
Laephotis cf. botswanae
Laephotis cf. botswanae

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Laephotis cf. botswanae – Setzer, 1971
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Laephotis – botswanae
Common Names: None available pending formal description
Synonyms: No Synonyms
Taxonomic Note:
Based on mtDNA sequence and craniometric data, the name L. botswanae sensu stricto (Setzer, 1971; described from Botswana) is synonymous with the savanna-associated species, L. angolensis (Monard, 1935; described from Angola), while specimens of L. botswanae senso lato (i.e., previously assigned to L. botswanae) from localities in South Africa comprise a uniquely distinct clade, here termed L. cf. botswanae, pending formal naming of this species (Kearney & Seamark 2005; Taylor et al. 2022; 2024).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.J.1, Howard, A.1, Balona, J.2 & da Silva, J.M.3
Reviewers: Moir, M.4 & Richards, L.R.5
Institutions: 1University of the Free State, 2Gauteng and Northern Regions Bat Interest Group, 3South African National Biodiversity Institute, 4Stellenbosch University, 5Durban Natural Science Museum
Previous Assessors and Reviewers: Taylor, P., Schoeman, C., Jacobs, D., MacEwan, K., Richards, L, Cohen, L., Sethusa, T. & Monadjem, A.
Previous Contributors: Roxburgh, L., Raimondo, D. & Child, M.F.
Assessment Rationale
Endemic to the assessment region. Although only known from three localities in Limpopo Province from the previous assessment (2004), subsequent field surveys have located it at two more and at another site in Gauteng Province, as well as in southern KwaZulu-Natal (KZN) and the Eastern Cape province (Moir et al. 2020a), revealing a wider distribution in the assessment region extending to, with an extent of occurrence of 379,451 km2. It is patchily distributed and is losing habitat in parts of its range due to forest degradation. Due to its wide distribution and in the absence of population data, we list this species as Least Concern given its wide distribution. However, further data on subpopulation sizes and trends are needed and this species should be reassessed when such data are available.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Taylor PJ, Howard A, Balona J & da Silva JM. 2025. A conservation assessment of Laephotis cf. botswanae. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is endemic to South Africa (Taylor et al. 2024). The species has been recorded from Limpopo, KwaZulu-Natal (KZN), Gauteng and Eastern Cape provinces (Moir et al. 2020a), although it was only known from Limpopo in the 2004 assessment (Friedmann & Daly 2004). Although the species has not been recorded in Eswatini or Lesotho, predictive distribution models indicate the presence of suitable habitat, particularly in Eswatini (Monadjem et al. 2010, 2021).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 193 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1614 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
