Blainville’s Beaked Whale
Mesoplodon densirostris

2025 Red list status
Data Deficient
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Mesoplodon densirostris – (Blainville, 1817)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – ZIPHIIDAE – Mesoplodon – densirostris
Common Names: Blainville’s Beaked Whale, Atlantic Beaked Whale, Dense Beaked Whale (English), Blainville se Snoetwalvis (Afrikaans), Ballena de Pico de Blainville, Zifio de Blainville (Spanish; Castilian), Blainvilles næbhval (Danish), Blainvilles spisshval (Norwegian), Kobuha kujira (Japanese), Króksnjáldri (Icelandic), Mésoplodon de Blainville (French)
Synonyms: Delphinus densirostris Blainville, 1817
Taxonomic Note:
Blainville’s Beaked Whales (Mesoplodon densirostris) is one of 15 species of Mesoplodon with a worldwide range in tropical and warm-temperate waters. No subspecies are currently recognised.
Red List Status: Data deficient
Assessment Information
Assessors: James, B.S.1 & da Silva, J.M.2
Reviewers: Patel, T.3 & Purdon, J.4,5
Institutions: 1University of Cape Town, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust, 4TUT Nature Conservation, 5University of Pretoria
Previous Assessors & Reviewers: Relton, C., Cockcroft, V. & Hofmeyr, G.J.G.
Previous Contributors: Elwen, S., Findlay, K., Meÿer, M., Oosthuizen, H., Plön, S. & Child, M.F.
Assessment Rationale
There is no information pertaining to the population abundance of beaked whales within the assessment region, and they are generally considered to be naturally rare. The main current threats for this species are climate change, whaling, military sonar, vessel noise, entanglement, depredation, persistent organic pollutants, toxic metals, plastics and oil spills. In addition, marine noise pollution, usually in the form of seismic surveys, navy operations and marine construction have been identified as emerging and escalating threats to beaked whales. Anecdotal evidence suggests that beaked whales are more vulnerable to marine noise (particularly mid-frequency active sonar) than other cetaceans. The compounding influences of these threats could potentially cause beaked whale population declines. With the exception of the Southern Bottlenose Whale, beaked whales in the assessment region are listed as Data Deficient, which highlights the need for additional research, specifically on assessments of abundance, changes in abundance, distribution and anthropogenic threats, including marine noise pollution.
Regional population effects: Beaked whales are considered to be wide-ranging, seasonally migrating species. Those present within South African waters in summer presumably spend winters in the southern oceans, thus there are no barriers to dispersal, and rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: James BS & da Silva JM. 2025. A conservation assessment of Mesoplodon densirostris. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
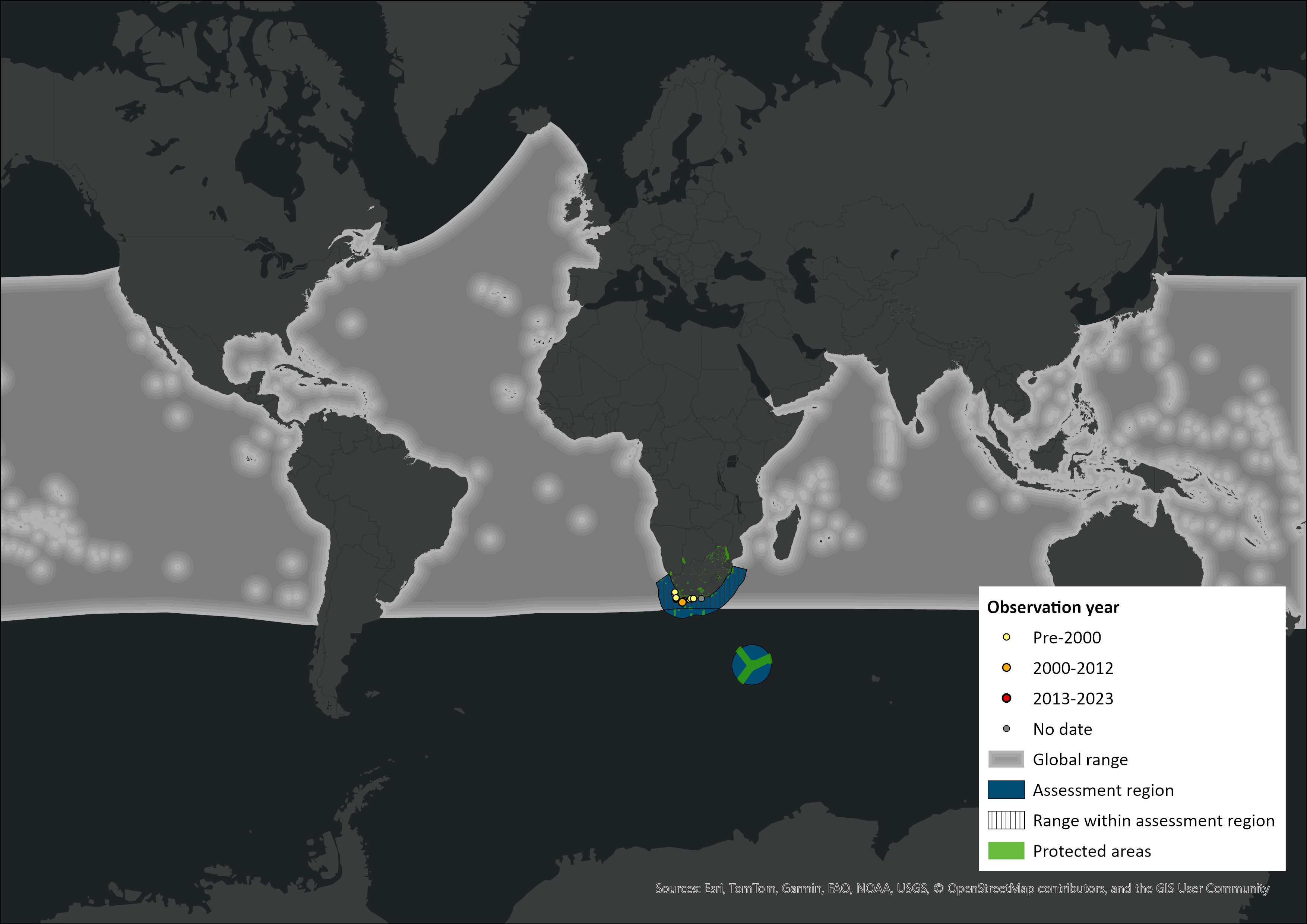
M. densirostris is considered the most widely distributed species of this genus (Pitman 2002; Macleod et al. 2006), occurring in all oceans except the Arctic with a preference for temperate and tropical waters of the Atlantic, Pacific and Indian Oceans (Mead 1989; Allen et al. 2011). Sightings and strandings are common around oceanic islands and archipelagos, for example Hawaii, the Society Islands, Mauritius and the Seychelles. As one of the most tropical beaked whales, this species often occurs within enclosed, deep, warm waters, including the Caribbean Sea, the Sea of Japan and the Gulf of Mexico. Within the assessment region, this species may utilise the warm waters of the Agulhas Current as a channel to the coast from tropical waters (Ross 1984). It is the beaked whale most recorded stranded on the South African coastline, these events having a primarily warm temperate distribution (Hofmeyr et al. 2014). Strandings have been recorded on both the east and west coasts of South Africa however Blainville’s beaked whales are more likely to be found in the warm waters of the Agulhas current on the south and east coasts (Ross 1984; Findlay et al. 1992).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical, Australasian, Indomalayan, Nearctic, Neotropical, Oceanian, Palearctic
Map
Figure 1. Distribution records for Blainville’s Beaked Whale (Mesoplodon densirostris) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Anguilla | Presence Uncertain | Native | – | – |
| Antigua and Barbuda | Presence Uncertain | Native | – | – |
| Argentina | Presence Uncertain | Native | – | – |
| Aruba | Presence Uncertain | Native | – | – |
| Australia | Extant | Native | – | – |
| Australia -> New South Wales | Extant | Native | – | – |
| Australia -> Northern Territory | Extant | Native | – | – |
| Australia -> Queensland | Extant | Native | – | – |
| Australia -> South Australia | Extant | Native | – | – |
| Australia -> Tasmania | Extant | Native | – | – |
