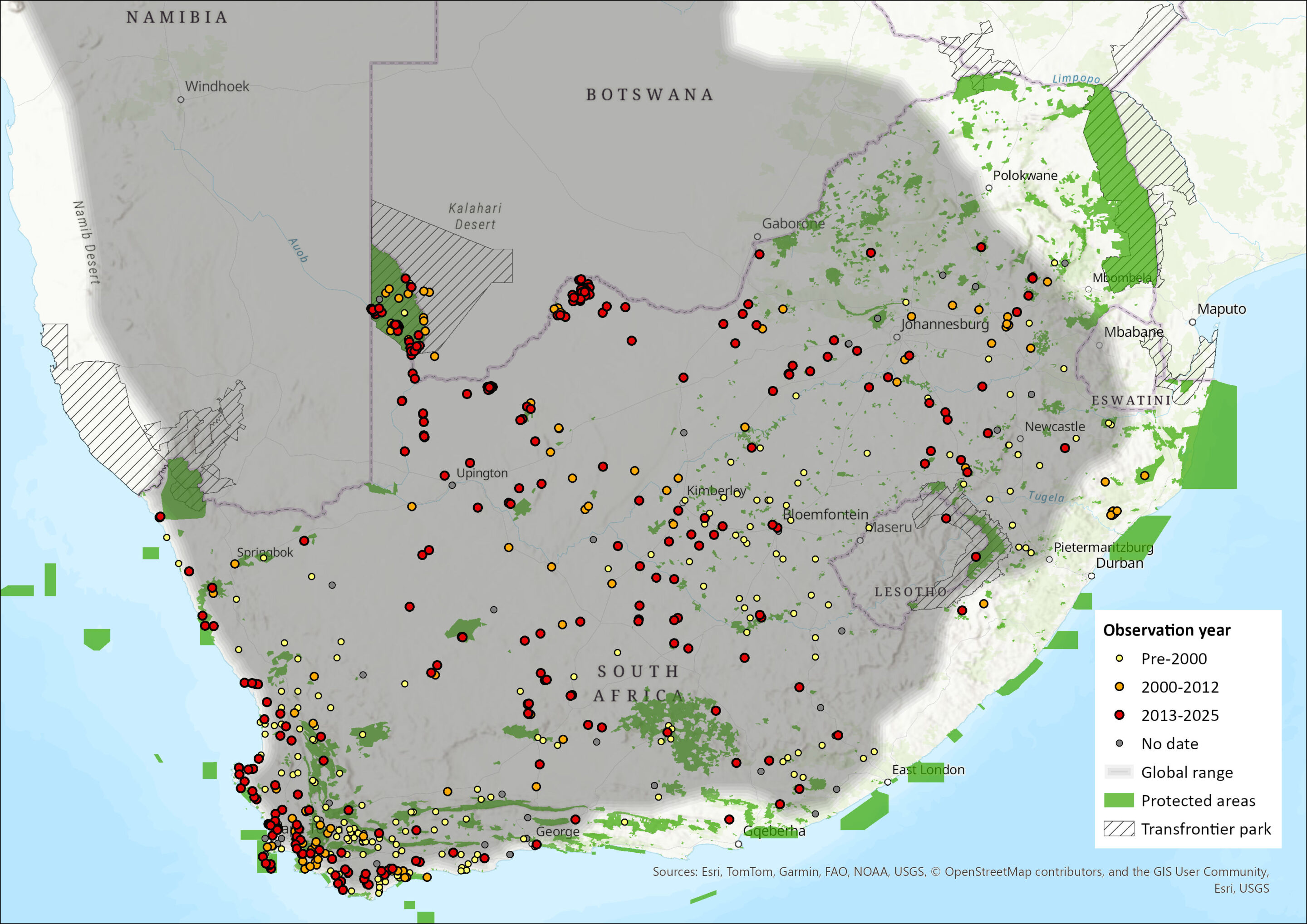
The species is widespread in the central and western regions of southern Africa. It mainly occupies arid and semi-arid areas, of Botswana, Namibia, and in parts, such as the Fynbos biome of South Africa’s Western Cape Province, the species enters areas receiving higher precipitation and denser vegetation (Stuart & Stuart 2008; Kamler et al. 2016). The species has recently been documented to have exhibited a genuine range expansion into Zimbabwe’s Hwange National Park (Loveridge et al. 2019).
The distribution of the Cape Fox in Lesotho was uncertain (Lynch 1994), although it appears as if it has been confirmed recently (Figure 1). The species occurs in the Highveld of Eswatini (Monadjem 1998), despite no known recent records.
The species is still present in neighbouring regions of north-western KwaZulu-Natal (Rowe-Rowe 1992), and appears to now occur in the lower foothills skirting the Drakensberg to connect to the Eastern Cape it seems (see Map). In KwaZulu-Natal, Mpumalanga, Limpopo and Gauteng provinces, Cape Foxes are limited to highland grassland (Kamler et al. 2016).
The species is still present in the Eastern Cape Province when compared to earlier baselines (Hey 1964; Stuart 1981; Lloyd & Millar 1984), though was curiously not detected in the Mountain Zebra National Park in a survey (Smith et al. 2023) but appears to be holding the same range since the last assessment (Figure 1). In the Northern Cape, they are widespread, especially in the Kalahari (Figure 1), and are common throughout the Kgalagadi Transfrontier Park (Kamler et al. 2016), and they also occur in the Tswalu Kalahari Reserve (Smith et al. 2023). Corroborating this, a survey in North West Province noted that the species was more common in the western Kalahari than the southern grasslands (Power 2014), although they occur over most of the province (Power et al. 2019), they specifically occur in the west and south (Figure 1). The species still occurs across the Free State (cf Lynch 1975) (Figure 1). They were recorded on the Mpumalanga Highveld, and occurred at Verloren Vallei Nature Reserve (Loock & Swanepoel 2019), although was not detected at the Telperion Nature Reserve, on the edge of the savannas (see Webster et al. 2021), although it had been observed on the outskirts of the reserve. In Limpopo, the species is marginal (Kamler et al. 2016), but there does appear to be what could be a marginal range expansion on the southern margins of the province (Figure 1).
It is unclear whether the Cape Fox historically occurred in the Cape Peninsula (Boshoff & Kerley 2001), but it is thought that sightings by early settlers were recorded as “jackal” or “jakkals”, and not as “foxes”. Other than there being no mention of the Cape Fox on the Cape Peninsula (Skead 2011). The mammals of Kirstenbosch have been described in detail and there was no historical account of the Cape Fox, or any canid mentioned (Bolus 1921). Similarly, in the divisional council situated around Cape Town, the species was reported by only 0.3% of landowners in 1974 (Lloyd & Millar 1984). This suggests they were likely not ever present in the Cape Peninsula area, or rare at best.
