Blue Wildebeest
Connochaetes taurinus

2025 Red list status
Least Concern
Regional Population Trend
Increasing
Change compared
to 2016
No Change
Overview
Connochaetes taurinus – (Burchell, 1823)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Connochaetes – taurinus
Common Names: Blue Wildebeest, Blue and White-bearded Wildebeest, Brindled Gnu, Brindled Wildebeest, Common Wildebeest (English), Blouwildebees (Afrikaans), Inkonkoni (Ndebele, Xhosa, Zulu), Imbudumo, Imbuduma ehlaza (Ndebele), Kgôkông (Sepedi, Setswana), Kgokong, Kgaranyane (Sesotho), Ingongoni, Ngongoni (Swati), Khongoni, Khongoini (Venda), Hongonyi (Xitsonga), Inkonkoni enombala oluhlaza (Zulu)
Synonyms: albojubatus, babaulti, borlei, cooksoni, corniculatus, fasciatus, gorgon, hecki, henrici, johnstoni, lorenzi, mattosi, mearnsi, reichei, rufijianus, schulzi
Taxonomic Note:
Generally, five subspecies are recognised the Western White-bearded Wildebeest (C. t. mearnsi), the Eastern White-bearded Wildebeest (C. t. albojubatus), Nyassa Wildebeest (C. t. johnstoni), Cookson’s Wildebeest (C. t. cooksoni), and the Blue Wildebeest (C. t. taurinus) (Ansell 1972, Grubb 2005).
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Patel, T.1 & da Silva, J.2
Reviewer: Anderson, J.3,4
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3International Conservation Services, 4IUCN SSC Antelope Specialist Group
Previous Assessors: Viljoen, P., Tambling, C. & Kotze, A.
Previous Reviewer: Child, M.F.
Previous Contributor: Relton, C.
Assessment Rationale
Listed as Least Concern as this species is numerous and occurs extensively throughout the assessment region and is present within many protected areas across its range. The largest subpopulation in Kruger National Park alone has a population size of 12,555 mature individuals in 2017. Thus, we infer there are well over 13,000 mature individuals in the assessment region. There are no real threats to this subspecies, however, incidences of hybridisation with Black Wildebeest (Connochaetes gnou), with fertile offspring, do occur. Additionally, it is unknown whether any exotic subspecies have been introduced into the region with resultant reduction in genetic integrity of C. taurinus taurinus. Selective breeding for colour variants is common practice in the wildlife industry. These threats should be monitored, and translocation regulations enforced to prevent compromised individuals entering formally protected areas.
