Brown Hyaena
Parahyaena brunnea

2025 Red list status
Near Threatened
Regional Population Trend
Stable
change compared
to 2016
No Change
Overview
Parahyaena brunnea – (Thunberg, 1820)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – HYAENIDAE – Parahyaena – brunnea
Common Names: Brown Hyaena (English), Bruinhiëna, Strandjut, Strandwolf (Afrikaans), Impisi, Ipisi enzotho (Ndebele), Sephiribjôkwane, Phiribjôkwane (Sepedi), Phiribjokwane, Phiri, Thamahane (Sesotho), Lefiritshwana, Mosonokwane, Mosonolokwane, Phiri, Phiri êntshonyana, Phiritshwana, Sethenekwane, Setinikwana (Tswana), Imphisi (Swati), Mhisi, Mhisana (Tsonga), Tshivhingwi (Venda), Ingqawane, Inchuka (Xhosa), Isidawana (Zulu)
Synonyms: Hyaena brunnea Thunberg, 1820
Taxonomic Note: Although previously classified under the genus Hyaena (e.g. Jenks & Werdelin 1998), Koepfli et al. (2006) placed this species in its own genus Parahyaena (also see Wozencraft 1993). The sister taxon is the Striped Hyaena (Hyaena hyaena) with which it last shared a common ancestor roughly 4.2 mya (Koepfli et al. 2006). The clade containing Brown and Striped Hyaenas last shared a common ancestor with the Spotted Hyaena (Crocuta crocuta) roughly 8.6 million years ago (Koepfli et al. 2006). Of interest, based on dentition of extinct Pachycrocuta spp, a change of genus is proposed for the Brown Hyaena, which would see it as integrated into Pachycrocuta (Pérez-Claros 2024).
Red List Status: NT, Near Threatened C2a(i) + D1 (IUCN version 3.1)
Assessment Information
Assessors: Power, R.J.1, Jansen van Vuuren, A.2 & da Silva, J.M.3
Reviewers: Nicholson, S.K.4, Do Linh San, E.5 & Williams, K.6
Contributor: Weise, F.W.7,8
Institutions: 1North West Provincial Government, 2Nelson Mandela University, 3South African National Biodiversity Institute, 4Endangered Wildlife Trust, 5Sol Plaatjie University, 6Cape Leopard Trust, 7Hyaena Specialist Group, 8Zambian Carnivore Programme
Previous Assessors and Reviewers: Yarnell, R., Richmond-Coggan, L., Bussiere, E., Williams, K., Bissett, C., Welch, R. & Wiesel, I.
Previous Contributors: Mills, M., Tambling, C., Avenant, N., Child, M.F., Do Linh San, E., Dell, S. & Dalerum, F.
Assessment Rationale
The Brown Hyaena is widespread and locally abundant, but rare within the assessment region. It is absent from Lesotho and possibly locally extinct in Eswatini. It faces multiple threats, especially across unprotected areas and in regions dominated by livestock and game ranching. The species is difficult to census due to it occurring at mostly low density and due to its nocturnal and secretive nature. The South African population baseline in 1998 was estimated to be 1,700 with a range of 800 to 2,200 individuals (Hofer & Mills 1998b), and the distribution has remained relatively unchanged. Recent studies have provided local density estimates that vary geographically and in relation to protection status, sympatric predator densities, local persecution intensity, carcass availability, habitat type, and fencing type. Such broad adaptability and the resulting variation in local density limit our ability to extrapolate an overall density with a corresponding national population estimate, and further density estimates are required from under-studied parts of the Brown Hyaena’s geographic range to provide a robust overall population estimate for South Africa. Additionally, researchers have added some more detailed population estimates to areas of their current distribution that were previously unstudied (particularly Limpopo, North West and Eastern Cape provinces) and shown that relatively high densities occur in some parts of these regions and that range expansions have occurred. These more recent studies suggest that the estimated South African population size of 1,700 is likely now an underestimate.
Despite the evidence of locally stable and increasing populations (i.e Madikwe, Pilanesberg), the species does face persistent threats of direct and indirect persecution within the assessment region. Quantifying the level of local Brown Hyaena persecution in relation to demographic rates is essential information required for future population assessments. Localised declines and/or extinctions from anthropogenic threats are possible – especially given the synergistic threats of incidental snaring, illegal hunting and poisoning – but evidence is currently lacking to prove a link between persecution (direct and indirect) and local population declines. Despite increasing evidence across South Africa, including range expansion, locally abundant populations that are not fragmented, the species is classified as Near Threatened based on fewer than 10,000 mature individuals across their global range, with a projected continuing decline in mature individuals outside protected areas. Thus, we retain the Near Threatened C2a(i)+D1 listing, in line with the global listing, under a precautionary purview. However, we note that once further field studies produce more robust population size and trend estimates, this species will need reassessment and could possibly be considered to be Least Concern.
Regional population effects: Movements between Namibia, Botswana, Zimbabwe and South Africa are possible in both directions, especially so from Botswana to neighbouring countries (Rich et al. 2017; Van der Weyde et al. 2022). However, it is unknown whether immigration is significant enough to secure isolated subpopulations within South Africa – particularly those located in the Western and Eastern Cape provinces. However, the northern subpopulations are well-connected to Botswana, Namibia and Zimbabwe and a rescue effect is very likely across the Limpopo and Molopo rivers on the borders of South Africa.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Power RJ, Jansen van Vuuren A & da Silva JM. 2025. A conservation assessment of Parahyaena brunnea. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Brown Hyaena is a near-endemic to southern Africa, largely due to the marginal extension into the arid parts of southwestern Angola where it occurs up to -15° latitude. The species mainly occurs in the semi-arid and arid regions of Namibia, Botswana, South Africa and Zimbabwe (Skinner & Chimbimba 2005). There are no resident populations of Brown Hyaena north of the Chobe River and Brown Hyaenas are probably extinct from Eswatini (HDMP 2018).
In South Africa, the Brown Hyaena’s distribution has diminished significantly since the 18th century when animals were recorded as far south as South Africa’s Western Cape (Stuart et al. 1985; Hofer and Mills 1998a; Mills 2013). After the 1850s, Brown Hyaena range retraction took place from the Western Cape in a north-easterly direction, and simultaneously, from the Eastern Cape, the range retracted in a north-westerly direction (Stuart et al. 1985). Brown Hyaena were extirpated from the Western Cape, and relicts remained in the northern part of the Eastern Cape, and most of the Northern Cape by the late 1960s (Stuart et al. 1985), and this remained into the 1970s (Lloyd & Millar 1984).
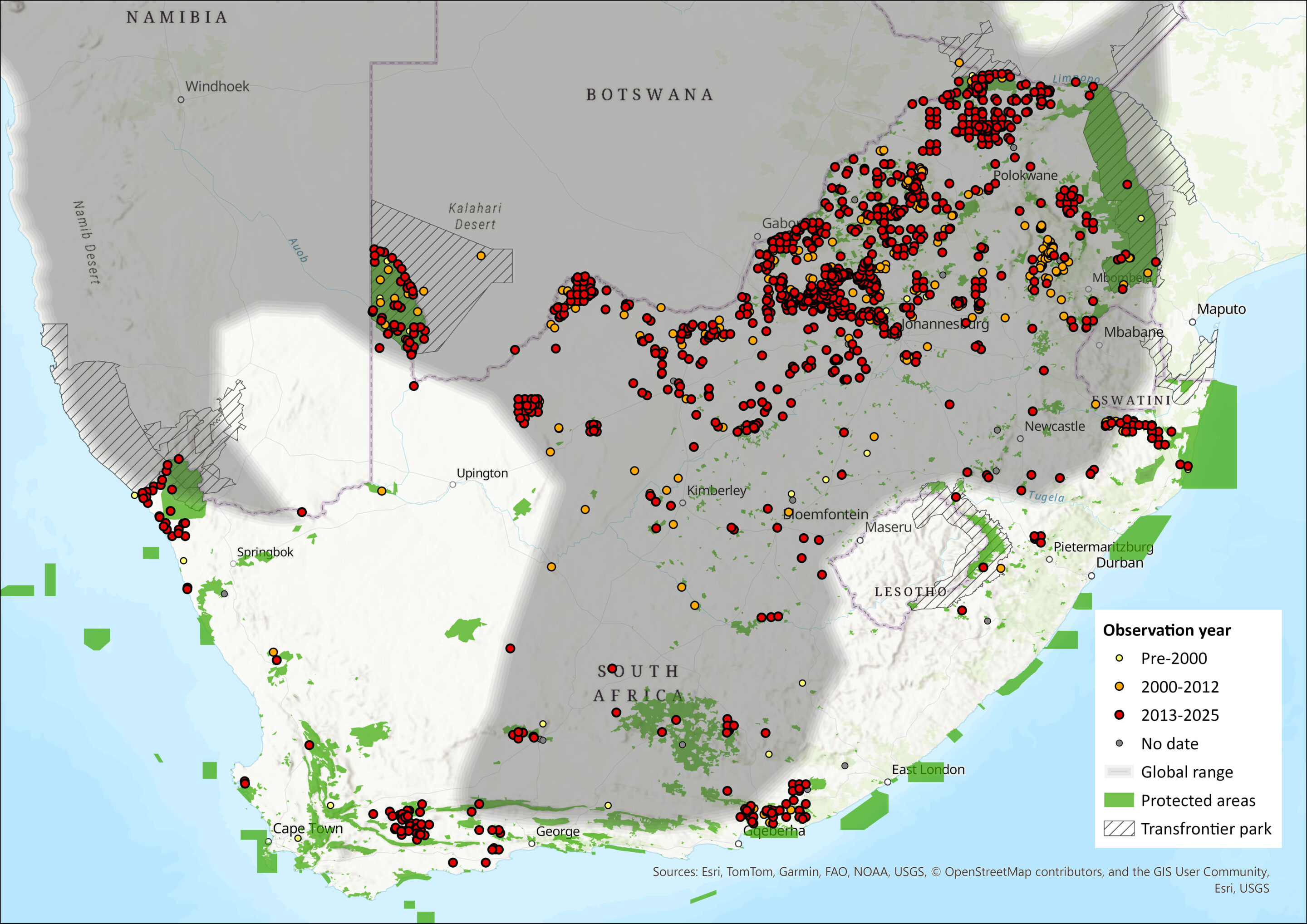
Today, this species remains relatively widespread in many parts of South Africa, with high levels of occupancy in the north of South Africa (Yarnell et al. 2016). Previously, Thorn et al. (2011) estimated that Brown Hyaena’s extent of occurrence in North West Province had increased by 45%, from 90,598 km2 in 2000 to 131,523 km2 in 2010, and the species occurs throughout the province (Power et al. 2019). Concomitantly, the species also appeared to have high levels of occupancy in Limpopo, Gauteng and Mpumalanga provinces (Richmond-Coggan 2014), and the species northern stronghold (Richmond-Coggan 2014), as deduced from recent occupancy surveys showing high occupancy rates (Williams et al. 2021a), suggests it to still be present and widespread there (Figure 1). Interestingly, it still also persists in Gauteng Province (Kuhn 2011; Richmond-Coggan 2014), and has for some time (Skinner 1976), no doubt aided by rescue effect by North West, Limpopo and Mpumalanga provinces. The species continues to be present across different geographic parts of South Africa, such as in the Northern Cape’s Tswalu Kalahari Reserve, Madikwe Game Reserve (Smith et al. 2023), and even in Mpumalanga’s Verloren Vallei Nature Reserve on the Highveld edge (Loock & Swanepoel 2019).
