Blue Duiker
Philantomba monticola

2025 Red list status
Vulnerable
Regional Population Trend
Declining
change compared
to 2016
No Change
Overview
Philantomba monticola – (Thunberg, 1789)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Philantomba monticola
Common Names: Blue Duiker (English), Blou Duiker (Afrikaans), Ipunzi Ehlaza (Ndebele), Phuti (Sepedi), Phuthi (Sesotho), Photi (Setswana), Imphunzi (Swati), Mhunti (Tsonga), Iphuti (Xhosa), Iphiti (Zulu)
Synonyms: Cephalophus monticola (Thunberg, 1789)
Taxonomic Note: The recognition of Philantomba as a separate genus to Sylvicapra and Cephalophus is contentious, but recent molecular evidence supports the genus as a basal clade (van Vuuren & Robinson 2001; Johnston & Anthony 2012). Thirteen subspecies have been named (Hart & Kingdon 2013), where P. m. monticola is isolated from the others, occurring from northern KwaZulu-Natal (KZN) to the Eastern and Western Cape Provinces of South Africa.
| Red List Status |
|
VU – Vulnerable C2a(i) (IUCN version 3.1) |
Assessment Information
Assessors: Landman, M.1, Ehlers-Smith, Y.2 & da Silva, J.3
Reviewer: Shrader, AM.4
Contributor: Patel, T.5
Institutions: 1Nelson Mandela University, South Africa, 2Ezemvelo KZN Wildlife, 3South African National Biodiversity Institute, 4University of Pretoria, South Africa, 5Endangered Wildlife Trust
Acknowledgements: Eastern Cape Parks and Tourism Agency and Ezemvelo KZN Wildlife provided records of occurrence for Blue Duiker.
Previous Assessors & Reviewers: Venter, J., Seydack, A., Ehlers-Smith, Y., Uys, R. & Child, M.F.
Previous Contributors: Jones, A., Roxburgh, L., Page-Nicholson, S., Selier, J. & Novellie, P.
Assessment Rationale
This sub-Saharan African species has a disjunct distribution between the eastern coastal forests of South Africa and the rest of its range. Within the assessment region, the species is inferred to be declining due to forest habitat loss from ongoing development along the coastal belt, illegal sand mining, and indigenous timber extraction. Increasing bushmeat poaching and hunting with domestic dogs are also suspected to be directly causing a decline in the number of mature individuals. Preliminary data indicate that around half the subpopulations on protected areas and private lands are declining or have unknown trends (see Population). The estimated area of occupancy (AOO) is 2,823 km2, using all forest patches within the extent of occurrence (EOO). Population estimates range widely: using a density range of 5–35 individuals / km2 yields a total mature population estimate of 7,063–49,403 individuals (using a 50% mature population structure). Blue Duiker are estimated to be unable to disperse further than 0.88 km between forest patches (Lawes et al. 2000). Using forest clusters that fall within this dispersal distance as proxies for subpopulations, the largest cluster was estimated to be 314–687 km2, which yields 785–12,023 mature individuals. Under a precautionary purview, we suspect the lower estimates are more realistic given the wide variation in density and occupancy between patches on fine spatial and temporal scale, combined with multiple ongoing threats that may be causing local subpopulation decline or extinction.
Thus, we list Blue Duiker as Vulnerable C2a(i) using the lower estimates of mature population size. Further surveys, density estimates and occupancy levels across its range are necessary to more accurately calculate key parameters. This species should be reassessed when such data are available. Key interventions include effective management of the inter-patch matrix by minimising poaching rates; enforcement of legislation prohibiting illegal sand mining, development and timber harvesting; and coastal forest conservation and restoration through biodiversity stewardship schemes. As such, this species remains conservation dependent.