Regional population effects: There is migration of individuals across transfrontier parks and the border between South Africa and Botswana, and no rescue effect is necessary as this subspecies is widespread and common within the assessment region. The historical migrations that existed in the Kalahari have been reduced due to the decimation of the Kalahari population (Spinage 1992) through the erection of veterinary fences and competition with cattle ranching (Estes & East 2009). The population in the Limpopo National Park all originate from the Kruger National Park (Estes & East 2009) and currently this population will most likely be a sink outside of the Kruger National Park rather than generating rescue effects for the assessment area. Wildebeest from South Africa have been introduced to Gorongoza National Park and Maputo Special Reserve as part of their rehabilitation programmes.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Patel T & da Silva JM. 2025. A conservation assessment of Connochaetes taurinus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
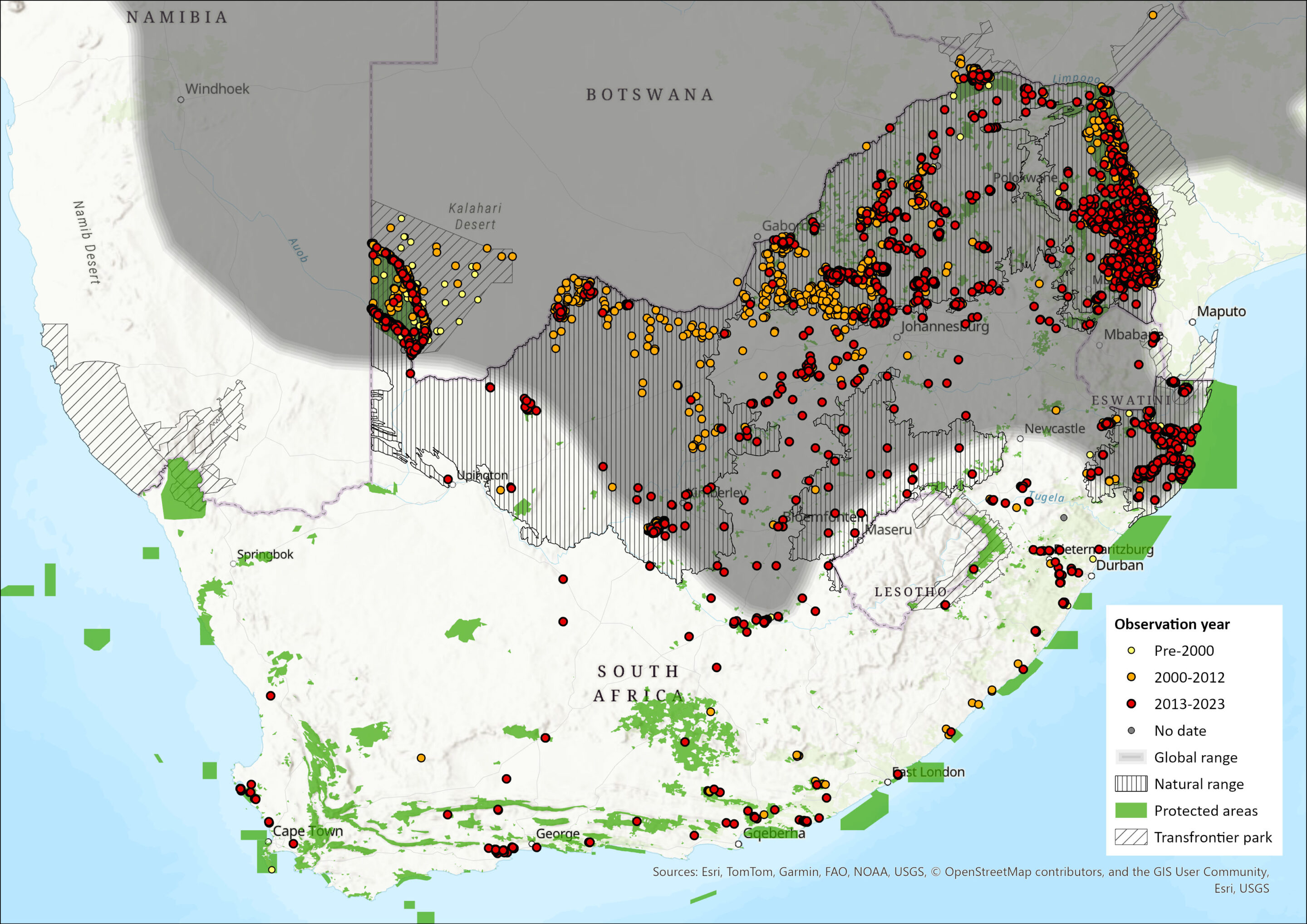
Regional Distribution and occurrence
Geographic Range
Formerly distributed from southern Kenya southwards to northern and eastern Namibia, Botswana, the Orange River in South Africa, and Mozambique (East 1999; Estes 2013; IUCN SSC Antelope Specialist Group 2016). Blue Wildebeest have also been introduced to regions outside of their former range, such as the Eastern Highlands of Zimbabwe, private farmland in Namibia and private game ranches south of their historical range in South Africa (East 1999; Estes & East 2009). They have never occurred in Lesotho (Lynch 1994).
C. t. taurinus occurs naturally in Namibia, South Africa to Mozambique north of the Orange River, from Mozambique to Zambia south of the Zambezi River, and from southwest Zambia to southeast and southern Angola (IUCN SSC Antelope Specialist Group 2016). This species was the inhabitant of the northern Kalahari savannahs, which is supported by naturally occurring populations in the Kgalagadi Transfontier Park (KTP) and Botswana’s Central Kalahari Game Reserve to the north (Smithers 1971; Skinner & Chimimba 2005), as well as in bushveld regions in northern Botswana.