Some evidence of range expansion has been recorded in the Western Cape (Figure 1; Yarnell et al. 2016) where it was previously believed to be extirpated (Hofer and Mills 1998a), with indication of increasing re-colonisation. The Western Cape supports a small subpopulation in the Little Karoo: in Sanbona Wildlife Reserve, Anysberg Nature Reserve and surrounding farmlands (Woodgate et al. 2023), and in Gondwana Game Reserve (Hall and Thatcher, 2021). It is possible that these records are vagrant individuals from this isolated population, or the species may have recolonised with dispersers from the Eastern Cape reintroductions (Hayward et al. 2007; Banasiak et al. 2021a). Within the Northern Cape, records are predominantly concentrated around the Kgalagadi Transfrontier Park and the |Ai-Ais/Richtersveld Transfrontier Park where Brown Hyaena populations are connected with extant populations in Botswana and Namibia respectively, and Tswalu Kalahari Reserve.
The species was deemed practically extinct in the Free State (Hofer and Mills 1998a), though there had always been relict populations along the periphery of the province with the exception of the northern fringe (Von Richter 1972). There are, however, likely to still be low levels of occupancy in the Free State (Richmond-Coggan 2014) as is depicted by occurrences throughout the central part of the province (see Figure 1), whether the species was increasing here or was previously under-recorded is unknown (Yarnell et al. 2016), but at least during the previous assessment it had increased since earlier baselines (see Von Richter 1972). Similarly, in KwaZulu-Natal, no resident populations were discovered in earlier assessments due to the reported high levels of shooting and trapping (Hofer and Mills 1998a), though these assessments were not thorough. At the time of the last assessment and more recently, there have been records of the species, mostly clustered in the northern parts of the province (Richmond-Coggan 2014; Yarnell et al. 2016; Williams et al. 2021a) with high levels of occupancy on some reserves (Williams et al. 2021a). Of interest, are the records occurring in the foothills of the Drakensberg with a southward extension (Figure 1), which links to where a small population was discovered on the KwaZulu-Natal-Eastern Cape border (Richmond-Coggan 2014; Yarnell et al. 2016). However, it was assumed that these sightings were either from game ranches that have reintroduced individuals to attract ecotourists and thus not representative of free-roaming recolonisation (Yarnell et al. 2016), while the Eastern Cape reintroductions cannot be discounted as a possible source of colonising individuals (Hayward et al. 2007; Banasiak et al. 2021a).
Other than a relict population in the north of the Eastern Cape (Lloyd & Millar 1984; Maddock 1993), Brown Hyaenas have also been reintroduced into a number of small reserves within the province (Hayward et al. 2007), and indications of secondary colonisation elsewhere are surmised in the same province (Banasiak et al. 2021a). The species is still present in Mountain Zebra National Park (Smith et al. 2023). Such reintroductions may be facilitating the recolonisation of the Brown Hyaena’s historical range in South Africa (Yarnell et al. 2016; Banasiak et al. 2021a). Killea (2020) modelled the distribution range using maximum entropy models using data from the Hyaena Distribution Mapping Project, and there appears to be differences in the populations in the arid west and mesic east. The distribution range is likely similar to that reported earlier (Yarnell et al. 2016), with some minor areas which may have been recolonised.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m asl
Elevation Upper Limit (in metres above sea level): 2200 m asl
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Brown Hyaena (Parahyaena brunnea) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Eswatini | Possibly extinct | Native | – | – |
| Mozambique | Presence Uncertain | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The effects of climate change are intertwined with a variety of additional anthropogenic effects on biodiversity (Trouwborst & Blackmore 2020). Even though some carnivore species are highly adaptable to a changing climate, others are more specialised and thus more sensitive to changes (Trouwborst & Blackmore 2020). It is important to consider a species’ ecological and biological traits and niches to determine the likely mechanisms of climate change impact and quantify these using expert knowledge (Foden & Young 2016), and empirical information, which may include biological information on the species (Mills 1984), as well as modelled distributions (Killea 2020). For instance, Brown Hyaenas in desert areas are more often found in areas with greater tree cover while hyaena presence in the savanna is correlated with relatively low tree cover (Killea 2020). Given the predictions of climate change warming/drying in southern Africa (Foden & Young 2016, and references within), the arid areas may likely become more unsuitable for the species, owing to a loss of vegetation, which has been found to be important to them (Welch et al. 2016), and so they may either perish or depart out of these areas, though it may only affect the western part of the Northern Cape. Conversely, it seems in the savannas, likely mesic ones, the species prefers lowered tree cover (Killea 2020), and eastward aridification may see an increase in suitable habitat for the species. It is possible that climate change, warming and drying, might facilitate a range expansion of the species (Trouwborst & Blackmore, 2020). Richmond-Coggan (2014) suggested that climate change impacts on Brown Hyaena ecology and their occurrence are worthy avenues of study. It has recently been hypothesised to occur more widely based on aridification-induced range expansions elsewhere in southern Africa (Weise, F. pers comm.), so this is entirely plausible.
Furthermore, climate change is not only predicted to cause changes in temperature, but it will also result in variability in rainfall (Déqué et al. 2017). Rainfall largely impacts the habitats and populations of hyaenas as it influences their feeding habits, recruitment, demography and conflict with humans (Kolowski & Holekamp 2009). A study by Ferreira and Viljoen (2022) in Kruger National Park indicated that periods of low rainfall are temporarily advantageous to large carnivores. During these periods, herbivores experience a decline in body condition as well as their anti-predator defences (Ferreira & Viljoen 2022). Therefore, increasing the hunting success of predators with a resulting increase of body condition and recruitment (Ferreira & Viljoen 2022). This will provide increased scavenging opportunities for species such as the Brown Hyaena. However, in the long term when cubs start dispersing from the dens, the increase in these carnivore populations will force individuals to explore outside their range (Ferreira & Viljoen 2022; Abade et al. 2014). This often leads them to alternative food sources, such as livestock and results in conflict with humans which may prove to be fatal (Ferreira & Viljoen 2022; Abade et al. 2014). Although the study was not modelled on the Brown Hyaena, a similar outcome may prevail.
Population information
The total global population size has been estimated as being between 4,365–10,111 mature individuals (Wiesel 2015), with Botswana harbouring the largest population (an estimated 3,173–4,048 animals); followed by Namibia (< 3000 mature individuals, Wiesel & Hanssen 2022), and then, South Africa (800–2,200). This national estimate is considered likely to be underestimated based on numerous recent local population densities and increasing extent of occurrence. Population estimates for Zimbabwe, Angola and Mozambique are unknown.
Quantitative population trends for two sites in North West Province, Pilanesberg National Park and a nearby private game reserve, suggest that these sites have been stable since the last assessment (Thorn et al. 2011b; Richmond-Coggan 2014; Yarnell et al. 2016), as is suggested by nationwide occupancy estimates (Williams et al. 2021a). It is also clear that Brown Hyaena densities are highest in protected areas compared to neighbouring unprotected rangelands (Thorn et al. 2011a; Yarnell et al. 2013; Richmond-Coggan 2014; Faure et al. 2022). The estimated Brown Hyaena density in agricultural land of the North West Province was 0.15/100 km2 which was an order of magnitude lower than in protected areas (Thorn et al. 2011a), and relative abundance is said to be four times lower outside protected areas in this province at least (Richmond-Coggan 2014). Similarly, and earlier in 2007, based on audio-play backs and sign surveys, a Brown Hyaena density range of 0.01 ±0.01/100 km2 – 0.16 ±0.04/100 km2 was determined across North West (Thorn et al. 2010), which is mostly applicable to ranchlands outside protected areas. These are very low estimates, but they may even be realistic estimates for low density populations in agricultural landscapes.
Brown Hyaena densities appear relatively higher in Limpopo and North West provinces, compared to the Kgalagadi Transfrontier Park (Mills 1990, Thorn et al. 2012, Yarnell et al. 2013; Richmond-Coggan 2014; Honiball 2021; Williams et al. 2021b) (Table 1). However, the highest South African densities have recently been estimated by Welch and Parker (2016) who found a density of 14–19 individuals/ 100km2 on Kwandwe Private Game Reserve (Eastern Cape). This unusually high density was attributed to the high density of mammalian predators in the reserve and the abundant scavenging opportunities afforded to Brown Hyaena by such predators, as well as the reserve’s well-maintained predator-proof fences (Welch and Parker 2016; Williams et al. 2021b). Thus, mirroring an equally high density elsewhere in southern Africa with similar conditions (see Edwards et al. 2019). However, a more recent estimate suggests that the Kwandwe population may have declined over time, from 15-16 Hyaenas/100 km2 (estimate in, Welch & Parker 2016) to 8.7 Hyaenas/ 100 km2 (Williams et al. 2021b). Of interest, Kwandwe only ranks 7th now, in a series of 15 reserves where density estimates were determined with a restriction on fence permeability (see Williams et al. 2021b). Elsewhere in the Eastern Cape, the density of Brown Hyaena in the Mountain Zebra National Park in 2015 was 6-10 hyaenas/100 km2, or an absolute abundance of 12 to 21 of them (Comley 2016).
A camera trap-based assessment of Brown Hyaenas on 15 fenced reserves nationwide revealed density estimates ranging from 2.55 to 15.06 Brown Hyaenas / 100 km2 when modelling assumed no fence permeability (Williams et al. 2021b). However, as Brown Hyaenas are known to traverse game fences utilising holes (Kesch et al. 2013), caution must be taken when calculating densities assuming no fence permeability. Density estimates were considerably lower when fence permeability was included in the models, generating estimates ranging between 0.17 to 1.59 Brown Hyaenas/100 km2 (Williams et al. 2021b). The highest recent density recorded was in 2016 for the Atherstone Nature Reserve, Limpopo Province, where 15 Hyaenas/100 km2 was determined (Williams et al. 2021b). In fenced reserves such as the Madikwe Game Reserve of the North West, in 2008, a small population of 11 to 13 Brown Hyaena was estimated, which was much smaller than what management anticipated (Barker 2012), amounting to 0.7 to 1.7 Hyaenas/100 km2. Camera trap data from 2016 indicated 9.49 Brown hyaenas/100 km2 in Madikwe Game Reserve assuming fence impermeability (Williams et al. 2021b). Subsequently, on the same reserve, and based on similar capture recapture frameworks from camera trap data, a population of 92±7.1 Brown Hyaena were deduced in 2019 (Honiball 2021), i.e. 13.5 Hyaenas/100 km2, which may be indicative of a population increase on this reserve. In the same province, in the Pilanesberg National Park, a camera trap-based approach determined 2.8 Hyaenas/100 km2 (Thorn et al. 2009), which later seemed to increase to 6 Hyaenas/100 km2 in 2011 (Yarnell et al. 2013), based on distance sampling estimation. The most recent camera trap-based study yielded a density estimate of 10.81 Hyaenas/100 km2 considering fence impermeability (Williams et al. 2021b), However, with variation in methods used, one should tentatively ascribe population trends, though it does appear there have been population increases on these two North West reserves.