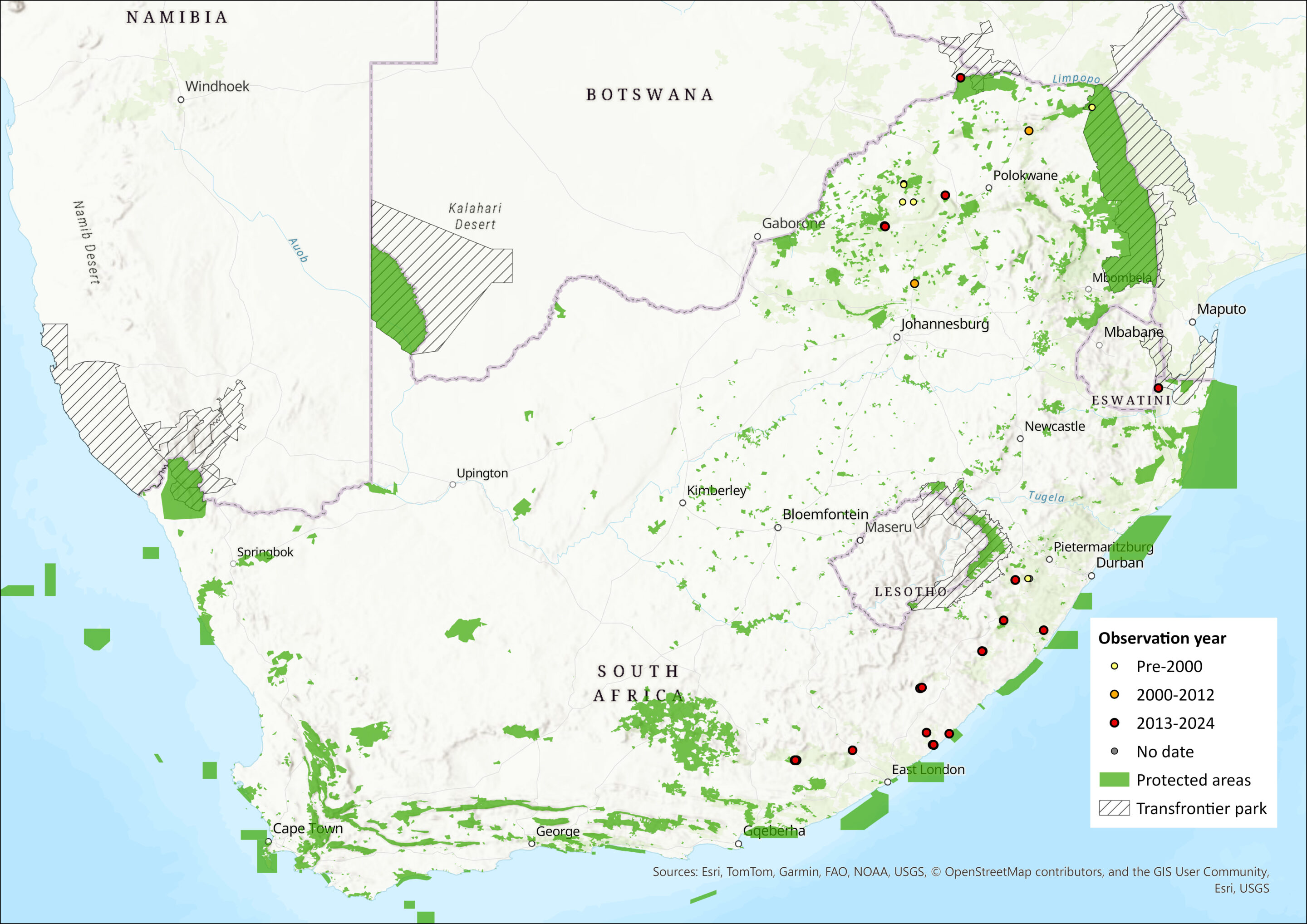 Figure 1. Distribution records for Laephotis cf. botswanae within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Laephotis cf. botswanae within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: (Not specified)
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
A study by Taylor et al. (2024) reported minimal range changes for this species under future climate models by suggesting the high-elevation mountains provide potential refugia for temperate montane species. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability, with increasing global aridity, which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020).
Population Information
The abundance of this species is not well known. Acoustic activity surveys conducted in forests in Eastern Cape and KwaZulu-Natal provinces showed a rate of approximately 0.02 passes per hour in both Eastern and Transkei Mistbelt forest types. While activity of this species was highest in Amatole Mistbelt forests (0.2 passes per hour), lowest in Pondoland Scarp forests and absent from Eastern Cape Dune and Albany Coastal forests (Moir et al. 2021). It is naturally rare with a few specimens available in museums. Further monitoring is needed to estimate population size and trend.
Current population trend: Unknown
Continuing decline in mature individuals? Cannot be determined in the absence of population estimate data.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: Presumed to belong to one population.
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Moir et al. (2020b) examined the genetic structure within this species using mitochondrial markers and found comparatively low genetic diversity and moderately high population genetic structure with differentiation between the northern (Limpopo) and eastern (Eastern Cape, KwaZulu-Natal) samples. This would suggest that at least two subpopulations exist; however, given that this species has low dispersal capabilities and prefers to fly under cover (Kearney 2013), and hence open habitat may pose a barrier to dispersal, more localised population structure might exist and could be uncovered with the use of fine-scale nuclear markers. Population genetics investigations show this species to depict a historical persistence in mistbelt and scarp forests of the Eastern Cape Province (Moir et al. 2020b). Estimates of contemporary Ne are not available for either subpopulation.