This species is migratory in the KTP between the three borders with Namibia and Botswana. There is also evidence of local migrations within Kruger National Park (KNP). In South Africa, the species was largely confined to the northern parts of the country north of the Orange River (Estes & East 2009). They still occur throughout most of their historical range (Skinner & Chimimba 2005), although have been exterminated in large proportions of the Northern Cape where, historically, they occurred extensively in the Kalahari Thornveld, Lowveld and Bushveld habitats (Estes & East 2009). They have been widely introduced to all provinces, despite their natural range excluding the Western and Eastern Cape.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 50m
Elevation Upper Limit (in metres above sea level): 1200m
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Blue Wildebeest (Connochaetes taurinus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Eswatini | Extant | Reintroduced | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extinct Post-1500 | Native | – | – |
| Mozambique | Extant | Native & Reintroduced | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Droughts tend to favour the species, particularly in the lowveld (KNP) where high rainfall years result in increased predation levels (Mills et al. 1995) and could contribute to local declines during prolonged periods of above average rainfall. As such, climate change is not predicted to be as threatening to this species as other species, as this species is well-adapted to variable environmental conditions, as is seen through successful reintroduction and adaptation to extralimital provinces, such as the Western and Eastern Cape.
With global temperature fluctuations becoming more apparent, understanding the implications that changing climate has on ungulates is important. In response to high temperatures Blue Wildebeest have a tendency to remain heat stressed throughout the year, which will influence their daily movement patterns (Shrestha et al. 2014). In addition, with the artificial southward spread of the species, individuals are being exposed to longer and colder winters which are considered sub-optimal for African antelopes (Shrestha et al. 2012). Although it is doubtful that these temperature-mediated impacts will influence wildebeest in their natural range, populations outside of their natural range may be compromised resulting in an artificial sink. However, within the assessment region, this is unlikely to have a severe impact on the local, wild population, and may only be a cause for concern for the long-term farming of this species.
Population information
A population estimate conducted during the late 1990s (mainly from aerial surveys) revealed an approximate global abundance of 1,298,000 C. taurinus (East 1999), with the Serengeti–Mara migratory population constituting 70% of that population (IUCN SSC Antelope Specialist Group 2016). Of these, 150,000 were estimated to be C. t. taurinus, half of which were in protected areas, and one quarter in conservancies or on private land (Estes 2013). The most current global population estimate of Common Wildebeest is approximately 1,550,000 individuals, largely attributed to the recovery of the Serengeti–Mara population to approximately 1,300,000 (Estes & East 2009). Subspecies estimates include 130,000 C. t. taurinus, 5,000–10,000 C. t. cooksoni, and 50,000–75,000 C. t. johnstoni (Estes 2013). A decline in the C. t. albojubatus population revealed a current estimate of 6,000–8,000 individuals (Estes & East 2009).
East (1999) indicates that population densities estimated by aerial surveys range from less than 0.15 animals / km² in areas such as Kafue, Etosha, Hwange and the central and southern Kalahari, to 0.6–1.3 animals / km² in areas such as KNP, North Luangwa, Selous and Kajiado, and 3.6 animals / km² in Tarangire (IUCN SSC Antelope Specialist Group 2016). Total counts in areas where the species was abundant have produced population density estimates as high as 34–35 animals / km²; for example, in the Serengeti and Ngorongoro Crater (IUCN SSC Antelope Specialist Group 2016). Within the assessment region, the overall population was stable or increasing with an estimated population size of well over 10,000 mature individuals at the time of the previous assessment. In South African national parks (six areas) alone, there were an estimated 8,818–18,318 animals (2013 count; Ferreira et al. 2013). Across the country, on both formally protected areas and private lands, there were at least 44,689 animals in 2013 occurring on 648 protected areas of wildlife ranches (Endangered Wildlife Trust unpubl. data). This total was reduced to 42,450 on 552 properties by only including areas within the natural distribution, which equated to 29,715 mature animals assuming a 70% mature population structure. The population estimate was similar to that estimated in 2005 of approximately 40,000 to 45,000 (Estes & East 2009). At the time of the 2005 assessment, the South African population was deemed to be increasing (Estes & East 2009). The largest subpopulation occurs on KNP, estimated at 6,058–10,896 animals in 2012 (Ferreira et al. 2013). In 2017, the KNP alone had 9,342—17,936 animals (SANParks data). The subspecies was especially numerous on private land: for example, while there were more than 3,600 individuals in North West Parks alone, there were a further 17,000 animals on private land in the province (Power 2014). Unfortunately, the numbers of both Black and Blue Wildebeest on all private properties were unknown. The occupancy of Blue Wildebeest is suspected to be an underestimate.
