Bryde’s Whale
Balaenoptera edeni brydei

2025 Red list
status
B. edeni brydei (inshore)

2025 Red list
status
B. edeni brydei (offshore)
Regional Population
Trend
Change compared
to 2016
Overview
Balaenoptera edeni brydei – (Olsen, 1913)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BALAENOPTERIDAE – Balaenoptera – edeni – brydei
Common Names: South African Bryde’s Whale, Bryde’s Whale Complex, Common Bryde’s Whale, Tropical Whale (English), Bryde se Walvis (Afrikaans)
Synonyms: Balaenoptera brydei, Olsen, 1913
Taxonomic Note: The taxonomic status of this species remains unresolved. However, recent mitochondrial DNA evidence supports that the offshore subpopulation is Balaenoptera edeni brydei. Additionally, the inshore subpopulation is more closely related to B. edeni brydei than to B.edeni edeni (Penry et al. 2018). New genetic work shows that both the inshore and offshore subpopulations are ‘brydei’ and fall outside of the ‘edeni’ group in phylogenetic trees (see Penry et al. 2018).
Internationally and regionally, the taxonomic status and population dynamics of the “Bryde’s Whale complex” remains unresolved. Globally, Bryde’s Whales, also known as Eden’s Whale, are currently classified as a single species (Balaenoptera edeni (Committee on Taxonomy, 2024), with two provisional subspecies, B. edeni edeni and B edeni brydei, which refer to the small coastal form and larger pelagic form, respectively (Kershaw et al. 2013, Rosel and Wilcox, 2014). Bryde’s Whales belong to the family Balaenopteridae (rorquals), of which there are nine accepted species (Committee on Taxonomy, 2024), the most recent being Rice’s Whale, previously referred to as the Gulf of Mexico Bryde’s Whale (Rosel et al. 2021). Within the assessment region, Best (1977) described two allopatric subpopulations (inshore and offshore) from South Africa that differ from each other in body size, migrations, reproductive seasonality, fecundity and prey types (Best 2001). These two subpopulations were subsequently referred to as B. edeni (Anderson 1878) and B. brydei (Olsen 1913) respectively, pending further investigation. Later comparisons suggest that Olsens’ (1913) description was not specified correctly (Best 2001; Yamada et al. 2008). It is currently accepted and supported by molecular work (Penry et al. 2018), that Olsen’s description of B. brydei from South Africa was based on a combination of features from both the coastal (inshore) and pelagic (offshore) subpopulations.
The general acceptance and use of the common name “Bryde’s Whale” for B. edeni has confused matters further, as has the discovery of at least two eco-types/ allopatric subpopulations within the approximate same geographic locations; for example, off South Africa, southwest Japan, and Oman (Best 1977; Kato et al. 1996 Mikhalev 2000). Comparisons of mitochondrial DNA (mtDNA) control region sequences from Bryde’s Whales sampled off South Africa with those already published for other Bryde’s Whale subpopulations and closely related species (Junge 1950; Omura et al. 1981; Árnason & Best 1991; Árnason et al. 1993; Sasaki et al. 2005, 2006), identified the South African offshore subpopulation (SE Atlantic stock) as Balaenoptera edeni brydei (ordinary type) (Penry et al. 2018). Notably, the inshore subpopulation was more similar to B. edeni brydei (Olsen 1913) than to B. edeni edeni (Anderson, 1878), with phylogenetic analyses grouping both the inshore and offshore subpopulation outside of the B. edeni edeni clade (Penry et al. 2018). Furthermore, ten fixed differences between the mtDNA control region sequences from the inshore and offshore subpopulations supported 100% diagnosability as subspecies (Penry et al. 2018). The morphological and genetic analyses of additional specimens of the reputed B. edeni (sensu Sasaki et al. 2006) from more localities is needed, as too is the identification and description of a type specimen for B. brydei (from South Africa, sensu Olsen 1913), before the taxonomy of the “Bryde’s Whale complex” can be resolved. Provisionally we use Balaenoptera edeni brydei for this assessment, while recognising that additional molecular markers and a larger sample size incorporating representative Bryde’s Whale samples from more geographic areas are needed (Constantine et al. 2018). Thus, following taxonomic revision, reassessment may be necessary.
Red List Status:
B. edenibrydei (inshore) – VU – Vulnerable, D1
B. edenibrydei (offshore) – DD – Data Deficient
(IUCN version 3.1)
Assessment Information
Assessors: Penry, G.1,2, Plön, S.3 & da Silva, J.4
Reviewer: Vermeulen, E.2
Contributor: Patel, T.5
Institutions: 1Institute for Coastal and Marine Research, Nelson Mandela University, 2Mammal Research Institute, University of Pretoria, 3University of Cape Town, 4South African National Biodiversity Institute, 5Endangered Wildlife Trust
Previous Assessors & Reviewers: Penry, G., Findley, K. & Best, P.
Previous Contributors: Relton, C., Child, M.F., Elwen, S., Meyer, M., Oosthuizen, H., Plön, S. & Collins, T.