Although only recorded from the Clanwilliam area (Shortridge 1942), it probably occurred throughout the Cape Floristic Region, particularly in the low-lying areas and open strandveld areas. The subpopulation in Table Mountain National Park is likely the result of introductions between 1960 and 1980, and the species still persists in the park, although only a few animals have been seen and recorded on camera traps (C. Cowell pers. obs. 2014).
The Cape Fox also occurs in both the Cape Winelands Biosphere Reserve and Kogelberg Biosphere Reserves (Nieman et al. 2021). In the Karoo, they occur throughout (Figure 1; Schurch et al. 2021; Woodgate et al. 2023), though may be locally rare in some of the protected areas, i.e. Anysberg Nature Reserve, and Sanbona Wildlife Reserve (see Woodgate et al. 2023).
The species distribution seems to have changed little since the last assessment, and there may even be a northward range expansion in the north-eastern part of the range (see Figure 1), which may be in tandem with a northerly range expansion into Zimbabwe (Loveridge et al. 2019).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Cape Fox (Vulpes chama) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Eswatini | Presence Uncertain | Native | – | Resident |
| Lesotho | Presence Uncertain | Native | – | Resident |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
It is important to consider a species’ ecological and biological traits to determine the likely mechanisms of climate change impact and quantify this using expert knowledge (Foden and Young 2016). An increase in shrub cover, as predicted by many climate change models (Foden and Young 2016), would negatively impact the Cape Fox. Given the species is indeed negatively affected by increasing shrub cover (Blaum et al. 2007), and it could then decline. On the other hand, given the prediction that the species range may increase with the advent of Karoo-like conditions (Von Richter 1972), it can also be predicted that they would be benefitted from increased drying out of central South Africa. It is unclear how climate change can affect the species, and it may even benefit from drying out conditions.
Population information
Estimated site sizes or numbers are not available for most areas, but it is thought that subpopulations are contiguous and currently stable across their entire range (Stuart & Stuart 2008). Density estimates were 5 and 14 individuals / 100 km2, respectively, during recent studies on two sites near Kimberley, South Africa (Kamler et al. 2012, 2013), whereas Bester (1982) estimated a density of 30 individuals / 100 km2 in the Free State Province, with a total population then of 31,000 individuals. In conducting a feasibility assessment aimed at the Cape Floristic Region in southern South Africa, based on literature, a Cape Fox density estimate for the area was 14.8 Foxes / 100 km2 (Boshoff et al. 2002). Though given densities in other more optimal habitats for the species (Kamler et al 2012), this may be an overestimation. Similarly, shortly after, a total population size of 16,863 was estimated for the species there (Kerley et al. 2003).
It is currently unclear how many Cape Foxes are killed annually in animal control operations, and how persecution may affect their populations. In the 1970s and 1980s, thousands of Cape Foxes were killed annually in control operations throughout South Africa (Stuart & Stuart 2004), but Bester (1982) thought that it resulted in no obvious decline in their overall population in the Free State. In 1956, 15,323 Cape Fox were killed in the old Cape Province, second only to the jackal of all the carnivores, and this was incentivised by a bounty system (Hey 1964). Similar to the situation in the Free State, despite the high offtakes of the species, the species has still persisted (Stuart 1981).
The Cape Fox also had a low reported relative abundance in the Cape Winelands Biosphere Reserve, but were reported to have high relative abundance in the Kogelberg Biosphere Reserve (Nieman et al. 2021).
Between 2013 and 2015, extensive camera trapping took place in the Western Cape Karoo and Cape Fox were detected only on commercial small livestock farmlands, while not on ecologically comparable private and state protected areas, while Black-backed Jackal has higher occupancy in these protected areas (Woodgate et al. 2023). The population in the Western Cape is stable or increasing due to agricultural transformation (which can favour the species) and the reduction in medium-sized predators such as Black-backed Jackals (Kamler et al. 2016). However, local groups may decline as jackals make a return due to more “holistic” farming methods and as part of wildlife ranching and ecotourism expansion (Kamler et al. 2016). Of course, recovery of Leopards (Panthera pardus) may counter the effects of jackals, and thus benefit Cape Foxes (see Kamler et al. 2020).
In an intensive camera trapping study spanning 2017 to 2019 in the Tswalu Kalahari Reserve (Northern Cape), the Cape Fox was detected at high frequencies, and so too was the Black-backed Jackal (Smith et al. 2023). Similarly, in the North West Kalahari, reproduction, and relatively high detections were obtained in reserves, but not outside reserves (NWPG 2016; 2017). In surveys assessing the impact of hunting and disturbance on Cape Fox inter alia, the control site (!Khamab Kalahari Reserve) in 2015, revealed a photo-capture rates (Relative Abundance Indices – RAI) of 0.6±1.1 foxes/100 trap nights, and none were recorded on adjacent hunting farms adjacent the reserve (NWPG 2016). A year later, at the same reserve, the RAI was higher at 5.3±8.5/100 trap nights, and naïve occupancy was 53% there, which is despite an increasing jackal population of 14% (if aerial count data is used, i.e. 125 jackals counted in 2015, and 109 in 2014) (NWPG 2017). Even at the nearby provincial reserve, also a control site (Molopo), the RAI was 2.8±5.4/1oo trap nights and naïve occupancy was 33% there, and similarly there were no detections for this species on its paired comparison nearby of a livestock farm and neither at the game farm which focusses on trophy hunting and is electrified (NWPG 2017).
However, in the North West, away from the Kalahari proper, on the Highveld, the species was not recorded on any reserves (NWPG 2012; 2013), and this was ascribed to high Black-backed Jackal abundances. Similarly, in other reserves with high prevalence of Black-backed Jackals, Cape Foxes have not been detected elsewhere on the Highveld (Webster et al. 2021), and from at least three other reserves in the Karoo (Smith et al. 2023; Woodgate et al. 2023).
On Benfontein Game Farm, estimated Cape Fox densities in 2008 were 0.49 fox/10 km2 (Kamler et al. 2012). Black-backed Jackals killed at least one of 11 radio-collared Cape Fox between 2005 and 2008, indicating clearly that there is potential interference competition (Kamler et al. 2012). In the absence of jackals, densities of Cape Foxes increased by 64% despite similar food and habitat resources between sites, indicating that jackals suppressed Cape Fox populations (Kamler et al. 2013).
The smaller home ranges on the private ranch were said to be due to a lower density of Cape Fox on the private ranch (Kamler et al. 2013), though there should be an inverse relationship, i.e. higher density when home-ranges are smaller. Black-backed Jackals appeared to suppress populations of Cape Foxes (Kamler et al. 2013).
