Brants’ Whistling Rat
Parotomys brantsii

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Parotomys brantsii – (A. Smith, 1834)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Parotomys – brantsii
Common Names: Brants’ Whistling Rat (English), Brants se Fluitrot (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note:
Molecular data suggest that Parotomys should be synonymised with Otomys (Taylor et al. 2011, 2014), but the genera are recognised as distinct pending a final phylogenetic analysis of species relationships within the Otomyinae (Monadjem et al. 2015). This species is similar to P. littledalei, but distinguishable, both by its dental morphology (grooved upper incisors) and by its whistle frequency and length (Le Roux et al. 2002; Monadjem et al. 2015).
Red List Status: LC – Least Concern
Assessment Information
Assessors: Schradin, C.1,2 & Da Silva, J.M.3
Reviewer: Smith, C.4
Institutions:1Institut pluridisciplinaire Hubert Curien,2University of the Witwatersrand,3South African National Biodiversity Institute,4Endangered Wildlife Trust
Previous Assessors and Reviewers: Schradin, C., Malan, G., Kerley, G. & Child, M.F.
Previous Contributors: Roxburgh, L., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Mondajem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern because of its wide distribution within the assessment region, its occurrence in several protected areas (including Kgalagadi Transfrontier Park, Goegap Nature Reserve and Tswalu Kalahari Reserve), and because there are no major documented threats that could cause range-wide decline. They are not exploited by humans. However, this is a species we need to flag as being potentially threatened due to projected aridification from climate change and overgrazing in some areas. This may affect the forage resources on which this species depends and may be exacerbated by the sensitivity of this rodent to high ambient temperatures that may limit foraging behaviour under hotter conditions. It became locally extinct in Goegap Nature Reserve after long periods of drought years in 2017 and only returned in low numbers in 2023. Given that this species displays a patchy distribution and undergoes population irruptions, it may qualify for listing under the C criterion in the future and will need to be reassessed once more data are generated. We recommend that more research and long-term monitoring of subpopulation trends, geographic distribution and threat level are undertaken.