Regional population effects: There are no confirmed records from southern Mozambique, which suggests a gap in distribution between South Africa and the rest of its range. Hence, there is no rescue effect possible. Unlike the central African scenario, Blue Duiker in the assessment region occur in relatively small patches of suitable habitat within a forest/non-forest mosaic, which makes recolonisation of locally depleted patches difficult.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Landman M, Ehlers-Smith Y & da Silva JM. 2025. A conservation assessment of Philantomba monticola. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
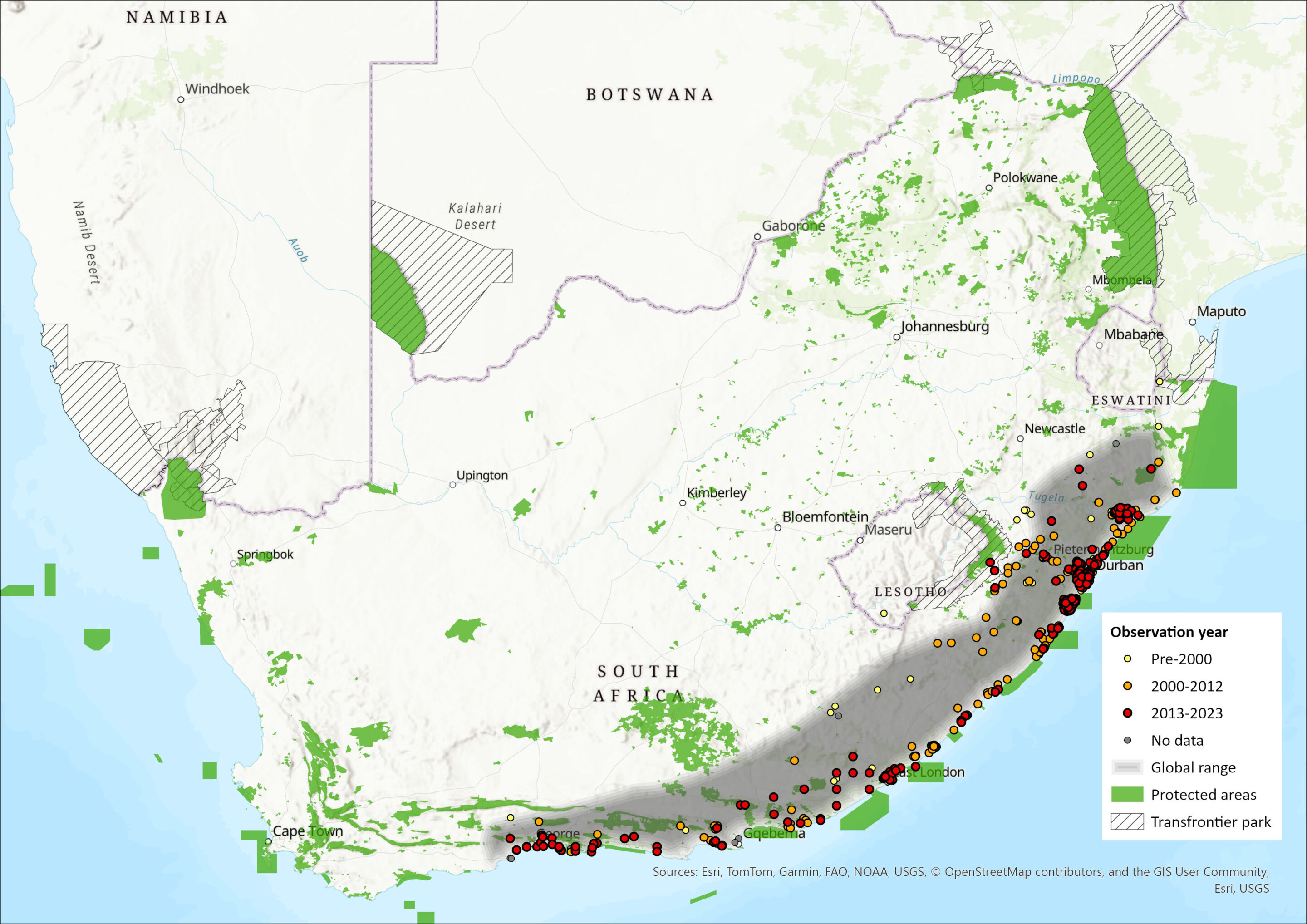
This species is thought to occur in forested and densely wooded areas throughout central, eastern, and southern Africa. Within southern Africa, it occurs in eastern Zimbabwe, central Mozambique, Eswatini, and along the eastern seaboard of South Africa. In South Africa, it is primarily confined to evergreen coastal and scarp forests and thickets from northern KZN southwards to the eastern Western Cape Province. Additionally, the species is suspected to have been introduced into captive-breeding systems across the country, with confirmed presence in the North-West Province at least (Power 2014). There are no contemporary confirmed records from southern Mozambique, which suggests a discontinuous distribution between South Africa, eastern Zimbabwe and central Mozambique. As a result, the South African population is considered isolated.