Generation length has been calculated as 8 years (Pacifici et al. 2013), which yields a 24-year three generation period (1991–2015). Most subpopulations in protected areas have increased over this period (for example; Nel 2015; Ferreira et al. 2016).
Historically, there have been substantial declines in private subpopulations (for example, in the Associated Private Nature Reserves, west of KNP), as well as in the western parts of the KNP due to fencing restricting movement during periods of drought (Walker et al. 1987), which may counterbalance increases in other subpopulations. The subpopulation in the KTP fluctuates considerably depending on local climatic variation and predation levels. Even so, the subpopulation is not expected to decline in the near future. Although most subpopulations in formally protected and private areas are fragmented and isolated due to fencing, the largest subpopulations (KTP and KNP) are free to follow local migrations, tracking recent rainfall. However, before the erection of fences these local migrations were much larger, with the KNP populations ranging to the foothills of the escarpment and those of KTP extending further into Botswana. Many subpopulations were confined and forced to be sedentary, resulting in local declines in abundance (IUCN SSC Antelope Specialist Group 2016).
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | – | – |
Population trend: Increasing
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Number of mature individuals in population: Approximately 40,000–45,000
Number of mature individuals in largest subpopulation: 12,555 in Kruger National Park (SANParks count, 2017)
Severely fragmented: Yes. Most subpopulations are restricted to fenced reserves or ranches.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Very Low)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Low)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
The five recognized subspecies are supported by morphological and genetic evidence (Liu et al. 2024). However, only moderate FST values (up to 0.35) were detected and relatively recent divergence times (65 kya) between ESUs, suggesting Blue Wildebeest subspecies are not highly differentiated. Given that only one ESU is present within the assessment region, it can be considered a single metapopulation and should be managed accordingly to mimic, as closely as possible, natural dispersal in order to maintain genetic diversity within the subspecies/ESU. This has implications on the selective breeding of colour morphs, which could act to lower or distort diversity levels in more natural populations should escapees interbreed with them. Given the species is highly managed, caution must be taken not to mix ESUs so that the genetic integrity of each can be conserved.
Assuming a single metapopulation and population estimates of approximately 40,000-50,000, the effective population size (Ne) for this population can be estimated after applying a conversion ratio of 0.1-0.3. This equates to an Ne between 4000 and 15000 animals.
Habitats and ecology
Blue Wildebeest are predominantly associated with savannah woodland and short grasslands, with the availability of shade and drinking water as essential habitat requirements (Skinner & Chimimba 2005; Estes 2013). Blue Wildebeest are grazers, but 10–20% of their diet can consist of browse (Gagnon & Chew 2000). In general, Blue Wildebeest prefer short grass areas with grass less than 100-150 mm in height (Skinner & Chimimba 2005). Blue Wildebeest tend to follow localized rainfall on shorter migrations (Skinner & Chimimba 2005). Bulls are territorial and will maintain the territory year-round unless drought forces the territories to be abandoned. Breeding herds consisting of females and associated young will move into male’s territories resulting in a territorial male associated with breeding herds. Rutting takes place from April to June and births occur in November and December. The natural population growth rate is approximately 20% / annum. Long term population trends of wildebeest appear to be correlated negatively with minimum temperature and positively with dry season rainfall, an artefact of population trends being related to the forage quantity and quality (Seydack et al. 2012).