Assessment Rationale
Although the Bryde’s Whale complex may be split into a number of species or subspecies pending further genetic analyses, this assessment differentiates between an inshore and offshore subpopulation in South African waters, focusing on the inshore subpopulation. The coastal, inshore subpopulation is estimated to have fewer than 1,000 mature individuals based on data from a 1982 line-transect, ship-based survey that covered the known Southern Hemisphere summer distribution of the subpopulation and resulted in an estimate of 582 ± 184 animals, where inclusion of secondary sightings might have increased this estimate by 29% (Best et al. 1984). A mark-recapture study in Plettenberg Bay conducted from 2005 to 2008 produced abundance estimates of between 150 and 250 individuals (Penry 2010), but how representative this is of the total sub population is unknown. Based on the assumption that the subpopulation size is fewer than 1,000 mature individuals, we list the inshore subpopulation as Vulnerable D1.
There are currently no assessments available for the offshore subpopulation (SE Atlantic), which was exploited by land-based whalers from at least 1911 to 1967 as an integral part of the catch, and more extensively between 1969 and 1976 by illegal unregulated pelagic whaling. Being largely restricted to the southern African shelf edge from Cape Point to at least the equator on the west coast; its environment is likely to have been exposed to oil and gas exploration activities, increased ship traffic, commercial fishing, and climate change effects for the last three decades. Given the suspected effects of seismic surveys on fish, it is possible (but unsupported by any data) that prey availability to this subpopulation may have been adversely affected. There are no abundance estimates, either before or after exploitation, or indications of trend and the offshore subpopulation must remain listed as Data Deficient.
This regional assessment echoes the global situation whereby the taxonomy (number and identity of species) is not yet resolved. If there is more than one species (strongly supported by a growing body of evidence), the less abundant species may be threatened. Taxonomic resolution and current estimates of population size and trends for both the inshore and offshore subpopulations are required and should result in a reassessment once such data are available.
Regional population effects: The inshore subpopulation is non-migratory and occurs year-round over the continental shelf of South Africa (Best 2001); the offshore subpopulation, occurring off the west coast of Southern Africa, appears to migrate northwards in autumn (Best 2001). There are no apparent barriers to the dispersal of either subpopulation, although prey distribution, habitat type, and the strong boundary currents (Agulhas and Benguela) off the southern African east and west coasts are likely to have an important influence. Similar oceanographic features occur off Japan, whereby the Kuroshio Current appears to act as a barrier between coastal and pelagic populations of Bryde’s Whales (Kato et al. 1996).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Penry G, Plön S & da Silva JM. 2025. A conservation assessment of Balaenoptera edeni brydei. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
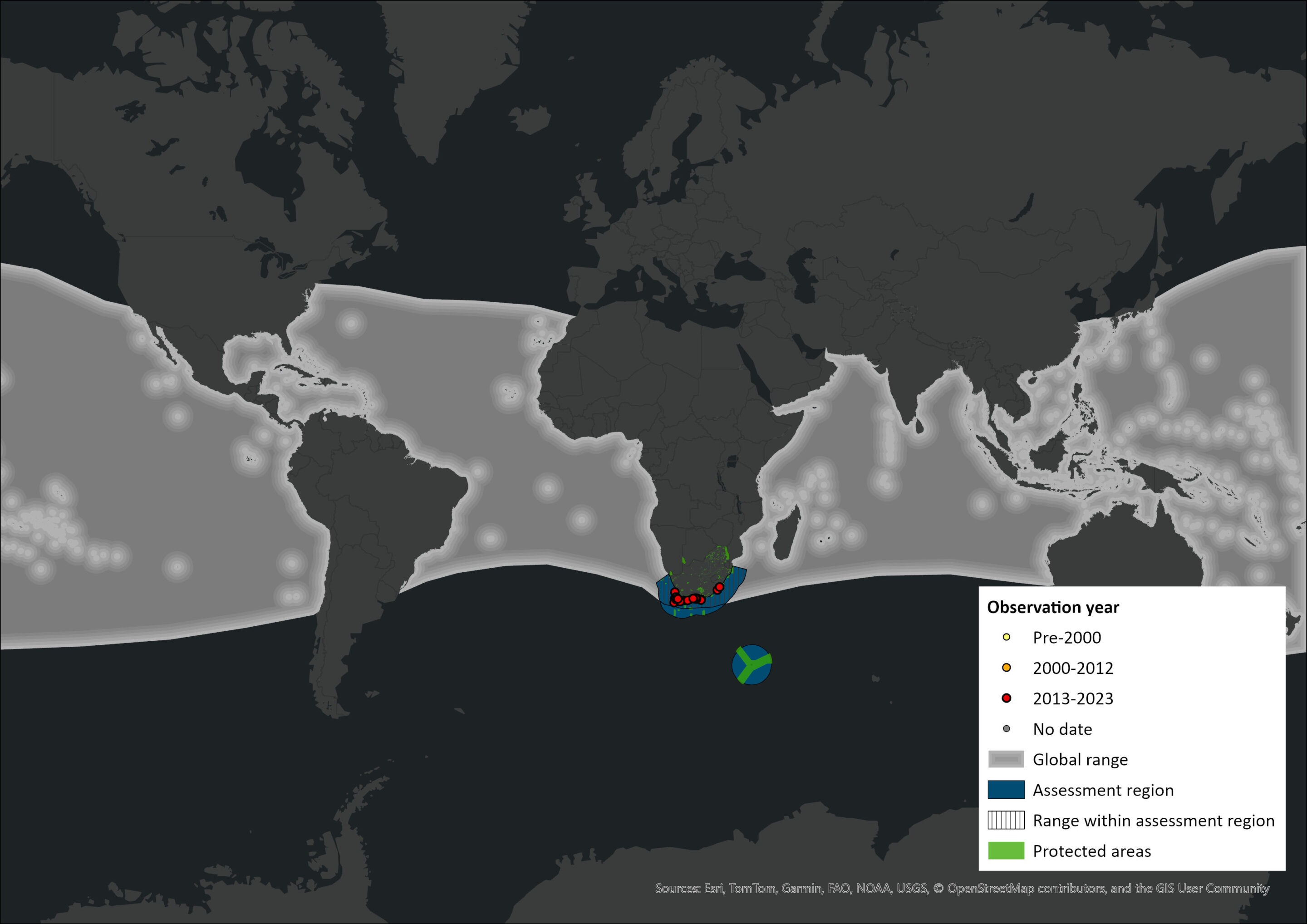
The known distribution of the inshore subpopulation is within approximately 20 nautical miles of the coast, over the continental shelf between Port St Johns on the east coast and Lambert’s Bay on the west coast; this roughly corresponds to the 200 m depth contour. The inshore subpopulation displays seasonal shifts in distribution, with most sightings occurring on the south-east coast of South Africa, between Cape Agulhas (20°E) and East London (~28°E) in summer and autumn (Best et al. 1984; Penry et al. 2011). Previously, they were shown to move up the west coast in winter; however, dedicated observations during the late 1990s recorded lower numbers of sightings than those