Regional population effects: The bulk of the population exists in South Africa, so extra-regional rescue effect is minimal. It is a rapid disperser over short distances (< 1 km), especially into areas that were recently overgrazed. Long distance dispersal ability is unknown and would be dependent on corridors of suitable habitat. The successful colonisation of poorly vegetated mine dumps (Desmet & Cowling 1999) suggests an ability to establish in harsh habitats provided deep soils are available.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Regional Distribution and occurrence
Geographic Range
This species is restricted to the arid regions of southern Africa occurring in southwestern South Africa, southern Namibia and extreme southwestern Botswana (Skinner & Chimimba 2005). It is associated with open habitats and hard, sandy substrates throughout the arid Karoo and southern Kalahari regions (Monadjem et al. 2015). It prefers dry, coarse, sandy soils with adequate forage in the more arid parts of the Nama-Karoo and Succulent Karoo biomes (Skinner & Chimimba 2005). It occurs from sea level up to 1,000 m asl and generally inhabits regions with an annual rainfall of 300–500 mm.
The species distribution marginally abuts the North West Province, and it has been tentatively mentioned in provincial documents, but during a recent mammal survey, Power (2014) found no evidence of their existence in the province. Similarly, there are no recent records from the Free State except for a handful of historical records from the southwestern corner of the province (N.L. Avenant pers. comm. 2014). Historically, it was also recorded 15 km west of Steytlerville, Eastern Cape Province, but is no longer present (G.I.H. Kerley unpubl. data).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 1000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
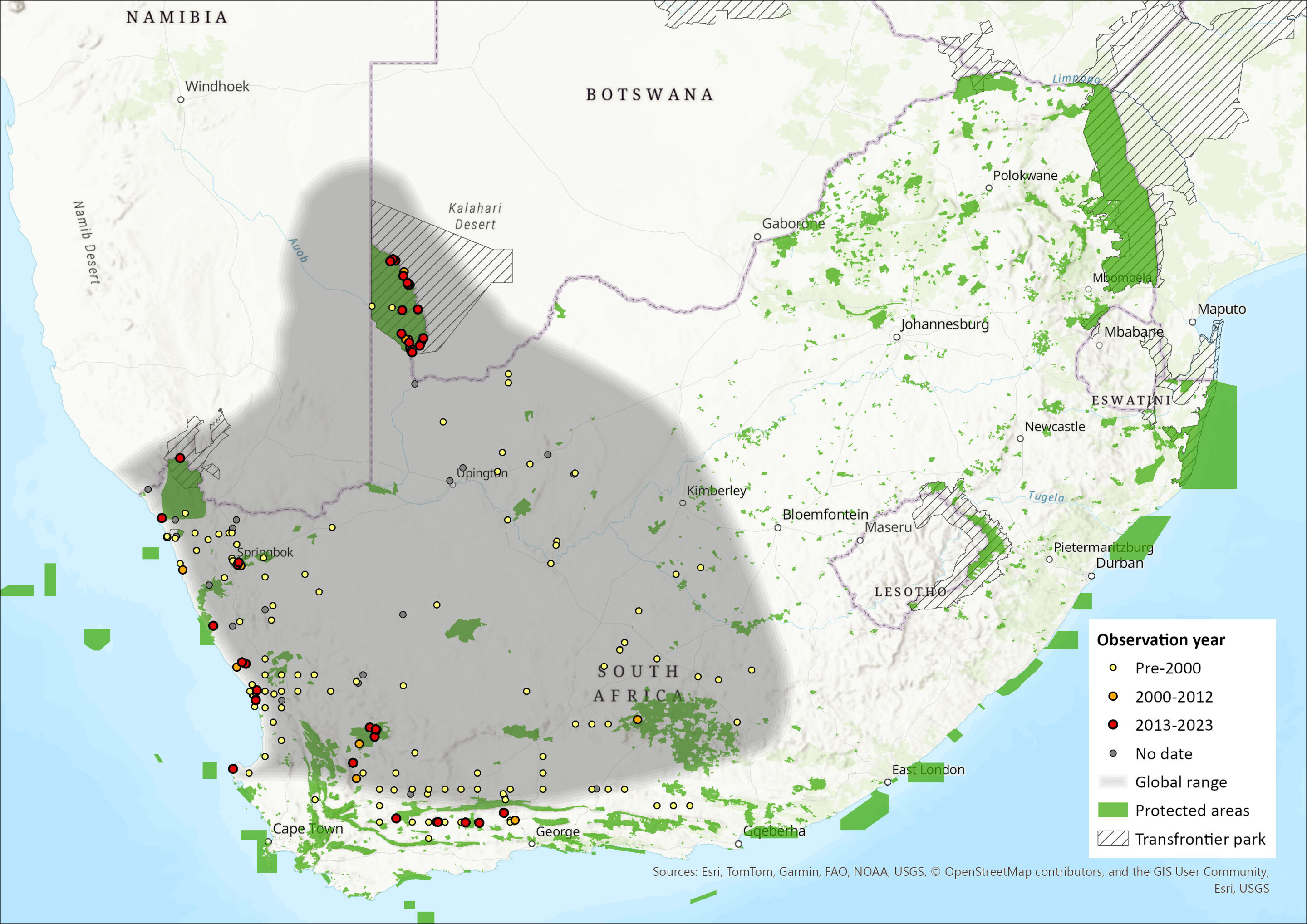
Map
Figure 1. Distribution records for Brants’ Whistling Rat (Parotomys brantsii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
|
Country |
Presence |
Origin |
Formerly Bred |
Seasonality |
|
Botswana |
Extant |
Native |
– |
– |
|
Namibia |
Extant |
Native |
– |
– |
|
South Africa |
Extant |
Native |
– |
– |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
An increase in the frequency and intensity of droughts due to climate change would increase extinct risk. As the species is mainly folivores, it might be especially vulnerable to month long droughts without green plant material, as it cannot change to a granivores diet. It is not known to hoard food.
Population
It can be very common locally, as it undergoes population irruptions in response to environmental conditions (Jackson 2013), and has a patchy distribution, linked to the distribution of deep sandy soils. This reflects its requirements for burrowing as the Brants’ Whistling Rat is an obligate burrower and uses burrows to avoid physiological extremes and predators (du Plessis & Kerley 1991). Thus, detecting a population trend is difficult, and long-term, systematic monitoring is necessary.