Table 1. Summary of range of density estimates for Brown Hyaena in different provinces (after Williams et al. 2021).
| Provinces | Reserves | Year(s) | Fence permeability (density – Hyaenas/100 km²) | Fence Impermeability (density – Hyaenas/100 km²) |
| Limpopo | 6 | 2013-2016 | 0,19-1,33 | 2,77-15 |
| Mpumalanga | 2 | 2016 | 0,17-0,63 | 2,55-2,8 |
| KwaZulu-Natal | 2 | 2014-2015 | 0,29-0,8 | 3,2-11,44 |
| Eastern Cape | 1 | 2017 | 1,09 | 8,73 |
| Gauteng | 1 | 2016 | 0,52 | 3,8 |
| North West | 3 | 2016 | 0,68-1,59 | 5,32-10,71 |
Using a novel approach i.e. applying a Distance sampling approach to camera trap data, 11.6 hyaenas/ 100 km2 were estimated in the Alldays area of Limpopo in 2019 (McKaughan et al. 2023). Nearby using spatially explicit capture-recapture methods, estimates of 10.8 hyaenas/ 100 km2 at Zingela Nature Reserve, and 5.6 hyaenas/ 100 km2 at Venetia Limpopo Nature Reserve were recorded (Williams et al. 2021b). The Distance sampling method is in its infancy, and it showed a few upward biases in estimating the species density in the before mentioned study; it may thus be better to rely upon spatially capture-recapture analysis approaches (McKaughan et al. 2023). If these trends are indeed true for the wider population, it would be fair to assume that the national population is likely increasing or stable at least. However, we must acknowledge that there are no data on Brown Hyaena population sizes or trends for more than half of its South African distribution. Thus, there is a large degree of uncertainty and trends may differ outside of protected areas. Like other large carnivores, brown hyaenas population density is less frequently studied outside of protected areas, but densities appear to be lower outside protected areas (Balme et al. 2010; Faure et al. 2022; Williams 2017; Williams et al. 2021b; Wilkinson et al. 2024). In Platjan in Limpopo Province, a predominantly agricultural, mixed land-use system, brown hyaena density was determined as 0.74 / 100 km2 (Faure et al. 2022) and an estimate from a mixed used landscape in the western Soutpansberg Mountains estimated 2.56 – 3.63 Brown Hyaenas / 100 km2 (Williams 2017).
Brown Hyaenas are typically able to penetrate most game fences by utilising holes dug by other mammals (Richmond-Coggan 2014; Kesch et al. 2015; Fink et al. 2020; Banasiak et al. 2021b) and as such habitat fragmentation in areas of cattle or game ranching is lessened. Some protected areas have well-maintained predator proof game fences and the Brown Hyaena subpopulations within these reserves are potentially at risk from inbreeding depression, as estimates suggest subpopulations of less than 40 individuals in each area (Yarnell et al. 2016; Honiball 2021; Williams et al. 2021b). The issue of confined reserves and the risk of inbreeding depression has been raised several times (Richmond-Coggan 2014; Comley 2016; Welch & Parker 2016; Edwards et al. 2019). Brown Hyaena in the rangelands of the North West Province and Limpopo Province typically have ranges up to of about 47 km2 and do move through game fences (Richmond-Coggan 2014), however, home ranges can range from 95,05 km2 up to 169,8 km2 in northern Limpopo (Williams 2017). Other isolated subpopulations, such as in the Little Karoo (Anysberg–Sanbona and surrounding farmland) are small with only 16 adult individuals being identified from camera trap photographs (Yarnell et al. 2016), while there are other private protected areas that also contribute to Brown Hyaena conservation. For example, Tswalu Kalahari Reserve and !Khamab Kalahari Reserves (Northern Cape and North West, respectively) are important due to their size and location. In the latter reserve, based on the last density estimate in 2016 (Williams et al. 2021b), an abundance estimate range of 10 –48 Brown Hyaenas was extrapolated. Research also shows that Brown Hyaenas in unprotected areas have smaller home ranges (Maude 2005; Richmond-Coggan 2014).
Other causes of fragmentation include urbanisation or land given to arable production which Brown Hyaena rarely frequent. Therefore, the level of fragmentation across the Brown Hyaena distribution is low overall in the majority of areas (Limpopo and North West provinces) where they currently exist. Subpopulation structure is difficult to determine for this species as not much is known about connectivity between regions and thus gene flow, though a maxent modelled distribution map shows what appears to be a contiguous population (Killea 2002). However, it is unlikely that there is natural dispersal from the main stronghold in the north to the southern provinces. Based on our limited knowledge of geographic range and connectivity, we tentatively define the following five subpopulations: northern (Northern Cape, Limpopo, Gauteng, Mpumalanga and North West provinces); the central interior (south-eastern Northern Cape and Free State); Klein Karoo in the Western Cape; the Eastern Cape reserves and farmland matrix; and KwaZulu-Natal. However, mindful of the mobility of the species, these populations may be relatively contiguous, particularly across the northern half.
Private land is especially important for the survival of Brown Hyaenas, providing sizeable areas where competitor species are largely absent (Kent and Hill 2013; Thorn et al. 2012). The conversion from livestock farming to wildlife ranching may be beneficial for the Brown Hyaena in terms of habitat conservation (Richmond-Coggan 2014) and wildlife ranching tends to have lower human disturbance and provide a greater prey base compared to livestock farming (Thorn et al. 2012), as well as high carcass availabilities (Maude & Mills 2005). Brown Hyaena occupancy in northern Limpopo Province was higher on properties with game fences compared to locations with cattle fences or no fences, indicating a preference for game farms over livestock farms (Williams 2017). However, where impenetrable fences are erected to protect valuable and rare game species, connectivity may be compromised (Pirie et al. 2017). Interestingly, in Botswana, Kent and Hill (2013) found that estimated densities of Brown Hyaenas were higher on farms used for livestock production than on those used for game farming, further corroborating that the species can tolerate land-use change where reliable alternative food resources exist and competition for carcasses is relaxed. In mixed farming landscapes with Leopard (Panthera pardus) populations, Brown Hyaenas also benefit from regular opportunities for carcass kleptoparasitism (Williams et al. 2018). Thus, livestock ranches and mixed wildlife/livestock areas could be improving habitat suitability and therefore, also connectivity. It remains to be seen whether this situation is replicated in South Africa, as all indications are to the contrary (Thorn et al. 2011a; Richmond-Coggan 2014; Faure et al. 2022).
Population Information
Current population trend: The population seems stable and even increasing in many parts, but is also described as decreasing in parts of South Africa though more recently, there has been no sign of a decrease of any sort.
Continuing decline in mature individuals: Yes
Extreme fluctuations in the number of subpopulations: Unknown
Number of mature individuals in population: 800-2,200, based on previous assessments.
All individuals in one subpopulation: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown, possibly five.
Severely fragmented: No, due to good dispersal abilities and large distributional range.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A fairly recent genomic study found the Brown Hyaena to harbour very low levels of genetic diversity likely related to a progressive decline in their historical effective population size approximately 1-2 million years ago (Westbury et al. 2018; Westbury et al. 2021). The levels of genetic diversity were low even compared against endangered species known to have extremely low levels of diversity (e.g., Cheetah). More surprising was that the species was also found to have no strong signs of inbreeding and high levels of connectivity across its range (Westbury et al. 2018; Westbury et al. 2021). Despite this connectivity, a weak signal of population genetic structure has detected (Westbury et al. 2018). Indeed, four genetic clusters have been identified corresponding to the geographic origins of the individuals, with South African animals grouping together and separately from the rest (Westbury et al. 2018). Considering the low levels of diversity and the low estimated census size within the assessment region, it is likely the effective population size (Ne) for the region is well below Ne 500, and likely at or below Ne 500 for the species as a whole. Direct analysis of Ne is needed to verify these estimates.
Habitats and ecology
The Brown Hyaena is widespread across southern Africa and is found in the following habitat types: desert areas with annual rainfall less than 100 mm (particularly along the Skeleton Coast in Namibia), semi-desert (such as the Kalahari), open scrub and open woodland savannah with a maximum rainfall up to about 700 mm (Skinner 1976; Skinner & Chimimba 2005). It even shows an ability to survive close to urban areas, in particular on the outskirts of suburban Johannesburg (Kuhn 2014), and can utilise agricultural and rangeland landscapes, but avoid highly urbanised areas (Skinner & Van Aarde 1987; Thorn et al. 2011a; Richmond-Coggan 2014; Faure et al. 2022). It requires some type of cover in which to lie up during the day. For this it favours rocky, mountainous areas with bush cover in the bushveld areas of South Africa (Skinner 1976; Williams 2017). Having said this, habitat selection for steep slopes and high elevation areas is confirmed for the species (Welch et al. 2016), and thus rugged terrain is important for Brown Hyaenas. In South African rangelands it prefers woodlands verses scrub (Richmond-Coggan 2014), and in protected areas, preferring scrub verses grassland (Thorn et al. 2009). In terms of finer scale habitat use, in a Karroid-like reserve, Brown Hyaena selected for denser vegetation (Welch et al. 2016), while in some areas they tend to also select drainage lines with elements of riparian vegetation (Welch et al. 2016; Fink et al. 2020), which speaks to their need for cover. In the Kgalagadi, they are found in open plains and dunefields (Mills 1984), and in general are said to be dependent on some form of light cover in the arid parts (Killea 2020). Interestingly, anthropogenic features in the landscape are also important to Brown Hyaenas (Welch et al. 2016). Roads are important in the use of space by Brown Hyaena, possibly in providing foraging routes and as possible sites for intra- and inter-clan communications. Additionally, they may facilitate interactions with other large predators that provide scavenging opportunities for Brown Hyaenas (Welch et al. 2016).
In the Kgalagadi Transfrontier Park, it has been shown to be primarily a scavenger consuming a wide range of vertebrate remains, which is supplemented by wild fruits, insects, birds’ eggs and the occasional small animal which is killed (Mills & Mills 1978), and it feeds on similar food resources elsewhere in its range (Maddock 1993; Burgener & Gusset 2003; Barker 2012; Yarnell et al. 2013; Slater & Muller 2014; Ramnanan et al. 2016; Comley et al. 2018; Williams et al. 2018; Faure et al. 2019; Van der Merwe et al. 2020; Honiball et al. 2021), in many cases, its prey use is a function of prey availability (Williams et al. 2028). Although reported widely to prey upon livestock such as sheep (Skinner 1976; Stuart 1981; Pirie et al. 2017), and cattle (Van As 2012; Badenhorst 2014), actual dietary studies , report either no use of livestock (Ramnanan et al. 2016), or very little use of it (Faure et al. 2019; Maude and Mills 2005; Stein et al. 2013; Williams et al. 2018) and in many occasions this is likely to have been acquired through scavenging (Williams et al. 2018), suggesting minimal impact on domestic livestock (Mills 1998; Maude 2005). However, despite posing a comparatively minor threat to livestock in many cases, perceived conflict or misidentification of the predator responsible for losses may be driving retributive killings of brown hyaenas (Marker et al. 2003). Given they feed on large mammals such as ungulates, most of which is likely scavenged after large predators have departed (Barker 2012; Yarnell et al. 2013; Comley et al. 2018; Williams et al. 2018; Faure et al. 2019; Van der Merwe et al. 2020; Honiball et al. 2021), this behaviour may bring them into conflict with game farming enterprises (Schepers 2016; Pirie et al. 2017).
