Brant’s Climbing Mouse
Dendromus mesomelas

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Dendromus mesomelas s.l. – (Brants, 1827)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – NESOMYIDAE – Dendromus – mesomelas
Common Names: Brant’s Climbing Mouse, Chestnut Tree Mouse (English), Brants se Klimmuis, Rooiboommuis (Afrikaans)
Synonyms: Mus mesomelas (Brants, 1827); Dendromus typus A. Smith, 1829; Dendromys (?) typus Wagler, 1830; Dendromys mesomelas Smuts, 1832; Dendromys typicus A. Smith, 1834; Dendromys pumilio J. A. Wagner, 1841; Dendromys mesomelas pumilio Fitzinger, 1867; Dendromus mesomelas Thomas & Schwann, 1906; Dendromus pumilio Wroughton, 1907; Dendromus ayresi A. Roberts, 1913; Dendromus mesomelas major St. Leger, 1930; Dendromus mesomelas pumilio Shortridge, 1942
Taxonomic Note:
Represents a species complex as a recent phylogenetic study by Voelker et al. (2021), identified eight distinct evolutionary lineages with distributions ranging from South Africa to Tanzania and Uganda. The species type locality is reported as “east of Port Elizabeth, Sunday’s River, Eastern Cape Province, South Africa,” however at least three separate genetic lineages have been described from South African samples alone (Voelker et al. 2021). As such, the taxon is referenced as D. mesomelas s.l., pending further taxonomic investigations. Although previously included within this species, D. insignis, D. oreas and D. vernayi are now considered valid species (Monadjem 2013; Monadjem et al. 2015). Chromosomal analysis by Solano et al. (2014) reveals further potential species splits; for example, populations in Mozambique, Zambia and Botswana that are isolated from those in South Africa may in fact reveal separate species. A comprehensive integrative taxonomic approach is urgently needed to resolve the present uncertainties regarding the evolutionary origins of southern African taxa within the genus Dendromus.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Richards, L.R.1, Naidoo T.1 & da Silva, J.M.2
Reviewers: Patel, T 3.
Institutions:1Durban Natural Science Museum,2South African National Biodiversity Institute,3Endangered Wildlife Trust
Previous Assessors and Reviewers: MacFadyen, D., Shoeman, C. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern, as this species is widespread along the eastern grassland regions of the assessment regions, occurs in many protected areas, and is fairly common. However, although there is no evidence for large-scale population decline at present, continued loss of grassland habitat would certainly impact it in the future. Although its fynbos habitat is well-protected in the south of its range, grassland habitats in the north are severely threatened and fragmented. This species needs high-quality grassland to survive. Loss of area of occupancy through grassland loss should be systematically monitored and this species should be reassessed when finer-scale data emerge. Taxonomic resolution, by assessing which populations / genetic clades qualify for being upgraded to species level, is required and may necessitate reassessment in future.
