
Bat-eared Fox
Otocyon megalotis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Otocyon megalotis – (Desmarest, 1822)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – CANIDAE – Otocyon – megalotis
Common Names: Bat-eared Fox, Delalande’s Fox (English), Bakoorjakkals, Bakoorvos, Draaijakkals (Afrikaans), Unga (Ndebele), Motlhosa (Sepedi), Phokojwee e ditsebe tsa mankgane, Motlhose (Sesotho), Udlamhloshana, Imphunushe (Swati), Xilwanandau, Xilwana-ndzawo (Tsonga), Motlhose, Motlhôse, Lethose, Tlhose, Tlhôsi (Tswana), Phunguhwe i re na Ndevhe Khulwane (Venda), Impungutye (Xhosa), Udlamhloshana (Zulu)
Synonyms: Canis megalotis Desmarest, 1822
Taxonomic Note:
This is a monotypic genus with two recognised subspecies, Otocyon megalotis megalotis in southern Africa and O. m. virgatus from East Africa (Nel & Maas 2013).
Red List Status: LC – Least Concern
Assessment Information
Assessor: Davis, R.S.1
Reviewers: Nicholson, S.K.2 & Do Linh San, E.3
Contributor: da Silva, J.M. 4
Institutions: 1Nelson Mandela University, 2Endangered Wildlife Trust, 3Sol Plaatjie University, 4South African National Biodiversity Institute
Previous Assessors and Reviewers: Dalerum, F., le Roux, A., Kamler, J.F., Page-Nicholson, S., Stuart, C., Stuart, M., Wilson, B. & Do Linh San, E.
Previous Contributors: Hoffmann, M., Collison, W., de Bruin, R. & Jumban, K.
Assessment Rationale
This species is listed as Least Concern as it is common in conservation areas and occurs widely on farms throughout the assessment region. Bat-eared Foxes are occasionally persecuted in agricultural areas, as they are mistaken for damage-causing animals, whilst roadkill is another significant threat to local population persistence. However, these threats, while suspected to cause local declines periodically, are not expected to be affecting the population trend overall. Monitoring, management and education are recommended to mitigate losses on farmlands, ranches and roads.
Regional population effects: There is a continuous population consisting of the assessment region and neighbouring countries. It is unlikely that the population in the assessment region is a sink. It is unknown whether or not immigrants from outside the region are frequent enough to enhance the regional population significantly. Bat-eared Foxes are efficient dispersers (Kamler et al. 2013a).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Davis RS. 2025. A conservation assessment of Otocyon megalotis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Bat-eared Fox has a disjunct distribution range, occurring across the arid and semi-arid regions of eastern and southern Africa in two discrete populations (representing each of the known subspecies) separated by about 1,000 km. Otocyon m. virgatus ranges from southern Sudan, Ethiopia and Somalia down through Uganda and Kenya to southwestern Tanzania; O. m. megalotis occurs from Angola through Namibia and Botswana to Mozambique and South Africa (Coetzee 1977; Kingdon 1977; Skinner & Chimimba 2005). The two ranges were probably connected during the Pleistocene (Coe & Skinner 1993). This disjunct distribution is similar to that of other endemic, xeric species, for example Aardwolf (Proteles cristatus) and Black-backed Jackal (Lupulella mesomelas). Bat-eared Foxes are considered widespread in both Namibia and Botswana and their distribution extends narrowly into Zimbabwe (Skinner & Chimimba 2005). In Mozambique, they are limited to the southwestern areas.