Brown Hyaenas compete with Spotted Hyaenas Crocuta crocuta (Mills 1984; Mills and Funston 2003), and on fenced reserve they avoid them both spatially and temporally (Barker 2012). Spotted Hyaena and human abundance negatively influenced Brown Hyaena occupancy across 25 protected and unprotected sites across South Africa (Williams et al. 2021a). While their diet may overlap, Brown Hyaenas have a broader dietary breadth than Spotted Hyaenas (Barker 2012; Honiball et al. 2021). Management practices have favoured the Spotted Hyaena in the Kruger National Park, leading to the Brown Hyaena being excluded as a breeding species (Mills 1990; Skinner & Chimimba 2005), which point to their local extinction, or uncertain presence at best. However, in the Kgalagadi Transfrontier Park it was found that although the Brown Hyaena outnumbers the Spotted Hyaena by a ratio of about 2:1, in areas well frequented by Spotted Hyaenas, for example around dens, Brown Hyaenas were less often sighted (Mills 1990), and the low numbers of Spotted Hyaena have minimal impact on the Brown Hyaena population (Mills 1984). In many rangelands outside protected areas in South Africa, large carnivores are often extirpated, and Brown Hyaenas are sympatric with Black-backed Jackals Lupulella mesomelas, and Leopards Panthera pardus (Thorn et al. 2011b). In these areas, Brown Hyaena and Black-backed Jackals show high food resource overlap and potential for competition (Yarnell et al. 2013; Ramnanan et al. 2016), which is only tempered, when apex predators are present, such as lions Panthera leo, for instance (Van der Merwe et al. 2009; Yarnell et al. 2013; Comley et al. 2018; Bashant et al. 2020). In this situation, when large apex hunting predators are present, Brown hyaenas become the dominant scavenger in a system, relegating jackals to switch prey, i.e. small mammal hunting (Van der Merwe 2009; Yarnell et al. 2013). In fact, Brown Hyaenas benefit from food facilitation from African Wild Dogs Lycaon pictus (Yarnell et al. 2023), lions (Yarnell et al. 2013; Comley et al. 2018, Bashant et al. 2020), Cheetahs (Slater & Muller 2014; Comley et al. 2018), and Leopards (Williams et al. 2018). Beyond stealing and feeding on abandoned carcasses, intensive farmland studies have also yielded evidence of regular kleptoparasitism of fresh Leopard prey by Brown Hyaenas (Weise, F. pers. obs.). Leopard had a positive effect on Brown Hyaena occupancy across their South African range (Williams et al. 2021a) and may function as a keystone species for Brown Hyaena on private land (Williams et al. 2018).
Although 65% of Brown Hyaenas live in mixed sex clans (Mills 1983) of 4–14 individuals (Mills 1982), they are solitary foragers who spend much of their time alone (Mills 1983; Owens and Owens 1996; Skinner and Chimimba 2005). A clan is generally composed of one dominant male and a dominant unrelated female (Mills 1982; Knowles et al. 2009), several additional females, natal male adults, subadults and cubs (Mills 1982; Owens and Owens 1996).
Brown Hyaenas are territorial (Mills 1983), and in addition to defecating at latrines (Mills 1990), they also mark their territories using pastings secreted from anal glands, and deposit these pastings in those areas in which they spend most of their time, which is normally in the central part of their territory (Mills et al. 1980). Brown Hyaenas increase the frequency of anal pasting when they visit their territorial boundaries (Mills et al. 1980).
Behavioural observations show that Brown Hyaenas can recognise different individuals’ pastings. Pasting could function to inform group members of each other’s movements as well as to inform outsiders that the territory is occupied (Mills et al. 1980). Brown Hyaenas also defecate in discrete latrines, which are periodically visited (Mills 1990), and in the Waterberg of the Limpopo, Brown Hyaena defecation rates at latrine sites were low i.e. 1 defecation/30 days, but visited sites significantly more often than they defecated at them (median: 2.6 visits/30 days) (Hulsman et al. 2010).
Figure 2. Brown Hyaena cub at a den that was established next to a tourist road during the Covid-19 Lockdown at the Botsalano Nature Reserve, April 2020 (Photo by North West Provincial Government).
The smallest recorded home-range of a Brown Hyaena is at Kwandwe Game Reserve in the Eastern Cape of 26.3 km2 (Welch & Parker 2016).
While even outside reserves, they can still be remarkably small, where they are estimated to be 2.5 times smaller than some protected populations (Richmond-Coggan 2014). In Limpopo and the North West, in unprotected areas, male home-ranges averaged 22.8 km2 and females averaged 38.4 km2 (Richmond-Coggan 2014), while they can range up to 169.8 km2 in northern Limpopo (Williams 2017). In protected areas of the North West, male home-ranges averaged 108.9 km2, based on two individuals, and females averaged 101 km2 (Richmond-Coggan 2014), which is interesting, as ranges appear larger inside protected areas, which is unexpected.
Ecosystem and cultural services: As a mammalian scavenger, the Brown Hyaena plays a key role in the ecosystem by cleaning up carrion. This service can prevent the spread of diseases and assist other scavengers or decomposers to meet their feeding requirements (Mills 1990; Yarnell et al. 2016). Brown Hyaena can thus play a role in the recycling of nutrients from crushed bones they consume (Abraham et al. 2022).
Many farmers, especially on hunting farms, make use of this service by leaving carcasses and offal in the veld for Brown Hyaenas and other scavengers to consume (Yarnell et al. 2016), or they are deliberately placed at another threatened vertebrate attraction site, a ‘vulture restaurant’ (Yarnell et al. 2015).
Brown Hyaenas are also competitors with mesocarnivores such as Black-backed Jackals (Van der Merwe et al. 2009; Yarnell et al. 2013) that can cause economic losses to farmers (Badenhorst 2014; Schepers 2016; Pirie et al. 2017) and thus Brown Hyaenas may be able to help to alleviate such losses.
Brown Hyaena are solitary scavengers which carry food back to their cubs at their natal dens (Fig 2), thereby contributing to extensive bone assemblages at den sites, which, in turn, reflect the faunal assemblage in adjacent areas to the den (Skinner 2006). These sites then over time can render insight into the palaeoecology of the area, with its attendant faunal assemblages, and not to mention Brown Hyaena behaviour (Berger et al. 2009), or their interactions with early hominins (Arriaza et al. 2021), or more contemporary ones (Klein et al. 1999).
As one of the few widely distributed large carnivores left in South Africa, and given their elusiveness, Brown Hyaenas can be considered charismatic and are used to educate children about the natural world and conservation (Yarnell et al. 2016).
While the Kgalagadi Transfrontier Park population has always been touted as the classic landscape to see the species for photographic tourists (Mills 1990), the larger populace of the country, can within a reasonable drive of two hours from the Gauteng cities, and at a reasonable cost even, get to see and photograph them in the Pilanesberg National Park. However, for more discerning visitors, seeking exclusivity, and the higher price, the Madikwe Game Reserve and Tswalu Kalahari Reserve can offer unrivalled viewing of the species, and with the expert attention of local guides.
The Brown Hyaena features in local stories and songs in northern Limpopo; however, these cultural connections are significantly more prominent with older people, which suggests that many of this species’ cultural links may phase out with the younger generation. Frequently, the hyaena is portrayed in local folklore as a character who is ignorant, easily fooled or strongly connected with witchcraft (Yarnell et al. 2016). In very rare instances, farm workers may cut small tufts of hair off the coat of a Brown Hyaena to brew a tincture that is dispensed around the farm house, in belief that it may keep away bad spirits and the Tokoloshe (Weise, F. pers. obs.).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.7. Shrubland -> Shrubland – Subtropical/Tropical High Altitude | – | Marginal | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 5.13. Wetlands (inland) -> Wetlands (inland) – Permanent Inland Deltas | – | Suitable | – |
| 6. Rocky areas (eg. inland cliffs, mountain peaks) | – | Suitable | – |
| 8.3. Desert -> Desert – Cold | – | Suitable | – |
| 12.1. Marine Intertidal -> Marine Intertidal – Rocky Shoreline | – | Suitable | – |
| 12.2. Marine Intertidal -> Marine Intertidal – Sandy Shoreline and/or Beaches, Sand Bars, Spits, Etc | – | Suitable | – |
| 12.5. Marine Intertidal -> Marine Intertidal – Salt Marshes (Emergent Grasses) | – | Marginal | – |
| 13.3. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Coastal Sand Dunes | – | Suitable | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Marginal | – |
Life History
Generation Length: 8.5
Age at Maturity: Female or unspecified: 3 years
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 16 years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 3 months
Reproductive Periodicity: Irregular
Average Annual Fecundity or Litter Size: 2-3
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: Not a Migrant although 8% of all subadults and adults become nomadic (Mills 1982) but has propensity to move over long distances.
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Although less coveted for the traditional medicine market than Spotted Hyaenas (Hofer and Mills 1998b), both glands and organs are utilised from specimens taken opportunistically, for example, as roadkill. However, some traditional healers and rural people in the Soutpansberg Mountains, Limpopo Province, stated that the Brown Hyaena has greater magical properties than Spotted Hyaena, but this may be explained by greater abundance and local availability (Williams 2017).
At the Faraday Market in Gauteng in 2005, 1 of 32 traders interviewed stock body parts of this species, and 1 body part in total were reported (Whiting et al. 2016). A questionnaire-based study in a part of the Western Cape that took place in 2018 obtained information on various species, and the parts of an animal used, as well as their purpose and price (see Nieman 2018; Nieman et al. 2019). There, Brown Hyaena skin were used for decorative purposes (Nieman et al. 2019), and could be sold for between R700 and R1,100. The subcutaneous fat of the same species would be used for a belief purpose and thus was to attract good fortune (Nieman et al. 2019), and its fat could be sold for between R550 and R800 (Nieman 2018). The Cultural Significance Index (CSI) was 1.75 for the species, but considered low when compared to the leopard at 15 (Nieman 2018). The use of the species may be low regionally, simply because of its rarity in the area (Nieman 2018), and it may be that there is more of a demand where the species is more prevalent.
Interviews in Limpopo Province indicate that Brown Hyaena body parts have various usages including to protect from theft or alternatively to enable house breaking and facilitate theft, to locate lost items, to improve business, to treat mental illness and as a hunting aid (Williams 2017). Sangomas in the Soutpansberg area of Limpopo province stated that they acquire parts from roadkill, people who catch and sell them, and from farmers who shoot the animals (Williams 2017).
This species is persecuted as a damage-causing animal (Stuart 1981; Thorn et al. 2012; Richmond-Coggan 2014; Badenhorst 2014; Pirie et al. 2017), but despite potentially high levels of persecution (Pirie et al. 2017), it still persists across a large proportion of South Africa, and an improvement in landowner attitudes towards the species would certainly enhance its population persistence (Thorn et al. 2012; Richmond-Coggan 2014; Yarnell et al. 2016).
Brown Hyaenas are not commonly hunted for trophies but with a permit, it is possible. Trophy hunting Brown Hyaenas is mainly of interest to hunters who are specifically aiming to collect a wide variety of species and will consequently target less commonly hunted species. The Brown Hyaena is protected under the United States Endangered Species Act (ESA) and, therefore, it is unlawful to import this species as a trophy into the United States, which may account for the low popularity in trophy hunting Brown Hyaena by American clients. For instance, in the North West province, since 2020, only 4% of Brown Hyaena hunting applicants were US citizens (n = 25, NW Directorate of Biodiversity Management records, North West Provincial Government). The Professional Hunters (PG) register should render insights into the trends of trophy hunting of the species, where in 2016, 60 were hunted, 41 in 2017, 30 in 2018, and 34 in 2019 (PH Register, SANBI & DFFE data bases). Of the 20 hunting applications to hunt the species in North West Province since 2020, 65% were recommended favourably, and this is tied to a policy guiding this process (NW Directorate of Biodiversity Management records, North West Provincial Government).