Habitats and ecology
Animals have been recorded from forests (Taylor et al. 2013; Moir et al. 2020) and grasslands (Howard et al. 2022). It is often found in the vicinity of rivers (Monadjem et al. 2010; Kearney 2013). Its association with rocky outcrops has also been noted in some localities (Herholdt 1989; Cotterill 1996). This species has been recorded from forests of the Southern Mistbelt Group, Pondoland Scarp, Transkei Coastal Scarp and Southern Coastal Forests. All specimens have been mist-netted or captured in harp traps, so there is no information on roosting sites (Monadjem et al. 2020).
Ecosystem and cultural services: Insectivorous bats are important regulators of insect populations (Boyles et al. 2011; Kunz et al. 2011). Bats feed on arthropods that damage crops, and thus agricultural areas with bats require less pesticides which may also be detrimental to human health (Frank 2024; Kunz et al. 2011).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1. Forest and Woodland – 1.6. Subtropical/Tropical Moist Lowland Forest | Suitable | ||
|
1. Forest and Woodland – 1.9. Subtropical/Tropical Moist Montane Forest |
Suitable | ||
| 4. Native Grassland – 4.7. Subtropical/Tropical High-Altitude Grassland | Suitable |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female mean forearm length = 3.66 ± 0.14 cm (Monadjem et al. 2020, may include data from L. angolensis individuals); Male mean forearm length = 3.55 ± 0.12 cm (Monadjem et al. 2020, may include data from L. angolensis individuals).
Size at Maturity (in cms): Female maximum forearm length = 3.82 cm; Male maximum forearm length = 3.70 cm) (Monadjem et al. 2020; may include data from L. angolensis individuals)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: Lactating females were captured from Nxumeni State Forest in December (M. Moir unpublished data), suggesting that breeding coincides with the austral summer and parturition takes place in December. Similar findings have been reported from Malawi (Kearney 2013).
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: No information available
Congregatory: No information available
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised.
Local Livelihood: None reported
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
As in conspecifics, it may depend on old indigenous trees for roosting. There is ongoing degradation of its forest habitat in the Eastern Cape, due to fires and subsistence harvesting. This is particularly severe for smaller forest fragments.
It has lost habitat in the past in the Midlands region of KwaZulu-Natal to plantation forestry.
Conservation
Recorded from Kruger National Park, Lapalala Wilderness Area (a private nature reserve in the Waterberg Mountains of Limpopo) and Dinokeng Game Reserve. In the Eastern Cape Province, this species has been recorded from Fort Fordyce Nature Reserve, Mkambati Nature Reserve, Silaka Nature Reserve, Dwesa Nature Reserve, Cape Morgan Nature Reserve, Woody Cape Nature Reserve, and The Island Nature Reserve. Additionally, this species has been recorded from Oribi Gorge Nature Reserve and Umtamvuna Nature Reserve in KwaZulu-Natal. It may occur in more protected areas, which should be documented. No direct interventions are necessary at present. Rather, studies further detailing the distribution, ecology and taxonomic resolution are needed to better inform conservation plans.
Recommendations for land managers and practitioners:
- Protection of riparian vegetation and natural habitat within the species known distribution range
- Reduce the use of pesticides and herbicides in agricultural landscapes or employ biocontrol.
Research priorities:
- Further field surveys are required to more accurately delimit the distribution of this species.
- Ecological studies are needed to determine the dietary and spatial ecology.
- Molecular studies are required to resolve the taxonomy between L. angolensis, and L. botswanae.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Ansell WFH, Dowsett RJ. 1988. Mammals of Malawi – an Annotated Checklist and Atlas. The Trendrine Press, Zennor, St Ives, Cornwall, UK.