Population data from Goegap Nature Reserve, Northern Cape Province, indicate that this species is generally more resilient and stable than P. littledalei to short term drought (extreme drought in 2003), with densities of 2–10 individuals / ha remaining stable and consistent between 2001 and 2014 (C. Schradin unpubl. data). However, successive drought years lead to local extinction in Goegap in 2017. Even though the drought ended in 2020, the species remained locally extinct until 2023, when it re-occurred at very low population density (1 individual / ha). This compares to a density of 15 individuals / ha being recorded for P. littledalei at the same site in 2001, and subsequently, not having been recorded until 2014, at a density of one individual / ha (C. Schradin unpubl. data). Similarly, in the Little Karoo, (33°30’–33°37’S; 21°43’–21°52’E) over 14 years (1988–2001), their numbers peaked in 1989 and 1990 but thereafter fluctuated between moderate and low densities (G. Malan unpubl. data). In the central Little Karoo (study site 108 km2 in size), low densities were categorised by isolated pockets of active warrens, and moderate densities had animals evenly spread across the study area (G. Malan unpubl. data). At high densities, the whistling rats were so abundant that they even burrowed in road verges and open disturbed areas (and commonly ran across roads), and their piercing whistles reverberated through the veld (G. Malan unpubl. data). In high-density years, in Karroid Broken Veld, the density was estimated at 128 active warrens / km2 (range 76–212 warrens / km2) (Malan 2001). In the neighbouring and more open Succulent Karoo, the density was estimated at 192 active warrens / km2 (range 96–388) (Malan 2001). In a low-density year (1991) in Karroid Broken Veld, the estimated density decreased to 40 active warrens / km2 (standard deviation = 28 warrens) (recalculated from Malan 2004). Jackson (2013) reports densities exceeding 50 individuals / ha in favourable conditions. At one adult per warren, the biomass estimate was 16 kg / km2 (Malan 2001).
Population Information
Current population trend: Stable
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While no population genetic studies have been undertaken on this species, it has been investigated in a phylogenetic context (e.g., Montgelard et al. 2023). While it is a rapid disperser over short distances (see Assessment Rationale), its long-distance dispersal ability is unknown and would be dependent on corridors of suitable habitat. Consequently, without dispersal or genetic studies, it is not possible to determine whether the species exists as a metapopulation or as several genetically isolated subpopulations.
Moreover, given the extreme fluctuations in densities recorded over the years, it is difficult to approximate overall census size (Nc) for the species, which prevents an estimate of effective population size (Ne) from being quantified.
Habitats and ecology
Brants’ Whistling Rat is a largely crepuscular species (Jackson 1998), with additional short bouts of activity during the night and day (du Plessis 1989). It has a patchy habitat distribution (reflecting forage availability and the need for deep soils). It is restricted to areas with consolidated sands in semi-desert landscapes, with a low percentage plant cover (plant cover = 34–40%) (du Plessis & Kerley 1991; Malan 2001). These open areas probably provide it with a clear view of approaching aerial and terrestrial predators. In the Kalahari, it occurs in dune swales associated with Driedoring (Rhigozum trichotomum) (Jackson 2013). It is not associated with grass patches in Goegap Nature Reserve (C. Schradin unpubl. data). It can also occur in rangelands or old fields. It feeds on a variety of annuals in spring, and mostly perennial succulents, grasses and shrubs in summer and winter (Jackson 1998, 2001). Perennial succulents may function as key resource areas for this species, and it is thus potentially sensitive to destruction of these areas. It is a central place forager; smaller food items are eaten in situ whilst larger items are eaten at burrow entrances or stored underground (Jackson 2001).
It lives in colonies in extensive tunnel systems or warrens in sandy soils (De Graaff 1981; du Plessis et al. 1991), especially in the relatively stone-less soil of Mima-like mounds or heuweltjies (Lovegrove & Siegfried 1986; Cox et al. 1987). It relies on the cooler temperatures of burrows to escape high temperatures (du Plessis & Kerley 1991; Jackson et al. 2002). Heuweltjies are earth mounds, 25–32 m in diameter and 1.25–2.25 m in height, and are dispersed in a uniform fashion 41–51 m apart (measured centre to centre) (Cox et al. 1987; Lovegrove & Siegfried 1989). The highest heuweltjie density recorded in its Western Cape distribution range was in the eastern Little Karoo at 500 mounds / km (Lovegrove & Siegfried 1989). Densities of heuweltjies accounted for 56% of the variation in the density of P. brantsii warrens (Malan 2001). In the shallow soils (< 40 cm) of the central Little Karoo, it is an obligatory burrower that prefers deeper soils (77 cm) (du Plessis & Kerley 1991). In this area, 58% of heuweltjies surveyed contained active warrens (N = 524), whereas 97% of warrens not on heuweltjies were inactive (N = 145) (Malan 2001). The relationship between heuweltjie and P. brantsii densities can therefore be explained by the dependence of P. brantsii on the suitable heuweltjie soils.
It has a range of aerial and terrestrial predators and uses an “urgency-based” alarm call to alert conspecifics of predators depending on the immediate threat (Jackson 2001).