reported during commercial whaling in the 1960s (Best 2001) when high numbers were caught further north on the west coast in autumn and winter (Best 1977). This shift may reflect changes in the availability of small-pelagic fish, with a general south and eastward shift in the distribution of sardine (Sardinops sagax) and anchovy (Engraulis capensis), their main prey (Roy et al. 2007; Coetzee et al. 2008). Bryde’s Whales also follow the annual northward migration of small-pelagic fish along the east coast of South Africa (commonly known as the “Sardine Run”) in winter, but the number of individual whales involved, and the geographic limits of their movements remain unclear (Caputo et al. 2017).
Sightings are extremely rare in Namibian waters (pers comm S Elwen); the reasons for this are not understood. Analyses of the mtDNA control region of two individuals (one from Walvis Bay, Namibia and one from the southwest Indian Ocean, south of Madagascar) sampled from outside the currently accepted distributional range revealed an identical haplotype to individuals sampled on the south coast of South Africa, suggesting that they possibly have a wider distribution than is currently understood (Penry et al. 2018). Further sampling and genetic analyses are needed to fully understand their distributional range limits. Within South African coastal waters, inshore Bryde’s Whales were encountered more frequently during summer and autumn in Plettenberg Bay (south-east coast of South Africa), with peak encounter rates in April (Penry et al. 2011). These findings support those of Best (2001) who described a seasonal movement of inshore Bryde’s Whales down the west coast towards the Agulhas Banks in spring.
The offshore subpopulation ranges from 34°S to equatorial latitudes on the west coast of Southern Africa and was reported to occur off the west coast of South Africa in the austral summer and off Gabon in winter. All recorded catches were made within 300 nautical miles of the coast. The geographical ranges of the two subpopulations may occasionally overlap, but the level of genetic differentiation between them suggests that they do not interbreed (Penry et al. 2018). This new evidence further supports the allopatric separation of the two subpopulations in reproduction, feeding and seasonal migrations (Best 1977).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Bryde’s Whale (Balaenoptera edeni brydei) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Climate change
Climate change affects cetaceans through a reduction in prey availability and a shift in the distribution of prey species (Kebke et al. 2022). This in turn results in a change in distribution range. In addition to affecting distribution range and abundance, climate change can also impact migration timing and behaviour (Kebke et al. 2022). With the reduction in food availability comes a change in body condition, leading to a decline in reproductive success. However, these impacts are all species-specific. Specific research needs to be carried on the impact of climate change on Bryde’s Whale.
Population information
Within the assessment region, two subpopulations occur; the South African inshore (coastal) and the southeast Atlantic (offshore/pelagic) subpopulation. For the inshore subpopulation, a range-wide line-transect survey estimated approximately 582 ± 184 individuals in the early 1980’s (Best et al. 1984). Thirty years later, Penry (2010) used photo-identification data collected in Plettenberg Bay to estimate the abundance of Bryde’s Whales using the south and east coast of South Africa. Mark-recapture analyses produced estimates of between 158 (SE=17) and 248 (SE = 93) individuals for this area between 2003 and 2007; however, it is acknowledged that the study was spatially restricted (Penry 2010). Molecular analyses indicate that the resident South African inshore subpopulation is significantly differentiated from the migratory offshore subpopulation, which is thought to have higher population numbers (Best 1996; Penry et al. 2018).