The effects of wildlife ranching are currently unknown, and further research is required to determine the impacts of wildlife ranching on ranched species and non-ranched or associated species. However, we have observed that Brown Hyaenas can do well on game/wildlife farms, if the property is lightly managed (Richmond-Coggan 2014). For example, if supplementary feeding is set at a vulture restaurant Brown Hyaenas are likely to increase in local density (Yarnell et al. 2015). Ranches may also provide important pathways for dispersal (Richmond-Coggan 2014; Yarnell et al. 2016). One ranch in particular, Mafunyane Game Reserve in the North West, did in 2008, avail three brown hyaenas (1 adult female, 1 adult male, and 1 sub-adult female) that were reintroduced to Mountain Zebra National Park, and so too did Thaba Tholo assist in Limpopo too (Comley 2016; Welch et al. 2016).
However, management recommendations should focus more on reducing persecution (Thorn et al. 2012; Richmond-Coggan 2014; Yarnell et al. 2016; Faure et al. 2022).
Subsistence: No
Rationale: –
Local Commercial: Yes
Further detail including information on economic value if available: Traded for medicinal use and ceremonies. Limited trophy hunting.
National Commercial Value: Unknown
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 3. Medicine – human & veterinary | true | – | – | – |
| 15. Sport hunting/specimen collecting | – | – | true | – |
| 17. Other (free text) | true | – | – | traditional rituals |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: Majority harvested for medicine trade.
Threats
Threat categories have been assessed for various carnivore species in Africa, and each species is differentially impacted by a set of ten external threats commonly encountered by African carnivores (Ray et al. 2005). Each species was subjected to a scoring process for each threat that assessed the relative severity (its effect on the species), urgency (timescale over which it is most likely to occur), probability (likelihood of occurrence), and geographical extent of the threat through the species’ range in Africa. Of 20 larger carnivores assessed in Africa, the Brown Hyaena was ranked 10th in terms of its threat score, and 4th in terms of vulnerability (Ray et al. 2005). This pan-African assessment suggests the species to be relatively at risk.
Human-wildlife conflict
Most Brown Hyaenas in the north of South Africa, and assumedly across the country, live on private land outside of protected areas where they may come into conflict with humans (Richmond-Coggan 2014). They are often shot, poisoned, trapped, snared and hunted with dogs in an attempt to reduce livestock predation events (Stuart 1981; Mills 1998; Badenhorst 2014; Richmond-Coggan 2014; Schepers 2016; Pirie et al. 2017). St John et al. (2011) and Thorn et al. (2012) showed that poisoning, shooting and hunting of Brown Hyaenas frequently occurs in Limpopo and North West provinces, but with relatively fewer ranchers killing Brown Hyaena compared to Leopard (Panthera pardus), Black-backed Jackal and Caracal (Caracal caracal) (St John et al. 2012; Thorn et al. 2012). In North West Province, a few collared animals on unprotected rangelands were shot and reported on in the last assessment (Yarnell et al. 2016). Brown hyaenas are implicated in cattle depredations of 4% in North West Province (Badenhorst 2014), 7% in the Northern Cape (Badenhorst 2014), 7% in the Eastern Cape (Badenhorst 2014), 5-6% in Limpopo (Badenhorst 2014, Richmond-Coggan 2014), 13% in KwaZulu-Natal (Richmond-Coggan 2014), 3-11% in the Free State (Badenhorst 2014; Richmond-Coggan 2014), and as many as 20% for Mpumalanga (Badenhorst 2014). It is surprising that the species is an infrequent problem with cattle in the northern provinces, i.e. North West and Limpopo (see Badenhorst 2014; Richmond-Coggan 2014).
Sheep depredation has had serious consequences for Brown Hyaena historically (Skinner 1997; Stuart 1981), and interestingly, 17% of sheep losses are ascribed to the Brown Hyaena in the Western Cape (Richmond-Coggan 2014), which is important considering the species has re-colonised some parts of the province since the mid-2000s (Richmond-Coggan 2014; Yarnell et al. 2016). Regionally across all livestock, Brown Hyaena are implicated in 12% of losses in the North West province (Thorn et al. 2012). Locally, in Mpumalanga, in the Lydenburg area, Brown Hyaena account for as many as 25% of livestock head removed (Pirie et al. 2017), and it seems the Mpumalanga is the most problematic province for depredations by this species.
Since game has commercial value, almost on a par with livestock (Pirie et al. 2017), Brown Hyaenas are also blamed for depredations upon game kept by landowners on game farms (Richmond-Coggan 2014; Schepers 2016). Locally, in Mpumalanga, 11% of farmers reported game losses to Brown Hyaena (Pirie et al. 2017), which is 25% at the provincial level for small to large game kept (Schepers 2016). Brown Hyaenas are implicated in the large game losses of 33.3 % in KwaZulu-Natal, 3.1 % in Free State, 6.5 % in North West, and 22.4 % of Limpopo farms (Schepers 2016). For small game kept, Brown Hyaena ascribed losses occur on 22.9 % of Limpopo farms, 6.5 % of North West farms and 1.5% of Free State farms (Schepers 2016). As for the more scarce and rare colour variant game, Brown Hyaenas are implicated in losses on 0.5% of Limpopo farms, 1.6% North West farms, 1.5 % of Free State farms (Schepers 2016) and the lessened values are obviously related to the efforts landowners make to protect them from predation.
Regionally, in 2008 in the North West province, Brown Hyaena were estimated to be removed by farmers at a rate of 0.1 Hyaenas/100 km2 per annum, leading to 55 removed provincially per year (Thorn et al. 2012). While nationally, in a 2011 study, throughout South Africa, Brown Hyaena were estimated to be removed through retaliatory killing at 0.09 Hyaenas/100 km2/ per annum (Richmond-Coggan 2014). Simultaneously, to this latter study, Badenhorst (2014) gathered the same information, from whence similar spatially related off-takes could be determined retrospectively. Between 2011 and 2012, the following removals of Brown Hyaenas were estimated to have been undertaken by farmers in response to cattle predation, in Hyaenas removed/100 km2: North West -0.15, Limpopo – 0.12, KwaZulu-Natal – 0.05 and Eastern Cape – 0.02 (after Badenhorst 2014).
Similarly, in 2015, through extrapolation based on recorded off-takes of Brown Hyaena from retaliatory killings in the Lydenburg area of Mpumalanga (Pirie et al. 2017), it was estimated that 0.5 Brown Hyaenas were removed/ 100 km2 which was higher than the regional assessments. Similarly, in the Waterberg of the Limpopo in 2011, 4.4 % of large livestock (cattle), and 2.2% of game losses were ascribed to the Brown Hyaena, respectively (Thorn et al. 2013) which was a relatively small impact. Despite this low predation rate, the off-take of Brown Hyeana from Waterberg ranches in the Limpopo in 2011 was estimated at 1,17 Brown Hyaena / 100 km2 (Thorn et al. 2013). Since this approximates the density determined at Lapalala five years later of 1,33 Hyaenas/100 km2 (Williams et al. 2021), this suggests the population may exist in a perpetual sink and may be under pressure. The higher off-take removal rate in the Waterberg (Thorn et al. 2013), can be related to scale, where this estimate is lower when upscaled to the level of a province (Thorn et al. 2012; Badenhorst 2014) and country (Richmond-Coggan 2014).
It is remarkable that the regional off-takes of Brown Hyaenas for North West alone can vary from 0.1-0.15 Hyaenas/100 km2 (Thorn et al. 2012, Badenhorst 2014), and yet these studies were done independently of one another. In the North West Province, since 2020, there have been 25 complaints regarding Brown Hyaena, where in most cases correct husbandry is advised upon, but at the extreme, 4 individuals have had to be translocated owing to human-wildlife conflict (Directorate of Biodiversity Management, North West Provincial Management records), while no destruction permits have been issued.
Thirty-two per cent of interviewed landowners or managers on commercial farms living in and around the Soutpansberg Mountains, Limpopo Province, reported livestock or game losses caused by Brown Hyaenas. The majority of these respondents did not respond with lethal measures. In this area, conflict with Leopards was much more problematic, affecting 58% of respondents, and this may have deferred the focus away from Brown Hyaenas (Williams 2017). Of all carnivores enquired upon in South Africa, only the Brown Hyaena appeared to divide opinion with 25 % of respondents stating they were highly favourable compared to 26 % stating they were highly unfavourable (Richmond-Coggan 2014), so there is uncertainty with regard tolerance thresholds with the species, which can only be improved with education.
By 2016, five of six Brown Hyaena reintroduction sites in the Eastern Cape had reported breakouts (Banasiak et al. 2021b). There, Brown Hyaenas were the most frequently reported predator to escape from reserves where they have been reintroduced to as 38.1% of neighbouring properties report this species has entered their properties (Banasiak et al. 2021b). On these peripheries of reserves, this species was reported to have attacked sheep, goats, cattle and game (Banasiak et al. 2021b).
In the North West province, and northern South Africa in general, the large number of snares used for bushmeat is still cause for concern (Yarnell et al. 2016), making it increasingly likely that snaring and other illegal persecution by humans is a major cause of unrecorded mortality that is likely to suppress Brown Hyaena population numbers. In the North West Province, since 2020, one Brown Hyaena was rescued from a snare and repatriated with the assistance of the North West Parks and Tourism Board, which is far fewer than the number of Leopards that are annually rescued from snares (Directorate of Biodiversity Management, North West Provincial Government). However, it is unknown whether such levels of persecution are having a tangible impact on populations, and further research is needed in this area.
If future landowners maintain similar attitudes towards Brown Hyaena then it is likely that these threats will persist. The threats are ongoing, and it is unknown whether they are likely to increase or cease in the future.
Roadkill
Brown Hyaena are sometimes killed on roads (Collinson et al. 2015; Weise et al. 2015), although the severity of this to populations is not always known. However, in a study of roadkill by Collinson et al. (2015) in northern Limpopo Province (Venetia Limpopo Nature Reserve) along paved and unpaved roads (sampling distance was 14,400 km over 120 days split by season: 40 days of sampling in each season from October 2011 to July 2012), and over this period of 10 months, two Brown Hyaena carcasses were detected as being killed by a motor vehicle on the road transect driven. Given the 10-month study period, this would amount to 0.76 Hyaenas killed by roads per annum which may have an impact, but may not be that severe, with all things being equal. If the Brown Hyaena relationship with roads is similar outside of protected areas, it could represent a considerable threat to the species from road-related mortality and warrants further investigation (Welch et al. 2016). Though road related mortality does not seem to pose a risk to the species in South Africa.
Use and trade
In the Western Cape, in a survey amongst traders, Brown Hyaena parts were reported from 23.5% community respondents (Nieman 2018), which was a high frequency, considering the rare and threatened status of the species (Nieman et al. 2019). The species was not recorded locally (Nieman 2018), but is increasingly being found to occur in the wider region of the province (Yarnell et al. 2016), and given the language of interviewees, who were both Sesotho and Xhosa, but mostly Xhosa (Nieman et al. 2019), an hypothesis could be that body parts of the species have been transported into the area from their stronghold ranges to the north and east (Figure 1).
Genetics
Small, isolated subpopulations in reserves surrounded by predator-proof fencing may be at risk of inbreeding depression impacting the populations and some management to maintain genetically diverse populations is recommended (Richmond-Coggan 2014, Comley 2016, Welch & Parker 2016, Williams et al. 2021b). This is mindful of the already low genetic heterogeneity the species possesses (Westbury et al. 2018).