| Australia -> Western Australia | Extant | Native | – | – |
| Bahamas | Extant | Native | – | – |
| Bangladesh | Presence Uncertain | Native | – | – |
| Barbados | Presence Uncertain | Native | – | – |
| Belize | Extant | Native | – | – |
| Benin | Presence Uncertain | Native | – | – |
| Bermuda | Presence Uncertain | Native | – | – |
| Bonaire, Sint Eustatius and Saba | Extant | Native | – | – |
| Brazil | Extant | Native | – | – |
| British Indian Ocean Territory | Presence Uncertain | Native | – | – |
| Brunei Darussalam | Presence Uncertain | Native | – | – |
| Cabo Verde | Extant | Native | – | – |
| Cambodia | Presence Uncertain | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Canada | Extant | Native | – | – |
| Canada -> Nova Scotia | Extant | Native | – | – |
| Cayman Islands | Extant | Native | – | – |
| Chile | Extant | Native | – | – |
| China | Extant | Native | – | – |
| Cocos (Keeling) Islands | Extant | Native | – | – |
| Colombia | Extant | Native | – | – |
| Comoros | Extant | Native | – | – |
| Congo | Presence Uncertain | Native | – | – |
| Congo, The Democratic Republic of the | Presence Uncertain | Native | – | – |
| Cook Islands | Presence Uncertain | Native | – | – |
| Costa Rica | Extant | Native | – | – |
| Cuba | Presence Uncertain | Native | – | – |
| Curaçao | Extant | Native | – | – |
| Côte d’Ivoire | Presence Uncertain | Native | – | – |
| Djibouti | Presence Uncertain | Native | – | – |
| Dominica | Presence Uncertain | Native | – | – |
| Dominican Republic | Presence Uncertain | Native | – | – |
| Ecuador | Extant | Native | – | – |
| El Salvador | Presence Uncertain | Native | – | – |
| Equatorial Guinea | Presence Uncertain | Native | – | – |
| Eritrea | Presence Uncertain | Native | – | – |
| Fiji | Extant | Native | – | – |
| France | Presence Uncertain | Native | – | – |
| French Guiana | Presence Uncertain | Native | – | – |
| French Polynesia | Presence Uncertain | Native | – | – |
| Gabon | Presence Uncertain | Native | – | – |
| Gambia | Presence Uncertain | Native | – | – |
| Ghana | Presence Uncertain | Native | – | – |
| Gibraltar | Presence Uncertain | Native | – | – |
| Grenada | Presence Uncertain | Native | – | – |
| Guadeloupe | Presence Uncertain | Native | – | – |
| Guam | Extant | Native | – | – |
| Guatemala | Extant | Native | – | – |
| Guinea | Presence Uncertain | Native | – | – |
| Guinea-Bissau | Presence Uncertain | Native | – | – |
| Guyana | Extant | Native | – | – |
| Haiti | Presence Uncertain | Native | – | – |
| Honduras | Extant | Native | – | – |
| Hong Kong | Presence Uncertain | Native | – | – |
| India | Extant | Native | – | – |
| Indonesia | Extant | Native | – | – |
| Iran, Islamic Republic of | Presence Uncertain | Native | – | – |
| Ireland | Presence Uncertain | Native | – | – |
| Jamaica | Presence Uncertain | Native | – | – |
| Japan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Kiribati | Extant | Native | – | – |
| Liberia | Presence Uncertain | Native | – | – |
| Madagascar | Extant | Native | – | – |
| Malaysia | Extant | Native | – | – |
| Maldives | Presence Uncertain | Native | – | – |
| Marshall Islands | Extant | Native | – | – |
| Martinique | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mauritius | Extant | Native | – | – |
| Mayotte | Extant | Native | – | – |
| Mexico | Presence Uncertain | Native | – | – |
| Micronesia, Federated States of | Extant | Native | – | – |
| Montserrat | Presence Uncertain | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Myanmar | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nauru | Extant | Native | – | – |
| New Caledonia | Extant | Native | – | – |
| New Zealand | Extant | Native | – | – |
| New Zealand -> Chatham Is. | Extant | Native | – | – |
| New Zealand -> North Is. | Extant | Native | – | – |
| Nicaragua | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Niue | Presence Uncertain | Native | – | – |
| Northern Mariana Islands | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Palau | Extant | Native | – | – |
| Panama | Extant | Native | – | – |
| Papua New Guinea | Extant | Native | – | – |
| Peru | Extant | Native | – | – |
| Philippines | Extant | Native | – | – |
| Portugal | Extant | Native | – | – |
| Portugal -> Azores | Extant | Native | – | – |
| Portugal -> Portugal (mainland) | Extant | Native | – | – |
| Puerto Rico | Extant | Native | – | – |
| Réunion | Extant | Native | – | – |
| Saint Barthélemy | Extant | Native | – | – |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | – |
| Saint Helena, Ascension and Tristan da Cunha -> Ascension | Extant | Native | – | – |
| Saint Kitts and Nevis | Presence Uncertain | Native | – | – |
| Saint Lucia | Presence Uncertain | Native | – | – |
| Saint Martin (French part) | Extant | Native | – | – |
| Saint Vincent and the Grenadines | Presence Uncertain | Native | – | – |
| Samoa | Presence Uncertain | Native | – | – |