The estimated EOO is 269,584 km2, which is unchanged from the previous assessment (Venter et al. 2016). Although previous assessments have asserted that the species exists inland in montane forest (Friedmann & Daly 2004; IUCN SSC Antelope Specialist Group 2008), there are no confirmed records for Afromontane forests along the foothills of the Drakensberg Mountains. Thus, this area is excluded from the AOO, estimated to be a maximum of 2,823 km2, which includes all remaining forest patches within the EOO as of 2018. Furthermore, within the remaining forest patches, the species is patchily distributed. Lawes et al. (2000) found that only 18% of patches of Balgowan and Karkloof forests in KZN were occupied. Blue Duiker were absent from forest patches smaller than 0.045 km2 or located further than 0.88 km from mainland patches. The probability of patch occupancy was greater than 50% when patches exceeded 0.05 km2 and were within 0.25 km of a mainland forest (Lawes et al. 2000). There are no current records of the species in the Kosi Bay area (S. Kyle pers. comm. 2015). Similarly, while the species was once described as “numerous” in Hluhluwe-iMfolozi and Mkhuze areas (Province of Natal 1935; Bourquin et al. 1971), they currently appear to be absent from these areas (Ezemvelo KZN Wildlife unpubl. data).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): Sea level
Elevation Upper Limit (in metres above sea level): 1200 m but extralimital
Depth Lower Limit (in metres below sea level): NA
Depth Upper Limit (in metres below sea level): NA
Depth Zone: NA
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Blue Duiker (Philantomba monticola) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
The long-term decline of at least one Blue Duiker population – the largest population in the assessment region, located in the Knysna and Tsitsikamma forests – has been linked to decreasing habitat quality, potentially associated with rising ambient temperatures driven by climate change (Seydack et al. 1998). Colyn et al. (2020) examined altitudinal patterns in forest contraction for forest specialist bird species. However, many of their findings are pertinent to the effects of climate change on habitat persistence for Blue Duiker. Their results show that mid- to high altitude Mistbelt and Afrotemperate forests (800-1800 m) are predicted to experience severe contractions (accounting for 79.9% of all contracted habitat), whereas coastal lowland and scarp forests had contraction rates in only 8.6% of core habitats. Their model also showed a moderate expansion (305 ha) in Afromontane forest above 2100m and larger expansion (1961 ha) in low altitude (0-200 m) coastal forests. These are similar to the results of Ehlers Smith, Y (unpublished data) that indicate a coastal shift in habitat suitability for Blue Duiker. However, habitat suitability alone does not ensure persistence. On-going land-use change along the coast will likely be the most important factor determining the future of these lowland forests.
Population information
Blue Duiker have been recorded in at least two national parks (Addo Elephant and Garden Route) and 38 provincial nature reserves across the Western Cape, Eastern Cape, and KZN Provinces. However, it is a rare and secretive species, which makes population counts difficult. Additionally, density estimates can vary considerably depending on the survey method. For example, in Kenneth Stainbank Nature Reserve, strip transects estimated densities of 1.44-1.55 individuals/km², whereas dung heap counts and territory mapping suggested much higher densities of 2.38 and 2.70 individuals/km², respectively (Bowland 1990, Bowland & Perrin 1995). Consequently, population estimates for the species within the assessment region vary widely, ranging from 7,063–49,403 mature individuals, with a potential effective population size between 706-14,821 individuals. Field surveys are urgently needed to estimate subpopulation sizes across habitat types and land uses within the species’ range.
Blue Duiker maintain very small territories (approximately 0.6 ha in coastal forests; Bowland and Perrin 1995) and live in small family groups typically comprising one adult male, one adult female, one subadult, and one infant/juvenile. There is a large overlap in offspring due to relatively short lambing intervals (approximately 265 days), and females have a gestation period of 207 days (range = 196–216; Bowland 1990; Skinner & Chimimba 2005; Pacifici et al. 2013). This reproductive strategy means that relatively large numbers can persist in suitable habitats where threats (e.g., illegal harvesting) are low (e.g., Seydack et al. 1998).
Throughout its continental range, Blue Duiker densities range between 5–35 individuals/km² (references within Hart & Kingdon 2013). Within the assessment region, which lies at the extreme southern limit of their distribution, densities are comparable: 9–55 individuals/km² in the Knysna Forest and 13–19 individuals/km² in the Tsitsikamma Forest (Hanekom & Wilson 1991). However, population densities in the Knysna Forest declined sharply between 1970 and 1980, and a similar decline occurred in the Tsitsikamma Forest after 1992 (Seydack et al. 1998). Before these declines, Blue Duiker were found at relatively high densities in moister forests, whereas, after the decline, population persistence was associated with drier forests (Seydack 1984; Seydack et al. 1998). The decline is not thought to be caused by habitat loss or fragmentation. Instead, nocturnal warming – affecting the digestible non-structural carbon content of forage – has been implicated (A. Seydack et al. unpubl. data), but this requires further testing. These density estimates are now severely outdated, and it is not known whether these populations have stabilised at the lower densities. Nevertheless, they remain the best available information for what is likely the largest population in the assessment region.