Conservation
Brown Hyaenas occur in a number of large conservation areas, including the most important of which are the following: the Kgalagadi Transfrontier Park (South Africa and Botswana), Pilanesberg National Park, Madikwe Game Reserve, Marakele National Park, Mapungubwe National Park, Karoo National Park, Greater Addo Elephant National Park, and Mountain Zebra National Park to name a few. Additionally, they are found outside protected areas (Thorn et al. 2011b, Richmond-Coggan 2014). Furthermore, private landowners also play their part in nurturing existing populations such as Tswalu Kalahari Reserve and !Khamab Kalahari Reserve.
Legal protection is in place for the Brown Hyaena making it illegal to kill the species without a permit. However, most killing occurs without a permit (Thorn et al. 2012; Badenhorst 2014; Schepers 2016; Pirie et al. 2017), though only 6% of farmers have admitted to this, which is less than for transgressions with leopard (St John et al. 2011). Educational campaigns should thus be conducted to educate landowners and farmers that Brown Hyaenas are predominantly scavengers and that they do not pose a threat to humans. They are shy and elusive and usually do not prey on large animals (Faure et al. 2019). The threat that Brown Hyaenas pose to livestock remains uncertain and misinforming farmers may go against the desired outcomes (Yarnell et al. 2016). Holistic management methods, such as the use of livestock guarding dogs, should also be encouraged (see Fink et al. 2020).
Maude (2005) and Mills (1990) showed that hunting is unspecialised and opportunistic, making it possible for Brown Hyaenas to show different hunting habits in different environments and under different constraints: for example, where vegetation structure allows predator ambush and where natural food source is scarce. Using the Problem Animal Control Registers from the Kweneng District of Botswana, dated between 2000 and 2001, Schiess-Meier et al. (2007) showed that 12% (269 heads) of all livestock losses were attributed to Brown Hyaenas. The latter could be over-estimated due to the Brown Hyaena scavenging presence at carcasses.
Brown Hyaena depredations upon both game and livestock are prevalent in South Africa (Thorn et al. 2012; Badenhorst 2014; Richmond-Coggan 2014; Schepers 2016; Pirie et al. 2017). In response to this, farmers have resorted to persecute Brown Hyaena through the use of shooting, trapping, dog hunting and poisons (Badenhorst 2014; Richmond-Coggan 2014; Schepers 2016). Although Brown Hyaena are adept at escaping any fenced areas (Hayward et al. 2007; Richmond-Coggan 2014; Kesh et al. 2015; Banasiak et al. 2021b), there are some highly fortified fences where it may be near impossible to breach (Edwards et al. 2019; Williams et al. 2021b). In addition, it is important to reiterate that maintaining the integrity of a predator-proof fencing is crucial in protecting livestock as displayed by one particular predation event during a study involving a Brown Hyaena which entered a camp by creeping under a hole of an unmaintained fence (Fink et al. 2020).
Livestock Guarding Dogs (LDGs) are also highly recommended for mitigating human-wildlife conflict (Fink et al. 2020; Spencer et al. 2020). Across farms in northern Limpopo, Brown Hyaena occupancy was positively associated with the presence of LDGs (Spencer et al. 2020), suggesting their presence allowed them to persist in the landscape.
To quantify livestock predation risks posed by carnivores in Namibia, Fink et al. (2020) developed occupancy models to make inferences about the factors influencing presence and temporal activity patterns of two carnivore species. They found that livestock were most at risk from predation from Brown Hyaenas at night and in riparian habitats. Their results suggested that farmers can reduce risks by implementing animal husbandry practices to include protecting livestock at night using methods such as night-time corrals and LGDs, or herders (Fink et al. 2020).
Spatial and temporal information about local predator visitation rates on a farm should guide preventative management. They recommend preventing livestock from entering high-risk areas during risky times. Corralling livestock from 17h00 hours until 07h59 hours would reduce exposure to predation, given Brown Hyaena were both nocturnal and partly crepuscular, as in active at dawn (Fink et al. 2020).
Landowners should also be encouraged to form conservancies to enhance the rangelands and provide a safe contiguous area for Brown Hyaena populations to thrive. Landowners should conduct regular snare sweeps and improve anti-poaching measures on their properties. Initiating a widespread anti-snaring programme and improving education on the negative effects snaring has on non-target animals is also necessary (Yarnell et al. 2016).
Reintroductions, however, are not encouraged as a conservation tool as they are not needed and may divert conservation resources away from priority conservation measures (Yarnell et al. 2016). However, it must however be praised that the Eastern Cape reintroductions have been a success, albeit the authors describe this as uncertainty or moderate success (Banasiak et al. 2021a), given the difficulty to monitor the species, and the low priority the species is at, given the other charismatic carnivores (see Hayward et al. 2007; Banasiak et al. 2021a), some local populations have thrived (Welch & Parker 2016), which in part could be seen as successful. Given the concerns of inbreeding depression from perpetual confinement on reserves (Richmond-Coggan 2014; Comley 2016; Welch & Parker 2016), the principles of the metapopulation should be considered in some circumstances, as has been done with other larger carnivores (see Akçakaya et al. 2007).
In some cases, translocations of problem Brown Hyaenas have occurred but these are isolated examples which are prone to failure due to the social characteristics and territorial nature of the species (Skinner & Van Aarde 1987; Weise et al. 2015), and at least in the North West Province, some of the past translocations have had limited success with Hyaenas establishing away from the release sites (Skinner & Van Aarde 1987), and more recent ones in the North West, having been rather haphazard with only one of the four being collared only (Cheetah Outreach Trust & Directorate of Biodiversity Management, North West Provincial Government).
Notwithstanding, the small reserves in the Eastern Cape that have reintroduced Brown Hyaenas have subpopulations that are thriving as these have been well co-ordinated, documented and studied (see Hayward et al. 2007; Comley 2016; Welch & Parker 2016; Welch et al. 2016; Banasiak et al. 2021a; Williams et al. 2021b). If any translocations or reintroductions are proposed, they should follow the IUCN guidelines for translocation and reintroduction of species (IUCN 2013) and seek expert guidance and government approval. The subpopulations should also be closely monitored thereafter and any NGOs purporting to do this, should be pressurised to publish their outcomes, as these events are often used for short-term publicity gain, without any thought on actual long-term post-release monitoring needing to be done.
Recommendations for land managers and practitioners:
- Consult and follow the guidelines of the 1998 Hyaena Conservation Action Plan (Mills and Hofer 1998).
- Monitoring only occurs at a few study sites. A national monitoring programme would be welcomed. Most effective large-scale monitoring would involve sign-based (e.g., from scat) occupancy field surveys or landowner questionnaires, which can be very intensive on time and resources (see Thorn et al. 2011a), and understandably, authorities are averse to prioritise such.
- With the use of citizen science, and technologies such as camera traps, a multi-species carnivore monitoring programme could easily be initiated in South Africa.
- Secondary use of Panthera’s South African Leopard monitoring programme, where bycatch data on Brown Hyaena can be used, as it has already (Williams et al. 2021a; Williams et al. 2021b), herein a more priority, charismatic felid is ‘piggybacked’ by a less charismatic, but no less important species to accrue crucial population trend data.
- Establish ‘vulture restaurants’ with the view to locally enhance Brown Hyaena populations (Yarnell et al. 2015), and to dissuade them from foraging in areas where risks of poisoning and other persecution are high.
- Farm managers on livestock ranch lands to employ appropriate night-time kraaling, fence exclusion, human guarding and LGDs to minimise predation by Brown Hyaena (Fink et al. 2020; Spencer et al. 2020).
- Regular visible snare removal patrols should be incorporated in land management. Enforcement of a zero-tolerance policy for snaring is recommended. This includes forewarning all permanent and seasonal workers that snaring is illegal and will not be tolerated (include a clause in contracts).
- Where predators, and Brown Hyaenas are reintroduced, baselines need to be shifted (Banasiak et al. 2021b), in that neighbouring landowners of reserves should tolerate some degree of incursion of the species, and to appreciate it enough, so as to avert its being extirpated again.
Research priorities:
- Accurate population or occupancy assessments throughout the majority of the species range – particularly in unprotected rangelands.
- Accurate data on persecution rates and influence on population growth rates/population persistence.
- Effects of management regimes on private subpopulations.
- Assess the hunting abilities of the Brown Hyaena and the risk posed to small stock, especially in systems where natural food sources are scarce and the vegetation structure/landscape allows predator ambush.
- Genetic studies, especially on small, enclosed reserves (to determine the level of inbreeding) as well as across the species’ range (to determine movement between populations).
- To address research gaps and inform Hyaenidae management, it is recommended that there is to be increased applied research outside of protected areas using interdisciplinary and community-involved methods to increase foundational knowledge on understudied populations of the species (Wilkinson et al. 2024).
- To assess the impact of climate change on Brown Hyaenas (Richmond-Coggan 2014), and distribution modelling may be a good starting point (see Killea 2020).
Encourage citizen actions:
- Report sightings of free-roaming individuals both inside and outside protected areas on virtual museum platforms (e.g., iNaturalist, MammalMAP, African Carnivore Wildbook (https://africancarnivore.wildbook.org/submit.jsp)).
- Do not set out poison or snares on properties, and remove all snares encountered, and to join volunteer initiatives to rid areas of snares. Report snares found in the Cape provinces (https://app.capeleopard.org.za/).
- Drive slowly and carefully through Brown Hyaena habitats to avoid hitting and injuring / killing animals.
Bibliography
Abade L, MacDonald DW, Dickman AJ. 2014. Using landscape and bioclimatic features to predict the distribution of lions, leopards and spotted hyaenas in Tanzania’s Ruaha landscape. PLoS One 9(5): e96261.
Abraham, A.J., Webster, A.B., Jordaan, J., Prys-Jones, T.O., Ganswindt, A., De Jager, P.C. & Doughty, C.E. 2022. Hyaenas play unique ecosystem role by recycling key nutrients in bones. African Journal of Ecology 60(1): 81-86.
Akçakaya, H.R., Mills, G. & Doncaster, C.P. 2007. The role of metapopulations in conservation. Key topics in conservation biology 1: 64-84.
Arriaza, M.C., Aramendi, J., Maté-González, M.Á., Yravedra, J. & Stratford, D. 2021. The hunted or the scavenged? Australopith accumulation by brown hyenas at Sterkfontein (South Africa). Quaternary Science Reviews 273, 107252.
Badenhorst, C.G. 2014. The economic cost of large stock predation in the North West Province of South Africa. Msc Agric thesis, University of Free State, Bloemfontein.
Banasiak, N.M., Hayward, M.W. & Kerley, G.I. 2021a. Ten years on: have large carnivore reintroductions to the Eastern Cape Province, South Africa, worked?. African Journal of Wildlife Research 51(1): 111-126.
Banasiak, N.M., Hayward, M.W. & Kerley, G.I. 2021b. Emerging human-carnivore conflict following large carnivore reintroductions highlights the need to lift baselines. African Journal of Wildlife Research 51(1): 136-143.
Barker, N. 2012. Resource competition between spotted hyaenas, Crocuta crocuta,(Erxleben 1777), and brown hyaenas, Parahyaena brunnea,(Thunberg 1820). Master’s thesis, University of Pretoria, Pretoria.