| Sao Tome and Principe | Extant | Native | – | – |
| Senegal | Presence Uncertain | Native | – | – |
| Seychelles | Extant | Native | – | – |
| Sierra Leone | Presence Uncertain | Native | – | – |
| Singapore | Presence Uncertain | Native | – | – |
| Sint Maarten (Dutch part) | Extant | Native | – | – |
| Solomon Islands | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Spain | Extant | Native | – | – |
| Sri Lanka | Extant | Native | – | – |
| Suriname | Presence Uncertain | Native | – | – |
| Taiwan, Province of China | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Thailand | Presence Uncertain | Native | – | – |
| Timor-Leste | Presence Uncertain | Native | – | – |
| Tokelau | Extant | Native | – | – |
| Tonga | Extant | Native | – | – |
| Trinidad and Tobago | Presence Uncertain | Native | – | – |
| Turks and Caicos Islands | Presence Uncertain | Native | – | – |
| Tuvalu | Presence Uncertain | Native | – | – |
| United Kingdom of Great Britain and Northern Ireland | Extant | Native | – | – |
| United States Minor Outlying Islands | Extant | Native | – | – |
| United States Minor Outlying Islands -> Midway Is. | Extant | Native | – | – |
| United States of America | Extant | Native | – | – |
| Uruguay | Extant | Native | – | – |
| Vanuatu | Extant | Native | – | – |
| Venezuela, Bolivarian Republic of | Extant | Native | – | – |
| Viet Nam | Extant | Native | – | – |
| Virgin Islands, British | Presence Uncertain | Native | – | – |
| Virgin Islands, U.S. | Presence Uncertain | Native | – | – |
| Wallis and Futuna | Extant | Native | – | – |
| Western Sahara | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 21. Atlantic – northwest | Extant | Native | – | – |
| 27. Atlantic – northeast | Extant | Native | – | – |
| 31. Atlantic – western central | Extant | Native | – | – |
| 34. Atlantic – eastern central | Extant | Native | – | – |
| 37. Mediterranean and Black Sea | Extant | Vagrant | – | – |
| 41. Atlantic – southwest | Extant | Native | – | – |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 51. Indian Ocean – western | Extant | Native | – | – |
| 57. Indian Ocean – eastern | Extant | Native | – | – |
| 61. Pacific – northwest | Extant | Native | – | – |
| 71. Pacific – western central | Extant | Native | – | – |
| 77. Pacific – eastern central | Extant | Native | – | – |
| 81. Pacific – southwest | Extant | Native | – | – |
| 87. Pacific – southeast | Extant | Native | – | – |
Climate change
The specific effects of climate change on beaked whales is currently unknown however it has been suggested that similar to other cetaceans, beaked whales will likely undergo extensive range shift towards higher latitudes where they may be exposed to additional stressors such as increased noise exposure, interactions with fisheries, incidence of disease outbreaks and risk of ship strikes as well as reduced prey availability (Feyrer et al. 2024). Drastic reductions in suitable habitat and available prey for beaked whales due to climate change may result in future population declines, which would be difficult to quantify give the scarcity of abundance, life history and population level information we currently have for many beaked whale species.
Population information
M. densirostris is the most common species of its genus and is considered fairly common within tropical waters (Reeves et al. 2003), however, no abundance estimates are available for this species within the assessment region. In some regions such as off Hawaii both island associated and open ocean populations of Blainville’s beaked whales occur, further complicating assessments of abundance (Baird 2019).
Population Information
Continuing decline in mature individuals? (Not specified)
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Recent population genomic investigations have uncovered substantial structure within Blainville’s Beaked Whales, with animals being from the Atlantic and Indo-Pacific Ocean showing deep divergences (Carroll et al. 2021; Onoufriou et al. 2022). Moreover, further substructure was detected within each Ocean basin (Onoufriou et al. 2022), with two ESUs identified in the Indo-Pacific region (Central Pacific and southern Hemisphere) and two distinct subpopulations within the southern hemisphere ESU, of which animals around the assessment region form one. Unfortunately, no measure of effective population size (Ne) were reported; however, through reanalysis of the data, this metric should be obtainable.
Based on the available information, one of the two genetic indicators in the Convention of Biological Diversity’s Global Biodiversity Framework can be quantified – the complementary indicator- proportion of populations maintained with a species. Given that 1 genetic distinct subpopulation is known from the region and no other subpopulations are known to have gone extinct, this indicator would receive a value of 1.0 (1/1 subpopulation remains).