The highest Blue Duiker densities were recorded by Bowland (1990) and Bowland and Perrin (1995) in KZN coastal forests, ranging between 90–320 individuals/km². The lower estimate is based on four survey methods with multiple repeats over several years, whereas the upper limit is based on two survey methods that were not repeated (Bowland 1990). While the upper values may be outliers, considerable variability in Blue Duiker density and occupancy have also been observed between and within habitat patches over time (Bowland 1990). For example, Sosibo et al. (2022, 2023) recorded the species at only 13 out of 169 camera trap sites in Mist-belt forests of KwaZulu-Natal and Eastern Cape. Moreover, site occupancy in mist-belt forests (0.07 – 0.17) was markedly lower than in coastal forests (0.45 – 0.85; Ehlers Smith et al, 2018).
Subpopulations also exist in suitable habitat outside of protected areas, including on private land and within residential areas. Most of these populations are wild and free-roaming and therefore count towards this assessment, though numbers are unknown. A survey of 63 conservancies and private landowners in KZN showed that 47% confirmed the presence of Blue Duiker. However, 53% suspected that the subpopulation trend was either unknown or decreasing over the five-year study period (Jones 2015). Similarly, among 92 protected areas within the species’ range, only 36% confirmed Blue Duiker presence, and 42% reported an unknown or decreasing subpopulation trend (Jones 2015). This is supported by long-term game census data in KZN protected areas, which show declining subpopulations from the 1980/1990s to the present (Ezemvelo KZN Wildlife unpubl. data). These findings suggest continuing decline in mature individuals, likely driven by ongoing habitat loss and fragmentation. Elsewhere, numbers may be increasing, particularly where initiatives have been implemented to limit illegal harvesting activities (e.g., C. Hoskins, Crags View Rehabilitation Centre, pers. comm. 2015).
Throughout the continent, Blue Duiker is thought to be declining due to bushmeat hunting on both a subsistence and commercial scale (IUCN SSC Antelope Specialist Group 2016). Additionally, Blue Duiker are sensitive to land-use changes (e.g., Lawes et al. 2000). Although they can move through some transformed lands (e.g., plantations; Lawes et al. 2000) and well-wooded residential areas, these habitats should be considered low quality dispersal routes. Limited connectivity between suitable habitat patches or large isolation distances results in small, fragmented subpopulations that are unlikely to be genetically viable in the long-term. Consequently, many subpopulations are unlikely to contribute to the regional genetic diversity necessary for persistence of the species.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | Estimated | Snaring and dog-hunting leading to loss of individuals. |
Current population trend: Declining. Both in situ and from ongoing habitat loss and degradation.
Number of mature individuals in population: 7,063–49,403 mature individuals
Number of subpopulations: Unknown, but possibly as many as 51 (using occupied forest clusters as a proxy).
Severely fragmented: Yes. Fragmented forest habitats throughout the Eastern Cape and KZN provinces. Also occurs in coastal and dune thickets which are separated by coastal developments.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: 12,023 mature individuals
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Phylogenetics studies examining relationships within the Cephalophinae report the genus Philantomba to be separate from Sylvicapra and Cephalophus (van Vuuren & Robinson 2001; Johnston & Anthony 2012). However, no examination of the population genetic structure and diversity of Philantomba has been undertaken.
Population estimates for the species within the assessment region vary widely, ranging from 7,063–49,403 mature individuals. Assuming a single population and a Ne/Nc ratio of 0.1-0.3, this translates to a potential effective population size between 706-14821 individuals, which would suggest a genetically healthy population.
However, since the species is thought to occur as multiple subpopulations,it is highly likely that some subpopulations would fall below the Ne 500 threshold. A comprehensive genetic assessment is, therefore, needed to determine the extent of genetic connectivity and effective population size across the species’ range.
Based on its current distribution and occurrence inside and outside of protected areas, Blue Duiker dispersal between forest patches is considered unlikely when distances exceed 800 m (Lawes et al. 2000), as the areas between patches may be inhospitable. However, dispersal may still be possible where riverine vegetation provides corridors that facilitate movement. These patterns require confirmation through population genetic or genomic analyses.
Habitats and ecology
Blue Duiker occur across a wide range of forested habitats (both primary and secondary) and dense woodlands, from sea level up to 3000 m (Skinner & Chimimba 2005; Hart & Kingdon 2013). They commonly forage in forest gaps and open areas but rely on dense underbrush for resting and cover. Although Blue duiker can also be found in human-modified habitats, including near urban edges, the extent to which they are able to persist in these environments remains unclear. Surface water availability does not appear to be a critical habitat requirement.