Between 1911 and 1967, more than 2,000 Bryde’s Whales were recorded as having been caught by whaling operations off the Cape region of South Africa. This included 1,300 individuals caught between 1947 and 1967 (IWC 2006a), which are expected to have been from the inshore subpopulation (IWC 1980a, 1980b). Unfortunately, no estimates are currently available for the south-east Atlantic; however, between 1969 and 1976 the majority of the 2,536 “Sei” Whales caught by the pirate whaling ship Sierra in the South Atlantic are assumed to have been Bryde’s Whales from the offshore (SE Atlantic) subpopulation (1980b; Best 1996). This suggests that the offshore subpopulation was probably more abundant than the inshore subpopulation.
Bryde’s Whales were not consistently distinguished from Sei Whales in International Whaling Statistics, but in some cases a breakdown of “Sei” Whale catches into the two species can be determined from original records, or approximated, based on current knowledge of the geographical and seasonal occurrence of Sei and Bryde’s Whales, or from the compositions of later catches in the same area and season (IWC 1997, 2006b). Abundance estimates of Bryde’s Whales in the Southern Hemisphere are in urgent need of reassessment. Historic estimates of abundance revealed the following approximations: in the southern Indian Ocean – 13,854; in the western South Pacific – 16,585; and in the eastern South Pacific – 13,194 (IWC 1981). These estimations were not founded on recently approved survey methods, and consequently a “zero catch limit” was set by the IWC for all Bryde’s Whales, until a satisfactory estimation of population size has been established (IWC 1983).
Population Information
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Number of mature individuals in subpopulation: <1,000 (inshore); unknown (offshore)
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: There are no known subpopulations of either the inshore or offshore populations.
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Within the assessment region, two subpopulations currently exist; the South African inshore (coastal) and Southeast Atlantic (offshore/pelagic) subpopulations. These two subpopulations are defined based on significant differences in their morphometrics, diet, reproductive cycles, distributions, and most recently, mtDNA control region sequence analyses and phylogenetic comparisons (Best 1977, 2001; Penry et al. 2018).
While abundance data are limited, the South African inshore subpopulation is estimated at less than 1000 individuals (unpublished data; previous published numbers are 582 ± 184; Best et al. 1984). Because this subpopulation is thought to exhibit non-migratory behaviour and remains year-round over the continental shelf of South Africa (Best 2001), rescue effects from other subpopulations are not expected to occur, but this needs further investigation. Based on this information, the estimated effective population size is between 100-300 individuals (assuming an Ne/Nc between 0.1-0.3).
Population size for the offshore subpopulation is unknown; however, given that they are migratory, moving northwards in autumn (Best 2001), their population size and rescue effect is thought to be high.
Habitats and ecology
Bryde’s Whales are presumably able to satisfy their nutritional and reproductive needs within their warm, temperate distribution, freeing them from the need to make extensive latitudinal migrations (Bannister 2002). Although pelagic subpopulations (for example, the SE Atlantic subpopulation) undertake limited migrations towards the equator in winter and higher latitudes in summer, coastal subpopulations do not migrate and their movements are primarily alongshore, most likely governed by the distribution of their prey. Year-round occurrence has been reported from the coastal areas of south-western Japan, south-eastern Brazil and South Africa. South African inshore Bryde’s Whales feed at a constant and high rate throughout the year (Best 1967) and feeding events commonly involve multi-species aggregations (Best 2007; Penry et al. 2011). The inshore subpopulation is dependent on year-round prey availability and feed predominantly on small pelagic fish, such as anchovy and sardine.
The South African inshore subpopulation is resident over the Agulhas Bank (Best 2007), but shows a seasonal shift in distribution, with the majority of sightings on the southeast coast of South Africa, between Cape Agulhas and East London in the austral summer and autumn (Best 2007; Penry et al. 2011). Off South Africa, aggregation size of Bryde’s Whales was positively correlated with the occurrence of feeding behaviour. During winter there are increased sightings of Bryde’s Whales further north along the East Coast, frequently in association with Common Dolphins (Delphinus delphis) and Cape Gannets (Morus capensis) (Best et al. 1984; Best 2001; O’Donoghue et al. 2010; Caputo et al. 2017). This appears to coincide with the annual northward migration of sardines into KwaZulu-Natal waters (Fréon et al. 2010; Penry et al. 2011). In general, Bryde’s Whales do not display reproductive seasonality. However, this is more apparent in the offshore/pelagic subpopulation. Penry et al. (2011) found no seasonality in the occurrence of calves off South Africa. The inshore subpopulation is polyoestrous and has a high frequency of ovulation, possibly due to the year-round abundance of food (Best 2007). The period for lactation is not known.
The offshore subpopulation occurs on the edge of the continental shelf and migrates seasonally between the equator in the Southern Hemisphere winter and approximately 34°S in summer (Best 1996). Breeding can occur year-round but is more seasonally defined than in the inshore subpopulation, peaking in autumn/early winter. The offshore subpopulation is also less piscivorous than the inshore subpopulation, with euphausiids being regularly consumed and the fish eaten being more mesopelagic in nature: there also seems to be a marked seasonality in prey type, with euphausiids being relatively unimportant in autumn and winter, but predominating in summer (Best 2001). Because of its year-round presence in temperate and tropical waters, this subpopulation is subjected to an unusually high level of attack by what are believed to be cookie-cutter sharks (Isistius spp.), to such an extent that the scars eventually cover large areas of the flanks and belly, often obscuring the whale’s natural pigmentation (Best 1977).