Regional population effects: Populations are isolated within countries, and rescue effect is unlikely because there is a disjunct distribution between the assessment region and rest of the species range.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Richards LR, Naidoo T & da Silva JM. 2025. A conservation assessment of Dendromus mesomelas s.l.. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
While previously considered to occur widely across African highlands, recent molecular evidence suggests D. mesomelas is restricted to south of the Zambezi River while north occur several taxa now listed as valid species including D. insignis, D. nyasae, D. oreas and D. vernayi (Monadjem 2013, Monadjem et al. 2015). Within the assessment region, it occurs widely in the eastern and southwestern grassland regions of the country, including Eswatini and Lesotho along an altitudinal gradient, from sea level, in the southern portions of its range, to montane areas in the north (Skinner and Chimimba 2005, Monadjem 2013, Monadjem et al. 2015). The distribution is reported to marginally intrude into the North West Province (Skinner and Chimimba 2005), but was not detected during a 2014 survey (Power 2014). Further vetting of historical museum records is required to confirm this extension of their range.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m asl
Elevation Upper Limit (in metres above sea level): recorded at 2,400 m asl (Rowe-Rowe and Meester 1982)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
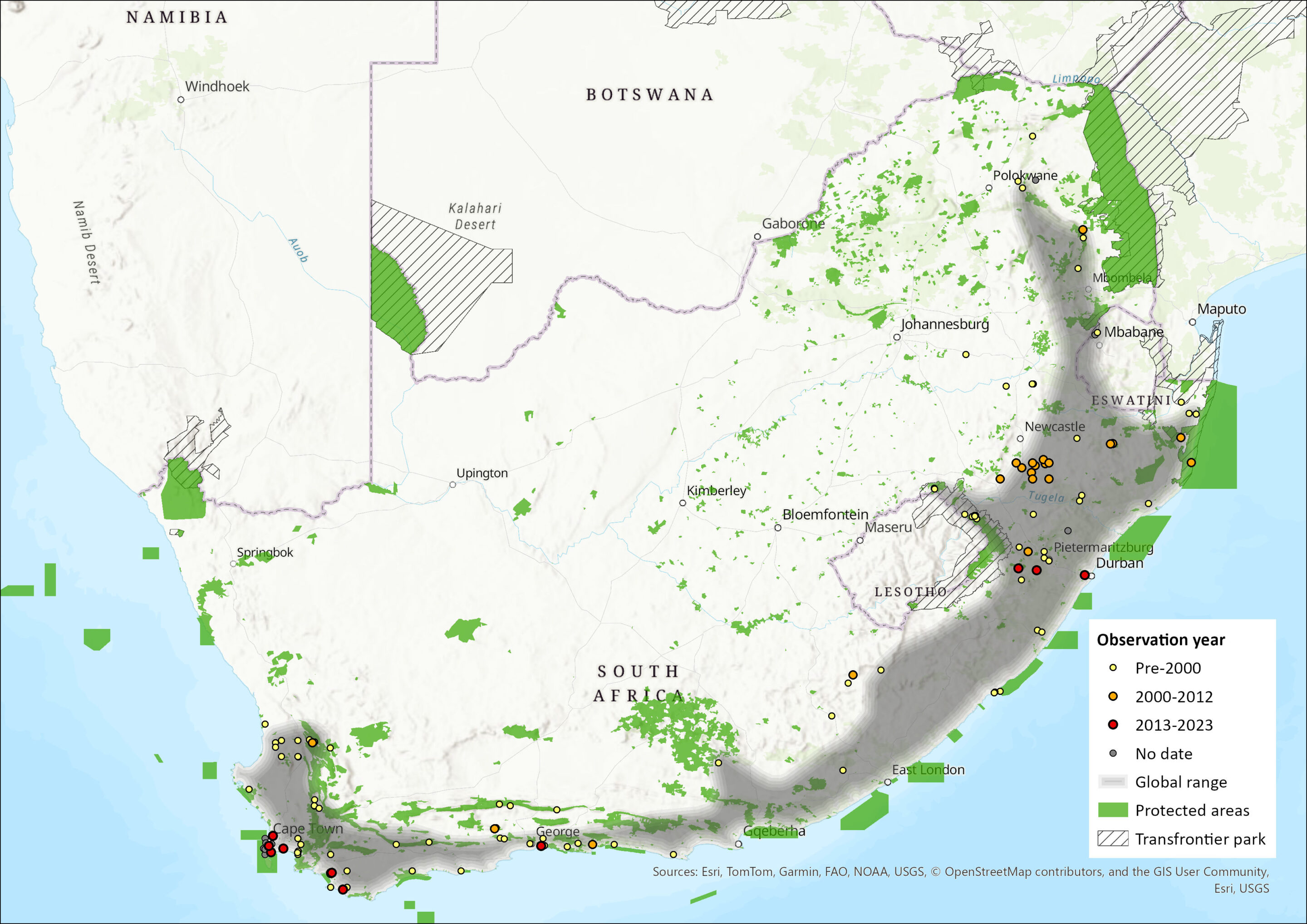
Map
Figure 1. Distribution records for Brant’s Climbing Mouse (Dendromus mesomelas) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Presence Uncertain | Native | – | – |
| Botswana | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
To date, no formal studies have examined the potential impacts of climate change, along with associated bioclimatic and environmental shifts, on the biology, ecology, or physiology of this species. The species is more commonly associated with grass-dominated landscapes and appears to be stricter in its habitat preference than other Dendromus species, exhibiting a strong affinity for moist habitats (Monadjem 2013; Monadjem et al. 2015). Midgeley and Thuiller (2010) predicted a 30% replacement of grassland-dominated habitats by more arid-adapted or tolerant vegetation types (e.g. thorn-scrub savanna). This may result in the displacement or range contraction of grassland-associated species, that may include Dendromus mesomelas s.l. within the assessment region. Rodent population declines tend to be more pronounced in habitats experiencing significant anthropogenic transformation or, alternatively, in high-altitude environments where the effects of climate change are likely to be intensified (Wan et al. 2022). Further research is required to assess the potential impacts of climate change on this species within the assessment region.