They are selective foragers, consuming a diversity of fruits and seeds, browse, flowers, fungi, gum, and forest litter (Bowland 1990; Hanekom & Wilson 1990; Fauri & Perrin 1993; Gagnon & Chew 2000). Their diet also includes animal matter such as bird eggs and insects, and they have been observed actively collecting ants from the ground using their tongues. Despite their small body size, Blue duiker appear to be capable of subsisting on lower quality diets than would typically be expected for herbivores of similar size (Shipley & Felicetti 2002).
They are generally active at dawn and dusk (Bowland & Perrin 1995), although nocturnal activity has been observed seasonally and in areas with high levels of disturbance (e.g., Ehlers Smith et al., 2019). Breeding occurs year-round, although birth rates tend to decline during the dry season (Hart & Kingdon 2013). A single offspring is born, and individuals often form stable pair bonds or small social groups (Dubost 1984; Bowland & Perrin 1995).
Blue Duiker populations are vulnerable to local extinction due to habitat loss and fragmentation (Lawes et al. 2000), overharvesting (Wilson 2001; Lindsey et al. 2013; SANBI 2017), and potentially interspecific competition with other ungulates, including Bushbuck Tragelaphus sylvaticus and Nyala Tragelaphus angasii (Bowland 1990; Hart & Kingdon 2013; Ehlers Smith et. al. 2020). Typical predators include the African Rock Python Python sebae, Crowned Eagle Stephanoaetus coronatus, Leopard Panthera pardus, and caracal Caracal caracal (Hanekom & Wilson 1991; Boshoff et al. 1994; Vernon 2001; Braczkowski et al. 2012; Reeves & Boshoff 2016).
Ecosystem and cultural services: As frugivores, Blue Duiker play an important role in seed dispersal, likely contributing to forest regeneration and ecosystem functioning (Brodie et al. 2009; Abernethy et al. 2013). Through their browsing of seedlings, they may also influence patterns of forest succession. In addition to their ecological roles, Blue duiker comprise a significant proportion of forest carnivore diets (e.g., Hanekom & Wilson 1991; Braczkowski et al. 2012), and they are an important source of bushmeat for many rural communities (Abernethy et al. 2013; Lindsey et al. 2013).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 14.6. Artificial/Terrestrial -> Artificial/Terrestrial – Subtropical/Tropical Heavily Degraded Former Forest | – | Marginal | – |
Life History
Generation length: 4.9 years
Age at maturity: female or unspecified: 9–12 months
Age at Maturity: Male: 12–18 months
Size at Maturity (in cms): Female, Mean total length: 70 cm; Mean shoulder height: 35 cm
Size at Maturity (in cms): Male, Mean total length: 72 cm; Mean shoulder height: 34 cm
Longevity: 10–12 years
Average Reproductive Age: (Not specified)
Maximum Size (in kgs): Males: 4.5 kg; Females: 5.4 kg
Size at Birth (in kg): 590– 950 g
Gestation Time: 207 days
Reproductive Periodicity: Year round
Average Annual Fecundity or Litter Size: Single fawn
Natural Mortality: Hunted by African rock python, Crowned eagle, Leopard.
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Non-migrant
Congregatory: Monogamous pairs
Systems
System: Terrestrial
General Use and Trade Information
Blue Duiker are hunted across their range for trophies, bushmeat, and other products, and are considered one of the most economically important wild ungulates in Africa (Wilson 2001). The bushmeat trade in Africa is expanding and, in many regions, has shifted from subsistence hunting to a commercial enterprise (Robinson & Bennett 2004; Lindsey et al. 2013). For example, nearly half of the 32 traders at the Faraday market in Johannesburg reported selling duiker products such as horn and skin, although the specific species were not identified (Whiting et al. 2011). More recently, Sosibo et al. (2022) conducted interviews around Mistbelt forests in the Eastern Cape and KwaZulu Natal and found that 15 out of 303 respondents reported hunting Blue duiker, whereas 124 had hunted bushbuck. These results likely reflect local availability, with bushbuck being the more abundant ungulate species in the region.
Illegal harvesting of Blue Duiker is likely widespread across the species’ range, with varying impacts on populations. For example, taxi-hunting — organised hunting using dogs (sensu Grey-Ross et al. 2010) — has been observed in two major reserves in southeastern KwaZulu Natal, where duiker densities are considerably lower, possibly as a result of this hunting pressure (Y. Ehlers-Smith unpublished data). Conversely, in areas where hunting with dogs is effectively managed through anti-poaching measures, Blue Duiker display 100% occupancy. In the absence of dog racing, taxi-hunting may expand as a commercial enterprise in the region and a source of income. When poachers are caught while hunting with dogs in parts of the Eastern Cape and KwaZulu Natal (such as the Umzimkhulu Valley), they are often found in possession of Blue duiker. The species is also frequently caught in snares intended for bushpig (D. de Villiers pers. comm. 2016).