Blue Wildebeest are a preferred prey species of Lions (Panthera leo) and exhibit cyclical predation patterns depending on the current climatic conditions. Short grass areas (grazing lawns and recently burnt patches) are targeted for nutrients but also appear to offer wildebeest protection from predators (Burkepile et al. 2013; Yoganand & Owen-Smith 2014). During wetter cycles, predation by Lions on Blue Wildebeest increases, whereas predation often declines during drier periods characterised by shorter grass (Mills et al. 1995). The increased predation on wildebeest during wet periods is expected to be the result of increased cover provided to stalking Lions and a fragmentation of the wildebeest herds (Smuts 1978).
Ecosystem and cultural services: The Blue Wildebeest is considered a keystone species in certain ecosystems such as the Serengeti where their migrations are the major drawcard for tourism revenue, as well as conserving the Serengeti ecosystem. Although large scale migrations occurred in the past, which included regions in the assessment areas, this aspect is not relevant for the assessment area. Within the assessment region, the Blue Wildebeest constitutes an important prey species for many large predators, including Spotted Hyaena (Crocuta crocuta), Lion (Hayward 2006), Leopard (Panthera pardus) (Hayward et al. 2006a), African Wild Dog (Lycaon pictus) (Hayward et al. 2006c) and Cheetah (Acinonyx jubatus) (Hayward et al. 2006b).
According to local folklore stories the wildebeest is a combination of the discarded parts of Zebra, Warthog (Phacochoerus africanus), Buffalo (Syncerus caffer) and Lion. The combination of the leftover stripes from the Zebra, the Warthog shape to its head, the neck shaped like that of a Buffalo and the left-over Lion’s mane was used to create the wildebeest. Alternate stories suggest the wildebeest is composed of the face of a mule, the beard of a goat, the horns of a cow and the body of a horse. Despite the conglomeration of parts, legend has it that the wildebeest was happy to be in the new world and had a personality of its own.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: Most conceive for the first time at 28 months
Age at Maturity: Male: Probably by 24 months
Size at Maturity (in cms): Female: shoulder height 135.3 cms; weight 214.4 kg
Size at Maturity (in cms): Male: shoulder height 147.2 cms; weight 251 kg
Longevity: 13-14 years in the wild, up to 24 years in captivity
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 250 days
Reproductive Periodicity: One per year, mostly in November-December
Average Annual Fecundity or Litter Size: One
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Full Migrant
Congregatory: Congregatory (and dispersive)
Systems
System: Terrestrial
General Use and Trade Information
This species is utilised widely in trophy hunting, biltong hunting (both local subsistence and local commercial), and live animal sales. These uses are not considered to impact negatively on the population, as it is expected to be increasing in abundance on private lands (Estes & East 2009). As such, this is a candidate species on which to base rural wildlife economies and sustainable food production.
The proportion of off-take from the wild and game ranches is difficult to determine. Growth rate is estimated at approximately 20% per annum (Bothma 2002) so under optimal conditions (as experienced on numerous game farms where water and forage will not be limiting, and predators are not present) offtake could be as high as the estimated growth rate.
The conversion from livestock to wildlife ranching is largely mitigating the decline in habitat quality caused by overgrazing. However, a number of threats associated with wildlife ranching have been specifically identified for Blue Wildebeest. These include bush encroachment, vulnerability to pathogens, and declines in genetic diversity and integrity, as a result of hybridisation, genetic isolation and inbreeding. Livestock farmers in the North West Province specifically have voiced concerns about the presence of this species on game farms owing to the harmful diseases they harbour (Bothma 2002), and the possibility of disease transmission between wildlife and cattle.
Golden Wildebeest (a colour variant of the Blue Wildebeest) are actively bred on wildlife ranches, primarily for live sales. Although these animals are economically valuable for wildlife ranchers, they may impact negatively on the conservation of the species.
Additionally, a large number of hybridisation events have been recorded between Blue and Black Wildebeest as a result of the artificial confinement of these species within the same enclosures on private lands across the Free State, North West and KwaZulu-Natal provinces. The resulting movement restriction and lack of access to mates leads to species-specific reproductive isolation, and eventual hybridisation (Grobler et al. 2011). Although historically, the natural ranges of these species overlapped, no hybridisation events were documented, as the animals were not restricted by fences (Grobler et al. 2011). The hybrids are fertile and, if mated back to a pure-bred individual, are extremely difficult to identify (Patterson & Khosa 2005).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Biltong hunting. | Yes | Trophy hunting, biltong hunting & live animal sales. |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | true | – |
| 15. Sport hunting/specimen collecting | – | true | true | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Trophy hunting, biltong hunting & live animal sales.