Population Information
Within temperate grasslands and moist montane habitats of the assessment region, this species occurs commonly. For example, they are relatively numerous in the leaf-litter of afromontane forest in Eswatini (Monadjem 2013). No density estimates are available. Subpopulations are separated by regions of unsuitable habitat and fluctuations occur seasonally.
Population Information
Current population trend: Stable
Continuing decline in mature individuals? Unknown, but unlikely.
Extreme fluctuations in the number of subpopulations: Cannot be determined as there is a general lack of information on population ecology.
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Unknown, pending phylogenetic and phylogeographic studies
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown, pending phylogenetic and phylogeographic studies
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unknown, but unlikely.
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Recent phylogenetic investigation of the genus Dendromus revealed cryptic diversity within D. mesomelas, with eight lineages currently included under this taxon (Voelker et al. 2021). The specific geographic distributional limits and ecological niches of these lineages remain unresolved and requires further investigation. Within the assessment region, several subpopulations may exist; however; it is unclear as to whether these form a single metapopulation or separate genetically distinct groups potentially due to habitat fragmentation and limited dispersal capabilities. A comprehensive population genetic study is recommended to elucidate the genetic structure and diversity within this species.
Habitats and ecology
Similar to other Dendromus species, Brants’ Climbing Mouse is associated with grassland or marsh vegetation, especially in tall grasses with shrub (Taylor 1998, Monadjem 2013). In the southern portions of its range, they have been recorded within forest – grassland mosaic and fynbos habitats. It has also been sampled in Palmveld (Hyphaene coriacea) in KwaZulu-Natal (Rautenbach et al. 2014), swamps and Afromontane forests (Rowe-Rowe and Meester 1982, Monadjem 1998). However, they appear to be more restricted to high quality grassland habitats further north. During recent surveys in Gauteng and Mpumalanga, this species was not recorded in the Rocky Highveld Grassland of Ezemvelo Nature Reserve (Gauteng) and Telperion (Mpumalanga) (MacFadyen 2014). This species prefers tall, rank grassland with a high plant biomass, but is also recorded in areas with low woody vegetation. The species has a reported preference for moist habitats, being notably absent from arid and semi-arid environments (Monadjem 2013). It appears more specific in its habitat requirement when compared with D. melanotis and D. mystacalis. As such, it does not occur in modified landscapes and thus loss of natural grasslands impacts severely on the species.
This omnivorous species feeds on seeds, as well as a wide range of invertebrates, which contribute to their diets at varying proportions depending on their local conditions. For example, in the Okavango Delta, where they have no access to seeds, they were found to feed exclusively on insects (Smithers 1971), however, in other regions, invertebrate consumption is likely correlated with seasonal changes in abundance (Monadjem 2013). In the Drakensberg and Eswatini, their diets comprised mostly of grass seeds (Rowe-Rowe 1986, Monadjem 1997). Like other species of climbing mice, this nocturnal and largely terrestrial species accesses seeds and invertebrates in low bushes and grass stalks, using its prehensile tail to steady itself. The species is usually solitary, in pairs or small family groups.
Similar to other southern African Dendromus species, these rodents construct round grass nests with entrances at both ends (Roberts, 1954). Nests may be positioned either above or below ground (Kingdon 1974). They are also known to utilize abandoned weaver and warbler nests as sites for raising their young or for resting (Roberts, 1954; De Graaff, 1981). The species is largely nocturnal but is known to be active during and may share the runways of Otomys in high altitudinal grasslands (Kingdon 1974).
Ecosystem and cultural services: There are no cultural services or folklores associated with this species. However, similar to other small mammals, this species plays a role in regulating invertebrate numbers, seed predation, nutrient cycling, and is an important prey species for predators.
