
Bushveld Horseshoe Bat
Rhinolophus simulator

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Rhinolophus simulator – K. Andersen, 1904
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – simulator
Common Names: Bushveld Horseshoe Bat (English), Bosveldsaalneusvlermuis (Afrikaans)
Synonyms: Rhinolophus simulator Sanborn, 1936 ssp. alticolus
Taxonomic Note:
Although no subspecies were recognised by Meester et al. (1986), more recently two subspecies are tentatively recognised: Rhinolophus simulator simulator from eastern Africa, extending from Ethiopia to KwaZulu-Natal, South Africa; and R. s. alticolus from Guinea, Liberia, Nigeria and Cameroon (Cotterill & Happold 2013). Future research may confirm that R. s. alticolus is a distinct species (Cotterill & Happold 2013).
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.1, Lötter, C.A.2, Balona, J.3. & da Silva, J.M.4
Reviewers: Howard, A.1, Mutumi, G.5 & Bastian, A.6
Institutions: 1University of the Free State, 2Inkululeko Wildlife Services (Pty) Ltd, 3Gauteng and Northern Regions Bat Interest Group, 4South African National Biodiversity Institute, 5Weblyfe Biological Consulting; University of California, 6University of KwaZulu-Natal
Previous Assessors and Reviewers: Jacobs, D., Taylor, P., Richards, L.R., Cohen, L., MacEwan, K., Schoeman, C., Sethusa, T. & Monadjem, A.
Previous Contributors: Relton, C. & Raimondo, D.
Assessment Rationale
Listed as Least Concern in view of its sizeable distribution in the assessment region (extent of occurrence is estimated as 528,973 km²), its presumed large population, ability to utilise modified habitats for roosting, and, although declining in some parts of its range, the overall population within the assessment region is not suspected to be declining fast enough to qualify for listing in a threatened category. Savannah habitats within the assessment region are generally well protected.
Regional population effects: The range of this species extends to the moist savannah and bushveld regions of Zimbabwe and Mozambique. However, while dispersal rates are unknown, dispersal capacity is assumed to be low due to its low wing-loading (Cotterill & Happold 2013), and thus rescue effects are uncertain.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Taylor P, Lötter CA, Balona J & da Silva JM. 2025. A conservation assessment of Rhinolophus simulator. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species has a broad distribution across sub-Saharan Africa. In West Africa, there are records from the Wonegizi Mountains and Mount Nimba (Guinea and Liberia), from central Nigeria and Cameroon, which refer to the subspecies R. s. alticolus (Denys et al. 2013). There are many more records in East Africa, where this species ranges from Ethiopia and southern Sudan, southwards through Uganda, Kenya and Tanzania to Zambia, Malawi, Zimbabwe, Mozambique, southern Botswana to the eastern regions of South Africa, including Eswatini (ACR 2015). It has possibly been overlooked in southern Mozambique and eastern Botswana (Monadjem et al. 2020). In the assessment region, R. simulator occurs in the Limpopo, Mpumalanga, Gauteng, North West and KwaZulu-Natal provinces of South Africa as well as Eswatini (Monadjem et al. 2020). The extent of occurrence in the assessment region is 528,973 km².