Some foxes in a population may breed as yearlings indicating Cape Foxes have high reproductive potential (Kamler & Macdonald 2014). Two adult female Cape Foxes maintained their territories after their mates died, whereas two adult males dispersed soon after their mates died, indicating they likely have a female-based social organization (Kamler & Macdonald 2014). Cape Fox dispersal was male biased, as all juvenile males (n = 6) dispersed when 9–11.5 months old, whereas 3 of 4 juvenile females remained philopatric as either breeders or non-breeding associates (Kamler & Macdonald 2014).
Unfortunately, there are no recent population estimates, only estimates of relative abundance which can be gleaned (i.e. camera trap detections etc) to infer population status.
Cape Fox moving a pup to a new den in the !Khamab Kalahari Reserve, circa 2016 (Photo courtesy of Panthera).
Population Information
Current population trend: Stable, based on wide habitat tolerance and extent of occurrence.
Continuing decline in mature individuals: Yes, due to direct or indirect persecution.
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown, but likely to be Kgalagadi Transfrontier Park.
Number of subpopulations: Unknown
Severely fragmented: No. Can exist in multiple habitats, including agricultural lands.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
The complete mitogenome of V. chama has been assembled have been investigated and used to explore the phylogenetic relationships within Canidae (Gray et al. 2024). This study revealed that V. chama diverged early within the Vulpes genus. To date, however, no comprehensive population genetic/genomic study has been undertaken on the species to understand its current structure and diversity.
Despite this, given the dispersal capabilities of the species (capable of dispersing across regional borders and not being restricted by fences) and its continuous distribution across southern Africa, it is expected that the species exists as a single metapopulation, likely connected to neighbouring countries. Assuming no subpopulations have gone extinct, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – could be estimated at 1.0 (all populations remaining).
Recent population size estimates are unavailable for the species, however, relative estimates from camera traps suggest the population likely exceeds 10,000 individuals within the assessment region. Assuming a 0.1-0.3 Ne/Nc conversion ratio, Ne is estimated to exceed 1,000-3,000 individuals, which is above the Ne 500 threshold, indicating the species is likely genetically healthy and stable into the long-term. Based on this information, the GBF’s headline genetic indicator – proportion of populations with an Ne > 500 would be 1/1 population above Ne 500, equating to a score of 1.0. It should be noted however, that DNA-based Ne estimates should be prioritised given the potential impact of predators and animal control operations.
Habitats and ecology
Cape Foxes mainly occur in open country with habitats including grassland, grassland with scattered thickets, and lightly wooded areas, particularly in the dry Karoo regions, the Kalahari and the fringes of the Namib Desert (Stuart & Stuart 2008). Other vegetation types that the Cape Fox has been recorded in include the Kimberley Thornveld, Eastern Mixed Kama Karoo and dry Western Highveld Sandy Grasslands (Kamler et al. 2016), as well as Gordonia Duneveld and Kalahari Karroid Shrubland vegetation types (Bullock et al. 2011). These include Karroid shrublands, open thornveld and Highveld grasslands.
Along the eastern areas of the Namib Desert (Namibia) they occupy rock outcroppings and inselbergs, ranging out onto bare gravel plains at night (Stuart 1975), which may be similar to areas where this landscape enters South Africa. In Botswana, the species occurs in Acacia scrubland, short grassland (specifically on the fringes of shallow seasonal pans), and cleared and overgrazed areas (Smithers 1968; Skinner & Chimimba 2005). In the Western Cape, they also occur in moderately dense vegetation in the lowland fynbos (Stuart 1981). Cape Foxes also occur in extensive agricultural lands, lying in surviving patches of natural vegetation during the day and foraging on arable and cultivated fields at night (Stuart 1981). In the central Karoo area of South Africa, Cape Foxes occupy the plains and the low rocky ridges and isolated rock outcroppings (Kamler et al. 2016). Lynch (1975) recorded that in the Free State, Cape Foxes were most abundant in areas which had less than 500 mm of rainfall. However, in KwaZulu-Natal they have been recorded between 1,000 and 1,500 m asl, where rainfall is roughly 750 mm per year, and there they preferred lightly treed savanna (Rowe-Rowe 1992). They also occur in highland grasslands (Rowe-Rowe 1992; Monadjem 1998; Loock and Swanepoel 2019).
In a semi-arid savanna, at the home-range scale, habitat selection by Cape Foxes was used in proportion to availability for Cape Foxes at least on a private ranch (Kamler et al. 2013), but in essence there was no overt selection. Den site habitat selection of Cape Foxes was in proportion to availability on a comparable private ranch, whereas on Benfontein Game Farm, the dens of Cape Foxes were selected positively for particular habitats, specifically, savanna type habitat there (Kamler et al. 2012), suggesting the importance of cover. Cape Foxes also showed selection for habitats with pan slopes (Kamler et al. 2013), which suggests pans are important landscape features to them.
They are good diggers and often dig their own dens for shelter, or use those of other species, such as Springhares (Pedetes capensis), especially during the pup-rearing period (Kamler & Macdonald 2014). At other times of the year, they are more likely to use daytime shelter above ground in thickets, dense bushes, and hollowed termite mounds (Skinner & Chimimba 2005; Kamler & Macdonald 2014).
Cape Foxes were found to be mostly nocturnal (Kamler et al. 2012), while in the absence of jackals, Cape Foxes were marginally more active during daytime (Kamler et al. 2013). Cape Foxes exhibited different activity patterns in the absence of jackals, and differences were consistent across seasons, and the presence of jackals appeared there to at least influence activity patterns of Cape Foxes (Kamler et al. 2013). The absence of jackals resulted in smaller home-range sizes for the Cape Fox (Kamler et al. 2013), and population density appeared to increase.
They were consistently more active during the day and less active at night on a nearby private ranch compared with Benfontein Game Farm (Kamler et al. 2013). During the night they hunt in a variety of habitats, typically using habitat types in proportion to availability within their home ranges, although they generally prefer more open habitats at the landscape level (Kamler et al. 2012), though appear to be sensitive to an increase in cover (Blaum et al. 2007). Due to habitat transformation by humans, they have been found in more mountainous or rocky areas compared to their historical occurrence (Kamler et al. 2016). At Benfontein, on the fringe of the Free State and Northern Cape, the mean annual home-range sizes for Cape Fox were 27.7 km2 (Kamler et al. 2012). In the Free State, on a private ranch, home-range sizes for Cape Fox averaged 9.20 ±0.82 km2 (n=8) while it was higher on Benfontein Game Ranch, at 27.68±1.45 km2, n = 5) (Kamler et al. 2013). The smaller home ranges on the private ranch were said to be due to lower density of Cape Fox on the private ranch (Kamler et al. 2013), though this is meant to read that it is due to a higher density of foxes, which is the norm for many carnivores where an inverse relationship between home-range size and density exists.