Threats
Blue Wildebeest once occurred in substantially large concentrations, migrating extensively in response to rainfall. Following the compounding anthropogenic impacts, such as landscape transformation, the erection of fences, elimination of natural water sources, poaching and the spread of disease, the historic global distribution and abundance of Blue Wildebeest has shown substantial decline (East 1999; Estes & East 2009). However, since the 1970s and 1980s, the establishment and management of conservation areas, privately owned game farms, as well as national and provincial protected areas, lead to an increase in the financial value and abundance of large antelope species in South Africa (Estes & East 2009). Thus, there are currently no major threats affecting the survival of this species. However, a number of threats, related predominantly to small, fenced ranches and reserves have been identified, such as genetic isolation, inbreeding, possible crossbreeding with exotic subspecies, and hybridisation with Black Wildebeest (see Use and Trade). Poaching for bushmeat may also be a localised threat in some areas. For example, on Borakalalo National Park in North West Province, Blue Wildebeest are the most poached antelope (Nel 2015).
The erection of fences, which restricted the natural movements and migrations of Blue Wildebeest between wet and dry season ranges, in association with periodic drought conditions and the provision of artificial waterholes, lead to significant mass mortality events in southern Africa (Owen & Owen 1980; Walker et al. 1987; Knight 1995). During a drought period in the 1980s, tens of thousands of Blue Wildebeest perished as a result of starvation in the Kalahari, due to veterinary cordon fences preventing their movement into areas of more substantial water and forage availability (Spinage 1992; Knight 1995). The provision of artificial waterholes attracts herbivores to areas of unsustainable forage resources, leading to a reduction in habitat quality and an increase in the risk of predation (Knight 1995).
The possible emerging threat of disease (especially Bovine Tuberculosis and Bovine Malignant Catarrhal) is a concern across the country, and thus authorities should be mindful of this concern when issuing such permits. Studies have shown the first confirmed infection of Mycobacterium bovis in Mpumalanga, a finding that suggested that Bovine tuberculosis was more widespread than previously believed in this province (Hlokwe et al. 2014). The wildebeest was infected with a novel strain of M. bovis which indicates the introduction of a new M. bovis genotype in the Greater Kruger National Park Complex ecosystem, which could be the result of repeated translocations of Blue Wildebeest (Hlokwe et al. 2014). Consistent with previous wildebeest infections of M. bovis in the Serengeti ecosystem in Tanzania (Cleaveland et al. 2005), the wildebeest did not have visible lesions and the M. bovis was isolated from the lung tissue (Hlokwe et al. 2014).
Conservation
This species occurs widely across many protected areas, with the bulk of the formally protected population in the KNP. Hluhluwe-iMfolozi Park (HiP) also contains an important population with over 2,000 estimated to occur in the park (Cromsight et al 2017). North West Parks reserves (primarily Molopo and Madikwe Parks are estimated to account for approximately 3,000 individuals (North West data, 2021). No direct interventions are necessary. However, the loss of range, the erection of fences and the provision of artificial waterholes has resulted in the replacement of large migratory Blue Wildebeest herds with smaller resident populations within protected areas. Continued establishment of transfrontier conservation areas is thus key to the long-term prosperity of this subspecies. Landowners are encouraged to drop fences to form conservancies and create conservation corridors which will enable further migratory behaviour and establish resilient subpopulations within the assessment region. Landowners should also reduce the number of artificial water-points on their lands to improve habitat quality and encourage migratory behaviour.
Ranch managers should consider the importance of maintaining genetic diversity in isolated localities of Blue Wildebeest, especially on game farms running breeding programmes selecting for particular traits or colour variants, such as the Golden Wildebeest. Genetic variability plays a vital role in continued reproductive success and fitness, which becomes principally important during adverse environmental conditions. Translocations of individuals should be adequately controlled and stringently recorded so as to maintain genetically viable populations, and prevent hybridisation between Blue and Black Wildebeest, and among the five ESUs. Additionally, due to the threats associated with hybridisation, this species should not be enclosed on the same property as the closely related Black Wildebeest, and furthermore, due to the threat of fence breakouts, ideally these species should not be kept on neighbouring properties (Grobler et al. 2011). Suspected hybrid individuals should be isolated, so as to prevent continued hybridisation with pure individuals (Grobler et al. 2011).