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 40 m asl (Mondajem et al. 2024)
Elevation Upper Limit (in metres above sea level): 3,869 m asl (Mondajem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
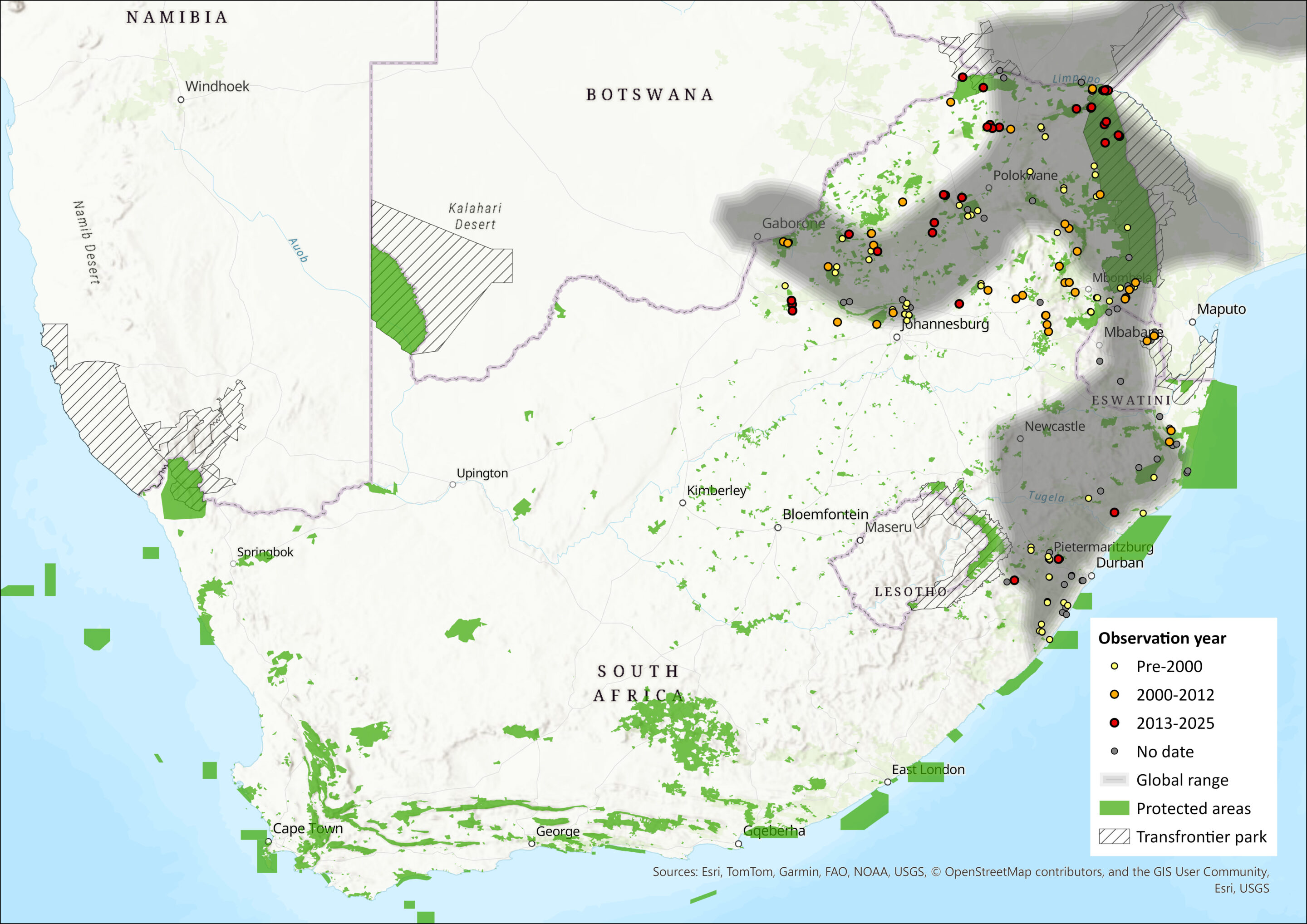 Figure 1. Distribution records for Bushveld Horseshoe Bat (Rhinolophus simulator) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Bushveld Horseshoe Bat (Rhinolophus simulator) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Botswana | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Using future climate Maxent models, Taylor et al. (2024) found that bat species with mesic savanna distributions on lower-elevation plateaux and highlands (including R. simulator) showed some contraction of their ranges in central Africa by 2070, but not as severe as predicted by the Köppen–Geiger climate models. In a review study on the impact and implications of climate change for bats, Sherwin et al. (2012) identified seven risk factors for bats encompassing three broad aspects: 1) biogeography – small range size, high latitude or high altitude range and a range occupying a geographic area likely to become water stressed; 2) foraging niche – frugivory and species restricted to aerial hawking; and 3) dispersal ability – species with restricted dispersal behaviour. These identified risk factors suggest that R. simulator might be impacted by climate change less significantly than congeners with smaller distribution ranges and more specific ecological requirements (such as Rhinolophus smithersi).
Population Information
This species is thought to be common, with 300 specimens examined in Monadjem et al. (2020). Colonies of up to 300 individuals have been recorded in caves and mine adits (Rautenbach 1982; Inkululeko Wildlife Services unpubl. data), but R. simulator also roosts singly or in smaller groups within rocky outcrops, caverns, culverts under the road, and buildings (Monadjem et al. 2020; Inkululeko Wildlife Services unpubl. data). There are limited population estimates available for this species, but they are considered locally common in Zimbabwe (Cotterill & Happold 2013).
