Black-tailed Tree Rat
Thallomys nigricauda

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Thallomys nigricauda – (Thomas, 1882)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Thallomys – nigricauda
Common Names: Black-tailed Tree Rat (English), Swartstertboomrot, Swartstertrot (Afrikaans), Khuruvuru (Tsonga)
Synonyms: No Synonyms
Taxonomic Note:
The type locality of this species is from the Hountop (Hudup or Hutop) River, west of Gibeon in Namibia (Skinner & Chimimba 2005). Although some suggest that this species should be treated as a species complex under Thallomys paedulcus sensu lato, Taylor et al. (1995) describe how T. paedulcus and T. nigricauda are distinguishable on chromosomal grounds, and partially distinguishable on morphometric grounds (the distribution maps based on morphometric discriminant analysis in Taylor et al. (1995) are not reliable). Thallomys shortridgei was previously included within T. nigricauda, but following Musser and Carleton (2005), T. shortridgei is currently recognised as a distinct species. The relationship between the species of this genus require resolution, and a molecular analysis of the genus is urgently required (Nel 2013; Monadjem et al. 2015).
Red List Status: LC – Least Concern
Assessment Information
Assessor: Taylor, P.1
Reviewers: Kraai, M.2 & Smith, C.3
Institutions:1University of the Free State, 2Sol Plaatjie University,3Endangered Wildlife Trust
Previous Assessors and Reviewers: Child, M.F., Power, J. & Mondajem, A.
Previous Contributors: Relton, C., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
The illegal harvesting of Acacia trees, especially Vachellia (previously Acacia) erioloba and Senegalia (previously Acacia) mellifera, across the range of this species is a major threat, as it reduces habitat quality through the destruction of nesting sites and food resources. Although widely distributed across the Acacia woodlands and shrublands of the northwestern parts of South Africa (including Northern Cape, North West and Limpopo provinces), this species was not recorded at all during a recent survey in North West Province, despite trapping effort of 380 trap nights and extensive spotlighting in the Molopo Bushveld vegetation type (R.J. Power unpubl. data) and the species was last recorded in Molopo in 2002. This suspected reduction in population over the last decade might qualify the species as Near Threatened A2c, but applying the regional criterion retains a Least Concern listing: the species is a good disperser, and habitat is contiguous across both the Namibian and Botswana borders with presumed dispersal across countries. Additionally, it has a large potential area of occupancy (84,860 km2) based on remaining natural woodland habitats within its range.
Although the species is benefitted by legislation in place to protect Acacia trees (harvesting requires a permit), ongoing illegal harvesting could be causing a continuing decline in habitat quality. For example, the Camel Thorn Tree (V. erioloba) is a protected species in terms of the National Forests Act (No. 84 of 1998), but it is not always enforceable. More research needs to be done to quantify the impact of this threat on population size and trend and provide evidence for effective conservation interventions. This species should be reassessed as more data become available.
Regional population effects: There is likely dispersal from Namibia and Botswana, and there is contiguous habitat across the borders to South Africa. Black-tailed Tree Rats are considered good dispersers as long as there is tree cover.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Taylor P. 2025. A conservation assessment of Thallomys nigricauda. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This largely southern African species has been recorded from western Angola in the north of its range, southwards through much of Namibia and Botswana to the northwestern parts of South Africa where it inhabits arid Acacia savanna. The precise delineation of this species’ range remains controversial, as it is often mistaken for T. paedulcus (Skinner & Chimimba 2005). Thallomys paedulcus may be restricted to the eastern regions of southern Africa, and T. nigricauda to the western regions (see Monadjem et al. 2015). Further vetting of museum records is required to delimit the respective distributions of T. nigricauda and T. paedulcus. Similarly, the records on or south of the Gariep River must be vetted to ensure they do not refer to T. shortridgei (Monadjem et al. 2015). Currently, the degree of allopatry with T. shortridgei is unknown (Nel 2013).