According to the Non-Detrimental Findings (NDF) for Blue Duiker, the current legal trade in live animals, both domestically and internationally, as well as the export of hunting trophies, presents a moderate to high risk to the species’ survival in South Africa (SANBI 2017). This largely stems from insufficient monitoring of wild populations and levels of harvesting. Legal hunting of the species is not permitted without an approved adaptive harvest management model.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat hunting which is prevalent in the former Transkei and Ciskei regions as well as certain regions in Kwazulu-Natal. | Yes | International and national trophy hunting. |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 15. Sport hunting/specimen collecting | – | true | true | – |
Is there harvest from captive/cultivated sources of this species? Not known
Harvest Trend Comments: No extensive systems for Blue Duiker production are known. Anecdotal evidence suggests extensive captive-bred populations across the country.
Threats
Within the assessment region, the primary threat to Blue Duiker is habitat loss and fragmentation. This is driven by expanding agriculture (e.g., macadamia nut and sugar cane farming, and plantation forestry), unsustainable harvesting of indigenous timber, illegal sand mining (e.g., Masalu 2002), and the spread of alien invasive plant species. Additionally, expanding human development and urban sprawl is contributing to the loss of remaining habitat fragments through housing and infrastructure development. However, Blue Duiker can exist on mixed land-use areas. For example, forest patches surrounded by exotic timber plantations seem to have less of a negative effect on Blue Duikers than other forest-dwelling species (e.g., Tree Hyraxes Dendrohyrax arboreus (Lawes et al. 2000; Sosibo et al. 2022).
Blue Duiker are widely hunted (legally, illegally) and traded, which poses an ongoing threat to the species (SANBI 2017). They are particularly vulnerable to snaring and hunting with dogs (whether for subsistence or sport), which can lead to local population declines and extirpations (Jones 2015; Y. Ehlers-Smith unpublished data). Their use of well-defined paths between bedding and feeding sites makes them especially susceptible to snaring (Skinner & Chimimba 2005), with individuals often found dead or dying in snares. However, where snaring is effectively managed (e.g., through continuous snare removal), local populations have shown signs of recovery (C. Hoskins, Crags View Rehabilitation Centre, pers. comm. 2015).
Natural predation (e.g., by leopard, caracal) is rarely considered a threat to Blue Duiker populations (Hanekom & Wilson 1991; Jones 2015). However, more recently, Ehlers Smith et al. (2019) found that both caracal and black-backed jackal (Canis mesomelas) numbers have been increasing along the KwaZulu Natal coastal belt, which has been associated with reduced Blue Duiker occupancy. Similarly, increasing caracal numbers in Krantzkloof Nature Reserve area have coincided with the absence of blue duiker sightings (W. Bekker, Ezemvelo KZN Wildlife pers. comm.).
Conservation
Within the assessment region, Blue Duiker occurs across several state- and privately-owned protected areas. Outside of protected areas, they are generally resilient to moderate anthropogenic disturbance and can occur in small (few hectares) forest and woodland patches, provided that threats in the surrounding matrix habitats are low, thereby facilitating movement between patches (Lawes et al. 2000). While commercial plantations may function as temporary movement corridors, they are unlikely to support the persistence of Blue duiker (Lawes et al. 2000; Y. Ehlers-Smith unpublished data). Consequently, alternative strategies to enhance landscape connectivity outside of protected areas are recommended. Biodiversity-orientated land-uses and biodiversity stewardship agreements could play a crucial role in protecting movement corridors for blue duiker. Additionally, reducing the threats of illegal hunting (e.g., by enforcing hunting regulations and implementing effective anti-poaching measures) will further improve connectivity between habitat patches.
Legal hunting of the species should not be permitted without an approved adaptive harvest management model (SANBI 2017) and clearly defined thresholds should be established to guide harvest management in all areas where Blue duiker are hunted (SANBI 2017).
Education and awareness campaigns targeting communities adjacent to key habitat patches are essential to raise awareness of the species’ conservation needs and reduce sport hunting and snaring. These initiatives should be supported by developing alternative livelihoods to address the socioeconomic drivers of poaching.
Stronger enforcement by government agencies is urgently needed to address illegal land development, sand mining, and indigenous timber harvesting.
Recommendations for land managers:
- Develop and implement a long-term monitoring system for Blue Duiker, with appropriate feedback to the relevant managing authority. Annual sampling of abundances is required and the impacts of threats should be identified and documented.