Current population trend: Stable
Continuing decline in mature individuals? (Not specified)
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
With respect to molecular evidence, only phylogenetic studies have been conducted on this species (e.g., Dool et al. 2016; Demos et al. 2019; Taylor et al. 2024). Mitochondrial evidence suggests historical introgression between R. simulator and R. rhodesiae, where they overlap in distribution, resulting in low genetic differentiation between them (Dool et al. 2016; Taylor et al. 2024). A molecular investigation using a wider range of genetic markers may be able to better resolve the differentiation between these two species as alternative evidence (echolocation, morphological) support strong differentiation between these species (e.g., Dool et al. 2016; Taylor et al. 2018).
Based on the current distribution of the species, it is likely the species exists as a single metapopulation; however, this will need to be confirmed through a population genetic study. Considering the species is common and colonies can be occupied by several hundred individuals, and several colonies are known for this species, the total number of mature individuals for the species in the assessment region is thought to exceed 5000; therefore, the effective population size is also thought to exceed 500.
Habitats and ecology
This species typically occurs in caves within areas of moist savannah but has also been documented in bushveld regions adjacent to rivers and savannah woodland (for example, Brachystegia woodland in Zimbabwe) (Skinner & Smithers 1990), montane habitats and coastal mosaics (Cotterill & Happold 2013). It is most commonly associated with riparian forest and along wooded drainage lines (Monadjem et al. 2020). The availability of diurnal roosting sites, such as caves and mine adits, is an essential component of its habitat requirements (Taylor 1998). In KwaZulu-Natal it is often associated with Valley Bushveld regions, which occur along the lower reaches of east-flowing rivers (Taylor 1998). It is absent from deserts and rare in semi-arid savannahs (Cotterill & Happold 2013).
Rhinolophus simulator is an insectivorous clutter forager (Skinner & Chimimba 2005; Monadjem et al. 2020), feeding on a range of soft-bodied insects (Rautenbach 1997); it is known to hunt insect prey amongst dense vegetation and even on the ground (Rautenbach 1997). It feeds most commonly on Lepidoptera, but to a lesser extent, is also known to take Hemiptera, Isoptera, Orthoptera and Coleoptera (Whitaker & Black 1976; Findley & Black 1983; Schoeman 2006). This is a slow, but acrobatic, flier (Rautenbach 1997) with low aspect ratio, very low wing-loading and rounded wing tips (Cotterill & Happold 2013). In spring, females have been observed migrating to maternity roosts to give birth, before returning to the colony with their young (Wingate 1983). In KwaZulu-Natal, pregnant females were observed in September and October (Skinner & Chimimba 2005). A single, naked young is born in summer, usually in late November or early December (Rautenbach 1997). Young are weaned after a period of four to six weeks (Rautenbach 1997).
Ecosystem and cultural services: As this species is insectivorous, it may play an important role in controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Often, bats prey on the insect species that destroy crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can result in a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | Yes |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | Yes |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: Forearm mean = 4.51 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Forearm mean = 4.41 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: Parturition, which is highly synchronised, occurs from around mid-November (Cotterill 1998 cited in Monadjem et al. 2020).
Average Annual Fecundity or Litter Size: 1
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Unknown (likely to be limited).
Congregatory: Colonies may comprise up to 300 individuals (Rautenbach 1982 cited in Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that the species is traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Although there are no major threats to this species, it is locally threatened by habitat loss resulting from mining and agriculture. Extensive transformation of natural habitat, particularly in KwaZulu-Natal (Jewitt et al. 2015), is likely to be causing declines where the insect prey base is depleted as a result of loss of native vegetation or the use of pesticides. The development of the Musina – Makhado Special Economic Zone, will result in extensive clearing of habitat for industrial development and coal mining. Additionally, as a cave-roosting species, it may be threatened by human disturbance at roost sites as a result of traditional ceremonies and tourism activities. Implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021) could impact colonies that are roosting in old mine tunnels, if mitigatory approaches are not efficiently communicated. Future renewable energy development especially in association with mines in the Mpumalanga, Gauteng, North West, Limpopo, and KwaZulu-Natal provinces of South Africa may disturb roosts of this species. However, due to the clutter foraging habits of this species, a “Low” risk of fatality from wind turbines can be expected (MacEwan et al. 2020), if clutter plots are adequately managed.