Cape Foxes were socially monogamous and territorial, with annual home ranges of mated pairs (n = 8) overlapping 80% on average, compared to a mean overlap of 3% between foxes in adjacent ranges (Kamler & Macdonald 2014).
Members of mated Cape Fox pairs always foraged alone, however they used the same or nearby (<100 m apart) day rests 81% of the time when pups were 0–4 months of age, but only 28% of the time during other months of the year (Kamler & Macdonald 2014).Cape Fox dispersal was male biased, as all juvenile males (n = 6) dispersed when 9–11.5 months old, whereas 3 of 4 juvenile females remained philopatric as either breeders or non-breeding associates (Kamler & Macdonald 2014).
Cape Foxes have omnivorous diets, although their main prey items are small rodents, and other common food items are hares, insects, birds, reptiles, wild fruit, and carrion (Bothma 1966; Skinner & Chimimba 2005; Kamler et al. 2012; Klare et al. 2014). Although Cape Foxes have been documented to kill lambs (Stuart 1981; Bester 1982), the level of predation is uncertain as much of the consumption may be from carrion (Stuart 1981; Klare et al. 2014; Skinner & Chimimba 2005). On Benfontein Game Farm, Northern Cape, Cape Fox did not feed on carrion (Klare et al. 2014), perhaps to reduce encounters with jackals, however elsewhere they do feed on carrion (see Bothma 1971; Lynch 1975).
They forage solitarily (Kamler & Macdonald 2014) and primarily at night (Kamler et al. 2021). Cape Foxes live in monogamous pairs, with occasional helpers, and have stable home ranges (Kamler & Macdonald 2014), that vary from 3–30 km depending on local food resources and numbers of Black-backed Jackals (Bester 1982; Kamler et al. 2013; Kamler & Macdonald 2014). Although humans are likely their main source of mortality, natural mortalities result from predation by larger carnivores, especially Black-backed Jackals (Kamler et al. 2012; 2013; Kamler & Macdonald 2014). Two adult female Cape Foxes maintained their territories after their mates died, whereas two adult males dispersed soon after their mates died, indicating they likely have a female-based social organisation (Kamler & Macdonald 2014).
Annual Cape Fox survival was estimated at 64%, and predation from larger carnivores, primarily Black-backed Jackals was responsible for 71% of mortalities (Kamler & Macdonald 2014). Cape Foxes coexisted with jackals by exhibiting high levels of spatial, habitat, temporal, and dietary partitioning (Kamler et al. 2012). They are classified as a fugitive species i.e., species that forages widely and selectively uses areas with low occurrences of intraguild predators (Kamler et al. 2012). Jackals have the potential to suppress fox populations, yet foxes, in turn, use different mechanisms to coexist with a dominant canid (Kamler et al. 2012).
Interestingly, Cape Foxes and Bat-eared Fox (Otocyon megalotis) exhibited positive associations with each other and did not show avoidance of one another (Kamler et al. 2012), however in the absence of jackals, there was more niche segregation observed (Kamler et al. 2013). Cape Foxes appear to coexist well with Black-footed Cats (Felis nigripes) by partitioning space, but not habitats, activity, or diets (Kamler et al. 2015). The Cape Fox has been observed in a foraging association with the Striped Polecat (Ictonyx striatus) which appeared to be mutually beneficial to each (Panaino et al. 2022).
Ecosystem and cultural services: This is a relatively charismatic and endearing species and can be a potential flagship species (for example, Kgalagadi Transfrontier Park). They can be endearing to observe and photograph, especially at known den sites.
Ecologically, individual Cape Foxes consume nearly 4,000 rodents / year, therefore this species is likely beneficial to farmers and livestock owners in South Africa (Klare et al. 2014); for example, in vineyards in the Western Cape, where rodents are considered problematic. On Benfontein Game Farm, Northern Cape, the estimated annual consumption per Cape Fox was 3,861 rodents (Klare et al. 2014). It was therefore suggested that they might be beneficial to livestock and game farm owners in southern Africa (Klare et al. 2014), particularly in relation to vegetative cover protection in this case.
In terms of crop-based agriculture, since gerbils Gerbillurus sp are a known problem species in crop cultivation areas of the North West Province (Power 2014; Foster, McIntyre & Haussmann 2022), as well as the Western Cape (Hey 1964), and that they are sought after prey by Cape Fox (Klare et al. 2014), the species can be useful in such agroecosystems. However, farmers in the Ottosdal area of the North West tend to use chemical control, with only some amount of biological control. Out of 44 farmers interviewed, to control the species 28 used chemical methods, 12 mechanical and only 9 biological means of control (Foster et al. 2022). In this South African province, there is thus an under-appreciation of the role the species can play to control pest rodent species such as this.
Cape Foxes sometimes dig holes in hard-capped soils in Karoo shrubland and inter-dune pans to create microsites where detritus and water accumulate and seed germination is significantly increased (Dean & Milton 1991), thus promoting habitat structure. In inter-dune pans, 96% of all seedlings found were growing in diggings, and soils were deeper where diggings were made (Dean & Milton 1991).
The Nama name of the Cape Fox is !Khamab (see Skinner & Chimimba 2005; Skead 2011). This is also the name of an important private conservation area in North West, the !Khamab Kalahari Reserve of North West which not only bears the species name as the reserve’s name, but it is also in its branded logo.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 6. Rocky areas (eg. inland cliffs, mountain peaks) | – | Suitable | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Suitable | – |
Life History
Generation Length: 3.7 years
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
The Cape Fox is persecuted as a damage-causing animal and also killed incidentally by indiscriminate trapping and poisoning (Stuart & Stuart 2004).
Traditional medicine use
The Cape Fox appears in traditional medicine markets in Eastern Cape (Simelane & Kerley 1998), Gauteng (Whiting et al. 2016), and more recently, the Western Cape (Nieman et al. 2018). The species appeared in 5% of herbalist shops in the Eastern Cape (Simelane & Kerley 1998), while as many as 35% of them in the Western Cape (Nieman 2018).
At the Faraday Market in Gauteng in 2005, traders interviewed were stocking body parts of this species for sale (Whiting et al. 2016). In the Western Cape, the entire skin of the Cape Fox can be used decoratively and as clothing (Nieman et al. 2018) and can be sold for R500-R600 (Nieman 2018). The brain and urine of the Cape Fox is used as it is thought to improve cognition ability, while the urine only supposedly can be used as a headache treatment (Nieman et al. 2018) and can be sold for R50-R100 (Nieman 2018). The Cape Fox is used mostly in the traditional medicine trade in the Western Cape, followed by bushmeat secondarily only (Nieman 2018).