Ecosystem and cultural services: Desmet and Cowling (1999) suggested that the nutrient-rich soil patches around P. brantsii burrows may facilitate the re-vegetation of mine dumps. This should be interpreted with some caution as the rats require forage and would only recolonise areas where there is at least some plant cover. It is a key prey species for Pale Chanting Goshawks (Melierax canorus) and Booted Eagles (Aquila pennatus) in the Little Karoo, where the reproductive fitness of the latter is associated with the Karoo Broken Veld vegetation type that contains the tall shrubs and heuweltjies necessary for high Otomyine density (Malan 2001). Pale Chanting Goshawks catch P. brantsii individuals in proportion to the warren densities, as the birds may find it easier to catch P. brantsii on or near their warrens that are normally devoid of any vegetation (Malan 2001). At high rodent densities, Jackal Buzzard (Buteo rufofuscus), Black Harrier (Circus maurus) and Lanner Falcon (Falco biarmicus) move into the area to hunt P. brantsii (Malan 2000). Reptiles found in warrens include the Cape Cobra (Naja nivea) and Mole Snake (Pseudaspis cana) (G. Malan pers. obs. 2014).
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland |
– |
Suitable |
– |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major identified threats to this species. It is sometimes considered to be a pest in agricultural areas during population irruptions, but this is not expected to impact the population significantly. However, this is a species that should be flagged as being potentially threatened due to projected aridification from climate change (Boko et al. 2007), which may affect the forage resources on which this species depends through increased frequency and duration of drought conditions. This impact of global climate change may be exacerbated by the sensitivity of this rodent to high ambient temperatures (Du Plessis et al. 1989), which may limit foraging behaviour under hotter conditions. For example, it became locally extinct in Goegap Nature Reserve after a severe drought in 2003. It returned in low numbers seven years later, and the population has since fully recovered (C. Schradin unpubl. data). Thus, this species might be vulnerable to an increase in intensity of droughts. However, it is suspected to be less threatened by droughts than the closely related Littledale’s Whistling Rat.
Similarly, overgrazing in some areas reduces habitat quality for this species. Whereas grazing should be encouraged to decrease the bush encroachment (see Habitats and Ecology), overgrazing should be avoided, especially in the more open vegetation types, to limit direct competition between farm animals and P. brantsii (Malan & Crowe 1996). As such, the proliferation of wildlife ranching should be monitored for its potential negative impacts as overgrazing may impact key vegetation types that the species requires. More research is required to understand the net effects of local overgrazing on this species. For example, a study found that, contrary to prediction, grass cover has increased, and dwarf shrub cover has decreased in the Nama and Succulent Karoo, which is attributed to a general decrease in stocking rate in the area (Masubelele et al. 2014).
Habitat trend: Stable. Although the extent of habitat is not expected to decline, there may have been a decrease in habitat quality due to overgrazing – especially of the key resource areas listed above. Human settlements are not expected to encroach on this species’ habitat. Protected area expansion in the area should sustain strong subpopulations across the range. For example, the Goegap Nature Reserve increased in size from 150 to c. 250 km2 in 2014 by including several neighbouring farms. The effects of climate change on this species should be monitored.
Conservation
It occurs commonly in protected areas across the range, for example, Kgalagadi Transfrontier Park, Goegap Nature Reserve and Tswalu Kalahari Reserve (Pienaar et al. 2010). No specific interventions are necessary at present. However, the species would benefit from continued protected area expansion to enable it to track shifting habitats caused by climate change.
Recommendations for land managers and practitioners:
- Land managers should decrease stocking rates to conserve key resource areas.
- Long-term, systematic monitoring is needed to establish subpopulation trends and threat levels.
- During population irruptions, land managers can install perches near the warrens for perch-hunting raptors to sit on (Malan & Marais 2002).
Research priorities:
- Effects of overgrazing and climate change on key resource area quality and subpopulation size.
- Effect of extended drought periods on population dynamics over a larger geographic scale.
- The ecological factors that drive the population irruptions and crashes seen in this species.
- The genetic implications of population fluctuations, and the subsequent re-colonisations and abandonment of areas within its distribution range.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Boko M, Niang I, Nyong A, Vogel C, Githeko A, Medany M, Osman-Elasha B, Tabo R, Yanda P. 2007. Africa. Climate Change 2007: Impacts, Adaptation and Vulnerability. In: M. L. Parry, O. F. Canziani, J. P. Palutikof, P. J. van der Linden, and C. E. Hanson (eds), Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, pp. 433–467. Cambridge University Press, Cambridge, UK.
Cox GW, Lovegrove BG, Siegfried WR. 1987. The small stone content of mima-like mounds in the South African Cape region: implications for mound origin. Catena 14: 165–176.
De Graaff, G. 1981. The Rodents of Southern Africa. Butterworths, Durban, South Africa.