Boyles JG, Cryan PM, McCracken GF, Kunz TH. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Cotterill FPD. 1996.. New distribution records of insectivorous bats of the families Nycteridae, Rhinolophidae and Vespertilionidae (Microchiroptera: Mammalia) in Zimbabwe. Arnoldia Zimbabwe 10: 71–89.
Driver A, Sink KJ, Nel JN, Holness S, van Niekerk L, Daniels F, Jonas Z, Majiedt PA, Harris L, Maze K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Frank, E.G. 2024. The economic impacts of ecosystem disruptions: Costs from substituting biological pest control. Science 385,0344.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. CBSG Southern Africa, IUCN SSC Conservation Breeding Specialist Group, Endangered Wildlife Trust, South Africa.
Happold, D. C. D. and Happold, M. 1997. Conservation of mammals on a tobacco farm on the Highlands of Malawi. Biodiversity and Conservation 6: 837-852.
Herholdt EM. 1989. A new distribution record for the Botswana long-eared bat in South Africa. South African Journal of Wildlife Research 19: 86–87.
Kearney T. 2013. Laephotis botswanae Botswana Long-eared Bat. Pages 581–582 in Happold M, Happold D, editors. Mammals of Africa. Volume IV. Hedgehogs, shrews and bats. Bloomsbury Publishing, London, UK.
Kearney TC, Seamark EC. 2005. Morphometric analysis of cranial and external characters of Laephotis Thomas, 1901 (Mammalia: Chiroptera: Vespertilionidae) from southern Africa. Annals of the Transvaal Museum 42: 71–87.
Kunz TH, Braun de Torrez E, Bauer D, Lobova T, Fleming TH. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Moir, M., Richards, L.R.., Rambau, R.V., Cherry, M.I. 2020a. Bats of Eastern Cape and southern KwaZulu-Natal forests, South Africa: diversity, call library and range extensions. Acta Chiropterologica, 22(2):365-381.
Moir, M., Richards, L.R., Cherry, M.I., Rambau, R.V. 2020b. Demographic responses of forest-utilizing bats to past climate change in South Africa. Biological Journal of the Linnean Society, 130(4): 850-868.
Moir, M., Richards, L.R.R., Rambau, R.V., Cherry, M.I. 2021. Functional diversity and trait filtering of insectivorous bats relate to forest biogeography and fragmentation in South Africa. Journal of Biogeography, 48(5): 1170-1182
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman, M.C. 2020. Bats of Southern and Central Africa: A Biogeographic and Taxonomic Synthesis. University of the Witwatersrand Press, Johannesburg, South Africa.
Peterson RL. 1973. The first known female of the African Longeared Bat Laephotis wintoni (Vespertilionidae: Chiroptera. Canadian Journal of Zoology 51: 601–603.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N. 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Pierce M. W., Kearney T., Seamark E. C. J., Curran M. Kopp M., Keith M. 2012. New records and echolocation information of Laephotis botswanae (Chiroptera, Vespertilionidae) from southern Africa, Durban Natural Science Museum Novitates 34.
Taylor, P. J., Markotter, W., Strydom, E., Kearney, T., Cotterill, F. P. D., Cory Toussaint, D., Weier, S. M., Keith, M., Neef, G., Richards, L., Howard, A., Mamba, M., Magagula, S., Monadjem, A. 2022. Integrative taxonomic analysis of new collections from the central Angolan highlands resolves the taxonomy of African pipistrelloid bats on a continental scale. Zoological Journal of the Linnean Society 196: 1570-1590, https://doi.org/10.1093/zoolinnean/zlac071
Taylor, P. J., Kearney, T. C., Clark, V. R., Howard, A., Mdluli, M. V., Markotter, W. Geldenhuys, M., Richards, L. R., Rakotoarivelo, A. R., Watson, J., Balona, J., and Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate-driven diversification and a buffer against future climate change in bats, Global Change Biology, 30:e17344, https://doi.org/10.1111/gcb.17344.