A recent survey covering all vegetation types in the North West Province did not record this species, but did record T. paedulcus (Power 2013, 2014). It was last documented at Molopo Nature Reserve in the early to mid 2000’s (Eccard et al. 2006; Coleman & Downs, 2010). Power (2014) speculated that an extensive fire in 1997, which burnt the whole northern half of the reserve, could have led to a local extinction, but this was disproved by evidence of subpopulations present in 2002 (see Eccard et al. 2006; Meyer et al. 2008) and 2006-2007 (Coleman & Downs 2010). Further field surveys are required from across its range to determine area of occupancy and other local extinctions.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
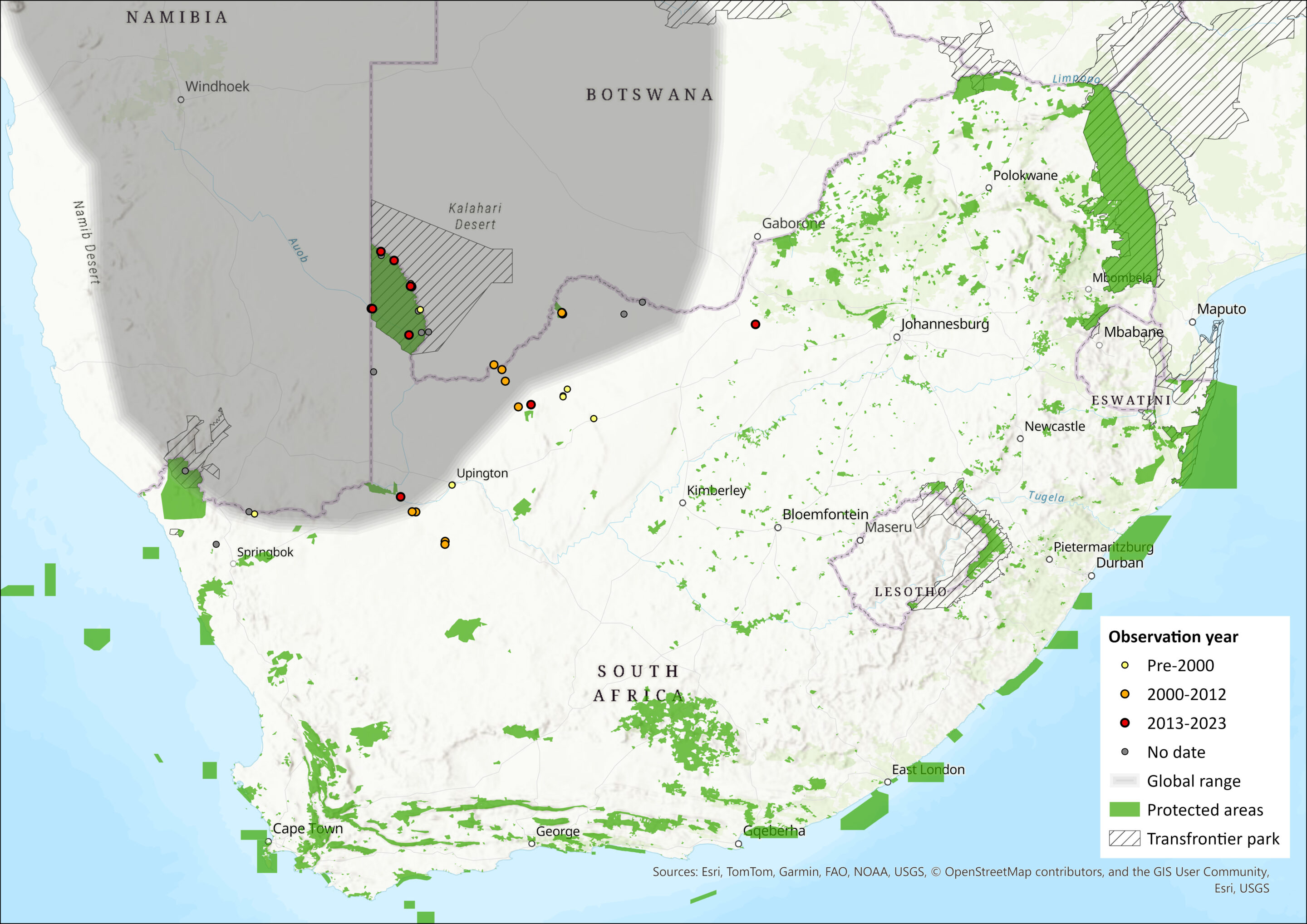
Map
Figure 1. Distribution records for Black-tailed Tree Rat (Thallomys nigricauda) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
The impact of climate change on arid Acacia savanna woodland is unknown but denudation of woodlands and especially large trees with cavities for fuelwood extraction would have a much larger impact on populations. In situ measurements of body temperature in tree rats from Molopo Nature Reserve showed physiological responses to a highly variable thermal environment, e.g. the amplitude of their diurnal cycle was 259.7% of that expected. Individuals forage nocturnally and are able to lower their body temperature whilst resting in cavities during the day (Coleman & Downs 2010). Whilst cavities provide moderate thermal buffering effect to allow behavioural thermoregulation, more extreme high temperatures in the future may minimise this energy saving and could potentially place animals under high physiological stress.