Habitats and ecology
Mesoplodon species occur commonly in deep-waters and along continental slopes, where prey availability may be enhanced by the interactions between ocean currents and topography (MacLeod & Zuur 2005). Off the Bahamas for example, Blainville’s Beaked Whales showed a strong preference for areas with specific seabed aspects (northeast), gradients (68 to 296 m/km) and depths (136 to 1319 m) likely driven by high prey abundance associated with these areas (Allen et al. 2011). Habitat partitioning driven by age class or dominance hierarchy may also be present in this species in some parts of their range (Allen et al. 2011). Baird (2019) also suggests that sympatric Cuvier’s and Blainville’s Beaked Whales also partition their habitat to avoid direct competition between the species, with Cuvier’s beaked whales found in deeper waters and diving deeper than Blainville’s Beaked Whales. Beaked Whales are believed to be suction feeders (Heyning & Mead 1996), feeding predominantly on squid and deep-water fish. M. densirostris is no exception. Diving data from Hawaii suggest that Balinville’s Beaked Whales can dive for 83 minutes and to depths of 1599m (Baird 2019). During these long foraging dives individuals consume on average 25 small prey items including deepwater cephalopods and fish (Baird 2019). Sekiguchi et al. (1992) and Ross (1984) reported stomach contents containing the remains of Buttersnoek (Lepidopus caudatus) and lanternfish (Lampanyctus spp.). Off Hawaii, schools of between three and seven Blainville’s Beaked Whales were observed by Shallenberger (1981) while Baird (2019) observed group sizes of 1 to 11 (mean 3.8) individuals in the same population, with mean group sizes of 4.1 reported from the northern Bahamas (Allen et al. 2011). Baird (2019) suggests that groups primarily form for functional purposes such as predator avoidance or to provide mating opportunities, but that these social relationships are ephemeral in nature.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Marginal | – |
| 10.2. Marine Oceanic -> Marine Oceanic – Mesopelagic (200-1000m) | – | Suitable | Yes |
| 10.3. Marine Oceanic -> Marine Oceanic – Bathypelagic (1000-4000m) | – | Marginal | – |
Life History
Generation Length: Not specified
Age at Maturity: Female or unspecified: 9 years, Mead 1984
Age at Maturity: Male: Not specified
Size at Maturity (in cms): Female: Not specified
Size at Maturity (in cms): Male: Not specified
Longevity: Not specified
Average Reproductive Age: Not specified
Maximum Size (in cms): Females: 471 cm; Males: 473 cm, Mead 1984
Size at Birth (in cms): 261 cm
Gestation Time: Not specified
Reproductive Periodicity: Not specified
Average Annual Fecundity or Litter Size: Not specified
Natural Mortality: Not specified
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? Yes
Movement Patterns
Movement Patterns: Not specified
Congregatory: Not specified
Systems
System: Marine
General Use and Trade Information
In general, beaked whales in the southern hemisphere are not utilised or traded commercially, although some small-scale subsistence utilisation of Blainville’s Beaked Whale by artisanal fisheries has been recorded.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
– |
– |
– |
National Commercial Value: No
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There appear to be no widely distributed major threats to beaked whales. The impact of potential threats are unknown but, considering that most Ziphiidae species are naturally rare, especially within the assessment region, they may have unsustainable impacts on local populations and further research is required. The main current threats for Blainville’s Beaked Whale are climate change, whaling, military sonar, vessel noise, entanglement, depredation, persistent organic pollutants, toxic metals, plastics and oil spills (Feyrer et al. 2024).
Anthropogenic noise pollution has become an increasing and well-known threat to beaked whales, as they appear to be more vulnerable to noise pollution than other cetacean species (Dalebout et al. 2005). A number of mass stranding events involving beaked whales, most commonly Blainville’s and Cuvier’s beaked whales, have been attributed to seismic exploration and high-powered navy sonar (Simmonds & Lopez-Jurado 1991; Mignucci-Giannoni 1996; Frantzis 1998, 2004; Balcomb & Claridge 2001; Jepson et al. 2003; Cox et al. 2006). Although the exact mechanistic causes are not clearly understood, the formation of gas bubbles (Fernández et al. 2005), appears to be attributed to sonar activities and noise pollution (Cox et al. 2006). Jepson et al. (2003) described the physiological damage, including acute and chronic tissue damage, inflicted on beaked whales by the deployment of military sonar at the Canary Islands. Repeated exposure to high-intensity anthropogenic noise can also have sublethal effects on animals through cessation of foraging or abandonment of critical foraging habitat which can ultimately impact reproduction and overall survival (Baird 2019). In 2004 a moratorium on naval activities in the Canary Islands was enforced by the Spanish government, and since then no mass stranding events have occurred in this area (Fernández et al. 2013). Within the assessment region, marine noise pollution is intensifying due to coastal industrial development, shipping traffic and energy exploration, and thus represents a potentially severe threat.
Plastic pollution is a large-scale and increasing problem in all marine environments. The ingestion of plastic marine pollution has been documented in several species of beaked whales and may eventually lead to mortality as a result of choking, a reduction in appetite or starvation (e.g. Scott et al. 2001). A dead adult Blainville’s Beaked Whale found on a beach in southern Brazil in 1993 was found to contain a bundle of blue plastic threads within its primary stomach chamber (Secchi & Zarzur 1999). Based on the lack of food and parasites within its stomach and gut, it is likely that this individual had not fed for some time. Chemical pollution (POPs and heavy metals) may also impact beaked whales due to negative effects on reproduction and overall health (Feyrer et al. 2024).
Accidental entanglement of beaked whales in fisheries is widespread, particularly in deep-water gillnets, although the number of recorded mortalities is not high. Entanglements involving at least 15 species of beaked whales have been documented globally (Feyrer et al. 2024). Extensive gillnet and longline fishing practises throughout the ranges of many beaked whales may become an increasing risk to these species as a result of accidental entrapment and drowning. Depredation of fish from fishing gear or nets has been reported for at least five species of beaked whales which may increase the incidence of entanglements, ingestion of inappropriate food items (hooks or line), ship strikes or injuries incurred due to negative interactions (retaliation) with fishers (Feyrer et al. 2024).