- Develop an adaptive harvest management model to guide trophy hunting, with quotas based on local subpopulation sizes and recruitment rates.
- Landowners are encouraged to remove fences to enhance landscape connectivity and facilitate Blue duiker movements. Biodiversity-orientated land-uses and biodiversity stewardship agreements could play a crucial role in protecting these movement corridors.
- Register captive breeding programs and animals in captivity.
Research priorities:
- A comprehensive genetic assessment is needed to determine genetic diversity, extent of genetic connectivity, and effective population size across the species’ range.
- Population surveys are required to refine estimates of distribution and abundance, thereby enabling population and habitat viability analyses.
- Quantifying threats, specifically the impacts of habitat loss, transformation, and both legal and illegal harvesting, on population dynamics.
Encouraged citizen actions:
- Report Blue Duiker sightings to virtual museum platforms (e.g., iNaturalist), especially outside protected areas.
Bibliography
Abernethy, K.A., Coad, L,. Taylor, G., Lee, M.E. and Maisels, F. 2013. Extent and ecological consequences of hunting in Central African rainforests in the twenty-first century. Philosophical Transactions of the Royal Society of London B: Biological Sciences 368: 20120303.
Boshoff AF, Palmer NG, Vernon CJ, Avery G. 1994. Comparison of the diet of crowned eagles in the Savanna and Forest Biomes of south-eastern South Africa. South African Journal of Wildlife Research 24: 26–31.
Bourquin O, Vincent J, Hitchins PM. 1971. The vertebrates of the Hluhluwe Game Reserve – Corridor (State-Land) – Umfolozi Game Reserve Complex. The Lammergeyer 14: 5-58.
Bowland AE, Perrin MR. 1995. Temporal and spatial patterns in blue duikers Philatomba monticola and red duikers Cephalophus natalensis. Journal of Zoology 237: 487-498.
Bowland AE, Perrin MR. 1994. Density estimate methods for blue duikers Philantomba monticola and red duikers Cephalophus natalensis in Natal, South Africa. Journal of African Zoology 108: 505 –519
Bowland, A.E. 1990. The ecology and conservation of blue duiker and red duiker in Natal. Ph.D. Thesis, University of Natal.
Braczkowski A, Watson L, Coulson D, Randall R. 2012. Diet of leopards in the southern Cape, South Africa. African Journal of Ecology 50: 377-380.
Brodie JF, Helmy OE, Brockelman WY, Maron JL. 2009. Bushmeat poaching reduces the seed dispersal and population growth rate of a mammal-dispersed tree. Ecological Applications 19: 854-863.
Colyn, R.B., Ehlers Smith, D.A., Ehlers Smith, Y.C., Smit‐Robinson, H. and Downs, C.T., 2020. Predicted distributions of avian specialists: A framework for conservation of endangered forests under future climates. Diversity and Distributions, 26(6), pp.652-667.
Dubost G. 1984. Comparison of the diets of frugivorous forest mammals of Gabon. Journal of Mammalogy 65: 298–316.
Ehlers Smith, Y.C., Ehlers Smith, D.A., Ramesh, T. et al. Forest habitats in a mixed urban-agriculture mosaic landscape: patterns of mammal occupancy. Landscape Ecol 33, 59–76 (2018). https://doi.org/10.1007/s10980-017-0580-1https://doi.org/10.1007/s10980-017-0580-1
Ehlers Smith, Y.C., Ehlers Smith, D.A., Ramesh, T. & Downs, C.T. 2020. Co‐occurrence modelling highlights conservation implications for two competing spiral‐horned antelope. Austral Ecology, 45(3), pp.305-318.
Faurie AS, Perrin MR. 1993. Diet selection and utilization in blue duikers (Cephalophus monticola) and red duikers (Cephalophus natalensis). Journal of African Zoology 107: 287–299.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Gagnon M, Chew AE. 2000. Dietary preferences in extant African Bovidae. Journal of Mammalogy 81: 490-511.
Grey-Ross R, Downs CT, Kirkman K. 2010. An assessment of illegal hunting on farmland in KwaZulu-Natal, South Africa: implications for oribi (Ourebia ourebi) conservation. South African Journal of Wildlife Research 40: 43-52.
Hanekom N, Wilson V. 1991. Blue duiker Philantomba monticola densities in the Tsitsikamma National Park and probable factors limiting these populations. Koedoe 34: 107-120.
Hart, J.A. and Kingdon, J. 2013. Philantomba monticola Blue Duiker. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. Volume VI: Pigs, Hippopotamuses, Chevrotain, Giraffes, Deer and Bovids, pp. 228-234. Bloomsbury Publishing, London.