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
1.9. Forest -> Forest – Subtropical/Tropical Moist Montane |
– |
Suitable |
– |
|
2.1. Savanna -> Savanna – Dry |
– |
Suitable |
– |
|
3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation |
– |
Suitable |
– |
|
4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude |
– |
Suitable |
– |
Life History
Generation Length: Unknown
Age at maturity: female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean head and body length= 7.40 cm (De Graaf 1981)
Size at Maturity (in cms): Male: Mean head and body length= 7.60 cm (De Graaf 1981)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Female maximum head and body length = 8.5 cm; male maximum head and body length = 8.0 cm (De Graaf 1981)
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Animals in breeding condition have been recorded in November (Rowe-Rowe and Meester 1982). Likewise, pregnant females have been collected during the same month (Taylor 1998).
Average Annual Fecundity or Litter Size: 2-6 pups per litter (Hanny 1965, Taylor 1998).
Natural Mortality: Dendromus spp. skeletal remains recovered from Barn Owl (Tyto alba; Vernon 1972; Durban Natural Science Museum specimen records), Grass Owl (Tyto capensis; Davis 1973), and Black-winged Kite (Elanus caeruleus; Mendelsohn 1982) regurgitated pellets. Also eaten by a variety of meso-carnivores, including Black-backed jackal (Lupelella mesomelas; Rowe-Rowe 1983).
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: N/A
Does the species have a free-living larval stage? N/A
Does the species require water for breeding? N/A
Movement Patterns
Movement Patterns: Movement and dispersal patterns remain poorly understood / unreported.
Congregatory: The species typically occurs alone, in pairs, or in small family groups. These family groups may continue to use the grass nest even after the young have been weaned (Skinner & Chimimba, 2005).
Systems
System: Terrestrial
General Use and Trade Information
There is no known subsistence or commercial use of this species.
Local Livelihood: None reported or known at present.
National Commercial Value: None reported or known at present.
International Commercial Value: None reported or known at present.
End Use: None reported or known at present.
Is there harvest from captive/cultivated sources of this species? None reported or known at present.
Harvest Trend Comments: None reported or known at present.
Threats
Although not believed to be currently declining, threats to this species are grassland habitat loss from agricultural expansion, forestry and mining developments. Loss of habitat quality from overgrazing and incorrect fire management may also cause local declines. Climate change is also expected to reduce afromontane grassland (Taylor et al. 2024), further jeopardising the species similar to other afromontane species (Taylor et al. 2016). The indiscriminate use of chemicals in agricultural industry and urbanisation is also expected to have an impact on this species. The impact of these threats on the population remains to be quantified.
Conservation
This species occurs in several protected areas within its range in the assessment region including the Cedarberg Wilderness Area, Tsitsikamma National Park, uKhahlamba Drakensberg Park, and iSimangaliso Wetland Park. Protected area expansion to incorporate grassland habitats is a key intervention for the species, specifically to create habitat linkages where connectivity between natural habitat patches has been lost. Planners should prioritise previously cultivated areas for development instead of remaining natural areas. For example, previously ploughed areas now left fallow make up 8.9% of the grassland biome in Mpumalanga, and these areas should be prioritised for further development (Lötter et al. 2014). Grassland restoration projects are also likely to benefit this species. Landowners are responsible for managing the land and vegetation, ensuring that under- or overutilisation of grasses is avoided. This species would also benefit from correct fire and land management practices that conserve grassland habitats.
Recommendations for land managers and practitioners:
- Landowners should promote and maintain corridors of grassland between cultivated areas.
- Employ correct land management practices to prevent overgrazing and indiscriminate burning.
- Protected areas expansion through provincial biodiversity stewardship programmes.
- Reduce over-stocking on cattle and game ranches.
Research priorities:
- This is a species complex that requires taxonomic revision. The species might be split along biome boundaries.
- Conduct phylogeographic and landscape genetic analyses to elucidate the genetic structure of animal populations within the assessment region.
- Land cover maps and climate-based modelling must be used to project future decline in the Grassland Biome and potential impact on the distributional range of the taxon within the assessment.
- Quantifying habitat preferences and understanding local movements and dispersal mechanisms between habitats; quantifying the species’ habitat under formal protection.
- Research into the reproductive biology and physiology of the species and the potential impacts of climate change associated extreme fluctuations in temperature and precipitation seasonality on the species.
- Research into the contribution of this species complex to ecosystem functioning.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP), especially outside protected areas.
- Deceased specimens should be submitted to the nearest accredited museum or biological repository for proper documentation and preservation.
- Plant indigenous species in gardens.
- Create corridors to offset the impacts of urbanisation and ensure movement and gene flow between populations.
- Reduce use of insecticides and herbicides.