Bushmeat
Cape Fox were occasionally entrapped in wire snares intended for bushmeat poaching (i.e. smaller ungulates) in the Boland Mountain Complex of the Western Cape. However, it did not seem as though they were targeted and were thus likely caught as by-catch (Nieman 2018).
Trophy hunting
The species is trophy hunted but it does not seem to be significant. Sport hunting and trophy hunting of the species is not very important and likely to be of minimal consequence. The Eastern Cape has been the most important province where they are hunted, though this has declined. Interestingly, Limpopo is the second most important province, with mass off-takes taking place in 2015. This is unusual as the species has marginal occurrence there (see Figure 1), and this could possibly be a mistaken identity. Since 2020 in the North West, there have been three issued permits to hunt the species, one in 2021, and two in 2022 (NW Directorate of Biodiversity Management records, 2020-2023). There has not been a drastic increase in the demand to hunt the species over time since the last assessment, though there has been a regional increase in hunting of the species in Mpumalanga.
The effects of wildlife ranching are generally positive for Cape Foxes (see Kamler et al. 2013; NWPG 2017; Schurch et al. 2021). It is observed that Cape Foxes can do well on game/wildlife farms, but fox densities are dependent on the level of jackal control (Blaum et al. 2009; Kamler et al. 2013), especially if apex predators are absent (see also Kamler et al. 2020).
Ecotourism
The species is highly endearing to see, and most members of the public can be afforded the best viewing in the Kgalagadi Transfrontier Park. For the more affluent, the Tswalu Kalahari Reserve does offer unrivalled viewing of the species. There, the species has offered opportunities to observe unique and interesting behaviours (Panaino et al. 2021).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| No | – | Yes | Skins are traded locally. |
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
Habitat loss
Habitat loss/changes are not a major factor influencing the conservation status of the Cape Fox (Stuart & Stuart 2008). Conversely, in the Western Cape and elsewhere, changing agricultural practices have resulted in range extensions for this species (Stuart 1981). In the Western Cape, Cape Foxes have adapted well to agricultural transformation, especially in the vineyards. As a result of desertification, the semi-arid karroid vegetation has expanded eastwards (Von Richter 1972; Kraaij & Milton 2006), resulting in an extension of the Cape Fox range. However, based on habitat transformation in the Cape Floristic Region, the Cape Fox population was said to have declined by 39.6 % (Kerley et al. 2003). This is largely due to their preferred habitat being in the lowlands where habitat transformation more readily occurs. In Kalahari rangelands, Cape Foxes were negatively affected by increasing shrub cover (Blaum et al. 2007), and their abundance was lower where stocking rate was high (Blaum et al. 2009), which was likely related to cover and a loss in prey species.
Road related mortality
In 2007, eight roadkills of Cape Fox were recorded while carrying out mortality surveys along the R360 main road in the Northern Cape for a total distance of 2796 km (Bullock et al. 2011). There are several roadkill records from the Northern Cape (Bullock et al. 2011) and recently in the North West (NWPG 2024). This may be more important than previously considered (Kamler et al. 2024). Several of the distribution records are linearly arranged which points to roadkill to at least be important to an extent (see Figure 1). In the North West, on a commissioned study, of 81, 673 km driven on randomly driven roads in the Highveld and Kalahari ecoregions of the North West Province from 2020 to 2024, there were eight Cape Fox roadkills recorded, equally divided into those on tarred and gravel roads (NWPG 2024, and previous annual reports). Locally, in the same province, on a systematic Lichtenburg-Mahikeng-Ottoshoop roadkill survey (150 km circuit), conducted from 2021 to 2024, 13, 200 km was driven, and the circuit was driven on 100 occasions, there were no mortalities recorded of the Cape Fox over this time period (NWPG 2024, and previous annual reports), despite the species occurring in the area. Given the species propensity to reproduce (Kamler & Macdonald 2014), the threat of roadkills may be locally important only.
 Cape Fox killed on the R360 road between Upington and Askham (Photo by Emmanuel Do Linh San)
Cape Fox killed on the R360 road between Upington and Askham (Photo by Emmanuel Do Linh San)
Body parts trade
In the Western Cape, the Cape Fox had a Cultural Significance Index (CSI) of 2.25, which was low amongst several others that are a lot more important such as the Leopard for instance which scores at 15 (Nieman 2018). The threat is low for the area, but it is higher than the threat of bushmeat trade at least for this species (Nieman 2018). The species was not deemed important enough to assess to assess its threat status, unlike several other mammal species (Nieman 2018). The trade in medicinal uses of the species is higher than what was previously reported (Kamler et al. 2016), it can be seen as an emerging threat, though not overly catastrophic.
Directed persecution and incidental losses
Cape Foxes are hunted and poisoned, directly and killed as bycatch, throughout most of their range in South Africa (Stuart & Stuart 2008), primarily due to their perceived predation on lambs (Stuart 1981; Bester 1982), although there is little evidence for this (Klare et al. 2014). Although Cape Foxes can survive on transformed lands, and are often seen on the farms around Namaqua National Park, they are also severely affected by pesticides used to poison their prey (rodents and insects) on farmlands (Kamler et al. 2016). The illegal but widespread and indiscriminate use of agricultural poisons on commercial farms poses perhaps the greatest threat (Kamler et al. 2016). On a private ranch near Kimberley, sheep were found in 19% of Cape Fox scats, but in relatively low amounts per scat, indicating sheep were likely scavenged rather than predated upon (Klare et al. 2014). Domestic fowl are also a potential prey species, and even with them, there are suspicions of them being scavenged (Klare et al. 2014).
In some parts of the country, they are a low conflict species (Nieman 2018; Foster et al. 2022). In the Western Cape, at least in the Boland Mountain Complex area it is at least a low conflict species there (Nieman 2018). In a study from the Ottosdal area of the North West, out of 5, the mean intolerance score for the Cape Fox by farmers was low at 2.46 – understandably the jackal was much higher (Foster et al. 2022). Only 1 of 44 farmers perceived them to cause damage to livestock/poultry, as well as damage to livestock falling into their holes on occasion (Foster et al. 2022). The species is sometimes overlooked, and resembles a juvenile jackal, so this is to its detriment, as mistaken identity is a recurring means in which they are wrongly persecuted (Power 2014). Mistaken identity as a jackal may be an unintended consequence of jackal control.
There is however some evidence that the Cape Fox occasionally catches small livestock (Stuart 1981; Bester 1982), particularly those up to three weeks (Stuart 1981), though the frequency of this is low, and economic impact likely low too (Somers et al. 2015). However, heavy direct and indirect problem animal control measures do not seem to have had a major impact on the Cape Fox, even though they have resulted in local declines. For example, annual offtake resulting from problem animal control programmes averaged roughly 16% of the total population in the Free State up until 1985, with no obvious declines in overall numbers (Bester 1982).