The expansion of high-latitude fisheries, such as those directed at Antarctic Toothfish (Dissostichus mawsoni), which are largely unregulated and illegal, threaten the food stocks available for large cetaceans such as beaked whales. There is substantial evidence of large-scale reductions in many predatory fish populations (Baum et al. 2003, 2005; Polacheck 2006; Sibert et al. 2006), over-fishing and the collapse of several important “prey” fish stocks world-wide (e.g. Jackson et al. 2001). Although the effects of anthropogenic fish exploitation and the subsequent ecosystem changes on beaked whales is considered to be fairly low in comparison to other cetaceans in the Pacific Ocean (Trites et al. 1997), the degree of impact associated with high-latitude fisheries world-wide is largely unknown and could result in population declines.
The marine-related threats associated with global climate change may pose unquantified and complex threats to beaked whales, particularly within cool temperate and cold Antarctic habitats (Learmonth et al. 2006). Increasing ocean temperatures may result in range shift or contraction (Learmonth et al. 2006); however, no direct predictions pertaining to the direction or size of these shifts in range are currently known.
Unlike many whale species, beaked whales have not experienced large-scale historic or recent exploitation for meat or other products. This may be attributed to their general scarcity and inconspicuous nature, deep-sea distributions and/or deep-diving behaviour.
Conservation
More research into the distribution, abundance, migration patterns, bycatch rate and diet of beaked whales is essential for the effective development of species-specific mitigation measures for these species in South African waters. Mitigation measures associated with anthropogenic marine noise is probably most vital for Ziphiidae species locally and world-wide. The avoidance of beaked whale habitats in South African waters is currently challenging due to their wide distribution, and the lack of data pertaining to habitat preferences and geographical extent across this region.
Passive acoustic monitoring is a valuable technique used to detect marine mammals in order to modify marine activities so as to avoid the animals, decrease the amplitude or temporarily stop the source of sound when animals are within a critical distance (Barlow & Gisiner 2006). Although beaked whales are acoustically difficult to detect, all species are assumed to give off echolocation clicks, some may also produce whistles (Dawson et al. 1998; MacLeod & D’Amico 2006). However, Cuvier’s and Blainville’s Beaked Whales have been found to only produce echolocation clicks when they are several hundred metres deep. Generally, the clicks of Ziphiidae species are more narrow-banded than those of other marine mammals of a similar frequency, thus electronic filtering methods may be more effective than other methods (Barlow & Gisiner 2006).
Maintaining sightings records of beaked whales, during ship-based surveys directed at other species, is a valuable means with which to monitor the distribution and abundance of these cryptic and unknown species in South African waters.
All Ziphiidae species within the assessment region are listed either on Appendix I or II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES).
Recommendations for managers and practitioners:
- Critical beaked whale habitats, and areas of high beaked whale concentration should be identified, so as to effectively mitigate the effects of noise pollution.
- Although species-specific monitoring is deemed unnecessary for Ziphiidae species in the assessment region, sightings data should be recorded during systematic monitoring of other cetacean species.
- Establish a nationwide strandings network and databases (comprised of whale-watching operators, coastal protected areas, police stations, hotels, etc.) to gather and pool information.
Research priorities:
- Population size and trend estimates.
- Effects of marine noise pollution and plastic pollution on beaked whale populations.
- The identification of high concentration areas in South African waters, including distributional limits, seasonal movements and diving behaviour.
- Diet, reproduction and general biology
Encouraged citizen actions:
- Report strandings east of Mossel Bay to the Port Elizabeth Museum, and west of Mossel Bay to Iziko Museums, Cape Town.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) to help with mapping geographical distribution.
- Avoid using plastic bags.
- Save electricity and fuel to mitigate CO2 emissions and hence the rate of climate change.
Bibliography
Allen, B.M., Mead, J.G. and R.L. Brownell. 2011. Species review of Blainville’s beaked whale, Mesoplodon densirostris. IWC Scientific Committee documents SC/63/SM16. p. 18 (accessed on 14 March 2025).
Anderson, R. C., Clark, R., Madsen, P. T., Johnson, C., Kiszka, J. and Breysse, O. 2006. Observations of Longman’s beaked whale (Indopacetus pacificus) in the Western Indian Ocean. Aquatic Mammals 32(2): 223-231.
Baird, R.W. 2019. Behavior and ecology of not-so-social odontocetes: Cuvier’s and Blainville’s beaked whales. In: Würsig, B. (ed) Ethology and behavioral ecology of odontocetes. Springer, Cham, p 305– 329
Baker AN. 1983. Whales and Dolphins of New Zealand and Australia. An Identification Guide. University Press, Wellington, Victoria.
Balcomb, K. C. 1989. Baird’s beaked whale Berardius bairdii Stejneger, 1883: Arnoux’s beaked whale Berardius arnuxii Duvernoy, 1851. In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Vol. 4: River dolphins and the larger toothed whales, pp. 261-288. Academic Press.
Balcomb, K.C. and Claridge, D.E. 2001. A mass stranding of cetaceans caused by naval sonar in the Bahamas. Bahamas Journal of Science 8(2): 2-12.
Barlow, J. 1999. Trackline detection probability for long-diving whales. In: G. W. Garner, S. C. Amstrup, J. L. Laake, B. J. F. Manley, L. L. McDonald and D. G. Robertson (eds), Marine mammal survey and assessment methods, pp. 209-221. Balkema Press, Netherlands.
Barlow, J. 2006. Cetacean abundance in Hawaiian waters estimated from a summer/fall survey in 2002. Marine Mammal Science 22(2): 446-464.
Barlow, J. and Gisiner, R. 2006. Mitigating, monitoring and assessing the effects of anthropogenic sound on beaked whales. Journal of Cetacean Research and Management 7(3): 239-250.