Bashant, J., Somers, M., Swanepoel, L. & Dalerum, F. 2020. Facilitation or competition? Effects of lions on brown hyaenas and leopards. Diversity 12(9), 325.: https://doi.org/10.3390/d12090325.
Berger, L.R., Pickering, R., Kuhn, B., Backwell, L., Hancox, P.J., Kramers, J.D. & Boshoff, P. 2009. A Mid-Pleistocene in situ fossil brown hyaena (Parahyaena brunnea) latrine from Gladysvale Cave, South Africa. Palaeogeography, Palaeoclimatology, Palaeoecology 279(3-4): 131-136.
Burgener, N. & Gusset, M. 2003. The feeding habits of brown hyaenas (Hyaena brunnea) on a game ranch in Limpopo Province, South Africa. African Zoology 38(1): 181-184.
Collinson WJ, Parker DM, Bernard RTF, Reilly BK, Davies-Mostert HT. 2015. An inventory of vertebrate roadkill in the Greater Mapungubwe Transfrontier Conservation Area, South Africa. African Journal of Wildlife Research 45: 301–311.
Comley, J. 2016. Population assessment and feeding ecology of brown hyenas (Hyaena brunnea) in Mountain Zebra National Park, Eastern Cape, South Africa. Phd Doctoral dissertation, Rhodes University, Grahamstown.
Comley, J., Bissett, C., Tambling, C.J., Welch, R.J. & Parker, D.M. 2018. Diet of recently established brown hyaenas in the Eastern Cape, South Africa. African Journal of Wildlife Research 48(1): 1-7.
Déqué M, Calmanti S, Christensen OB, Aquila AD, Maule CF, Haensler A, Nikulin G & Teichmann C. 2017. A multi-model climate response over tropical Africa at +2°C. Climate Services, 7, 87–95.
Edwards, S., Noack, J., Heyns, L. & Rodenwoldt, D. 2019. Evidence of a high-density brown hyena population within an enclosed reserve: the role of fenced systems in conservation. Mammal Research 64(4): 519-527.
Faure, J.P.B., Holmes, N.J., Watson, L.H. & Hill, R.A. 2019. Brown hyaena (Parahyaena brunnea) diet composition from Zingela game reserve, Limpopo Province, South Africa. African Zoology 54(2): 119-124.
Faure, J.P.B., Swanepoel, L.H., Cilliers, D., Venter, J.A. & Hill, R.A. 2022. Estimates of carnivore densities in a human-dominated agricultural matrix in South Africa. Oryx 56(5): 774-781.
Ferreira SM and Viljoen P. 2022. African Large Carnivore Population Changes in Response to a Drought.”African Journal of Wildlife Research 52(1).
Fink, S., Chandler, R., Chamberlain, M., Castleberry, S. & Glosenger-Thrasher, S. 2020. Distribution and activity patterns of large carnivores and their implications for human–carnivore conflict management in Namibia. Human–Wildlife Interactions 14(2): 16. https://doi.org/10.26077/658e-e8a8
Foden, W.B. & Young, B.E. 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Gusset, M. & Burgener, N. 2005. Estimating larger carnivore numbers from track counts and measurements. African Journal of Ecology 43(4): 320-324.
Hall, A., & Thatcher, H. 2021. A confirmed sighting of brown hyaena, Parahyaena brunnea (Thunburg, 1820) in an extirpated area of the Western Cape, South Africa. African Journal of Ecology, 59(4): 1047-1050.
Hayward, M.W., Kerley, G.I., Adendorff, J., Moolman, L.C., O’brien, J., Sholto-Douglas, A., Bissett, C., Bean, P., Fogarty, A., Howarth, D. and Slater, R., 2007. The reintroduction of large carnivores to the Eastern Cape, South Africa: an assessment. Oryx 41(2): 205-214.
Hofer, H. and Mills, M.G.L. (eds). 1998. Hyaenas: Status Survey and Conservation Action Plan. pp. 154. IUCN, Gland, Switzerland and Cambridge, UK.
Hofer, H. and Mills, M.G.L. 1998a. Worldwide distribution of Hyaenas. In: M.G.L. Mills and H. Hofer (eds), Hyaenas. Status Survey and Conservation Action Plan, pp. 39-63. IUCN/SSC Hyaena Specialist Group. IUCN, Gland, Switzerland and Cambridge, UK.
Hofer, H. and Mills, M.G.L. 1998b. Population size, threats and conservation status of hyaenas. In: M.G.L. Mills and H. Hofer (eds), Hyaenas. Status Survey and Conservation Action Plan, pp. 64-79. IUCN/SSC Hyaena Specialist Group. IUCN, Gland, Switzerland and Cambridge, UK.
Honiball, T.L. 2021. Estimating the population size of three large carnivore species and the diet of six large carnivore species, in Madikwe Game Reserve. Msc Thesis, Nelson Mandela University, George.
Honiball, T.L., Somers, M.J., Fritz, H. & Venter, J.A. 2021. Feeding ecology of the large carnivore guild in Madikwe Game Reserve, South Africa. African Journal of Wildlife Research 51(1): 153-165.
Hulsman, A., Dalerum, F., Swanepoel, L., Ganswindt, A., Sutherland, C. & Paris, M. 2010. Patterns of scat deposition by brown hyaenas Hyaena brunnea in a mountain savannah region of South Africa. Wildlife Biology 16(4): 445-451.
IUCN/SSC. 2013. Guidelines for Reintroductions and Other Conservation Translocations. Version 1.0. Page viiii + 57 pp. IUCN Species Survival Commission, Gland, Switzerland.
Jenks, S. and Werdelin, L. 1998. Taxonomy and systematics of living hyaenas (family Hyaenidae). In: M.G.L. Mills and H. Hofer (eds), Hyaenas: status survey and conservation action plan, pp. 8-17. IUCN, Gland, Switzerland and Cambridge, UK.
Kent, V.T. and Hill, R.A. 2013. The importance of farmland for the conservation of the brown hyaena Parahyaena brunnea. Oryx 47(3): 431-440.
Kesch, M.K., Bauer, D.T. & Loveridge, A.J. 2015. Break on through to the other side: the effectiveness of game fencing to mitigate human-wildlife conflict. African Journal of Wildlife Research 45(1): 76-87.
Killea, A. 2020. Brown Hyena (Hyaena brunnea) Distribution: Nuances in Modeling a Generalist Species. Masters thesis project, Duke University, Durham, North Carolina.
Klein, R.G., Cruz-Uribe, K., Halkett, D., Hart, T. & Parkington, J.E. 1999. Paleoenvironmental and human behavioral implications of the Boegoeberg 1 late Pleistocene hyena den, Northern Cape Province, South Africa. Quaternary Research 52(3): 393-403.
Knowles JC, van Coeverden de Groot PJ, Wiesel I, Boag PT. 2009. Microsatellite variation in Namibian Brown Hyenas (Hyaena brunnea): population structure and mating system implications. Journal of Mammalogy 90: 1381–1391.
Koepfli, K., Jenks, S.M., Eizirik, E., Zahirpour, T., Van Valkenburgh, B. and Wayne, R.K. 2006. Molecular systematics of the Hyaenidae: Relationships of a relictual lineage resolved by a molecular supermatrix. Molecular Phylogenetics and Evolution 38(3): 603–620.
Kolowski J M and Holekamp KE. 2006. Spatial, temporal, and physical characteristics of livestock depredations by large carnivores along a Kenyan reserve border. Biological Conservation 128(4): 529-541.
Kuhn, B.F. 2014. A preliminary assessment of the carnivore community outside Johannesburg, South Africa. South African Journal of Wildlife Research 44(1): 95-98.
Lloyd, P. H. & Millar, J. C. G. 1984.A questionnaire survey (1969-1974) of some of the larger mammals of the Cape Province. Bontebok 3: 1-49.
Loock D, SwanepoeL L. 2019. Assessing local patterns in carnivore occupancy and richness by camera trapping: a monitoring of diversity at Verloren Vallei Nature Reserve, Dullstroom, Mpumalanga. Wildlife & Resource Association (WRA), Bloemfontein.
Maddock, A.H. 1993. Analysis of brown hyaena (Hyaena brunnea) scats from the central karoo, South Africa. Journal of Zoology 231(4): 679-683.
Marker, L.L., Mills, M.G.L. and Macdonald, D.W. (2003a) Factors influencing perceptions of conflict and tolerance toward cheetahs on Namibian farmlands. Conservation Biology 17, 1290-1298.
Maude, G. and Mills, M.G.L. 2005. The comparative feeding ecology of the brown hyaena in a cattle area and a national park in Botswana. South African Journal of Wildlife Research 35(2): 201-214.
McKaughan JE, Stephens PA, Hill RA. 2023. Estimating mesocarnivore abundance on commercial farmland using distance sampling with camera traps. Ecological Solutions and Evidence 4(2), p.e12229.
McManus JS, Dickman AJ, Gaynor D, Smuts BH, Macdonald DW. 2015. Dead or alive? Comparing costs and benefits of lethal and non-lethal human–wildlife conflict mitigation on livestock farms. Oryx 49: 687-695.
Mills MGL, Funston PJ. 2003. Large carnivore ecology and savanna heterogeneity. In: du Toit J, Rogers KH, Biggs HC. (ed.), The Kruger Experience, pp. 370–388. Island Press, Washington, DC, USA.
Mills MGL. 1982. Factors affecting group size and territory size of the brown hyaena, Hyaena brunnea, in the southern Kalahari. Journal of Zoology 198: 38–51.
Mills MGL. 1983. Behavioural mechanisms in territory and group maintenance of the brown hyaena, Hyaena brunnea, in the southern Kalahari. Animal Behaviour 31: 503–510.
Mills, M.L. 1984. The comparative behavioural ecology of the brown hyaena Hyaena brunnea and the spotted hyaena Crocuta crocuta in the southern Kalahari. Koedoe 27(2): 237-247.
Mills, M.G L. 1998. Brown hyaena Hyaena (Parahyaena) brunnea (Thunberg, 1820). In: M.G.L. Mills and H. Hofer (eds), Hyaenas. Status Survey and Conservation Action Plan, pp. 26- 29. IUCN/SSC Hyaena Specialist Group, Gland, Switzerland and Cambridge, UK.
Mills, M.G.L. 1990. Kalahari hyaenas: the comparative behavioural ecology of two species. Unwin Hyman, London.
Mills, M.G.L. 2013. Hyaena brunnea. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. Volume V: Carnivores, Pangolins, Equids and Rhinceroses, Bloomsbury Publishing, London.
Mills, M.G.L., Gorman, M.L. & Mills, M.E. 1980. The scent marking behaviour of the brown hyaena Hyaena brunnea. African Zoology 15(4): 240-248.
Mills, M.G.L. & Mills, M.E., 1978. The diet of the brown hyaena Hyaena brunnea in the southern Kalahari. Koedoe 21(1): 125-149.
Mills, M. G. L. & Mills, M.E.J. 1982. Factors affecting the movement patterns of brown hyaenas, Hyaena brunnea in the southern Kalahari. South African Journal of Wildlife Research 12(4): 111-117.
Nieman WA. 2018. Culture, Conflict, and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human Wildlife Interface. Msc thesis, University of Stellenbosch, Stellenbosch.
Nieman WA, Leslie AJ, Wilkinson A. 2019. Traditional medicinal animal use by Xhosa and Sotho communities in the Western Cape Province, South Africa. Journal of ethnobiology and ethnomedicine 15: 1-14.