IUCN SSC Antelope Specialist Group. 2008. Philantomba monticola. The IUCN Red List of Threatened Species 2008: e.T4143A10469452.
IUCN SSC Antelope Specialist Group. 2016. Philantomba monticola. The IUCN Red List of Threatened Species 2016: e.T4143A50183103.
Johnston AR, Anthony NM. 2012. A multi-locus species phylogeny of African forest duikers in the subfamily Cephalophinae: evidence for a recent radiation in the Pleistocene. BMC Evolutionary Biology 12: 120.
Jones AL. 2015. The potential habitat space of caracal and blue duiker in KwaZulu-Natal. M.Sc. Thesis. University of KwaZulu-Natal, Pietermaritzburg, South Africa.
Lawes, M.J., Meakin, P.E. and Piper, S.E. 2000. Patch occupancy and potential metapopulation dynamics of three forest mammals in fragmented Afromontane forest in South Africa. Conservation Biology 14: 1088-1098.
Lindsey, P.A., Balme, G., Becker, M., Begg, C., Bento, C., Bocchino, C., Dickman, A., Diggle, R.W., Eves, H., Henschel, P., Lewis, D., Marnewick, K., Mattheus, J., McNutt, J.W., McRobb, R., Midlane, N., Milanzi, J., Morley, R., Murphree, M., Opyene, V., Phadima, J., Purchase, G., Rentsch, D., Roche, C., Shaw, J., Van der Westhuizen, H.,Van Vliet, N. and Zisadza-Gandiwa, P. 2013. The bushmeat trade in African savannas: Impacts, drivers, and possible solutions. Biological Conservation 160: 80-96.
Masalu DCP. 2002. Coastal erosion and its social and environmental aspects in Tanzania: a case study in illegal sand mining. Coastal Management 30: 347-359.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Province of Natal. 1935. Report of the Game Reserves Commission 1935. The Natal Witness. Ltd, Pietermaritzburg, South Africa.
Reeves B, Boshoff AF. 2016. Is diet adaptability a reason for the persistence of African Crowned Eagles Stephanoaetus coronatus in altered habitats? Ostrich 87: 29-36.
Robinson JG, Bennett EL. 2004. Having your wildlife and eating it too: an analysis of hunting sustainability across tropical ecosystems. Animal Conservation 7: 397-408.
SANBI 2017. Non-detriment findings made by the scientific authority – summary report.
Seydack AH, Huisamen J, Kok R. 1998. Long-term antelope population monitoring in Southern Cape Forests. Southern African Forestry Journal 182: 9-19.
Seydack AHW. 1984. Application of a photo-recording device in the census of larger rain-forest mammals. South African Journal of Wildlife Research 14: 10-14.
Shipley L, Felicetti L. 2002. Fiber digestibility and nitrogen requirements of blue duikers (Cephalophus monticola). Zoo Biology 21: 123-134.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Sosibo, M.T., Ehlers Smith, D.A., Ehlers Smith, Y.C. et al. Mammalian assemblages in Southern Mistbelt Forests of the northern Eastern Cape, and southern KwaZulu-Natal Provinces, South Africa, and their response to bordering land-use. Mamm Biol 102, 429–440 (2022). https://doi.org/10.1007/s42991-022-00243-5https://doi.org/10.1007/s42991-022-00243-5
Sosibo, M.T., Ehlers Smith, D.A., Ehlers Smith, Y.C., Gumede, S.T., Ngcobo, S.P. and Downs, C.T., 2023. Influence of microhabitat structure on large‐and medium‐sized mammals in South African forests. African Journal of Ecology, 61(3), pp.617-627.
Whiting, M.J., Williams, V.L. and Hibbitts, T.J. 2011. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Journal of Zoology 284: 84-96.
Wilson, V.J. 2001. Duikers of Africa: Masters of the African Forest Floor. Directory Publishers, Bulawayo, Zimbabwe.
van Vuuren BJ, Robinson TJ. 2001. Retrieval of four adaptive lineages in duiker antelope: evidence from mitochondrial DNA sequences and fluorescence in situ hybridization. Molecular Phylogenetics and Evolution 20: 409-425.
Venter J, Seydack A, Ehlers-Smith Y, Uys R, Child MF. 2016. A conservation assessment of Philantomba monticola. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Vernon C. 2001. Crowned eagles and blue duikers. In VJ Wilson, editor. Duikers of Africa: Masters of the African Forest Floor (eds.). Chipangali Wildlife Trust, Zimbabwe, pp. 266–274.