Artificially elevated jackal abundances
Black-backed Jackal control operations may positively influence Cape Fox populations (Blaum et al. 2009). Nevertheless, range-wide declines in numbers of Cape Foxes were reported in the late 1980s, possibly associated with range-wide increases in numbers of Black-backed Jackals (Ginsberg & Macdonald 1990). In general, numbers of Cape Foxes and Black-backed Jackals are often inversely related (Blaum et al. 2009; Kamler et al. 2013), primarily due to the predation and spatial displacement of Cape Foxes by Black-backed Jackals (Kamler et al. 2012, 2013). Black-backed Jackals kill Cape Fox (Kamler et al. 2012) and suppressed their populations (Kamler et al. 2013). Annual Cape Fox mortality is 36 % (survival is the converse), and Black-backed Jackals predation is responsible for 71% for most mortalities (Kamler & Macdonald 2014).
In the North West Province, of the provincial reserves surveyed between 2011 and 2012, despite extensive camera trapping and spotlighting (Power et al. 2019), no Cape Foxes were detected on the following reserves: Barberspan, Botsalano, Mafikeng, Molemane eye (NWPG 2012). A year later, they were not found either on the edge of the Highveld and Kalahari, at the Bloemhof dam Nature Reserve and neither SA Lombard Nature Reserve. However, they were frequently recorded at the Molopo Nature Reserve and !Khamab Kalahari Reserve, which was unusual for the latter sites, as jackal abundance was equally numerous across all areas (NWPG 2013). It was hypothesised that hyper-protection of the reserves on the Highveld led to high abundances of Black-backed Jackal which may have excluded the Cape Fox, which is only ever reported on the edges of these reserves and regionally so. The areas on the outside of the reserves do carry out unrestricted control upon jackals.
It was unclear why the Cape Fox was still common on the protected landscapes of the Kalahari (Power 2-14; NWPG 2016; 2017), and it was hypothesised that the sandy Kalahari had more refugia from burrowing animals (i.e. Springhares), or at least softer ground for them to dig and thus coexist with Black-backed Jackals. So too, in the ecologically similar Tswalu Kalahari Reserve, it seems there is equitable coexistence of the Cape Fox and Black-backed Jackal (Smith et al. 2023). Similarly, between 2013 and 2015, extensive camera trapping took place in a part of the Western Cape Karoo and the Cape Fox were only detected on commercial small livestock farmlands (but see Schurch et al. 2021), while they were not detected on ecologically comparable private and state protected areas, while Black-backed Jackal had higher occupancies in these protected areas (Woodgate et al. 2023).
The consequence of mesopredator release across South Africa leads to high abundances of Black-backed Jackals (Nattrass et al. 2020), and in the absence of Leopard predation for instance (Kamler et al. 2020), probably results in artificially high densities of the species, which can be to the detriment of subordinate competitors, such as the Cape Fox.
Cape Fox captured on camera trap near Buhrmansdrif in the North West Province, just 15 km, south and north, of the Botsalano and Mafikeng Nature Reserves respectively. Interestingly, the species has to date not been recorded on these reserves.
Conservation
The Cape Fox has been recorded in many provincial and private nature reserves, as well as on game ranches in all South African provinces, although the species has a much more restricted range in Limpopo and KwaZulu-Natal (Lynch 1975; Stuart 1981; Rautenbach 1982; Rowe-Rowe 1992). In Eswatini, the species may occur in Nhlangano Nature Reserve in the southwest and pups have been successfully reared in Mlilwane Wildlife Sanctuary (Monadjem 1998). In the North West, there is likely low occupancy or absences in some reserves on the Highveld (NWPG 2012; 2013), though they are common in the Kalahari, where they occur on Molopo Nature Reserve and private !Khamab Kalahari Reserves (NWPG 2017), as well as the Northern Cape’s Tswalu Kalahari Reserve (Smith et al. 2023), and these may be one of the most important reserves for the species outside Kgalagadi Transfontier Park.
The Cape Fox is a Protected species on the national Threatened or Protected Species (TOPS) list (Government Gazette, No. 29657 of 2007), although it is still persecuted as a problem animal across much of its range in South Africa. Permits are required from authorities to kill Cape Foxes in problem animal control operations, but no protection measures are currently enforced, and at the present time, they do not appear to be necessary. However, the “holistic” approach to the management of damage-causing animals needs to be spread more widely to reduce bycatch (non-target species). This concept relates to the current understanding that not all individuals of a species are “problem animals”. A range of deterrents have been developed, such as Anatolian Sheep Dogs and loudspeakers broadcasting predator calls, as well as techniques to identify and remove only those animals that are responsible for “damage”. Evidence is beginning to mount that livestock guarding dogs both lower predation rates and reduce farm running costs in the long run (McManus et al. 2014), though the latter work aimed at Black-backed Jackals mostly.
As detailed, increases in Black-backed Jackal numbers may reduce local Cape Fox subpopulations. That said, because larger apex predators kill Black-backed Jackals (Kamler et al. 2007; 2020), the reintroduction of apex predators to more reserves may have a positive effect on local Cape Fox populations, via a reduction in jackal numbers, but more research is needed on this subject.
For isolated subpopulations, translocation should be used to improve gene flow. For example, SANParks managers need to supplement the Table Mountain National Park subpopulation by bringing in animals from other areas. However, there is no evidence that Cape Foxes occurred on the peninsula historically (Bolus 1921; Boshoff & Kerley 2001; Skead 2011), although it is considered to occur throughout the Cape Floristic Region today (Kamler et al. 2016), the peninsula is a minor salient of the greater area. There is a strong indication that they were introduced in the 1960s and 1970s (Kamler et al. 2016), and it is intriguing why it is not documented, as the authorities then were meticulous in record keeping for the carnivores (see Stuart 1981) Furthermore, there are potential negative impacts of a novel predator on local “naive” populations of prey species, for example Krebs’s Fat Mouse (Steatomys krebsii) (H. Langley pers. comm. 2014, In: Kamler et al. 2016), and several other endemic vertebrates on the Cape peninsula too (i.e. Golden moles even). Management of Cape Fox on Table Mountain National Park thus needs to be researched and considered carefully, especially given ever more restrictions on conservation resources.
Recommendations for land managers and practitioners:
Currently, there are no conservation plans for Cape Foxes and they do not require a management plan at this stage. Cape Foxes have persisted in farming areas despite direct and indirect persecution during control operations. Therefore, no management actions are required. If Cape Foxes become a conservation concern, then managers need to consider controlling Black-backed Jackal numbers, in addition to food, habitat, and stocking rate adjustments, to increase Cape Fox numbers (Blaum et al. 2009; Kamler et al. 2012). Private reserves may want to consider jackal control with the objective in mind to enhance Cape Fox status, though this would likely be too controversial for state-owned reserves to consider, especially given likely backlash from the public for such actions there.