Owens D, Owens M. 1996. Social dominance and reproductive patterns in brown hyaenas, Hyaena brunnea, of the central Kalahari desert. Animal Behaviour 51: 535–551.
Pérez-Claros, J.A. 2024. Unravelling the origin of the brown hyena (Parahyena brunnea) and its evolutionary and paleoecological implications for the Pachycrocuta lineage. Palaeontologia Electronica 27(1): 1-25.
Pirie, T.J., Thomas, R.L. & Fellowes, M.D. 2017. Increasing game prices may alter farmers’ behaviours towards leopards (Panthera pardus) and other carnivores in South Africa. PeerJ 5: p.e3369.
Power, R.J., Van Straaten, A., Schaller, R., Mooke, M., Boshoff, T. and H. P. Nel. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Pringle, J.A. 1977. The distribution of mammals in Natal. Part 2. Carnivora. Annals of the Natal Museum 23(1): 93-115.
Ramnanan, R., Thorn, M., Tambling, C.J. & Somers, M.J. 2016. Resource partitioning between black-backed jackal and brown hyaena in Waterberg Biosphere Reserve, South Africa. Canid Biol. Conserv. 19: 8-13.
Ray JC, Hunter L, Zigouris J. 2005. Setting Conservation and Research Priorities for Larger African Carnivores. WCS Working Paper No. 24. Wildlife Conservation Society, New York.
Rich, L.N., Winterbach, C.W., Maude, G., Klein, R., Mahupeleng, G.N., Boast, L. & Somers, M.J. 2017. Conservation implications of brown hyaena (Parahyaena brunnea) population densities and distribution across landscapes in Botswana. Koedoe: African Protected Area Conservation and Science 59(2): 1-16.
Richmond-Coggan L. 2014. Comparative abundance and ranging behaviour of brown hyaena (Parahyaena brunnea) inside and outside protected areas in South Africa. Ph.D. Nottingham Trent University.
Rowe-Rowe, D.T. 1992. The carnivores of Natal. Natal Parks Board, Pietermaritzburg.
Rust NA, Whitehouse-Tedd KM, MacMillan DC. 2013. Perceived efficacy of livestock-guarding dogs in South Africa: implications for cheetah conservation. Wildlife Society Bulletin 37: 690–697.
Schepers, A. 2016. The economic impact of predation in the wildlife ranching industry in Limpopo, South Africa. Msc Agric thesis, University of Free State, Bloemfontein.
Schiess-Meier M, Ramsauer S, Gabanapelo T, König B. 2007. Livestock predation – Insights from problem animal control registers in Botswana. Journal of Wildlife Management 71: 1267–1274.
Skinner, J D. 1976. Ecology of the brown hyaena Hyaena brunnea in the Transvaal with a distribution map for Southern Africa. South African Journal of Science 72: 262-269.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skinner, J.D. & Aarde, R.V. 1987. Range use by brown hyaenas Hyaena brunnea relocated in an agricultural area of the Transvaal. Journal of Zoology 212(2): 350-352.
Slater, K. & Muller, K. 2014. The diet of brown hyaenas (Hyaena brunnea) in Shamwari Game Reserve, Eastern Cape, South Africa. Koedoe: African Protected Area Conservation and Science 56(1),: 1-5.
Smith K, Venter JA, Peel M, Keith M, Somers MJ. 2023. Temporal partitioning and the potential for avoidance behaviour within South African carnivore communities. Ecology and Evolution. 13(8):e10380.
Spencer, K., Sambrook, M., Bremner-Harrison, S., Cilliers, D., Yarnell, R.W., Brummer, R. & Whitehouse-Tedd, K. 2020. Livestock guarding dogs enable human-carnivore coexistence: First evidence of equivalent carnivore occupancy on guarded and unguarded farms. Biological Conservation 241, p.108256.
St John, F.A.V., Keane, A.M., Edwards-Jones, G., Jones , L., Yarnell, R.W. and Jones, J.P.G. 2011. Identifying indicators of illegal behaviour: carnivore killing in human-managed landscapes. Proceedings of the Royal Society Series B-Biological Sciences 279(1729): 804-812.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Stuart CT, Macdonald IAW, Mills MGL. 1985. History, current status and conservation of large mammalian predators in Cape Province, Republic of South Africa. Biological Conservation 31: 7–19.
Thorn M, Green M, Bateman PW, Cameron EZ, Yarnell RW, Scott DM. 2010. Comparative efficacy of sign surveys, spotlighting and audio playbacks in a landscape-scale carnivore survey. South African Journal of Wildlife Research 40(1): 77-86.
Thorn, M., Green, M., Bateman, P.W., Waite, S. & Scott, D.M. 2011a. Brown hyaenas on roads: estimating carnivore occupancy and abundance using spatially auto-correlated sign survey replicates. Biological Conservation 144(6): 1799-1807.
Thorn, M., Green, M., Dalerum, F., Bateman, P.W. and Scott, D.M. 2012. What drives human-carnivore conflict in the North West Province of South Africa? Biological Conservation 150: 23-32.
Thorn, M., Green, M., Keith, M., Marnewick, K., Bateman, P.W., Cameron, E.Z. and Scott, D.M. 2011b. Large-scale distribution patterns of carnivores in northern South Africa: implications for conservation and monitoring. Oryx 45(4): 579-586.
Thorn M, Green M, Scott D, Marnewick K. 2013. Characteristics and determinants of human-carnivore conflict in South African farmland. Biodiversity and conservation 22: 1715-1730.
Thorn, M., Scott. D.M., Green, M., Bateman, P.W. and Cameron, E.Z. 2009. Estimating brown hyaena occupancy using baited camera traps. South African Journal of Wildlife Research 39(1): 1-10.
Van As, M. 2012. Livestock, leopards and brown hyaenas: conflicts of cohabitation in the Roodewalshoek Conservancy, Mpumalanga. Msc thesis, University of Free State, Bloemfontein.
Trouwborst, A. and A. Blackmore (2020). “Hot Dogs, Hungry Bears, and Wolves Running Out of Mountain—International Wildlife Law and the Effects of Climate Change on Large Carnivores.” Journal of International Wildlife Law & Policy 23(3): 212-238.
Van der Merwe, I., Tambling, C.J., Thorn, M., Scott, D.M., Yarnell, R.W., Green, M., Cameron, E.Z. & Bateman, P.W. 2009. An assessment of diet overlap of two mesocarnivores in the North West Province, South Africa. African Zoology 44(2): 288-291.
Van der Weyde, L. K., Tobler, M. W., Gielen, M. C., Cozzi, G., Weise, F. J., Adams, T., Bauer, D., Bennitt, E., Bowles, M., Brassine, A., Broekhuis, F., Chase, M., Collins, K., Finerty, G. E., Golabek, K., Hartley, R., Henley, S., Isden, J., Keeping, D., … Flyman, M. V. 2022. Collaboration for conservation: Assessing countrywide carnivore occupancy dynamics from sparse data. Diversity and Distributions (28): 917–929. https://doi.org/10.1111/ddi.13386
Von Richter, W. 1972. Remarks on present distribution and abundance of some South African carnivores. South African Journal of Wildlife Research 2(1): 9-16.
Weise FJ, Wiesel I, Lemeris Jr J, van Vuuren RJ. 2015. Evaluation of a conflict-related brown hyaena relocation in Central Namibia. South African Journal of Wildlife Research 45: 178–186.
Welch RJ, Parker DM. 2016. Brown hyaena population explosion: rapid population growth in a small, fenced system. Wildlife Research 43: 178–187.
Welch, R.J., Tambling, C.J., Bissett, C., Gaylard, A., Müller, K., Slater, K., Strauss, W.M. & Parker, D.M. 2016. Brown hyena habitat selection varies among sites in a semi-arid region of southern Africa. Journal of Mammalogy 97(2): 473-482.
Westbury, M.V., Hartmann, S., Barlow, A., Wiesel, I., Leo, V., Welch, R., Parker, D.M., Sicks, F., Ludwig, A., Dalén, L. & Hofreiter, M. 2018. Extended and continuous decline in effective population size results in low genomic diversity in the world’s rarest hyena species, the brown hyena. Molecular biology and evolution 35(5): 1225-1237.
Whiting MJ. Williams VL, Hibbits TJ. 2016. A picture of health? Animal use and the Faraday traditional medicine market, South Africa. Journal of Ethnopharmacology. 179:265-73.
Wiesel I. 2015. Parahyaena brunnea. The IUCN Red List of Threatened Species 2015.
Wilkinson, C.E., Dheer, A., Zett, T., Torrents‐Ticó, M., Yarnell, R.W., Bar Ziv, E., Bhandari, S., Jacobson, A. & Dloniak, S.M. 2024. Review of the global research on Hyaenidae and implications for conservation and management. Mammal Review 54(2): 193-212.
Wilkinson, C.E., Dheer, A., Zett, T., Torrets-Tico, M, Yarnell, R.W., Bar Ziv, E., Bhandari, S., Jacobson, A., Dloniak, S.M. 2024. Review of global research on Hyaenidae and implications for conservation and management. Mammal Review 54: 193-212.
Williams, K. S. 2017. Human-brown hyaena relationships and the role of mountainous environments as refuges in a postcolonial landscape. PhD thesis, Durham University, Durham.
Williams, K.S., Pitman, R.T., Mann, G.K., Whittington-Jones, G., Comley, J., Williams, S.T., Hill, R.A., Balme, G.A. & Parker, D.M. 2021a. Utilizing bycatch camera-trap data for broad-scale occupancy and conservation: a case study of the brown hyaena Parahyaena brunnea. Oryx 55(2): 216-226.
Williams, K.S., Williams, S.T., Fitzgerald, L.E., Sheppard, E.C. & Hill, R.A. 2018. Brown hyaena and leopard diets on private land in the Soutpansberg Mountains, South Africa. African Journal of Ecology 56(4): 1021-1027.
Williams, K.S., Williams, S.T., Welch, R.J.Marneweck, C.J., Mann, G.K., Pitman, R.T., Whittington-Jones, G., Balme, G.A., Parker, D.M. & Hill, R.A. 2021b. Assumptions about fence permeability influence density estimates for brown hyaenas across South Africa. Scientific Reports 11(620): https://doi.org/10.1038/s41598-020-77188-7.
Woodgate Z, Drouilly M, Distiller G, O’Riain MJ. 2023. The effect of multi-use landscapes on mammal assemblages and its implication for conservation. Land.12(3):599. https://doi.org/10.3390/land12030599
Wozencraft, W.C. 1993. Order Carnivora. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World: A Taxonomic and Geographic Reference. Second Edition, pp. 279-344. Smithsonian Institution Press, Washington, DC, USA.
Yarnell, R.W., Phipps, W.L., Burgess, L.P., Ellis, J.A., Harrison, S.W.R., Dell, S., MacTavish, D., MacTavish, L.M. and Scott, D.M. 2013. The influence of large predators on the feeding ecology of two African mesocarnivores: the black-backed jackal and the brown hyaena. South African Journal of Wildlife Research 43(2): 155-166.
Yarnell RW, Phipps WL, Dell S, MacTavish LM, Scott DM. 2015. Evidence that vulture restaurants increase the local abundance of mammalian Carnivores in South Africa. African Journal of Ecology 53: 287–294.
Yarnell RW, Richmond-Coggan L, Bussière E, Williams K, Bissett C, Welch R, Wiesel I. 2016. A conservation assessment of Parahyaena brunnea. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.