Desmet PG, Cowling RM. 1999. Patch creation by fossorial rodents: a key process in the revegetation of phytotoxic arid soils. Journal of Arid Environments 43: 35–45.
du Plessis A, Erasmus T, Kerley GIH. 1989. Thermoregulatory patterns of two sympatric rodents: Otomys unisulcatus and Parotomys brantsii. Comparative Biochemistry and Physiology, Series A.
du Plessis A, Kerley GIH, Winter PED. 1991. Dietary patterns of two herbivorous rodents: Otomys unisulcatus and Parotomys brantsii in the Karoo. South African Journal of Zoology 26: 51–54.
du Plessis A, Kerley GIH. 1991. Refuge strategies and habitat segregation in two sympatric rodents Otomys unisulcatus and Parotomys brantsii. Journal of Zoology 224: 1–10.
du Plessis A. 1989. Ecophysiology of the bush Karoo rat Otomys unisulcatus and the whistling rat Parotomys brantsii. M.Sc. Thesis. University of Port Elizabeth.
Jackson TP, Roper TJ, Conradt L, Jackson MJ, Bennett NC. 2002. Alternative refuge strategies and their relation to thermophysiology in two sympatric rodents, Parotomys brantsii and Otomys unisulcatus. Journal of Arid Environments 51: 21–34.
Jackson TP. 1998. The diurnal activity of Brants’ Whistling Rat (Parotomys brantsii): the effect of seasonal and physical conditions. Transactions of the Royal Society of South Africa 53: 227–236.
Jackson TP. 2001. Factors influencing the food collection behaviour of a central place forager: Brants’ whistling rat, Parotomys brantsii. Journal of African Zoology 255: 15–23.
Jackson TP. 2013. Parotomys brantsii Brants’s Whistling Rat. In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, pp. 597–599. Bloomsbury Publishing, London, UK.
Le Roux A, Jackson TP, Cherry MI. 2002. Differences in alarm vocalizations of sympatric populations of the whistling rats, Parotomys brantsiiand P. littledalei. Journal of Zoology 257: 189–194.
Lovegrove BG, Siegfried WR. 1986. Distribution and formation of Mima-like earth mounds in the western Cape Province of South Africa. South African Journal of Science 82: 432–436.
Malan G, Crowe TM. 1996. The diet and conservation of monogamous and polyandrous pale chanting goshawks in the Little Karoo, South Africa. South African Journal of Wildlife Research 26: 1–10.
Malan G, Marais AvN. 2002. Guidelines for the design and management of artificial raptor perches and nest-tree stands on forestry estates in South Africa: management paper. Southern African Forestry Journal 2002: 49–54.
Malan G. 2000. Raptors (Falconiformes) of the Calitzdorp district, Little Karoo, South Africa (1988–1998). Durban Museum Novitates 25: 32–35.
Malan G. 2001. Otomyine rodent refuge habitat: does it represent habitat quality for pale chanting-goshawk families? South African Journal of Wildlife Research 31: 49–58.
Malan G. 2004. The influence of prey abundance and co-breeders on reproductive output of cooperative breeding Pale Chanting-goshawks. Ostrich 75: 44–51.
Masubelele ML, Hoffman MT, Bond WJ, Gambiza J. 2014. A 50 year study shows grass cover has increased in shrublands of semi-arid South Africa. Journal of Arid Environments 104: 43-51.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
Montgelard, C., Muller, T., Arnal, V., Maree, S., Taylor, P.J., Sands, A.F., Robinson, T.J. and Matthee, C.A., 2023. Diversification and evolutionary history of the African laminated-toothed rats (Rodentia, Otomyini). Molecular Phylogenetics and Evolution, 183, p.107779.
Pienaar C, Koen J, Badenhorst D. 2010. Evaluation of Tswalu Kalahari Reserve as a Protected Area. Department of Environment and Nature Conservation, Northern Cape, South Africa.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor PJ, Maree S, Cotterill FPD, Missoup AD, Nicolas V, Denys C. 2014. Peripatric speciation across a Neogene volcanic archipelago: molecular and morphological evidence for a Pleistocene radiation of laminate-toothed rats (Otomys: Rodentia) across equatorial Africa. Biological Journal of the Linnean Society 113: 320–344.
Taylor, P.J., Lavrenchenko, L.A., Carleton, M.D., Verheyen, E., Bennett, N.C., Oosthuizen, C.J. and Maree, S. 2011. Specific limits and emerging diversity patterns in East African populations of laminate-toothed rates, genus Otomys (Muridae: Murinae: otomyini): revision of the Otomys typus complex. Zootaxa 3024: 1-66.