Ecosystem and cultural services: Marine mammals integrate and reflect ecological variation across large spatial and long temporal scales and are therefore prime sentinels of marine ecosystem change; migratory mysticete whales may be used to investigate broadscale shifts in ecosystems (Moore 2008). The South African inshore Bryde’s Whale is the largest, resident predator in South African coastal waters. They are principally dependent on small pelagic fish stocks for their prey, and suitable/ safe habitat for breeding. Unlike most other large baleen whales that migrate between disparate feeding and breeding areas, the South African inshore subpopulation may have largely sympatric feeding and breeding areas within its range. Ecosystem services include revenue through tourism, food web stabilisation and ecosystem indicators.
Life History
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? Yes
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Marine
General Use and Trade Information
Bryde’s Whales were possibly less impacted by commercial whaling than some other large whale species owing to their tropical and subtropical distribution, although accurate stock assessments are lacking in most areas and are not straightforward given the (often) doubtful catch records involved. There has been no known direct whaling of this species in South African waters since 1976; however, the level of bycatch in coastal fishing gear has increased in the last two decades (Meÿer et al. 2011; Segre et al. 2022).
Bryde’s Whales are listed in Appendix I of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES), which prohibits commercial international trade (Cites.org).
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
Both inshore and offshore subpopulations were subjected to some whaling in the past, but no specific estimates of depletion exist. The Bryde’s Whale was the incidental beneficiary of IWC area restrictions on factory ship whaling that were originally designed to protect the low-latitude winter breeding grounds of other baleen whale species (Tønnessen & Johnsen 1982). This benefit was lost to the offshore subpopulation in 1969 when an illegal and unregulated pelagic operation commenced whaling in the southeast Atlantic (Best 1996). No direct takes are known in the area since 1976.
Like most cetaceans, Bryde’s Whales are susceptible to becoming bycaught in active or discarded (ghost) fishing gear. Between 2014 and 2020, an unusually high rate of entanglement was reported, with 17 Bryde’s Whales from the inshore subpopulation being entangled in fishing gear. Of these, 10 were fatal and seven of those were due to entanglement in bottom-mounted gear for an exploratory octopus fishery (Segre et al. 2022). A study was conducted to determine the reasons for this gear being a particular hazard for inshore Bryde’s whales. It was found that the whales perform high-speed chases along the seafloor and then lunge upwards into the water column, mouths open, to engulf prey (Segre et al. 2022). This explained why most fatal entanglements involved gear (rope) through the mouth. Subsequently, the South African Minister of Forestry, Fisheries and the Environment (DFFE) implemented a moratorium on the fishery and new regulations were issued that required gear changes, including compulsory use of sinking line and acoustic releases for buoy lines. At the time of this assessment, there have been no further entanglements; however, only one permit holder has redeployed their amended gear, and it remains to be seen whether the measures are effective for preventing whale entanglements.
The unique behaviour described by Segre et al. (2022) could put inshore Bryde’s Whales at risk of entanglement in similar coastal bottom-mounted gear and it is strongly recommended that species-specific behavioural data is included in impact assessments when new economic activities are explored in the marine environment. Vessel strikes are not widely reported in South African waters but are likely to increase in frequency as coastal tourism, port expansions, and marine traffic are encouraged to grow under the government’s National Development Plan, Oceans Economy Lab, targets (National Planning Commission, 2012).
The inshore subpopulation potentially competes with commercial fisheries for sardine and anchovy, their main prey species (sensu Clapham et al. 1999). Given the suspected effects of seismic surveys on fish (Gordon et al. 2003; Koper & Plön 2012), it is possible that prey availability for this subpopulation may have been adversely affected. Disturbances from ecotourism ventures, such as the permitted boat-based whale watching industry and during the “Sardine Run”, while Bryde’s Whales are feeding on bait balls (roughly spherical, tightly packed formations of fish), have been reported. Climate change and natural environmental stochasticity may exacerbate existing prey depletions (Burns & Baker 2000).
In summary, the potential reduction in prey resources, their inherent low abundance and genetic isolation, and entanglement in fishing gear are the greatest threats to the inshore subpopulation. Threats to the offshore (SE Atlantic) subpopulation cannot be identified owing to insufficient data but are likely to be similar to other pelagic balaenopterids and include ship strikes, reduced prey availability, disturbance from increasing underwater noise, pollution (chemical and physical), and entanglement in active fishing gear or ghost gear.
Current habitat trend: Decline in habitat quality due to climate change, fishing pressure, increasing noise, chemical, and physical pollution, competition with commercially important small-pelagic fisheries, and increasing risk of injury from collisions with vessels.
