Bontebok
Damaliscus pygargus ssp. pygargus

2025 Red list status
Vulnerable
Regional Population Trend
Decline
change compared
to 2016
No Change
Overview
Damaliscus pygargus ssp. pygargus – (Pallas, 1767)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Damaliscus – pygargus – pygargus
Common Names: Bontebok (English), Bontebok (Afrikaans), Pitsi ya maronthonthwane (Sepedi), Inyamatane (Swati), Nglangu (Xitsonga)
Synonyms: Damaliscus dorcas ssp. dorcas Pallas, 1766
Taxonomic Note:
Appeared in the 1996 IUCN Red List as Damaliscus dorcas dorcas, but it is generally agreed that D. pygargus pygargus is the correct name (see Grubb 1993). Van der Walt et al. (2013) argue that the matter of species versus subspecies status is far from resolved. Their genetic study, based on 34 Bontebok and 42 Blesbok (D. p. phillipsi) individuals, indicated that the classification of alpha taxonomy should be reconsidered in this genus and that management should seek to avoid hybridisation and sustain remaining diversity in the Bontebok.
Red List Status: Vulnerable, VU B1ab(iii)+2ab(ii,iii) + D1
Assessment Information
Assessors: de Villiers, M.S.1, Kerley, G.I.H.2, Herbst, M.3, Jordaan, M.1, & da Silva, J.4
Reviewers: Selier, J.4 & Radloff, F.G.T5
Contributors: Forsythe, T.1, Roxburgh, L.6 & Patel, T.6
Institutions: 1CapeNature, 2Nelson Mandela University, 3SANParks, 4South African National Biodiversity Institute, 5Cape Peninsula University of Technology, 6Endangered Wildlife Trust
Previous Assessors: Radloff, F.G.T., Birss, C., Cowell, C., Peinke, D., Dalton, D. & Kotze, A.
Previous Reviewers: Kerley, G., Child, M.F. & Mallon, D.P.
Previous Contributors: Hignett, D., Kleinhans, D., Jacobs, L., R. Nowers.
Assessment Rationale
The Bontebok (Damaliscus pygargus pygargus) is endemic to the East Coast Renosterveld bioregion within the Cape Floristic Region of the Western Cape, South Africa. The Bontebok population reached a critical low in the early 1900s but was saved from extinction by private landowners in the Bredasdorp area and the subsequent declaration and establishment of a Bontebok subpopulation on Bontebok National Park.
Currently (2023), the core population – Bontebok on protected areas in the natural distribution range (NDR) -is estimated to comprise of 500 individuals, equivalent to 375 mature individuals using a 75% mature population structure. An extended Bontebok natural distribution range (ENDR) has been defined as those areas adjacent the NDR within fynbos habitat traditionally considered suitable for Bontebok. Within the ENDR, there are 234 Bontebok on four protected areas. If it is assumed that all of these subpopulations are hybrid-free (this is true for provincial and national park subpopulations, but not all privately-owned Contract Nature Reserve subpopulations have been tested) and if they are included in the population estimate, then the total population size increases to 734 (551 mature individuals).
There are also a large number of Bontebok on private land. In the NDR, there are 108 Bontebok on seven private properties while in the ENDR, there are 1,229 Bontebok on 78 private properties. However, 154 of the animals on private land are in subpopulations that are known to contain hybrids. Preliminary national estimates of privately-owned subpopulations suggest that only 33–39% of these qualify as “wild” (Child et al. 2019). If all subpopulations on private land in the NDR and ENDR except those on private land that have been shown to contain hybrids are included, and the wildness estimates applied, then the total population size is increased by 390-461, resulting in a maximum of 1,124-1,195 individuals, equivalent to 843-897 mature individuals.
According to the 2024 draft non detriment finding for Bontebok (Scientific Authority of South Africa 2024), there are also 7,243-8,493 animals outside the NDR and ENDR. The IUCN does not include extralimital subpopulations in red list assessments, therefore these are not included in our population estimates. Although Bontebok numbers within the ENDR and extralimital ranges (i.e., populations beyond the NDR and ENDR) are increasing, the core population within the natural range has decreased since 2004 (770 individuals in protected areas in 2004 compared to 686 individuals in 2014 and 500 in 2023). Possible drivers of this decrease are health and reproductive issues associated with inbreeding, lack of appropriate habitat management, parasites and disease.
Protected area expansion possibilities are limited within the NDR and ENDR. Currently, the extent of occurrence (EOO) within the natural range is estimated at 8,779 km² and the current observed area of occupancy (AOO) is 356 km² (using the IUCN method of occupancy within a 2 by 2 km square grid). Including all known Bontebok-containing areas within both the NDR and ENDR yields 1,064 km² of observed occupancy (using occurrence records from 2017 to 2024). Protected areas within the ENDR with observed records of Bontebok total 1,606 km², and within the NDR total 638 km² (2024 South African Protected Areas Database, DFFE). There has been an ongoing loss of natural habitat to agriculture and urban in the Western Cape. Between 2014 and 2020, 1.4% of the natural land area in the Western Cape was converted to transformed landcover types (South African National Land-Cover 2014 / 2020 Change Assessment). This is a total loss of 251 km² of natural habitat, at a rate of loss of 42 km²/year. In 2022, the area of natural habitat remaining in the ENDR and NDR was 10886 km², equivalent to 43% if the estimated original extent. An estimated 87% of the area now comprises Critically Endangered and Endangered vegetation types.
Given that the estimated mature population size (within the NDR and ENDR areas) is anywhere between 375 and 897 individuals, that the numbers of hybrids is unknown, and that there is no indication that the population has exceeded the threshold for Vulnerable for the last seven years (2016-2023), Bontebok is listed here as Vulnerable D1 (<1000 mature individuals). Additionally, because the number of individuals in each subpopulation is <1,000 individuals, we also list it under Vulnerable C. Furthermore, the area of occupancy (AOO) is estimated at 1,064 km² and that there is continuing loss of suitable habitat that limits population expansion within both the NDR and ENDR range, and that all subpopulations are fragmented by fencing with no metapopulation plan in place, Bontebok also qualifies for Vulnerable under criterion B.