Given the ecological similarity of the Cape Fox and the Black-footed Cat (Kamler et al. 2015), and the higher public profile of the Black-footed Cat, it may be that it can be an umbrella species in the arid lands, to which the species can benefit secondarily from (i.e. Black-footed Cat Working Group etc). Owing to the problem of accidental shooting of Cape Fox during jackal control (Power 2014), the De La Rey Branch of the South African Hunters and Game Conservation Association (SAHGCA) have proposed to resolve this with a training programme to upskill shooters to have an accompanying spotter whose role is species identification.
Research priorities:
The Cape Fox has been extensively studied in South Africa’s Free State Province (Lynch 1975; Bester 1982; Kok 1996; Kamler et al. 2013), as well as near the Kimberley area, along the Free State–Northern Cape border (Kamler et al. 2012, 2013; Kamler & Macdonald 2014; Klare et al. 2014). In contrast, there is little information for elsewhere within its range. Aspects such as diet and reproduction are well known, and recent studies have increased our knowledge about their socio-spatial ecology and behaviour in the wild (Kamler et al. 2012, 2013; Kamler & Macdonald 2014). However, research is needed on:
- The impacts and extent of persecution, both direct and as bycatch from snares, and use in traditional medicine.
- The role, if any, that this species plays in disease transmission.
- More broadly, the numbers and population trends of Cape Foxes throughout their range need to be determined.
- More research is needed on the effects of Black-backed Jackals and apex carnivores on Cape Fox subpopulations.
- Why does it seem that Cape Fox are immune to jackal abundance in the Kalahari basin, whilst elsewhere they seem to be significantly suppressed when at high jackal abundances.
- Continued population monitoring at Benfontein Game Farm, so as to be able to monitor population trends going forward.
- The consequences of apex predator reintroduction on mesopredators and Cape Foxes.
- To study by-catch detections of the species from the many camera trap surveys that take place across the country, with a particular focus on where the species is absent.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Create conservancies and install permeable fences between properties.
- Encourage apex predator conservation.
Bibliography
Bester, J. L. 1982. Die gedragsekologie en bestuur van die silwervos Vulpes chama (A. Smith, 1833) met spesiale verwysing na die Oranje-Vrystaat. M.Sc. Thesis, University of Pretoria.
Blaum N, Rossmanith E, Popp A, Jeltsch F. 2007. Shrub encroachment affects mammalian carnivore abundance and species richness in semiarid rangelands. Acta Oecologica (1):86-92.
Blaum, N., Tietjien, B., and Rossmanith, E. 2009. Impact of livestock husbandry on small- and medium-sized carnivores in Kalahari Savannah Rangelands. Journal of Wildlife Management 73: 60-67.
Bolus L. 1921. Mammals at Kirstenbosch. Veld & Flora. 7(1):19.
Boshoff, A.F. and Kerley, G.I.H. 2001. Potential distributions of the medium-to large-sized mammals in the Cape Floristic Region, based on historical accounts and habitat requirements. African Zoology 36: 245-273.
Boshoff AF, Kerley GI, Cowling RM. 2002. Estimated spatial requirements of the medium-to large-sized mammals, according to broad habitat units, in the Cape Floristic Region, South Africa. African Journal of Range and Forage Science. 19(1):29-44.
Bothma J du P. 1966. Food of the silver fox Vulpes chama. African Zoology 2: 205–221.
Bullock KL, Malan G, Pretorius MD. 2011. Mammal and bird road mortalities on the Upington to Twee Rivieren main road in the southern Kalahari, South Africa. African Zoology 46(1):60-71.
Coetzee, P. W. 1979. Present distribution and status of some of the mammals of Albany. Albany Divisional Council and Grahamstown Municipality, South Africa.
Dean WRJ, Milton SJ. 1991. Disturbances in semi-arid shrubland and arid grassland in the Karoo, South Africa: mammal diggings as germination sites. African Journal of Ecology 29: 11–16.
Foden WB, Young BE. 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Foster IB, McIntyre T, Haussmann NS. 2022. Understanding the relationship between farmers and burrowing mammals on South African farms: are burrowers friends or foes?. Pages 69-81. In: Kerr RB, Pendergrast TL, Smith BJ, Liebert J, Eds. Rethinking Food System Transformation. Springer, New York.
Ginsberg, J.R. and Macdonald, D.W. 1990. Foxes, Wolves, Jackals, and Dogs: An Action Plan for the Conservation of Canids. IUCN, Gland, Switzerland.
Gray A, Zhuk AS, Kamler JF, Edwards CW, Figueiro HV, Koepfli KP. 2024. The complete mitogenome of the Cape fox, Vulpes chama (Canidae: Mammalia). Frontiers in Ecology and Evolution. 12:1291268.
Hey D. 1964. The control of vertebrate problem animals in the province of the Cape of Good Hope, Republic of South Africa. In: Proceedings of the Vertebrate Pest Conference. 2(2): 57-70.
Kamler JF, Davies-Mostert HT, Hunter L, Macdonald DW. 2007. Predation on black-backed jackals (Canis mesomelas) by African wild dogs (Lycaon pictus). African Journal of Ecology 45: 667–668.
Kamler, J. F., Stenkewitz, U., Klare, U. Jacobsen, N. F., and Macdonald, D. W. 2012. Resource partitioning among cape foxes, bat-eared foxes, and black-backed jackals in South Africa. Journal of Wildlife Management 76: 1241-1253.
Kamler, J. F., Stenkewitz, U., and Macdonald, D. W. 2013. Lethal and sublethal effects of black-backed jackals on cape foxes and bat-eared foxes. Journal of Mammalogy 94(295-306).
Kamler, J. F., and Macdonald, D.W. 2014. Social organization, survival, and dispersal of cape foxes (Vulpes chama) in South Africa. Mammalian Biology 79(64-70).
Kamler JF, Stenkewitz U, Sliwa A, Wilson B, Lamberski N, Herrick JR, Macdonald DW. 2015. Ecological relationships of black-footed cats (Felis nigripes) and sympatric canids in South Africa. Mammalian Biology 80:122-127.
Kamler J, Palmer G, Cowell C, Mills MGL, Stuart C, Stuart M, Do Linh San E. 2016. A conservation assessment of Vulpes chama. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa
Kamler J, Loveridge A, O’Donnell H, Macdonald D. 2020. Effects of humans and large carnivores on the survival of black‐backed jackals. African Journal of Ecology.58(3): 557-562.