Conservation
Bryde’s Whales are protected under several international policy instruments, including being listed on Appendix I of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) – and are subject to international agreement/trade controls, thus international trade of Bryde’s Whale material is currently illegal. They are listed on Appendix II of the Convention of Migratory Species (CMS), and the International Whaling Commission recognises the Bryde’s Whale as “protected since the moratorium apart from some special permit catches in the North Pacific”.
In the assessment region, the “Sardine Run” along the southeast coast of South Africa is a critically important natural phenomenon for many marine top predators. In 2020, the ‘Southern Coastal and Shelf Waters of South Africa’ Important Marine Mammal Area (IMMA) was declared based on the importance of this region for several marine mammal species, and specifically the recognition of the Sardine Run as a spatially and temporally important feeding area for Bryde’s Whales, odontocetes, and Cape fur seals (IUCN_MMPATF, 2020). The dense shoals of sardine during this migration provide relatively easy access to a valuable, high-protein food source. Thus, the inshore subpopulation is heavily dependent on this annual phenomenon and fisheries should be regulated accordingly.
As such, the main intervention for this species is to estimate abundance and unravel the discrepancy around the taxonomy of the offshore and inshore subpopulations. Subsequently, the inclusion of the species’ energetic requirements (based on the findings of Best et al. 1984) in setting Total Allowable Catch limits (TACs) for the pelagic fishery would benefit the South African inshore population. Additionally, an ecosystem approach to fisheries management is necessary. This is expected to also benefit many other marine species. Additionally, the identification of specific breeding and feeding areas of the inshore subpopulation may prove beneficial to its conservation. However, habitat utilisation is poorly known for the offshore subpopulation, therefore no key areas can be identified.
Recommendations for managers and practitioners:
- Critical habitats need to be identified (breeding areas, important temporal and spatial feeding patterns and/or areas): satellite tagging could assist here.
- Systematic monitoring: Ship-based line transect sampling to establish population size and trends. This includes the need for a range-wide abundance estimate.
- Sightings submissions: Continual update of the photo-ID catalogue, in particular maximising the amount of data by involving commercial whale watching operators along the coast. Establishment of a national online data-basing system for citizen science, whale watch/sardine run operators, and research groups to upload sightings and photo-ID data with the aim of producing a population estimate every two years to identify trends in abundance.
- Regulation of the “Sardine Run” diving operators and their activities. Several anecdotal reports are that divers disrupt bait balls resulting in the predator dispersing without securing a meal. Following formal investigation into the severity of this threat, stricter permitting conditions and clear codes of conduct should be drawn up and implemented.
Research priorities: Current research priorities include:
- There is an urgent need for current size estimates for both the inshore and offshore subpopulations throughout their known distributional ranges. Ship-based line transect surveys are recommended for achieving the most reliable range-wide estimates. For the inshore subpopulation, mark-recapture abundance estimates using photographic-identification and individual genotypes are underway. Satellite tagging of individuals throughout their range to determine movements and spatial distribution of individuals.
- More detailed genetic studies to clarify the level of separation between the subpopulations within the assessment region (underway).
- Determination of their dietary composition using a combination of data on stomach contents from stranded animals and stable isotope analyses from biopsy sampled individuals and museum specimens. Are they able to adapt to changing prey availabilities? (underway).
- Global comparison of all available molecular data (mtDNA and nDNA) on Bryde’s type Whales for the purposes of taxonomic resolution of the complex. This will require collaboration, possibly with one person identified as the project coordinator by the IUCN/IWC/Society for Marine Mammalogy (SMM) taxonomy committee.
Specific priorities for the inshore subpopulation:
- The extent of their distributional limits: distribution may extend further up the west coast, into Namibian waters (satellite tagging or ship-based survey of extended range).
- Movements: recent photo-ID work suggests surprisingly limited coastwise mixing between bays (c.500 km apart); and satellite tagging could clarify the level of individual site fidelity, distributional range, and important habitats further offshore (e.g. the Agulhas Banks).
- Range-wide ship-based line transect surveys replicating those completed over 30 years ago (Best et al. 1984) are needed for a valid comparison of current subpopulation size estimates.
- Routine stable isotope analyses to detect trophic level changes in their dietary contribution.
- Data types – Satellite tagging, biopsy sampling for genetic and stable isotope studies, body condition and energetic requirements (suction-cup attached data loggers), ship-based line transect survey, and photo-ID data.
Specific priorities for the offshore subpopulation:
- Estimate abundance and trends.
- Genetic differentiation from other pelagic populations.
- Identification of threats and threat severity.
- Identification and collection of a full skeleton to be used as the B. brydei type specimen.
Encouraged citizen actions:
- Uploading location sightings to virtual museum platforms will help in determining the spatial and temporal distribution of the subpopulations. Commercial whale-watching and Sardine Run operators must be encouraged to submit records of location, numbers and ID photos.
- Reduce vessel speed in bays and harbours.
- When participating in whale/dolphin watching tours, only support permitted operators and ensure regulations are followed.
- Report any stranding, entanglement or ship strikes to the relevant local authorities.
Bibliography
Árnason Ú, Best PB. 1991. Phylogenetic relationships within the Mysticeti (whalebone whales) based upon studies of highly repetitive DNA in all extant species. Hereditas 114:263–269.