Conservation
This species is well protected and present in a number of protected areas, including Kruger National Park, iSimangaliso Wetland Park, Madikwe Nature Reserve, Pilanesberg Game Reserve, Oribi Gorge Nature Reserve and Songimvelo Nature Reserve. It is likely to benefit from protected area expansion, as well as the protection of key roost sites.
Recommendations for land managers and practitioners:
- Renewable energy development must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (see MacEwan et al. 2020), as well as implement mitigatory strategies and measures along potential corridors surrounding roosts
- Reduce pesticide use in agricultural landscapes and encourage regulations on types of pesticides to be used.
Research priorities:
- Taxonomic revision of the species and tentative subspecies to determine whether R. s. alticolus is a distinct species and to further determine its genetic relationship to R. rhodesiae.
- Population size and trends.
- Systematic monitoring to identify key roost sites.
Encouraged citizen actions:
- Limit disturbance to roost sites.
- Citizens can assist in the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution. This species is easily mistaken with R. darlingi, and can only be reliably separated by the position and presence of the minute first upper premolar (Monadjem et al. 2010).
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Cotterill, F.P.D. and Happold, M. 2013. Rhinolophus simulato Bushveld Horseshoe Bat. Pages 351–353 in Happold M, Happold DCD, editors. Mammals of Africa. Volume IV: Hedgehogs, Shrews and Bats. Bloomsbury Publishing, London, UK.
Demos, T.C., Webala, P.W., Bartonjo, M. and Patterson, B.D. 2018. Hidden Diversity of African Yellow House Bats (Vespertilionidae, Scotophilus): Insights From Multilocus Phylogenetics and Lineage Delimitation. Frontiers in Ecology and Evolution 6.
Denys, C., Kadjo, B., Missoup, A.D., Monadjem, A. and Aniskine, V. 2013. New records of bats (Mammalia: Chiroptera) and karyotypes from Guinean Mount Nimba (West Africa). Italian Journal of Zoology 80: 279–290.
Dool, S.E., Puechmaille, S.J., Foley, N.M., Allegrini, B., Bastian, A., Mutumi, G.L., Maluleke, T.G., Odendaal, L.J., Teeling, E.C. and Jacobs, D.S. 2016. Nuclear introns outperform mitochondrial DNA in inter-specific phylogenetic reconstruction: lessons from horseshoe bats (Rhinolophidae: Chiroptera). Molecular phylogenetics and evolution, 97, pp.196-212.
Findley, J.S. and Black, H. 1983. Morphological and dietary structuring of a Zambian insectivorous bat community. Ecology 64: 625–630.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association. South Africa.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2010. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Rautenbach, I.L. 1982. Mammals of the Transvaal. No. 1, Ecoplan Monograph, Pretoria, South Africa.
Rautenbach, N. 1997. Bushveld Horseshoe Bat Rhinolophus simulator. Page 100 in Mills G, Hes L, editors. The Complete Book of Southern African Mammals. Struik Publishers, Cape Town, South Africa.
Schoeman, M.C. 2006. The relative influence of competition and coevolution on the community structure of insectivorous bats in southern Africa. Ph.D. Thesis. University of Cape Town.
Sherwin, H.A., Montgomery, W.I., and Lundy, M.G. 2012. The impact and implications of climate change for bats. Mammal Review. 2012. 10.1111/j.1365-2907.2012.00214.x.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Skinner, J.D. and Smithers, R.H.N. 1990. The Mammals of the Southern African Subregion, second edition University of Pretoria, Pretoria. South Africa.
Taylor, P. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., Kearney, T.C., Clark, V.R., Howard, A., Mdluli, M.V., Markotter, W., Geldenhuys, M., Richards, L.R., Rakotoarivelo, A.R., Watson, J., Balona, J., Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate‐driven diversification and a buffer against future climate change in bats. Global Change Biology, 30(6), p.e17344.
Whitaker, J.O. and Black, H. 1976. Food habits of cave bats from Zambia, Africa. Journal of Mammalogy 57: 199–204.
Wingate, L. 1983. The population status of five species of Microchiroptera in Natal. M.Sc. Thesis. University of Natal.