In 2019, a Biodiversity Management Plan for Bontebok was gazetted (South Africa 2019) with the aim of securing the long-term survival of the subspecies in the wild. Threats identified were hybridisation with Blesbok (Damaliscus pygargus phillipsi), loss of genetic diversity, habitat loss, and disease and parasite issues. The need for a metapopulation management strategy was also stressed (South Africa 2019). CapeNature’s Bontebok Conservation, Translocation and Utilization Policy (CapeNature 2022) addresses some of these threats, however there is still a need to (a) determine the genetic status of all Bontebok subpopulations in the NDR and ENDR (allowing for a more accurate assessment of true population size), and (b) develop and implement an active metapopulation management plan to facilitate gene flow, prevent inbreeding and promote population growth. Landowners should be incentivised to become Bontebok stewards and to manage habitat to improve subpopulation growth, allowing for the expansion of the conservation estate for this subspecies within the NDR and ENDR.
Red List Index
Red List Index: No change
Recommended citation: de Villiers MS, Kerley GIH, Herbst M, Jordaan M & da Silva JM. 2025. A conservation assessment of Damaliscus pygargus ssp pygargus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Bontebok are endemic to the Western Cape Province, South Africa, although introductions have been made in most provinces. Historically, they were confined to the coastal plain (60–200 masl) east of the Kogelberg in the Western Cape, where they are thought to have concentrated on the renosterveld areas in the Fynbos Biome. It is assumed that they were mainly confined to the East Coast Renosterveld Bioregion due to its well-developed grass component, but to have also occurred in the less grassy South Coast Fynbos Bioregion (Boshoff and Kerley 2001, Skead 2011, Boshoff et al. 2016). Early settlers may have confused Bontebok and Blesbok (D. pygargus phillipsi), resulting in overestimation the historical distribution of Bontebok. In reality, the two subspecies were separated by more than 350 km at the time of European settlement (van Rensburg 1975, Boshoff et al. 2016).
Due to its restricted range, habitat transformation for agriculture, competition with domestic grazers and hunting pressure, the Bontebok population reached a critical low in the early 1900s. In response to this, the Bontebok National Park in the Bredasdorp district was established (Barnard and van der Walt 1961). Translocated Bontebok from the Bontebok National Park subsequently formed the nucleus of reintroduced populations in other protected areas.
All translocations in the Western Cape are subject to the CapeNature Bontebok Conservation, Translocation and Utilisation Policy (CapeNature 2016). Within the NDR, there is a high degree of habitat fragmentation and natural dispersal between localities with Bontebok is prevented by fences and other barriers. Habitat loss within the NDR has been so extensive that the area was estimated to be able to, at best, support 38% of the pre-transformation population potential (Kerley et al. 2003). The poor quality and limited availability of the remaining natural habitat within the NDR necessitated the recognition of additional suitable habitat (in terms of altitude, slope and major preferred vegetation types), in areas adjacent to the NDR. This is referred to as the extended natural distribution range (ENDR), and has been demarcated for regulatory purposes. This extension has enabled the establishment of the subspecies over a wider area which includes the West Coast and Table Mountain national parks. The population within the NDR is small and the ENDR concept is a mechanism to support a larger population, however there is no metapopulation plan to connect subpopulations within the NDR to the ENDR and no further reintroductions from protected localities in the NDR to those in the ENDR have occurred.
Suitable natural habitat within the NDR is limited to the remaining renosterveld patches and some small grassy habitat patches in the fynbos areas of the Overberg region. The EOO within the natural range is estimated to be 8,779 km²,and the current observed AOO is 356 km² (using the IUCN method of occupancy within a 2 by 2 km square grid). Including all known Bontebok-containing areas within both the NDR and ENDR yields 1,064 km² of observed occupancy (using occurrence records from 2017 to 2024). Protected areas within the ENDR with observed records of Bontebok total 1,606 km² (2024 South African Protected Areas Database, DFFE). We infer a continuing loss of habitat from ongoing agricultural and urban expansion within the Cape Floristic Region. Between 2014 and 2020, there was a conversion of 1.4% of the land area from natural to transformed landcover types in the Western Cape (South African National Land-Cover 2014 / 2020 Change Assessment). This is a total loss of 251 km² of natural habitat over a 6 year period, or a loss of 42 km² per year. In 2022, the area of natural habitat remaining in the ENDR and NDR was 10886 km², equivalent to 43% if the estimated original extent. An estimated 87% of the area now comprises Critically Endangered and Endangered vegetation types.