Baum, J.K., Kehler, D.G. and Myers, R.A. 2005. Robust estimates of decline for pelagic shark populations in the northwest Atlantic and Gulf of Mexico. Fisheries 2005: 27-29.
Best, P.B. 2007. Whales and Dolphins of the Southern African Subregion. Cambridge University Press, Cape Town, South Africa.
Boschema H. 1950. Maxillary teeth in specimens of Hyperoodon rostratus (Muller) and Mesoplodon grayi Von Haast stranded on the Dutch coasts. Koninklijke Nederlandse Akademie Wetenschappen 53: 775–786.
Brownell Jr., R. L., Yamada, T., Mead, J. G. and van Helden, A. 2006. Mass strandings of Cuvier’s beaked whales in Japan: U.S. naval acoustic link? International Whaling Commission.
Caretta JV, Price T, Petersen D, Read R. 2004. Estimates of marine mammal, sea turtle, and seabird mortality in the California drift gillnet fishery for swordfish and thresher shark, 1996–2002. Marine Fisheries Review 66: 21–30.
Carrol, EL. et al. 2021. Speciation in the deep: genomics and morphology reveal a new species of beaked whale Mesoplodon eueu. Proceedings of the Royal Society B 288: 1961. https://doi.org/10.1098/rspb.2021.1213
Cockcroft VG, De Kock AC, Ross GJB, Lord DA. 1990. Organochlorines in common dolphins caught in shark nets during the Natal “sardine run”. South African Journal of Zoology 25: 144-148.
Cox, T.M., Ragen, T.J., Read, A.J., Vos, E., Baird, R.W., Balcomb, K., Barlow, J., Caldwell, J., Cranford, T., Crum, L., D’Amico, A., D’Spain, A., Fernández, J., Finneran, J., Gentry, R., Gerth, W., Gulland, F., Hildebrand, J., Houser, D., Hullar, T., Jepson, P.D., Ketten, D., Macleod, C.D., Miller, P., Moore, S., Mountain, D., Palka, D., Ponganis, P., Rommel, S., Rowles, T., Taylor, B., Tyack, P., Wartzok, D., Gisiner, R., Mead, J. and Benner, L. 2006. Understanding the impacts of anthropogenic sound on beaked whales. Journal of Cetacean Research and Management 7(3): 177-187.
Culik, B. M. 2004. Review of small cetaceans: Distribution, behaviour, migration and threats. Marine Mammal Action Plan/Regional Seas Reports and Studies 177: 343 pp.
Dalebout, M. L. 2002. Species identity, genetic diversity, and molecular systematic relationships among the Ziphiidae (beaked whales). Thesis, University of Auckland.
Dalebout, M. L., Mead, J. G., Baker, C. S., Baker, A. N. and van Helden, A. L. 2002. A new species of beaked whale Mesoplodon perrini sp. n. (Cetacea: Ziphiidae) discovered through phylogenetic analyses of mitochondrial DNA sequences. Marine Mammal Science 18(3): 577-608.
Dalebout, M. L., Ross, G. J. B., Baker, C. S., Anderson, R. C., Best, P. B., Cockcroft, V. G., Hinsz, H. L., Peddemors, V. M. and Pitman, R. L. 2003. Appearance, distribution and genetic distinctiveness of Longman’s beaked whale, Indopacetus pacificus. Marine Mammal Science 19(3): 421-461.
Dalebout, M.L., Robertson, K.M., Frantzis, A., Engelhaupt, D., Mignucci-Giannoni, A.A., Rosario-Delestre, R.J. and Baker, C.S. 2005. Worldwide structure of mtDNA diversity among Cuvier’s beaked whales (Ziphius cavirostris): Implications for threatened populations. Molecular Ecology 14: 3353-3371.
Dalebout, M.L., Van Helden, A., Van Waerebeek, K. and Baker, C.S. 1998. Molecular genetic identification of southern hermisphere beaked whales (Cetacea: Ziphiidae). Molecular Ecology 7: 687-694.
Dawson S, Barlow J, Ljungblad D. 1998. Sounds recorded from Baird’s beaked whale, Berardius bairdii. Marine Mammal Science 14: 335-344.
Dayaratne, P. and Joseph, L. 1993. A study on dolphin catches in Sri Lanka. Bay of Bengal Programme, Madras, India.
Dixon, J. 1980. A recent stranding of the strap-toothed whale, Mesoplodon layardii (Gray) (Ziphiidae) from Victoria, and a review of Australian records of the species. Victorian Naturalist 97: 34-41.
Ferguson, M. C. and Barlow, J. 2001. Spatial distribution and density of cetaceans in the eastern Pacific Ocean based on summer/fall research vessel surveys in 1986-96. Southwest Fisheries Science Center Administrative Report LJ-01-04: 61 pp.
Fernández, A., Edwards, J. F., Rodriguez, F., Espinosa, A., De Los Monteros, Herraez, P., Castro, P., Jaber, J. R., Martin, V. and Arebelo, M. 2005. “Gas and fat embolic syndrome” involving a mass stranding of beaked whales (family Ziphiidae) exposed to anthropogenic sonar signals. Veterinary Pathology 42: 446-457.
Feyrer, L.J., Stanistreet, J.E. and H.B. Moors-Murphy. 2024-Navigating the unknown: assessing anthropogenic threats to beaked whales, family Ziphiidae. Royal Society Open Science 11: 240058.
Findlay KP, Best PB, Ross GJB, Cockcroft VG. 1992. The distribution of small odontocete cetaceans off the coasts of South Africa and Namibia. South African Journal of Marine Science 12: 237-270.
Frantzis A. 2004. The first mass stranding that was associated with the use of active sonar (Kyparissiakos Gulf, Greece, 1996). ECS Newsletter 42: 14–20.
Frantzis, A. 1998. Does acoustic testing strand whales? Nature 392(5): 29.
Gambell R, Best PB, Rice DW. 1975. Report on the International Indian Ocean whale marking cruise, 24th Nov 1973 – 3rd Feb 1974. Reports of the International Whaling Commission.