Population
The Black-tailed Tree Rat is locally common in stands of suitable trees (those containing cavities for nesting) but patchily distributed throughout its range. This species was not recorded at all during a recent survey of the North West Province despite the trapping effort of 380 trap nights and extensive spotlighting in the Molopo Bushveld vegetation type (Power 2014). Across two reserves, the Molopo Nature Reserve and Khamab Kalahari Reserve, a total of 1,226 km and 72 hours were recorded over 52 surveys, and no individuals were encountered (R.J. Power unpubl. data). If not a sampling bias, thre is apparent rarity in North West Province should be a cause for concern (Power 2014). Yet at the same location, in 2002 Meyer et al. (2008) recorded density estimates of 0.5–1 individual/ha. Males had a larger range (10 ha) than females (1 ha) suggesting a promiscuous mating system. Coleman & Downs (2010) collected 15 individuals during 2006-2007. Population declines can also occur after bush fires when smoke filters through the cavities as the number of inhabited trees in burnt areas (even where trees are just singed) are lower than unburnt areas (Nel 2013). Captive individuals were found to have a longevity of about 4 years (Jones 1982).
Population Information
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
One population genetic study has been undertaken on this species (Meyer et al. (2010). While the study was localised to Molopo Nature Reserve in Northwest Province, South Africa it provided insight into the kind of diversity available in a fairly localised area. High haplotype diversity (16 haplotypes; mean haplotype diversity 0.68) was found. Higher spatial differentiation amongst females compared with males was detected, likely owing to, at least in part, males having larger ranges than females and hence more male-biased dispersal.
Given that the species is considered to be a good disperser as long as there is tree cover present, it is possible the species exists as a single metapopulation, connected with populations in neighbouring countries.
The Convention on Biological Diversity’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (1/1 population remains).
Unfortunately, due to very limited information, the GBF’s headline indicator – proportion of species with an effective population size (Ne) greater than 500 – cannot be estimated. A population genetic study is highly recommended to clarify the population structure of the species and provide estimates of its effective population size and overall genetic health.
Habitats and ecology
This arboreal, nocturnal species is generally associated with arid savannahs, especially Acacia bushland habitats and Kalahari thornveld with Acacia erioloba, A. luederitzii, Boscia albitrunca and Terminalia sericea trees, with A. mellifera shrubs (Nel 2013). Occurs mostly in dense or open stands of trees but also in large, isolated trees (Nel 2013). Typically, it requires tall trees, particularly Camel Thorn Trees (A. erioloba) (Dean et al. 1999), but has also been found nesting in Leadwood (Combretum imberbe), Mopane (Colophospermum mopane) and other species of large trees. On Molopo Nature Reserve, it was found to be dependent on any dead or alive mature trees with cavities for nesting, as long as they were in close proximity to its favoured food plant, Acacia mellifera (Eccard et al. 2006). This may limit the choice of suitable nest sites, since A. mellifera was less likely to grow within a vegetation patch containing large trees than in patches without large trees (Power 2014). Generally, a nest is occupied by a pair and their young, but up to eight adults have been witnessed leaving a single nest (Smithers 1983). Thorns of Acacia trees may effectively protect tree rats from predators. For example, during a 143-day study in the Kalahari Thornveld, no predation of tree rats was observed (Meyer 2004). Additionally, Meyer et al. (2008) suggested that the dense shrub cover may protect this species from aerial predators during movement.
Predominantly vegetarian, they typically feed on the newly sprouting leaves of Acacia spp., as well as fine twigs and the outer green covering of seed pods (Coleman & Downs 2010). They prefer the arid savannahs of the western parts of South Africa (Skinner & Chimimba 2005). Although most rodents dwelling in xeric environments avoid the harsh environmental conditions by burrowing, this is one of a few species to have opted for an arboreal lifestyle (Coleman & Downs 2010). Nests are formed under loose bark or in the forks and hollows of trees using grass, sticks and leaves (Skinner & Chimimba 2005). They are able to survive independent of drinking water, meeting water requirements from their diet (Skinner & Chimimba 2005).
Black-tailed Tree Rats are seasonal breeders, and although no data are available for the assessment region, in the northern parts of southern Africa, young were born between August and April, and a mean litter size of 3.6 was observed (Skinner & Chimimba 2005).
Ecosystem and cultural services: Although this species may have a low predation rate in some parts of its range (Meyer 2004), it likely still utilised as a food source by many terrestrial or arboreal predators, such as small carnivores and snakes. On a small-scale, this species could aid in some degree of bush encroachment control, as it feeds on a major encroacher, the Black Thorn, A. mellifera.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 4 years in captivity
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: 3.6
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: yes
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The Black-tailed Tree Rat requires vegetation structure with intact woodlands for food and nesting sites. Resultantly, it is vulnerable to the harvesting of mature trees, especially V. erioloba and A. mellifera, for firewood or charcoal production (Eccard et al. 2006). This is a noted problem in the North West Province (DACE 2008), though not severe in extent, and commercial harvesting of Camel Thorn in the Northern Cape is thought to be largely restricted to dead trees (von Staden & Raimondo 2015).