Extra-limital subpopulations have been established on private farms or game ranches in the Eastern Cape, Northern Cape, Free State, Limpopo and North West provinces, some of which fall within the Blesbok NDR. Some extra-limital populations are at risk of hybridisation with Blesbok.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
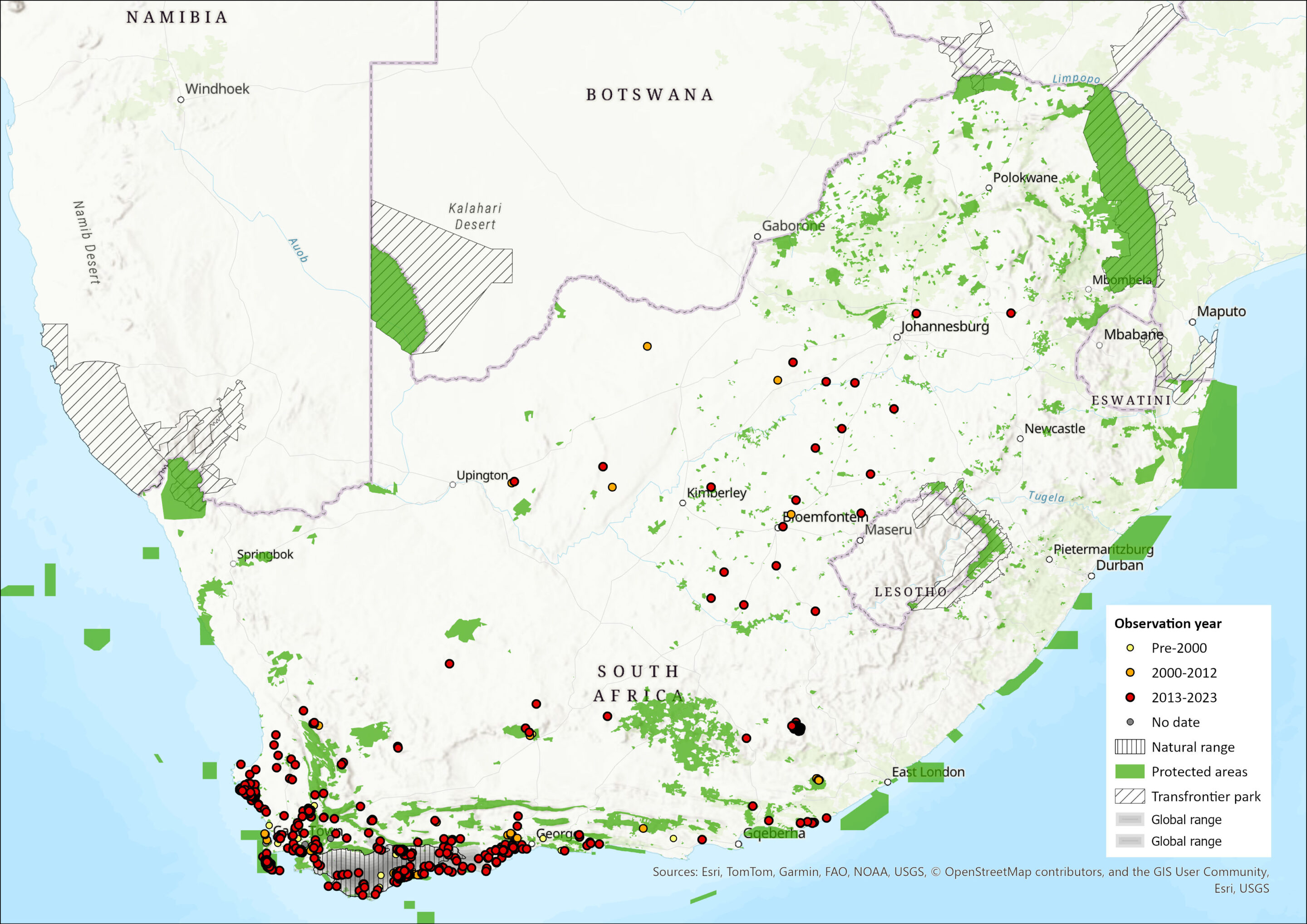
Figure 1. Distribution records for Bontebok (Damaliscus pygargus ssp. pygargus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | – |
| South Africa -> Eastern Cape Province | Extant | Introduced | – | – |
| South Africa -> Free State | Extant | Introduced | – | – |
| South Africa -> North-West Province | Extant | Introduced | – | – |
| South Africa -> Northern Cape Province | Extant | Introduced | – | – |
| South Africa -> Western Cape | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
Modelling of suitable habitat for Bontebok under future climate conditions showed no strong signal, with model outputs ranging from an increase of 1% in suitable habitat to a loss of 6% of suitable habitat (Cowan 2025). Fire and drought are key natural drivers of environmental processes in renosterveld, and both are predicted to be impacted by climate change. Top climate-related risks to the Fynbos Biome are predicted to be increased intensity and frequency of fires, and more “out of season” fires, an increase in alien invasive species, and increased habitat transformation and fragmentation, especially in the lowlands through agriculture and urbanization. This is expected to be exacerbated by changes in rainfall frequency, amount, and seasonality, most notably by a reduction in winter rainfall, a decrease in rainfall frequency and an increase in the intensity of rain events (DEA 2015, Engelbrecht et al. 2024). With environmental change, it is possible that disease vectors might change in distribution, thereby increasing the risk of disease transmissions. Species most at risk from climate change are those with restricted distributions and poor dispersal abilities, habitat specialists, and species that are responsive to specific disturbance regimes (DEA 2015). Bontebok qualify on all accounts. Landscape barriers such as fences and roads will prevent unassisted dispersal of Bontebok away from habitat that becomes less suitable into more suitable areas, and conservation translocations are likely to become an even more important aspect of Bontebok management than they are now.
Population information
The Bontebok population reached a critical low in the early 1900s. The Bontebok National Park was established with an initial population of 22 Bontebok (Bigalke 1955) (but note this figure was recorded as 17 by Barnard and van der Walt (1961)). At this time, there was also an unknown number of Bontebok on private land (van Rensburg 1975). The subpopulation subsequently grew but remained below 100 individuals. Bontebok at this site suffered from copper deficiency and high parasite infestation, specifically lungworm (Protostrogylus spp.). The park was also very small (6.8 km²) and was extensively covered (80%) by unpalatable renosterbos (Dicerothamnus rhinocerotis) (Barnard and van der Walt 1961). In 1960, the present site of Bontebok National Park was proclaimed close to Swellendam and 84 Bontebok were translocated there, of which 61 survived (Penzhorn 1971). At the new locality, numbers increased to 320 in 1981. This national park served as the source for the establishment of Bontebok at other localities.
Based on protected area game counts and information supplied by private landowners, the current number of Bontebok can be determined with reasonable accuracy. In 2023, it was estimated that there was a total of 500 individuals on two protected areas in the NDR (for comparison with earlier population estimates, this includes not only Bontebok National Park and De Hoop Nature Reserve, but also the privately owned Denel Overberg Test Range adjacent to De Hoop) (CapeNature, unpublished data). Assuming a mature population structure of 75%, it is thus estimated that there are 375 mature individuals on protected areas within the NDR in 2023. An extended Bontebok natural distribution range (ENDR) has been defined as those areas adjacent to the NDR within fynbos habitat traditionally considered suitable for Bontebok. Within the ENDR, there are 234 Bontebok on four protected areas (CapeNature, unpublished data). If it is assumed that all of these subpopulations are hybrid-free (this is true for provincial and national park subpopulations, but not all privately owned Contract Nature Reserve subpopulations have been tested) and if they are included in the population estimate, then the total population size increases to 734 (551 mature individuals).