- Proclaim private land nature reserves/protected areas under government stewardship program.
Bibliography
Davis, R.M. 1973. The ecology and life history of the vlei rat, Otomys irroratus (Brants, 1827), on the Van Riebeeck Nature Reserve, Pretoria. Doctoral Thesis, University of Pretoria (South Africa).
De Graaff G. 1981. The Rodents of Southern Africa: Notes on Their Identification, Distribution, Ecology and Taxonomy. Butterworths, Pretoria.
Jewitt D, Goodman PS, Erasmus BFN, O’Connor TG, Witkowski ETF. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kingdon J. 1974. East Arican Mammals. Volume II, Part B (Hares and Rodents). Academic Press, London.
Lötter MC, Cadman MJ, Lechmere-Oertel RG. 2014. Mpumalanga Biodiversity Sector Plan Handbook. Mpumalanga Tourism & Parks Agency, Mbombela, South Africa.
MacFadyen DN. 2014. The dynamics of small mammal populations in Rocky Highveld Grassland, Telperion, South Africa. Ph.D. Thesis. University of Pretoria, Pretoria, South Africa.
Mammal Diversity Database. 2025. Dendromus mesomelas. https://mammaldiversity.org/taxon/1003718. Accessed 14 November 2025.
Mendelsohn, J. 1982. The feeding ecology of the blackshouldered kite Elanus caeruleus (Aves: Accipitridae). Durban Museum Novitates, 13(8), pp.75-116.
Midgley, G.F. and Thuiller, W. 2011. Potential responses of terrestrial biodiversity in Southern Africa to anthropogenic climate change. Regional Environmental Change, 11(Suppl 1), pp.127-135.
Monadjem A. 1997. Stomach contents of 19 species of small mammals from Swaziland. South African Journal of Zoology 32: 23–26.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Monadjem A. 2013. Dendromus mesomelas Brants’s African Climbing Mouse. Pages 176–177 in Happold DCD, editor. Mammals of Africa. Volume III: Rodents, Hares and Rabbits. Bloomsbury Publishing, London, UK.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Power RJ. 2014. The Distribution and Status of Mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Rautenbach A, Dickerson T, Schoeman MC. 2014. Diversity of rodent and shrew assemblages in different vegetation types of the savannah biome in South Africa: no evidence for nested subsets or competition. African Journal of Ecology 52: 30-40.
Roberts A. 1954. The Mammals of South Africa. Second Edition. Trustees of the Mammal of South Africa Book Fund, South Africa
Rowe-Rowe DT, Meester J. 1982. Habitat preferences and abundance relations of small mammals in the Natal Drakensberg. South African Journal of Zoology 17: 202-209.
Rowe-Rowe DT. 1983. Black-backed jackal diet in relation to food availability in the Natal Drakensberg. South African Journal of Wildlife Research, 13(1):17-23.
Rowe-Rowe DT. 1986. Stomach contents of small mammals from the Drakensberg, South Africa. South African Journal of Wildlife Research 16: 32–35.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Solano E, Taylor PJ, Rautenbach A, Ropiquet A, Castiglia R. 2014. Cryptic speciation and chromosomal repatterning in the South African climbing mice Dendromus (Rodentia, Nesomyidae). PloS one 9: e88799.
Taylor PJ, Nengovhela A, Linden J, Baxter RM. 2016. Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia in press.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., Kearney, T.C., Clark, V.R., Howard, A., Mdluli, M.V., Markotter, W., Geldenhuys, M., Richards, L.R., Rakotoarivelo, A.R., Watson, J. & Balona, J. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate‐driven diversification and a buffer against future climate change in bats. Global Change Biology, 30(6):e17344.
Vernon, C.J. 1972. An analysis of owl pellets collected in southern Africa. Ostrich, 43(2), pp.109-124.
Voelker, G., Huntley, J.B., Denys, C., Sumbera, R., Demos, T.C., Lavrenchenko, L., Nicolas, V., Gnoske, T.P., Kerbis Peterhans, J.C. 2021. Molecualr systematics and biogeographic history of the African climbing-mouse complex (Dendromus). Molecular Phylogenetics and Evolution 161: 107166.
Wan, X., Yan, C., Wang, Z. and Zhang, Z. 2022. Sustained population decline of rodents is linked to accelerated climate warming and human disturbance. BMC Ecology and Evolution, 22(1), p.102.