Woodland clearing of Camel Thorn and Black Thorn (often with the use of pesticides) to enhance grassland establishment and increase grazing suitability for livestock may also cause a decline in habitat availability for this species. This occurs mainly in areas threatened by bush encroachment, as a result of poor land management (von Staden & Raimondo 2015). Incorrect fire regimes are also a root problem as it promotes overgrazing that removes the fuel load, which leads to bush encroachment (Driver et al. 2012).
Current habitat trend: Declining
Conservation
This species is present within some protected areas across the assessment region, for example Kgalagadi Transfrontier Park (de Graaff 1978) and Tswalu Kalahari Reserve. No direct interventions are necessary at present. However, conservationists should protect woodland patches and ensure connectivity through protected area expansion and biodiversity stewardship schemes. Additionally, harvesting of large trees should be regulated to make the practice sustainable. The protected status of A. erioloba in South Africa should be maintained, due to its ecological importance, cultural value and the threats associated with uncontrolled and illegal harvesting for commercial firewood (Seymour & Milton 2003). North West Province has enlisted the Black-tailed Tree Rat as Specially Protected in their current Biodiversity Act owing to the concern in the status of the species there.
Recommendations for land managers and practitioners:
- Subpopulations should be monitored to record any changes in abundance and distribution, and the monitoring of nest sites, and spotlight surveys, are suggested.
- Land managers should be encouraged to conserve woodlands and old trees.
Research priorities:
- Additional studies are needed into the taxonomic status of this species.
- Analysis of museum records to more accurately delimit distribution.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. However, this species cannot be readily distinguished from T. paedulcus based on morphological characteristics.
- Landowners can preserve Acacia trees, both living and dead.
Bibliography
Anderson MD, Anderson TA. 2001. Too much, too quickly? Doubts about the sustainability of the camelthorn wood harvest. African Wildlife 55: 21–23.
Coleman, J.C. and Downs, C.T. 2010. Does home range of the black-tailed tree rat (Thallomys nigricauda Thomas 1882) change with season along an aridity gradient? African Zoology 45: 177-188.
DACE. 2008. North West Province: Environment Outlook. North West Department of Agriculture, Conservation & Environment, Mmabatho, South Africa.
Dean WRJ, Milton SJ, Jeltsch F. 1999. Large trees, fertile islands, and birds in arid savanna. Journal of Arid Environments 41: 61–78.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Eccard JA, Dean WRJ, Wichmann MC, Huttunen SM, Eskelinen E, Moloney KA, Jeltsch F. 2006. Use of large Acacia trees by the cavity dwelling Black-tailed Tree Rat in the southern Kalahari. Journal of Arid Environments 64: 604–615.
Jones ML. 1982. Longevity of captive mammals. Zoologischer Garten 52: 113–128.
Meyer, J., Kohnen, A. & Brandl, R. 2010. Genetic differentiation in an arboreal rodent from African savannas. African Journal of Ecology 48, 831–836.
Meyer J, Raudnitschka D, Steinhauser J, Jeltsch F, Brandl R. 2008. Biology and ecology of Thallomys nigricauda (Rodentia, Muridae) in the Thornveld savannah of South Africa. Mammalian Biology 73: 111–118.
Meyer J. 2004. The impact of habitat structures on some small rodents in the Kalahari Thornveld (South Africa). Ph.D. Dissertation. University of Marburg.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Musser, G.G. and Carleton, M.D. 2005. Superfamily Muroidea. In: D.E. Wilson and D.A. Reeder (eds), Mammal Species of the World: a geographic and taxonomic reference, pp. 894-1531. The John Hopkins University Press, Baltimore, USA.
Nel JAJ. 2013. Thallomys nigricauda Black-tailed Acacia Rat (Black-tailed Thallomys). In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, pp. 559–560. Bloomsbury Publishing, London, UK.
Power RJ. 2013. Dr Ruth Segomotsi Mompata Mammal Report. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Seymour C, Milton S. 2003. A collation and overview of research information on Acacia erioloba (Camelthorn) and identification of relevant research gaps to inform protection of the species. Department of Water Affairs and Forestry, Pretoria, South Africa.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1983. The Mammals of the Southern African Subregion. University of Pretoria, Pretoria, South Africa.
Taylor, P. J., Rautenbach, I. L., Gordon, D., Sink, K. and Lotter, P. 1995. Diagnostic morphometrics and southern African distribution of two sibling species of tree rat, Thallomys paedulcus and Thallomys nigricauda (Rodentia: Muridae). Durban Museum Novitates 20: 49-62.
de Graaff G. 1978. Notes on the Southern African black-tailed tree rat Thallomys paedulcus (Sundevall, 1846) and its occurrence in the Kalahari Gemsbok National Park. Koedoe 21: 181–190.
von Staden L. and Raimondo D. 2015. Vachellia erioloba (E. Mey.) P.J.H. Hurter. National Assessment: Red List of South African Plants. South African National Biodiversity Institute, South Africa.