Recommendations for land managers and practitioners:
- Harvest and trade management, including the regulation of translocation is required to prevent the spread of disease and hybridisation with Black Wildebeest.
- The species is favoured by Lion (Hayward & Kerley 2005) and should be augmented as prey on all the parks with inflated free range lion populations. To ensure the health of the Blue Wildebeest herds, one or two focal herds should be monitored in terms of herd size and proportion of juveniles during calving and at intervals following calving.
- Identification of colour variants through stud books and parentage verification.
Research priorities:
- Assess the proportion of the population hybridised with Black Wildebeest, and assess areas where both species occur, whether on private or other properties. The distribution map of Blue Wildebeest is suspected to be an underestimate and although there are sufficient records of the population in protected areas, it is suspected that the true numbers and distribution on private lands is largely unknown. Possibility of incidences of extralimital subspecies crossing, due to the translocation of exotic subspecies.
- The geographical extent of anthropogenic translocation of this species into regions beyond its natural range, for example, into the Western and Eastern Cape provinces.
- Assessment of colour variants (Golden Gnu, Copper Gnu, Red Gnu) and their impact on wild populations.
- Current population trends and distribution on private lands.
- Harvest and use of this species, and its value as a source of sustainable source of protein for local communities.
- Research towards eliminating Snotsiekte or Bovine Malignant Catarrhal Fever (a Herpes virus) in wildebeest to reduce the risk of conflict with domestic cattle and stock owners.
Encouraged citizen actions:
- Landowners should create conservancies for this species and engage local stakeholders to create sustainable, wildlife-based rural economies.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. Citizens should also report sightings of mixed Blue and Black Wildebeest subpopulations.
Bibliography
Ansell, W.F.H. 1972. Part 2, 15 Family Artiodactyla. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-84. Smithsonian Institution Press, Washington, DC, USA.
Bothma J du P, du Toit JT, van Rooyen J. 2010. Buying and selling wild animals. In: Bothma J du P (ed.), Game Ranch Management, pp. 619–639 . Van Schaik Publishers.
Bothma J du P. 2002. Game ranch management, 4th edition. Van Schaik Publishers, Pretoria, South Africa.
Burkepile DE, Burns CE, Tambling CJ, Amendola E, Buis GM, Govender N, Nelson V, Thompson DI, Zinn AD, Smith MD. 2013. Habitat selection by large herbivores in a southern African savanna: the relative roles of bottom-up and top-down forces. Ecosphere 4: 1-19.
Cleaveland S, Mlengeya T, Kazwala RR, Michel A, Kaare MT, Jones SL, Eblate E, Shirima GM, Packer C. 2005. Tuberculosis in Tanzanian wildlife. Journal of Wildlife Diseases 41: 446-453.
Cromsigt.J.P.M, S. Archubald & N. Owen-Smith. 2017. Conserving Africa’s Mega-Diversity in the Anthropocene. Cambridge University Press. 406 pp.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Estes, R. D. 2013. Connochaetes taurinus Common Wildebeest. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa, pp. 533-543. Bloomsbury Publishing, London, UK.
Estes, R. D. and East, R. 2009. Status of the wildebeest (Connochaetes taurinus) in the wild 1967-2005. Wildlife Conservation Society, New York.
Ferreira S, Gaylard A, Greaver C, Hayes J, Cowell C, Bissett C. 2016. Large vertebrate abundances in Parks: 2015/2016. Internal Report 14/2016, Scientific Services, SANParks, Skukuza, South Africa.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Gagnon M, Chew AE. 2000. Dietary preferences in extant African Bovidae. Journal of Mammalogy 81: 490-511.