Within the assessment region, the species is absent from Lesotho, Eswatini and KwaZulu-Natal Province (Skinner & Chimimba 2005; Nel & Maas 2013). This could possibly be due to a lack of their preferred termite food source. Bat-eared Foxes are widespread throughout the rest of the assessment region, and indeed their distribution coincides mostly with that of harvester termites. There appears to have been a recent range expansion into the Gauteng and Mpumalanga provinces, as well as the southern parts of the Limpopo Province. The species occurs throughout the arid western parts of the North West Province (Power 2014) and, during above average rainfall years, abundance increases in arid savannahs (i.e. areas receiving < 300 mm rainfall / year) and decreases in mesic savannahs (> 500 mm / year) (Macdonald 1982), which would explain the species’ local abundance in any given year. In the Western Cape, bat-eared Foxes are found within the Cape Point Nature Reserve and along the coastal areas (e.g. West Coast National Park and Elandsbaai). In the Eastern Cape, they have a more restricted distribution – being confined to the western inland areas of the province (Skinner & Chimimba 2005).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map 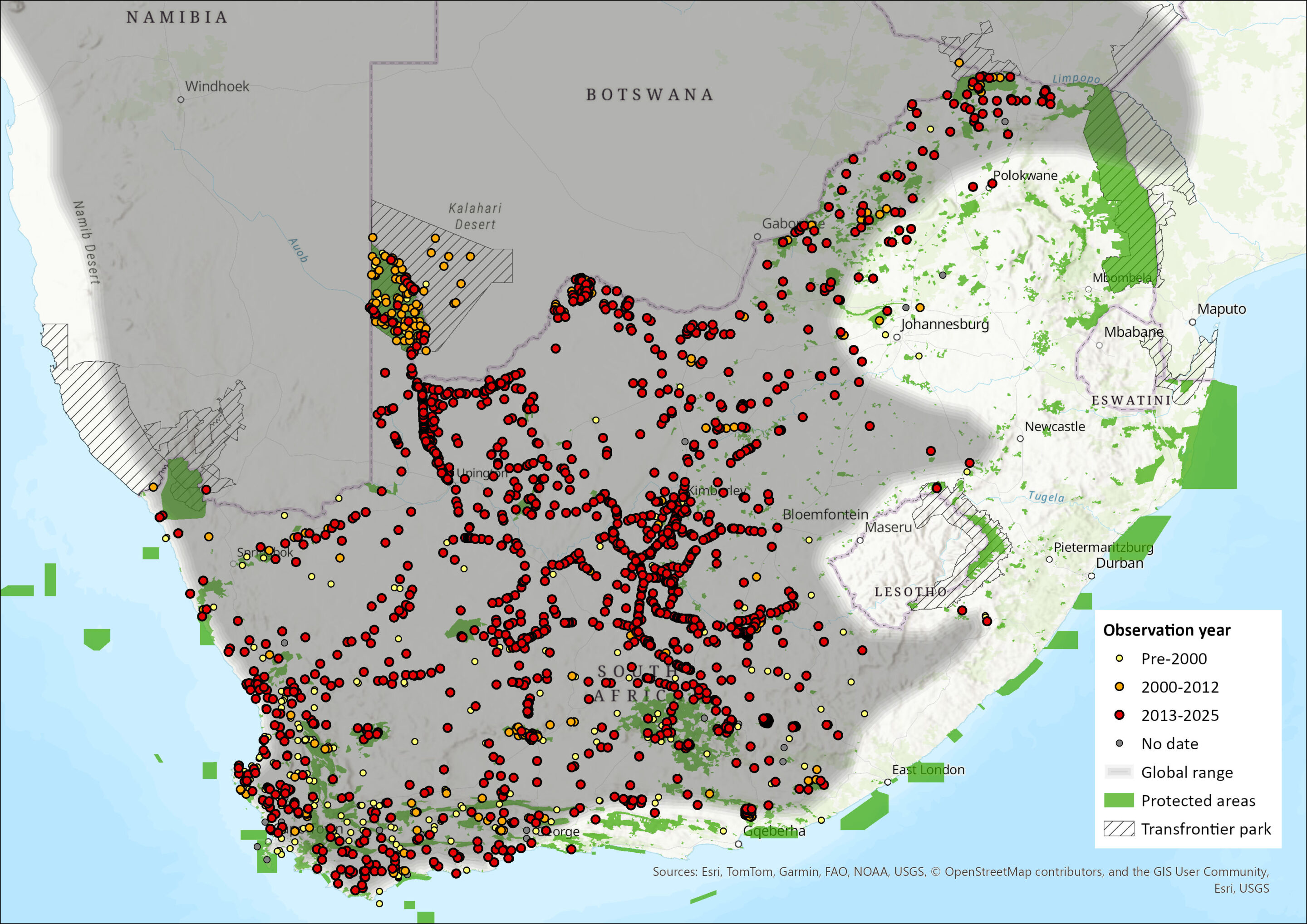
Figure 1. Distribution records for Bat-eared Fox (Otocyon megalotis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No modelling outputs are available to project and quantify direct and indirect impacts of climate change on Bat-eared Fox distribution and population persistence. As the species’ abundance is influenced by rainfall and aridity (Macdonald, 1982), shifts in climate patterns may influence the future distribution of Bat-eared Foxes. The birthing period for Bat-eared Foxes usually coincides with the onset of the wet season (October-December), when insect abundance is higher. Expected increases in aridity and reduced precipitation in southern Africa may negatively impact insect abundance and influence reproduction timing and juvenile survival. However, further research is needed to assess the response of Bat-eared Foxes to bioclimatic factors and the potential influence on population abundance and geographic distribution. Although the species does feed primarily on harvester termites, Bat-eared Foxes also consume other invertebrate species and fruits, meaning the species could adapt to changing foraging conditions (Klare et al. 2011; Kamler et al. 2020). Bat-eared Foxes have significantly larger home range sizes and group sizes during the dry season, which likely increases intragroup contact rates (Kamler et al. 2017). Consequently, increased periods of aridity and longer dry seasons could have implications for disease transmission, as the species is a primary host for rabies in South Africa.