Árnason U, Gullberg A, Widegren B. 1993. Cetacean mitochondrial DNA control region: sequences of all extant baleen whales and two sperm whale species. Molecular Biology and Evolution 10:960–970.
Bannister JL. 2002. Baleen Whales, Mysticetes. Pages 62–72 in Perrin WF, Würsig B, Thewissen JGM, editors. Encyclopedia of Marine Mammals. Academic Press, San Diego, California, USA.
Best PB. 1967. Distribution and feeding habits of baleen whales off Cape Province. Investigational Report No. 57. Division of Sea Fisheries, South Africa.
Best PB. 1977. Two allopatric forms of Bryde’s whale off South Africa. Reports of the International Whaling Commission, Special Issue 1:10–38.
Best PB. 1996. Evidence of migration by Bryde’s whales from the offshore population in the southeast Atlantic. Reports of the International Whaling Commission 46:315–331.
Best PB. 2001. Distribution and population separation of Bryde’s whale Balaenoptera edeni off southern Africa. Marine Ecology Progress Series 220:277–289.
Best PB. 2007. Whales and Dolphins of the Southern African Subregion. Cambridge University Press, Cape Town, South Africa.
Best PB, Butterworth DS, Rickett LH. 1984. An assessment cruise for the South African inshore stock of Bryde’s whales (Balaenoptera edeni). Reports of the International Whaling Commission 34:403–423.
Burns WC, Baker DJ. 2000. From the harpoon to the heat: Climate change and the International Whaling Commission in the 21st century. Journal of International Wildlife Law & Policy 3:50–72.
Caputo, M., Froneman, P., du Preez, D., Thompson, G., & Plön, S. (2017). Long-term trends in cetacean occurrence during the annual sardine run off the Wild Coast, South Africa. African Journal of Marine Science, 39(1), 83–94. https://doi.org/10.2989/1814232X.2017.1304451
CITES (Convention on the International Trade in Endangered Species of Wild Fauna and Flora. “Appendices | CITES”. cites.org. Retrieved 26 September 2024
Clapham PJ, Young SB, Brownell RL. 1999. Baleen whales: conservation issues and the status of the most endangered populations. Mammal Review 29:37–62.
Coetzee, J. C., Van Der Lingen, C. D., Hutchings, L., & Fairweather, T. P. (2008). Has the fishery contributed to a major shift in the distribution of South African sardine? ICES Journal of Marine Science, 65(9), 1676–1688. https://doi.org/10.1093/icesjms/fsn184
Committee on Taxonomy. 2024. List of marine mammal species and subspecies. Society for Marine Mammalogy, www.marinemammalscience.org, consulted on 10 September 2024.
Constantine R, Iwata T, Nieukirk SL and Penry GS (2018). Future Directions in Research on Bryde’s Whales. Front. Mar. Sci. 5:333. doi: 10.3389/fmars.2018.00333
Fréon P, Coetzee JC, van der Lingen CD, Connell AD, O’Donoghue SH, Roberts MJ, Demarcq H, Attwood CG, Lamberth SJ, Hutchings L. 2010. A review and tests of hypotheses about causes of the KwaZulu-Natal sardine run. African Journal of Marine Science 32:449–479.
Gordon J, Gillespie D, Potter J, Frantzis A, Simmonds MP, Swift R, Thompson D. 2003. A review of the effects of seismic surveys on marine mammals. Marine Technology Society Journal 37: 16–34.
IUCN (International Union for Conservation of Nature). 2012. Balaenoptera edeni. The IUCN Red List of Threatened Species. Version 3.1. http://www.iucnredlist.org. Downloaded on 21 February 2016.
IUCN-Marine Mammal Protected Areas Task Force, 2020. Southern Coastal and Shelf Waters of South Africa IMMA Factsheet. IMMA Factsheet Link.
IWC. 1980a. Report of the sub-committee on Bryde’s whales. Report of the International Whaling Commission 30:64–73.
IWC. 1980b. Report of the Special Meeting on Southern Hemisphere sei whales. Report of the International Whaling Commission 30:493–505.
IWC. 1981. Report of the subcommittee on other baleen whales. Report of the International Whaling Commission 31:122–132.
IWC. 1983. Chairman’s report of the 34th Annual Meeting. Report of the International Whaling Commission 33:20–42.
IWC. 1997. Report of the subcommittee on North Pacific Bryde’s whales. Report of the International Whaling Commission 46:163–168.
IWC. 2006a. Report of the workshop on the pre-implementation assessment of western North Pacific Bryde’s whales. Journal of Cetacean Research and Management 8:337–355.
IWC. 2006b. The IWC Summary Catch Database. International Whaling Commission.
Junge GCA. 1950. On a specimen of the rare Fin Whale, Balaenoptera edeni Anderson, stranded on Pulu Sugi Near Singapore. Zoologische Verhandelingen 9:26.
Kato, H., Shinohara, E., Kishiro, T. and Noji, S. 1996. Distribution of Bryde’s whales off Kochi, southwest Japan, from the 1994/95 sighting survey. Rep. int. Whal. Commn 46: 429–436.