Grobler JP, Rushworth I, Brink JS, Bloomer P, Kotze A, Reilly B, Vrahimis S. 2011. Management of hybridization in an endemic species: decision making in the face of imperfect information in the case of the Black Wildebeest—Connochaetes gnou. European Journal of Wildlife Research 57: 997-1006.
Grubb P. 2005. Artiodactyla. Pages 637-722. Mammal Species of the World. A Taxonomic and Geographic Reference. Johns Hopkins University Press, Baltimore, U.S.A.
Hayward MW, Henschel P, O’brien J, Hofmeyr M, Balme G, Kerley GIH. 2006a. Prey preferences of the leopard (Panthera pardus). Journal of Zoology 270: 298-313.
Hayward MW, Hofmeyr M, O’brien J, Kerley GIH. 2006b. Prey preferences of the cheetah (Acinonyx jubatus)(Felidae: Carnivora): morphological limitations or the need to capture rapidly consumable prey before kleptoparasites arrive? Journal of Zoology 270: 615-627.
Hayward MW, Kerley GI. 2005. Prey preferences of the lion (Panthera leo). Journal of Zoology 267: 309-322.
Hayward MW, O’Brien J, Hofmeyr M, Kerley GI. 2006c. Prey preferences of the African Wild Dog Lycaon pictus (Canidae: Carnivora): ecological requirements for conservation. Journal of Mammalogy 87: 1122-1131.
Hayward MW. 2006. Prey preferences of the Spotted Hyaena (Crocuta crocuta) and degree of dietary overlap with the Lion (Panthera leo). Journal of Zoology 270: 606-614.
Hlokwe TM, van Helden P, Michel AL. 2014. Evidence of increasing intra and inter-species transmission of Mycobacterium bovis in South Africa: Are we losing the battle? Preventive Veterinary Medicine 115: 10-17.
IUCN SSC Antelope Specialist Group. 2016. Connochaetes taurinus. The IUCN Red List of Threatened Species 2016: e.T5229A50185086. Available at: http://dx.doi.org/10.2305/IUCN.UK.2016- 2.RLTS.T5229A50185086.en..
Knight MH. 1995. Drought-related mortality of wildlife in the southern Kalahari and the role of man. African Journal of Ecology 33: 377-394.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Mills MGL, Biggs HC, Whyte IJ. 1995. The relationship between rainfall, lion predation and population trends in African herbivores. Wildlife Research 22: 75-87.
Nel, P. 2015. Population estimates for large herbivores and predators in protected areas in the North West Parks Board November 2015. North West Parks Board.
Owens, M. and Owens, D. 1980. Fences of death. Wildlife 214: 214-217.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Patterson C, Khosa P. 2005. Background Research Paper: A status quo on the professional and recreational hunting industry in South Africa. Report prepared for the panel of experts appointed by the Minister of Environmental Affairs and Tourism. TRAFFIC – The wildlife trade monitoring network.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Seydack AH, Grant CC, Smit IP, Vermeulen WJ, Baard J, Zambatis N. 2012. Large herbivore population performance and climate in a South African semi-arid savanna. Koedoe 54: 1-20.
Shrestha AK, van Wieren SE, van Langevelde F, Fuller A, Hetem RS, Meyer L, de Bie S, Prins HHT. 2014. Larger antelopes are sensitive to heat stress throughout all seasons but smaller antelopes only during summer in an African semi-arid environment. International Journal of Biometeorology 58: 41-49.
Shrestha AK, van Wieren SE, van Langevelde F, Fuller A, Hetem RS, Meyer LCR, de Bie S, Prins HHT. 2012. Body temperature variation of South African antelopes in two climatically contrasting environments. Journal of Thermal Biology 37: 171-178.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Smuts GL. 1978. Interrelations between predators, prey, and their environment. BioScience 28: 316-320.
Spinage CA. 1992. The decline of the Kalahari wildebeest. Oryx 26: 147-150.
Walker BH, Emslie RH, Owen-Smith RN, Scholes RJ. 1987. To cull or not to cull: lessons from a southern African drought. Journal of Applied Ecology 24: 381-401.
Yoganand K, Owen-Smith N. 2014. Restricted habitat use by an African savanna herbivore through the seasonal cycle: key resources concept expanded. Ecography 37: 969-982.