Population information
The species is common in conservation areas in southern Africa, and locally common in arid areas and on farms on the Karoo plateau. However, natural population fluctuations can complicate estimates of population trends. Within a circumscribed habitat, numbers can fluctuate depending on rainfall, food availability (Waser 1980; Nel et al. 1984), breeding stage and disease (Maas 1993a,b; Nel 1993). Persecution may cause local declines in other areas of their distribution. Living in nuclear family groups with high rates of amicable behaviour, Bat-eared Foxes may be particularly susceptible to local outbreaks of diseases such as canine distemper virus, canine parvovirus and rabies (Kamler et al. 2017). Populations in the Kimberley area (Northern Cape Province) that were being peripherally monitored during ongoing sympatric carnivore studies were seen to crash following such outbreaks, only recovering after a year or two (B. Wilson & F. Dalerum pers. obs. 2007–2012).
Population densities can exhibit significant variation, from Bat-eared Foxes being rare to abundant. The estimated density of Bat-eared Foxes on a sheep farm and a game farm near Kimberley, were estimated at 1.1 and 0.7 individual(s) / km2, respectively (Kamler et al. 2012, 2013b). In the southwestern Kalahari in dry riverbeds, Nel et al. (1984) recorded densities between 0.7–14 individual(s) / km2. Densities were higher in Limpopo Province at 5.7 individuals / km2. At Tussen-die-Riviere Game Reserve in the Free State, Mackie and Nel (1989) reported varying densities of 0.3–0.5 individual / km2 over a 3-year period, while in the central Karoo densities were higher with between 1.1 and 2.0 individuals / km2 (Kuntzsch & Nel 1992). However, it should be noted that there is a paucity of recent density estimates for the species in South Africa and available estimates are derived from traditional methods that may not accurately represent true density.
Population Information
Current population trend: Overall stable despite sometime drastic local fluctuations.
Continuing decline in mature individuals? Unlikely but difficult to determine based on lack of robust population monitoring data.