There are also 108 and 1,229 Bontebok that are not formally protected in the NDR and ENDR respectively, but 154 of these animals are in subpopulations that are known to contain hybrids (CapeNature, unpublished data). The occurrence of hybrids on other properties is largely unknown. Preliminary national estimates of privately-owned subpopulations suggest that only 33–39% of these qualify as “wild” (Child et al. 2019). If all subpopulations in the NDR and ENDR are included, but those on private land that are known to contain hybrids are excluded, and if the wildness estimates are applied, then the total population size is increased by 390-461, resulting in a maximum of 1,124-1,195 individuals, equivalent to 843-897 mature individuals.
According to the 2024 draft non detriment finding for Bontebok (Scientific Authority of South Africa 2024), there are also 7,243-8,493 animals outside the NDR and ENDR. The IUCN does not include extralimital subpopulations in red list assessments, therefore these are not included in our population estimates. Although Bontebok numbers within the ENDR and extralimital ranges have increased, the core population within the NDR has decreased since 2004 (770 individuals in formally protected areas in 2004 compared to 686 individuals in 2014 and 500 in 2023) and there are limited protected area expansion possibilities in both the NDR and ENDR.
Development and implementation of a Bontebok metapopulation management plan is a priority in order to sustain Bontebok genetic integrity and genetic diversity. Within the NDR, there are many small subpopulations that are at carrying capacity. For example, in 2022 the average subpopulation size on private properties in the Western Cape was 16 individuals (N = 153 properties), compared to 56 individuals in the Eastern Cape (N = 70 properties), 27 individuals in the Free State (N = 50 properties), 11-22 individuals in the Northern Cape (N = 115 properties), 10 individuals in the North West Province (N=3 properties), and four individuals in Limpopo Province (N=1 property) (Scientific Authority 2024). Although there is still scope for the core population to increase, state-owned and private protected areas need to significantly expand and be connected to localities in the ENDR in order to create a resilient population. Some extralimital subpopulations may be suitable for reintroductions or supplementation into the NDR and ENDR. A consideration is the time that has elapsed since removal of animals from the NDR, since selection pressures in different areas may differ significantly and this can lead to genetic erosion, genetic drift and inbreeding, potentially with the fixation of deleterious alleles, as well as behavioural changes (Russo et al. 2019). The shorter the time since the extralimital introduction, therefore, the higher the conservation value of the subpopulation.
Population Information
Current population trend: Recent decline in the core range.
Continuing decline in mature individuals? Recent decline in the core range.
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Number of mature individuals in population: 375 in the core range, and between 843 and 897 in the NDR and ENDR combined.
Number of mature individuals in largest subpopulation: 399 (De Hoop Nature Reserve and Overberg Test Range combined, 2023).
Number of subpopulations: Within the NDR, there are only two formally protected subpopulations in Bontebok National Park and Greater De Hoop Area (i.e. De Hoop Nature Reserve and Denel Overberg Test Range). There are an additional 7 private subpopulations. The total number of potential subpopulations within the natural range is thus 2–9.
Severely fragmented: Yes, at present, the dispersal of the Bontebok is restricted by fences and it is dependent on translocation for dispersal.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A recent molecular study of D. pygargus identified clear genetic structure between the two subspecies (D. p. pygargus and D. p. phillipsi) and their hybrids (Mogakala et al. 2023). This confirms that Bontebok and Blesbok are separate subspecies, as indicated by earlier studies (Essop et al. 1991, Van der Walt et al. 2001). Permits for translocations of Bontebok within and between the NDR and ENDR require proof of genetic integrity (no hybridisation with Blesbok), in order to reduce the risks of outbreeding.
Various studies have also shown that Bontebok have low levels of genetic diversity. Measures of genetic diversity in non-hybrid reference Bontebok were found to be lower than that of non-hybrid reference Blesbok (Van Wyk et al. 2013, Van Wyk et al. 2016), and there is low adaptive and neutral diversity in Bontebok (Mogokala et al. 2023 – but no estimates of the effective population size for Bontebok are reported by these authors, to allow for a full understanding of the extent of this low diversity). Low genetic diversity is the effect of the small size of the historical founder population, limited subsequent exchange of individuals between subpopulations, and the small size of many of the subpopulations with the NDR and ENDR. It could also reflect the long term (Pleistocene) isolation and smaller population of Bontebok since separation from the Blesbok form.
Bontebok and Blesbok can hybridise. Historically, hybrids were identified on the basis of phenotypic evaluation (Fabricius et al. 1989), but these are not reliable and have been replaced by genetic testing (Van Wyk et al. 2013, Van Wyk et al. 2016). In the NDR and ENDR, genetic testing has detected hybrids at three localities. Genetic testing has been conducted at several other properties, but not all of these results are available (CapeNature, unpublished data). Hybridisation is currently addressed through provincial and industry protocols including the Bontebok Protocol developed by Wildlife Ranching South Africa’s Bontebok Breeders Association, the Bontebok Conservation, Translocation and Utilisation Policy in the Western Cape (CapeNature 2022), the Norms and Standards for the Keeping and Management of Bontebok in the Free State, the Eastern Cape Bontebok Protocol: Operational Policy Guideline of the Chief Directorate: Environmental Affairs, and the Northern Cape Nature Conservation Act and subsequent Regulations (Scientific Authority 2024). All of these protocols call for DNA profiling of any animal translocated or exported as hunting trophies, using a validated microsatellite marker set, as well as microchipping for individual identification (Scientific Authority 2024). All test results are curated by SANBI in a national database, and this now contains 12,334 Bontebok DNA profiles. No CITES export permits are issued for hunting trophies of Bontebok-Blesbok or Bontebok-Tsessebe (Damaliscus lunatus) and identified hybrids must be culled (Scientific Authority 2024). This should serve as an incentive to private landowners to stock non-hybrid Bontebok.