Heyning, J. E. 2002. Cuvier’s beaked whale Ziphius cavirostris. In: W. F. Perrin, B. Wursig and J. G. M. Thewissen (eds), Encyclopedia of Marine Mammals, pp. 305-307. Academic Press, San Diego, USA.
Heyning, J. E. and Mead, J. G. 1996. Suction feeding in beaked whales: morphological and observational evidence. Natural History Museum of Los Angeles County, Contributions in Science 464: 12.
Heyning, J.E. 1989. Cuvier’s beaked whale Ziphius cavirostris G. Cuvier, 1823. In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, pp. 289-308. Academic Press.
Hobson, R. P. and Martin, A. R. 1996. Behaviour and dive times of Arnoux’s beaked whales, Berardius arnuxii, at narrow leads in fast ice. Canadian Journal of Zoology 74: 388-393.
Hofmeyr GJG, Findlay KP, Cockcroft VG, Kotze G, Meyer M, Plön S, Thornton M, Best PB. 2014. Spatial and temporal distribution of beaked whale strandings on the South African coast. South African Marine Sciences Symposium, Stellenbosch.
IUCN (International Union for Conservation of Nature). 2008. Mesoplodon mirus. Available at: http://www.iucnredlist.org. (Accessed: 21 February 2016).
IUCN (International Union for Conservation of Nature). 2012d. Mesoplodon densirostris . Available at: http://www.iucnredlist.org. (Accessed: 21 February 2016).
Jackson, J. B. C., Kirby, M. X., Berger, W. H., Bjorndal, K. A., Botsford, L. W., Bourque, B. J., Bradbury, R. H., Cooke, R., Erlandson, J., Estes, J. A., Hughes, T. P., Kidwell, S., Lange, C. B., Lenihan, H. S., Pandolfi, J. M., Peterson, C. H., Steneck, R. S., Tegner, M. J. and Warner, R. R. 2001. Historical overfishing and the recent collapse of coastal ecosystems. Science 293: 629-637.
Jefferson, T. A., Leatherwood, S. and Webber, M. A. 1993. Marine Mammals of the World: FAO Species Identification Guide. United Nation Environment Programme and Food and Agricultural Organization of the UN.
Jepson, P.D., Arebelo, M., Deaville, R., Patterson, I.A.P., Castro, P., Baker, J.R., Degollada, E., Ross, H.M., Herraez, P., Pocknell, A.M., Rodriguez, F., Howie, F.E., Espinosa, A., Reid, R.J., Jaber, J.R., Martin, V., Cunningham, A.A. and Fernandez, A. 2003. Gas-bubble lesions in stranded cetaceans. Nature 425: 575-576.
Julian, F. and Beeson, M. 1998. Estimates of marine mammal, turtle, and seabird mortality for two California gillnet fisheries: 1990-95. Fishery Bulletin 96: 271-284.
Kasamatsu, F. and Joyce, G. G. 1995. Current status of odontocetes in the Antarctic. Antarctic Science 7: 365-379.
Kasamatsu, F., Hembree, D., Joyce, G., Tsunoda, L., Rowlett, R. and Nakano, T. 1988. Distribution of cetacean sightings in the Antarctic: results obtained from the IWC/IDCR minke whale assessment cruises, 1978/79 to 1983/84. Report of the International Whaling Commission 38: 449–473.
Kasuya T, Brownell Jr RL, Balcomb III KC. 1997. Life history of Baird’s beaked whales off the Pacific coast of Japan. Report of the International Whaling Commission 47: 969–979.
Kasuya, T. 2002. Giant beaked whales Berardius bairdii and B. arnuxii. In: W. F. Perrin, B. Wursig and J. G. M. Thewissen (eds), Encyclopedia of Marine Mammals, pp. 519-522. Academic Press, San Diego, California, USA.
Koper RP, Plön S. 2012. The potential impacts of anthropogenic noise on marine animals and recommendations for research in South Africa. EWT Research & Technical Paper No. 1. Endangered Wildlife Trust, South Africa.
Learmonth, J.A., Macleod, C.D., Santos, M.B., Pierce, G.J., Crick, H.Q.P. and Robinson, R.A. 2006. Potential effects of climate change on marine mammals. Oceanography and Marine Biology: An Annual Review 44: 431-464.
MacLeod C.D. 2005. Niche partitioning, distribution and competition in North Atlantic beaked whales. PhD Thesis. University of Aberdeen.
MacLeod, C. D. and Zuur, A. F. 2005. Habitat utilization by Blainville’s beaked whales off Great Abaco, northern Bahamas, in relation to seabed topography. Marine Biology 147: 1-11.
MacLeod, C. D., M. B. Santos and G. J. Pierce. 2003. Review of data on diets of beaked whales: evidence of niche separation and geographic segregation. Journal of the Marine Biological Association of the United Kingdom 83: 651-665.
MacLeod, C. D., and D’Amico, A. 2006. A review of beaked whale behaviour and ecology in relation to assessing and mitigating impacts of anthropogenic noise. Journal of Cetacean Research and Management 7: 211-221.
Macleod, C. D., Perrin, W. F., Pitman, R. L., Barlow, J., Balance, L., D’amico, A., Gerrodette, T., Joyce, G., Mullin, K. D., Palka, D. L. and Waring, G. T. 2006. Known and inferred distributions of beaked whale species (Ziphiidae: Cetacea). Journal of Cetacean Research and Management 7(3): 271-286.
Malakoff, D. 2002. Suit ties whale deaths to research cruise. Science 298: 722-723.
McCann C. 1975. A study of the genus Berardius. Reports of the Whales Research Institute 27: 111–137.
Mead, J.G. 1984. Survey of reproductive data for the beaked whales (Ziphiidae). Reports of the International Whaling Commission, Special Issue, 6:91-96.
Mead, J.G. 1989. Beaked whales of the genus Mesoplodon. In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Vol. 4: River dolphins and the larger toothed whales, pp. 349-430. Academic Press.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Mignucci-Giannoni AA. 1996. Marine mammal strandings in Puerto Rico and the United States and British Virgin Islands. Ph.D. Thesis. University of Puerto Rico.