Kato H, Perrin WF. 2018. Bryde’s Whales Balaenoptera edeni / brydei. Pages 158–163 in Perrin WF, Würsig B, Thewissen JGM, editors. Encyclopedia of Marine Mammals. Third Edition. Academic Press, San Diego, California, USA.
Kershaw F et al. 2013. Population differentiation of 2 forms of Bryde’s Whales in the Indian and Pacific Oceans. Journal of Heredity 104:755–764.
Koper RP, Plön S. 2012. The potential impacts of anthropogenic noise on marine animals and recommendations for research in South Africa. EWT Research & Technical Paper No. 1. Endangered Wildlife Trust, South Africa.
Kebke A, Samarra F, Derous D. 2022 Climate change and cetacean health: impacts and future directions. Phil. Trans. R. Soc. B 377: 20210249. https://doi.org/10.1098/rstb.2021.0249
MacLeod CD. 2009. Global climate change, range changes and potential implications for the conservation of marine cetaceans: a review and synthesis. Endangered Species Research 7:125–136.
Meÿer, M. A., Best, P. B., Anderson-Reade, M. D., Cliff, G., Dudley, S. F. J. and Kirkman, S. P. (2011). Trends and interventions in large whale entanglement along the South African coast. African J. Mar. Sci. 33, 429–439.
Mikhalev YA. 2000. Whaling in the Arabian Sea by the whaling fleets “Strava” and “Sovietskaya Ukraina.” Pages 141–181 in Yablokov AV, Zemsky VA, editors. Soviet Whaling Data (1949- 1979). Centre for Russian Environmental Policy, Moscow.
Moore SE. 2008. Marine mammals as ecosystem sentinels. Journal of Mammalogy 89:534–540.
National Planning Commission [NPC] (2012). National Development Plan 2030: Our Future – Make It Work. Pretoria: National Planning Commission.
O’Donoghue SH, Whittington PA, Dyer BM, Peddemors VM. 2010. Abundance and distribution of avian and marine mammal predators of sardine observed during the 2005 KwaZulu-Natal sardine run survey. African Journal of Marine Science 32:361–374.
Olsen O. 1913. On the external characters and biology of Bryde’s Whale (Balaenoptera brydei), a new rorqual from the coast of South Africa. Proceedings of the Zoological Society of London 83:1073–1090.
Omura H, Kasuya T, Kato H, Wada S. 1981. Osteological study of the Bryde’s Whale from the Central South Pacific and Eastern Indian Ocean. Scientific Report from the Whales Research Institute of Tokyo 33:1–26.
Paynee DK 2022. MSc thesis. Genetic structure and diversity of South Africa’s inshore Bryde’s whale population
Penry GS. 2010. The Biology of South African Bryde’s Whales. Ph.D. Thesis. University of St. Andrews, Scotland. https://hdl.handle.net/10023/921
Penry GS, Cockcroft VG, Hammond PS. 2011. Seasonal fluctuations in occurrence of inshore Bryde’s whales in Plettenberg Bay, South Africa, with notes on feeding and multispecies associations. African Journal of Marine Science 33:403–414.
Rosel PE, Wilcox LA. Genetic evidence reveals a unique lineage of Bryde’s whales in the northern Gulf of Mexico. Endangered Species Research. 2014 Jul 31;25(1):19-34.
Rosel, P.E., Wilcoz, L.A., Yamada, T.K. & Mullin, K.D. 2021. A new species of baleen whale (Balaenoptera) from the Gulf of Mexico, with a review of its geographic distribution. Marine Mammal Science, 37(2):577-610.
Roy, C., Van Der Lingen, C. D., Coetzee, J. C., & Lutjeharms, J. R. E. (2007). Abrupt environmental shift associated with changes in the distribution of Cape anchovy Engraulis encrasicolus spawners in the southern Benguela. African Journal of Marine Science, 29(3), 309–319. https://doi.org/10.2989/AJMS.2007.29.3.1.331
Sasaki T, et al. 2005. Mitochondrial phylogenetics and evolution of mysticete whales. Systematic Biology 54:77–90.
Sasaki T, Nikaido M, Wada S, Yamada TK, Cao Y, Hasegawa M, Okada N. 2006. Balaenoptera omurai is a newly discovered baleen whale that represents an ancient evolutionary lineage. Molecular Phylogenetics and Evolution 41:40–52.
Segre PS, di Clemente J, Kahane‐Rapport SR, Gough WT, Meÿer MA, Lombard AT, Goldbogen JA, Penry GS. High‐speed chases along the seafloor put Bryde’s whales at risk of entanglement. Conservation Science and Practice. 2022 May;4(5):e12646. https://doi.org/10.1111/csp2.12646
Tønnessen JN, Johnsen AO. 1982. The History of Modern Whaling. University of California Press, Berkeley and Los Angeles, CA, USA.
Yoshida, H. & Kato, H., 1999. Phylogenetic relationships of Bryde’s Whales in the Western North Pacific and adjacent waters inferred from mitochondrial DNA sequences. Marine Mammal Science, 15(4), pp.1269-1286.
Yamada TK, Kakuda T, Tajima Y. 2008. Middle sized balaenopterid whale specimens in the Philippines and Indonesia. Memoirs of the National Museum of Nature and Science 45:75–83.