Habitats and ecology
The historic distribution range of Bontebok is very closely associated with the East Coast Renosterveld bioregion; in 2022, 26% of the original extent of this bioregion remained (CapeNature, unpublished data). The bioregion comprises four different vegetation types: Western Rȗens Shale Renosterveld, Central Rȗens Shale Renosterveld, Eastern Rȗens Shale Renosterveld and Mossel Bay Shale Renosterveld (Mucina and Rutherford 2006).
Bontebok are almost exclusively grazers (Beukes 1984) and are highly selective foragers, with a preference for short grass and recently burnt veld (Beukes 1987, Novellie 1987, Kraaij and Novellie 2010). Bontebok are water-dependent, and animals stay within 1.5 km from surface water during the dry season (Van Zyl 1978, Luyt 2005, David and Lloyd 2013). Within their natural range on the coastal lowlands, grassy areas are predominantly found in the renosterveld areas, but Bontebok also make use of small grassy microhabitats or recently burnt fynbos and strandveld habitats (Scott 1993, Radloff 2008). Suitable natural habitat for Bontebok is thus predominantly limited to the remaining renosterveld patches in the Overberg region. In the NDR and ENDR, subpopulations are primarily found on suboptimal habitat but also old, cultivated lands, where they appear to do relatively well. Bontebok prefer open areas with low shrubs and avoid tall, woody vegetation where visibility is low, and areas with steep slopes (Novellie 1987). Watson et al. (2011) showed that in the Bontebok National Park, Bontebok are more closely associated with burnt veld than Cape Mountain Zebra. Four to five years after fire, they no longer show interest in burnt areas and revert to grazing lawns of Cynodon dactylon. There is presently very little young veld available on De Hoop Nature Reserve and Overberg Test Range. In 2022, veld age on both properties was mostly 6-10 years old or older (15-25 years in the areas utilised by bontebok), with a limited area of <2 old years present on Overberg Test Range (CapeNature, 2024). However, it was observed that after one controlled burn on De Hoop Nature Reserve, Bontebok did not move into recently burnt limestone fynbos areas in large numbers (F. Radloff, pers. comm., 2023). It may be that there is a difference in the preference for young veld between Bontebok National Park and De Hoop Nature Reserve, related to differences in the geology of two protected areas, with the former having more silcrete and higher nutrient richness (F. Radloff, pers. comm., 2023).
A detailed 15-month study of Bontebok behaviour in Bontebok National Park during 1969 and 1970 revealed that the social structure comprises bachelor herds, nursery herds and territorial males (David 1973). Territories were few and small, leaving large areas where bachelor herds could roam with little or no harassment. Males defended their territories throughout the year through ritualised chasing, and physical fighting was not observed. The average nursery herd size was three adult females with 1.5 lambs. De Graaff et al. (1976) found a similar figure of 3 ± 2.2 (N = 18 herds) females per breeding herd. Bachelor herds consisted of males of all ages older than one year, as well as yearling females (David 1973). Bachelor herd size was 8.5 ± 6.3 (N = 12 herds) (De Graaff et al. 1976), and David (1973) reported as many as 75 animals together. Young males and females left the nursery herd on their own accord and could remain solitary but usually joined bachelor groups (David 1973). Males did not seem to establish territories before the age of five to six years of age, and returned to bachelor herds once displaced from their territories (David 1973). Bontebok are strongly seasonal breeders with the main calving season being September–October and the rutting season January to mid-March (David 1973).
Ecosystem and cultural services: Bontebok are a flagship subspecies of the Cape Floristic Region, particularly for renosterveld habitat, and can be used in ecotourism ventures. Rarity may increase trophy hunting and live sale value, especially of genetically tested non-hybrid Bontebok (Kerley et al. 2003).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
Life History
Generation Length: Unknown but likely similar to Blesbok (8.4 years)
Age at Maturity: Female or unspecified: Sexual maturity 2 years, social maturity 3 years
Age at Maturity: Male: Sexual maturity 2 years, social maturity 5-6 years
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 15 years in captivity
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 240 days
Reproductive Periodicity: Lambs born in spring and early summer, rutting in mid to late summer.
Average Annual Fecundity or Litter Size: One calf per adult female
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: Herd-forming
Systems
System: Terrestrial
General Use and Trade Information
There is no national quota for Bontebok harvest. Within all provinces in South Africa, restricted activities relating to Bontebok are permitted and well-managed (Scientific Authority 2024). Bontebok are utilised for trophy hunting, local hunting, photographic tourism, live sales, and recreation (e.g. captive populations). No hunting of Bontebok currently occurs on any national parks or provincial reserves, and harvesting of bontebok on protected areas is mainly in the form of live sales and translocation (Scientific Authority 2024). From 2017-2022, 261 Bontebok permits were issued in the Western Cape. The majority of these were for Certificates of Adequate Enclosure and transport, but four were for export from the province and 25 were for hunting. Bontebok are widely hunted and traded outside of the natural range, where numbers have increased dramatically (CapeNature, unpublished data).
Landowners maintain hunting records, but these are generally not accessible by conservation authorities. There is thus no official, consolidated record of offtakes to inform metapopulation management decisions (Scientific Authority 2024). Between 2010 and 2022, about 1,909 Bontebok were hunted by international clients (Scientific Authority 2024). Data from the DFFE Professional Hunters’ Register indicates a decline in hunting demand: more than 300 per year in 2011 and 2012, about 100 in 2019, five in 2020, and 54 in 2022 (Scientific Authority 2024). According to the CITES trade database, approximately 1,844 trophies and 35 live animals were exported between 2010 and 2021 (Scientific Authority 2024). Approximately 56% of all trophy hunts take place in the Eastern Cape, with less than 2% in the Western Cape (Scientific Authority 2024).