Kerley GI, Pressey RL, Cowling RM, Boshoff AF, Sims-Castley R. 2003. Options for the conservation of large and medium-sized mammals in the Cape Floristic Region hotspot, South Africa. Biological Conservation 112(1-2):169-190.
Klare U, Kamler JF, Macdonald DW. 2014. Seasonal diet and numbers of prey consumed by Cape foxes Vulpes chama in South Africa. Wildlife Biology 20: 190–195.
Kok, O. B. 1996. Diet composition of different carnivores in the Free State, South Africa. South African Journal of Science 92: 393-398.
Kraaij T, Milton SJ. 2006. Vegetation changes (1995–2004) in semi-arid Karoo shrubland, South Africa: effects of rainfall, wild herbivores and change in land use. Journal of Arid Environments 64: 174–192.
Lloyd PH, Millar JCG. 1984. A questionnaire survey (1969-1974) of some of the larger mammals of the Cape Province. Bontebok 3: 1-49.
Loock D, SwanepoeL L. 2019. Assessing local patterns in carnivore occupancy and richness by camera trapping: a monitoring of diversity at Verloren Vallei Nature Reserve, Dullstroom, Mpumalanga. Wildlife & Resource Association (WRA), Bloemfontein.
Loveridge AJ, Seymour-Smith JL, Sibanda AL, Mabika C, Ngosi E, Macdonald DW. 2019. First confirmed record of a Cape fox, Vulpes chama, in Zimbabwe. African Journal of Ecology 57(3).
Lynch, C. D. 1975. The distribution of mammals in the Orange Free State, South Africa. Navorsinge van die Nasionale Museum, Bloemfontein 3: 109-139.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
McManus JS, Dickman AJ, Gaynor D, Smuts BH, Macdonald DW. 2015. Dead or alive? Comparing costs and benefits of lethal and non-lethal human–wildlife conflict mitigation on livestock farms. Oryx 49: 687-695.
Monadjem A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41(2): 45-59.
Nattrass N, Conradie B, Stephens J, Drouilly M. 2020. Culling recolonizing mesopredators increases livestock losses: Evidence from the South African Karoo. Ambio 49:1222-1231.
Nieman WA. 2018. Culture, Conflict, and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the HumanWildlife Interface. Msc thesis, University of Stellenbosch, Stellenbosch.
Nieman, W.A., Leslie, A.J. and Wilkinson, A., 2018. The Use of Animals and Animal-Derived Constituents in African Traditional Medicine and Other Cultural Applications: Townships in the Western Cape Province. Culture, Conflict and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human-Wildlife Interface, p.215.
Nieman WA, Schultz BC, Wilkinson A, Leslie AJ. 2021. Stakeholders’ perceptions of mammal occurrence and abundance on agricultural properties bordering the Boland Mountain Complex, South Africa. African Zoology 56(2): 104-116.
NWPG (North West Provincial Government) 2012. Ngaka Modiri Molema District Mammal Report Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2013. Dr Ruth Segomotsi Mompata District Mammal Report Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2016. Report examining the status of small carnivores and the detriment of hunting them in the Molopo Bushveld of the Dr Ruth Segomotsi Mompati District of the North West. Department of Rural, Environment and Agricultural Development, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2017. The status of free ranging carnivores in Bokone Bophirima and impacts of hunting them. Department of Rural, Environment and Agricultural Development, North West Provincial Government, Mahikeng.
NWPG (North West Provincial Government) 2024 The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Panaino W, Lai S, Sliwa A. 2022. Interspecies teamwork: Evidence of interspecific foraging associations between Cape foxes and striped polecats in the southern Kalahari. African Journal of Ecology. 61(1):217-20.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Power RJ, Van Straaten A, Schaller R, Mooke M, Boshoff T, Nel HP. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Rowe-Rowe, D.T. 1992. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Schurch MP, McManus J, Goets S, Pardo LE, Gaynor D, Samuels I, Cupido C, Couldridge V, Smuts B. 2021. Wildlife-friendly livestock management promotes mammalian biodiversity recovery on a semi-arid Karoo farm in South Africa. Frontiers in Conservation Science 2:6. https://doi.org/10.3389/fcosc.2021.652415
Shortridge, G.C. 1942. Field notes on the first and second expeditions of the Cape Museums mammal survey of the Cape Province, and descriptions of some new subgenera and subspecies. Annals of the South African Museum 3: 281-291.
Skead, C.J. 2011. Historical incidence of the larger land mammals in the broader Western and Northern Cape provinces.In: Boshoff A.F., Kerley G.I.H, Lloyd P. (ed.), Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smith K, Venter JA, Peel M, Keith M, Somers MJ. 2023. Temporal partitioning and the potential for avoidance behaviour within South African carnivore communities. Ecology and Evolution. 13(8):e10380.
Smithers, R.H.N. 1968. A check list and atlas of the mammals of Botswana. The Trustees of The National Museums of Rhodesia, Salisbury, Rhodesia.
Somers M, Davies-Mostert H, Mzileni N, Swanepoel LH, Do Linh San E, Botha AJ, Tjelele J, Dumalisile L, Marnewick K, Tafani M, Hunnicut MA 2018. Biology, ecology and interaction of other predators with livestock. In Kerley, GIH, Wilson SL, Balfour D (eds), Livestock predation and its management in South Africa: A scientific assessment. Centre for African Conservation Ecology, Nelson Mandela University, Port Elizabeth.Pages 228-254.
Stuart C, Stuart M. 2004. Cape fox Vulpes chama. In: Sillero-Zubiri C, Hoffmann M, Macdonald DW. (ed.), Canids: Foxes, Wolves, Jackals and Dogs. Status Survey and Conservation Action Plan, pp. 189–193. Gland, Switzerland and Cambridge, UK.
Stuart C, Stuart M. 2008. Vulpes chama. IUCN 2013. IUCN Red List of Threatened Species.
Stuart, C. T. 1975. Preliminary notes on the mammals of the Namib Desert Park. Madoqua 4: 5-68.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Von Richter, W. 1972. Remarks on present distribution and abundance of some South African carnivores. South African Journal of Wildlife Research 2(1): 9-16.
Webster AB, Pretorius ME, Somers MJ. 2021. The determinants of mesocarnivore activity patterns in highveld grassland and riparian habitats. African Journal of Wildlife Research. 51(1):178-92.
Whiting MJ. Williams VL, Hibbitts TJ. 2016. A picture of health? Animal use and the Faraday traditional medicine market, South Africa. Journal of Ethnopharmacology. 179:265-73.
Woodgate Z, Drouilly M, Distiller G, O’Riain MJ. 2023. The effect of multi-use landscapes on mammal assemblages and its implication for conservation. Land.12(3):599. https://doi.org/10.3390/land12030599