Miyazaki N, Wada S. 1976. Observation of Cetacea during whales cruise in the western tropical Pacific. Scientific Reports of the Whales Research Institute 30: 179–195.
Moore JC. 1968. Relationships among the living genera of beaked whales. Fieldiana Zoology 53: 209–298.
Moore SE. 2008. Marine mammals as ecosystem sentinels. Journal of Mammalogy 89: 534-540.
Moritz, C. 1994. Defining ‘Evolutionarily Significant Units’ for conservation. Trends in Ecology and Evolution 9: 373–375.
Nishiwaki, M. and Oguro, N. 1972. Catch of Cuvier’s beaked whales off Japan in recent years. Scientific Reports of the Whales Research Institute 24: 35-41.
Omura, H., Fujino, K. and Kimura, S. 1955. Beaked whale Berardius bairdi of Japan, with notes on Ziphius cavirostris. Scientific Reports of the Whales Research Institute 10: 89-132.
Onoufriou, AB, et al. 2022. Biogeography in the deep: Hierarchical population genomic structure of two beaked whale species. Global Ecology and Conservation 40: e02308. https://doi.org/10.1016/j.gecco.2022.e02308
Paterson RA, van Dyck S. 1990. Records of beaked whales in Queensland. Scientific Reports of Cetacean Research 1: 63–77.
Paterson, R. A. and Parker, A. E. 1994. Aerial observations of large ziphiid whales, possibly Berardius arnuxii, off the southern coast of New South Wales. Memoirs of the Queensland Museum 37: 301-306.
Pitman, R. L. 2002. Mesoplodont whales Mesoplodon spp. In: W. F. Perrin, B. Wursig and J. G. M. Thewissen (eds), Encyclopedia of Marine Mammals, pp. 738-742. Academic Press.
Pitman, R. L., Palacios, D. M., Brennan, P. L. R., Brennan, B. J., Balcomb III, K. C. and Miyashita, T. 1999. Sightings and possible identity of a bottlenose whale in the tropical Indo-Pacific: Indopacetus pacificus. Marine Mammal Science 15(2): 531-549.
Podesta, M.D., Amico, A., Pavan, G., Drougas, A., Komnenou, A. and Portunato, N. 2005. A review of Cuvier’s beaked whale strandings in the Mediterranean Sea. Journal of Cetacean Research and Management 7(3): 251-261.
Polacheck, T. 2006. Tuna longline catch rates in the Indian Ocean: did industrial fishing result in a 90% rapid decline in the abundance of large predatory species? Marine Policy 30: 470-482.
Reeves, R.R., Smith, B.D., Crespo, E.A. and Notarbartolo di Sciara, G. 2003. Dolphins, Whales and Porpoises: 2002-2010 Conservation Action Plan for the World’s Cetaceans. IUCN, Gland, Switzerland and Cambridge, UK.
Rice, D.W. 1998. Marine Mammals of the World: Systematics and Distribution. Society for Marine Mammalogy, Special Publication Number 4, Lawrence, Kansas.
Ross GJ. 1984. Smaller cetaceans of the south east coast of southern Africa. Annals of the Cape Provincial Museums. Natural History 16: 309-319.
Ross GJB, Cockcroft VG, Cliff G. 1985. Additions to marine faunas of South Africa and Natal. Lammergeyer 35: 36–40.
Ross GJB. 1970. The occurrence of Hector’s beaked whale Mesoplodon hectori (Gray) in South African waters. Annals of the Cape Provincial Museums (Natural History) 8: 195–204.
Scott, M. D., Hohn, A. A., Westgate, A. J., Nicolas, J. R., Whitaker, B. R. and Campbell, W. B. 2001. A note on the release and tracking of a rehabilitated pygmy sperm whale (Kogia breviceps). Journal of Cetacean Research and Management 3(1): 87-94.
Secchi, E.R. and Zarzur, S. 1999. Plastic debris ingested by a Blainville’s beaked whale, Mesoplodon densirostris, washed ashore in Brazil. Aquatic Mammals 25(1): 21-24.
Sekiguchi, K., Klages, N. T. W. and Best, P. B. 1996. The diet of strap-toothed whales (Mesoplodon layardii). Journal of Zoology (London) 239: 453-463.
Sekiguchi, K., Klages, N.T.W. and Best, P.B. 1992. Comparative analysis of the diets of smaller odontocete cetaceans along the coast of southern Africa. South African Journal of Marine Science 12: 843-861.
Shallenberger EW. 1981. The status of Hawaiian cetaceans. Marine Mammal Commission.
Sibert, J., Hampton, J., Kleiber, P. and Maunder, M. 2006. Biomass, size, and trophic status of top predators in the Pacific Ocean. Science 314: 1773-1776.
Simmonds, M. P. and Lopez-Jurado, L. F. 1991. Whales and the military. Nature 351: 448.
Slip, D. J., Moore, G. J., and Green, K. 1995. Stomach contents of a southern bottlenose whale, Hyperoodon planifrons, stranded at Heard Island. Marine Mammal Science 11: 575-584.
Taylor RJF. 1957. An unusual record of three species of whale being restricted to pools in Antarctic sea-ice. Proceedings of the Royal Society of London 129: 325–331.
Trites AW, Christensen V, Pauly D. 1997. Competition between fisheries and marine mammals for prey and primary production in the Pacific Ocean. Journal of Northwest Atlantic Fishery Science 22: 173-187.
Yamada TK. 2002. On an unidentified beaked whale found stranded in Kagoshima. National Science Museum, Tokyo.
Yang, W.-C., Chou, L.-S., Jepson, P. D., Brownell Jr., R. L., Cowan, D., Chang, P.-H., Chiou, H.-I., Yao, C.-J., Yamada, T. K., Chiu, J.-T., Wang, P.-J. and Fernandez, A. 2008. Unusual cetacean mortality events in Taiwan, possibly linked to naval activities. Veterinary Record 162: 184-186.