In June 2024, the local trophy hunting price for Bontebok was R14,375 and the international price was $1,300 (a decrease of 53% from 2023) (R. Nowers, unpublished data). In South Africa, live sale prices for Bontebok rams increased sharply in 2015, reaching an all-time high average of R78,000/ram. Prices subsequently dropped, with the average cost of a ram being R2,500 in 2021 (the lowest recorded in the period 2004-2023) and R5,981 in 2023 (R. Nowers, unpublished data). There is no available information on the market value of genetically proven non-hybrid Bontebok versus untested Bontebok and together with current low prices for live animals and trophies, it acts as a disincentive to landowners to conserve the subspecies and its habitat.
There has been negligible illegal offtake of bontebok on protected areas, and poaching occurs at very low levels on other properties. SANBI has not recorded any forensic cases in recent years (Scientific Authority 2024), but there is evidence of at least one illegal harvesting incident in the Western Cape (three Bontebok, 2018) and illegal harvesting might be a threat on the Bontebok National Park.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| No | Low incidence of illegal poaching | Yes | Trophy hunting and live sales |
National Commercial Value: Yes
International Commercial Value: Yes
End Use: (Hunting trophies and meat)
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Formally protected areas sell animals to the private sector
Threats
The major threats to this subspecies are habitat loss in the NDR (thus limiting population expansion), low genetic diversity, and hybridisation with Blesbok. Each of these are described below. A major concern is the lack of a metapopulation management strategy, potentially resulting in mismanagement of the subspecies and its habitat, and misunderstanding between stakeholders. Such a strategy should include incentives for landowners to continue managing and conserving Bontebok and it should build on the strengths of the previous success of saving this subspecies.
Habitat loss and degradation has resulted in fragmented and isolated subpopulations, which exist primarily on (apparently) suboptimal habitat and have limited opportunities for growth. Although suitable natural habitat is limited in the NDR and ENDR, habitat management has the potential to stimulate population performance. For the two core populations, i.e. Bontebok National Park and the Greater De Hoop Conservation Area, one such action is the implementation of appropriate fire regimes to ensure that there is sufficient young veld available for Bontebok. However, Bontebok are not the only management priority for these protected areas and that there are other factors which will influence fire management strategies, e.g. populations of plants of conservation concern. Furthermore, there is a lack of information on appropriate fire regimes for renosterveld. On these core areas, other management actions that might benefit Bontebok include the provision of mineral supplements (in particular Copper; Barnard & van der Walt 1961)), population control of competing herbivore species, and parasite control. Stress related to poor body condition might alter sex ratios at birth, resulting in skewed sex ratios and decreased population performance. Nutritional stress can also increase susceptibility to parasites and associated diseases, and Bontebok are known to be prone to these (references in South Africa 2019). More information on the local significance of these issues is needed and can be obtained through targeted research and monitoring.
There is suspected low genetic diversity within the core population and thus presumably a lack of resilience to environmental change, including climate change. The historical genetic bottlenecks are currently exacerbated by the fragmented and isolated nature of the subpopulations, which can lead to decreased genetic variation within individual subpopulations due to inbreeding and genetic drift. Additionally, selection by some game farmers for coat colour, pattern and horn sizes threatens the genetic integrity and viability of the population through inbreeding depression.
Hybridisation with Blesbok has occurred due to human-mediated mixing of the subspecies, although the extent to which this has occurred is unknown. Preliminary research showed that a high proportion of Bontebok could be hybrids, with hybridisation detected in 33% of Bontebok samples (van Wyk et al. 2013). Indications are that recent regulatory interventions and increased awareness has reduced the risk of hybridisation, and van Wyk et al. (2016) detected no Fi hybrids in their more recent and extensive survey.
Conservation
Currently, key protected areas for Bontebok are Bontebok National Park (genetically certified subpopulation within the NDR), Table Mountain National Park (genetically certified subpopulation within the ENDR), and De Hoop Nature Reserve and Denel Overberg Test Range (largest subpopulation in the NDR). Although there are currently no Bontebok on Agulhas National Park, this protected area in the NDR has the potential to support a sizeable subpopulation. Although some subpopulations have exhibited positive growth rates, offtakes and translocations out of the NDR have suppressed core population growth to some extent. More importantly, the lack of additional suitable natural habitat within the NDR has severely inhibited population expansion and the area of occupancy has actually decreased since the previous red list assessment. The net effect is that the core Bontebok population has decreased within the NDR since the 2004 assessment (Friedmann and Daly 2004). This concern is addressed in the Bontebok Biodiversity Management Plan (South Africa 2019), with key actions listed in the implementation framework. Some of these actions have been implemented but others have not, and there should now be a focus on the latter (South Africa 2019), with key actions listed in the implementation framework. Some of these actions have been implemented but others have not, and there should now be a focus on the latter.
Recommendations for national and provincial conservation entities:
- Evaluate the implementation of the 2019 Bontebok Biodiversity Management Plan and address those actions which have not yet been addressed.
- Develop a Bontebok metapopulation management strategy/ guideline. This should aim to ensure that translocations sustain or improve genetic diversity and integrity, and increase the number and size of subpopulations within the NDR and ENDR. The strategy should identify key subpopulations for genetic testing, and make recommendations for genetic bolstering. These subpopulations will mostly be those in the NDR and ENDR but should also include recently established ex situ/extralimital populations.
- Secure additional natural habitat for Bontebok in the NDR and ENDR. This should be incorporated into the Western Cape Protected Area Expansion Plan. Expansion of existing protected areas should continue until there is sufficient habitat in the NDR to support at least 2,000 Bontebok, and links between protected areas should be created wherever possible (Kerley et al. 2003). The Agulhas National Park has been identified as ideal habitat to establish a subpopulation of Bontebok but the park currently has no fencing, and funding for this should be sourced. Conservancies and corridors linking existing Bontebok localities should be created. This subspecies can act as a flagship for renosterveld conservation, and landowners in the NDR and ENDR should be incentivised to become Bontebok stewards. The importance of biodiversity stewardship to Bontebok conservation is illustrated by the subpopulation established on the Nuwejaars Special Management Area through the introduction of 38 animals in 2010. Bontebok are tolerant of human activities and readily utilise short grass areas and transformed landscapes, and this should expediate stewardship opportunities.
- Introduce mechanisms to slow transformation and loss of renosterveld habitat, apply offsets against the transformation of natural veld to agriculture.
Recommendations for land managers and practitioners:
- Restore renosterveld habitat. Possible mechanisms include the control or eradication of invasive alien plant species, implementation of fire and/or other disturbance regimes that are appropriate to the available veld types. Prudent veld management is needed, however, as improving grass cover within renosterveld could result in the loss of plant species diversity, including species classified as rare or threatened (Novellie and Kraaij 2010).
- Prevent overstocking of livestock and or/game, especially selective grazers, on properties with Bontebok. Population control can be achieved through sustainable harvesting, which provides a financial incentive to conserve the subspecies.
- Preferably only translocate animals within and between the NDR and ENDR, and incorporate this objective into wildlife management plans.
- Only introduce Bontebok where genetic tests show that these are not hybrids.
- Follow the recommendations of the metapopulation management strategy/guidelines, once these have been produced, to maintain or improve genetic diversity.
- Avoid stocking Blesbok in proximity to Bontebok.
Research priorities:
- Determine the size, demography, location, health and reproductive success of genetically tested, hybrid-free Bontebok subpopulations, and quantify the effective population size for the subspecies. This information should be incorporated into the metapopulation management strategy/guidelines. Information on which populations are stable or increasing and can be incorporated as core sites into the metapopulation is crucial.
- Determine the appropriate fire intervals for different types of renosterveld. This will help to inform fire management practices to sustain not only Bontebok but also botanical taxa of conservation concern. This information is urgently required to inform protected area management.
- Investigate the impacts of climate change on Bontebok habitat and population vigour. There is evidence that climate change will affect tick species composition and distribution, as well as the occurrence of tick-borne diseases (Nuttal 2022) and it is expected that new tick species and related diseases will invade parts of South Africa (Makwarela et al. 2023). Knowledge about these processes and their implications in the Bontebok NDR and ENDR is needed.
- There is a concern that low concentrations of certain essential micronutrients could be affecting the health of Bontebok in the core protected area subpopulations. Research on this, and other factors which impact Bontebok health, e.g. parasite loads, will inform management actions to mitigate these threats.
- Evaluate the demographic consequences of inbreeding and outbreeding (hybridisation) for Bontebok.
Bibliography
Allendorf, F.W., Leary, R.F., Spruell, P. and Wenburg, J.K. 2001. The problems with hybrids: setting conservation guidelines. Trends in Ecology & Evolution 16: 613-622.
Barnard PJ, van der Walt DK. 1961. Translocation of the bontebok (Damaliscus pygargus) from Bredasdorp to Swellendam. Koedoe 4: 105-109.
Beukes, P.C. 1984. Sommige aspekte van die ekologie van die Vaalribbok (Pelea Capreolus, Forster 1790) in die Bontebok Nasionale Park. University of Stellenbosch.
Beukes, P.C. 1987. Responses of grey rhebuck and bontebok to controlled fires in coastal renosterveld. South African Journal of Wildlife Research 17: 103-108.
Bigalke, R. 1955. The bontebok (Damaliscus pygargus (Pall.)) with special reference to its history and preservation. Fauna Flora 6: 95-116.
Boshoff AF, Landman M, Kerley GIH. 2016. Filling the gaps on the maps: historical distribution patterns of some larger mammals in part of southern Africa. Transactions Royal Society of South Africa 70: 1-65.
Boshoff, A.F. and Kerley, G.I.H. 2001. Potential distributions of the medium-to large-sized mammals in the Cape Floristic Region, based on historical accounts and habitat requirements. African Zoology 36: 245-273.
Broders HG, Mahoney SP, Montevecchi WA, Davidson WS. 1999. Population genetic structure and the effect of founder events on the genetic variability of moose, Alces alces, in Canada. Molecular Ecology 8: 1309-1315.
Broders HG, Mahoney SP, Montevecchi WA, Davidson WS. 1999. Population genetic structure and the effect of founder events on the genetic variability of moose, Alces alces, in Canada. Molecular Ecology 8: 1309-1315.
CapeNature. 2022. Bontebok conservation, translocation and utilisation policy. Version 5. Internal document, CapeNature, Cape Town.
Child MF, Selier SAJ, Radloff FGT, Taylor WA, Hoffmann M, Nel L, Power RJ, Birss C, Okes NC, Peel MJ, Mallon D & Davies-Mostert H. 2019. A framework to measure the wildness of managed large vertebrate populations. Conservation Biology 33(5): 1106-1119.
Cowan, O.S. 2025. Modelling the change in potential species distribution of Bontebok (Damaliscus pygargus pygargus) under predicted future climate change scenarios. Technical Report, Endangered Wildlife Trust.
Cowell C, Birss C. 2013. Resource Information for Bontebok (Damaliscus pygargus pygargus) in South Africa. CRC-11-2013. Cape Research Centre, South African National Parks, Cape Town, South Africa.
David JHM. 1973. The behaviour of the Bontebok, Damaliscus dorcas dorcas, (Pallas 1766), with special reference to territorial behaviour. Zeitschrift für Tierpsychologie 33: 38-107.
David, J. and Lloyd, P. 2013. Damaliscus pygargus. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa, Academic Press, Amsterdam, The Netherlands.
De Graaff G, van der Walt PT, van Zyl LJ. 1976. Populasie-samestelling van die bontebok Damaliscus dorcas dorcas in die Bontebok Nasionale Park gedurende Januarie 1974. Koedoe 19: 67-74.
Engelbrecht, F.A., Steinkopf, J., Padavatan, J., and Midgley, G.F. 2024. Projections of Future Climate Change in Southern Africa and the Potential for Regional Tipping Points. In: von Maltitz, G.P., et al. Sustainability of Southern African Ecosystems under Global Change. Ecological Studies, vol 248. Springer, Cham. https://doi.org/10.1007/978-3-031-10948-5_7
Fabricius C, van Hensbergen HJ, Zucchini W. 1989. A discriminant function for identifying hybrid bontebok x blesbok populations. South African Journal of Wildlife Research 19: 61-66.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. CBSG Southern Africa, IUCN SSC Conservation Breeding Specialist Group, Endangered Wildlife Trust, South Africa.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Grubb P. 1993. Order Artiodactyla. In: D.E. Wilson & D.M. Reeder (ed.), Mammal Species of the World: A Taxonomic and Geographic Reference, 2nd edition., Smithsonian Institution Press.
IUCN Standards and Petitions Subcommittee. 2014. Guidelines for Using the IUCN Red List Categories and Criteria. Version 11.
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-2. Available at: www.iucnredlist.org. (Accessed: 14 September 2017).
Kerley GIH, Pressey RL, Cowling RM, Boshoff AF, Sims-Castley R. 2003. Options for the conservation of large and medium-sized mammals in the Cape Floristic Region hotspot, South Africa. Biological Conservation 112: 169-190.
Kraaij, T. and Novellie, P.A. 2010. Habitat selection by large herbivores in relation to fire at the Bontebok National Park (1974–2009): the effects of management changes. African Journal of Range & Forage Science 27: 21-27.
Luyt ED. 2005. Models of Bontebok (Damaliscus pygargus pygargus, Pallas 1766) habitat preferences in the Bontebok National Park and sustainable stocking rates. MSc. Thesis. University of Stellenbosch, Stellenbosch, South Africa.
Makwarela, T.G.; Nyangiwe, N.; Masebe, T.; Mbizeni, S.; Nesengani, L.T.; Djikeng, A.; Mapholi, N.O. Tick Diversity and Distribution of Hard (Ixodidae) Cattle Ticks in South Africa. Microbiol. Res. 2023, 14, 42-59. https://doi.org/10.3390/microbiolres14010004.
Mogakala MR, Smith RM, Mavimbela C, Dalton DL. Identification of low levels of neutral and functional genetic diversity in South African bontebok (Damaliscus pygargus pygargus). Ecol Evol. 2024 Mar 6;14(3):e10962. doi: 10.1002/ece3.10962.
Mucina, L. and Rutherford, M.C. 2006. The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria, South Africa.
Novellie P, Kraaij T. 2010. Evaluation of Themeda triandra as an indicator for monitoring the effects of grazing and fire in the Bontebok National Park. Koedoe 52: 1-5.
Novellie, P. 1987. Interrelationships between fire, grazing and grass cover at the Bontebok National Park. Koedoe 30: 1-17.
Nuttal P.A. 2022. Climate change impacts on ticks and tick-borne infections. Biologia 77: 1503-1512.
Penzhorn BL. 1971. A summary of the re-introduction of ungulates into South African National Parks (to 31 December 1970). Koedoe 14: 145-159.
Radloff FGT. 2008. The ecology of the large herbivores native to the coastal lowlands of the Western Cape, South Africa. Ph.D. Thesis. University of Stellenbosch, Stellenbosch, South Africa.
Scientific Authority of South Africa. 2024. Non-detriment finding for Damaliscus pygargus pygargus (bontebok). Draft report, reference number Dam_pyg_pyg_Feb2024.
Scott HA. 1993. An investigation into possible reasons for a decline in numbers of bontebok Damaliscus dorcas dorcas (Pallas, 1766) in the De Hoop Nature Reserve, Southwestern Cape. M.Tech Thesis. Port Elizabeth Technikon, Saasveld School of Forestry, South Africa.
Skead CJ. 2011. Historical incidence of the larger land mammals in the broader Western and Northern Cape provinces. In: A. Boshoff, G. Kerley, and P. Lloyd (eds), Historical incidence of the larger land mammals in the broader Western and Northern Cape provinces, Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth, South Africa.
South Africa. 2019. National Environmental Management: Biodiversity Act, 2004 (Act No. 10 of 2004) Biodiversity Management Plan for the bontebok (Damaliscus pygargus). Government Gazette No. 42887, 6 December 2019.
Watson LH, Kraaij T, Novellie P. 2011. Management of rare ungulates in a small park: habitat use of Bontebok and Cape Mountain Zebra in Bontebok National Park assessed by counts of dung groups. South African Journal of Wildlife Research 41: 158-166.
van der Walt J, Nel LH, Hoelzel AR. 2013. Differentiation at mitochondrial and nuclear loci between the blesbok (Damaliscus pygargus phillipsi) and bontebok (D. p. pygargus): implications for conservation strategy. Conservation Genetics 14: 243-248.
van der Walt, J.M., Nel, L.H. and Hoelzel, A.R. 2001. Characterization of major histocompatibility complex DRB diversity in the endemic South African antelope Damaliscus pygargus: a comparison in two subspecies with different demographic histories. Molecular Ecology 10: 1679-1688. van Rensburg APJ. 1975. Die geskiedenis van die Nasionale Bontebokpark, Swellendam. Koedoe 18: 165-190.
van Rensburg APJ. 1975. Die geskiedenis van die Nasionale Bontebokpark, Swellendam. Koedoe 18: 165-190.
van Wyk AM, Kotzé A, Randi E, Dalton DL. 2013. A hybrid dilemma: a molecular investigation of South African bontebok (Damaliscus pygargus pygargus) and blesbok (Damaliscus pygargus phillipsi). Conservation Genetics 14: 589-599.
Van Wyk, A., Dalton, D., Hoban, S., Bruford, M., Russo, I-R., Birss, C., Grobler, J., Jansen van Vuuren, B. & Kotzé, A. (2016). Quantitative evaluation of hybridization and the impact on biodiversity conservation. Ecology and Evolution. 7. 10.1002/ece3.2595.
van Zyl LJ. 1978. Die waterbehoeftes en drinkgewoontes van die Bontebok Damaliscus dorcas dorcas (Pallas, 1766) in die Bontebok Nasionale Park. MSc. thesis. University of Stellenbosch, Stellenbosch, South Africa.
von Hase A, Rouget M, Maze K, Helme N. 2003. A finescale conservation plan for Cape lowlands renosterveld. Technical Report. Botanical Society of South Africa, Kirstenbosch, Cape Town.