Extreme fluctuations in the number of subpopulations: Unlikely
Continuing decline in number of subpopulations: Unlikely based on distribution data
All individuals in one subpopulation: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: It is currently not possible to determine the extent or number of subpopulations.
Severely fragmented: No. Although this species seems to largely depend on the presence of harvester termites, its distribution is relatively continuous as favourable habitats are generally connected.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
While comprehensive population genetics studies have not been undertaken across the species range or even across the assessment region, more localised studies have examined the genetic structure and differentiation between reserves (Kamler et al. 2013; Granell-Ruiz et al. 2021). Both studies found limited genetic differentiation between sites, with reserves sharing haplotypes and revealing signs of isolation by distance. Based on this evidence, while kin groups may exist, the overall species structure represents a metapopulation structure, with sufficient gene flow between sites due to dispersal of individuals (of both sexes and ages classes). Based on this information, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – can be estimated. The single metapopulation remains; therefore, the indicator gets a value of 1.0 (1/1 populations remaining).
Considering the relatively widespread distribution of the species in South Africa, and the species’ ability to persist and disperse in areas outside of formal protection (Kamler et al. 2013a), it is not thought that the species is at risk of losing genetic diversity and is stable into the long-term. Translating this into genetic terms, this would suggest that the metapopulation is assumed to possess an effective population size (Ne) greater than 500. The headline genetic indicator in the GBF – proportion of populations within a species with Ne > 500 – would therefore receive a score of 1.0 (1/1 populations with Ne > 500). This is only a gross estimate, Consequently, it is highly recommended a population genetic study be undertaken to obtain a more precise estimate of Ne.
Habitats and ecology
In southern Africa, the prime habitat is mainly short-grass plains and areas with bare ground (Mackie & Nel 1989), but Bat-eared Foxes are also found in open scrub vegetation and arid, semi-arid or winter rainfall (Fynbos or Cape Macchia) shrub lands, and open arid savannah. They are absent from true desert or afforested areas. The range of both subspecies overlaps almost completely with that of Hodotermes and Microhodotermes termite genera prevailing in the diet (Mackie & Nel 1989; Maas 1993a). However, recent studies in the Kalahari have indicated that Bat-eared Fox did not exhibit strong habitat preferences and the species’ space use is not entirely dictated by the distribution of termite species (Periquet & Le Roux, 2018). Recent empirical evidence confirms that these foxes are primarily acoustic foragers (Renda & Le Roux 2017; Renda et al 2023), unlike many other canids who rely on visual and olfactory cues while hunting.
Density, home range size, and group size appear to be affected by both bottom-up (i.e. termite abundance; Maas 1993b) and top-down factors (i.e. Black-backed Jackal numbers; Kamler et al. 2013b). Group size varies with the time of the year, with the monogamous pair being accompanied by up to six cubs prior to the dispersal period (Kamler et al. 2017). Groups forage as a unit in home ranges that may increase during the dry season, due to changes in food resources and group sizes. These home ranges are patrolled and urine-marked and will be defended, with the outcome determined by group size. These family groups may be extended by philopatric daughters from several generations (Maas 1993b). In the Serengeti, Tanzania, behavioural evidence on group and pair formation and the existence of “super families”, consisting of one male and up to three closely related breeding females, raises interesting questions about regular inbreeding between males and their daughters from several generations (see Maas 1993a). That said, dispersal patterns by all sex and age groups, especially adult females, appears to reduce the opportunities for inbreeding (Kamler et al. 2013a).
Pair-bonding and mating peak during late winter and early summer, with births occurring at the onset of the wet season, in September to December. A single litter of 1–6 cubs (usually 5) is produced annually after a 60 to 75-day gestation period. Extra-pair paternity is low in this species, presumably because the diet of Bat-eared Foxes allows pair members to maintain proximity, while discouraging roaming (Wright et al. 2010). Males play an important role in the raising of the young, spending more time than females with cubs, grooming, guarding and playing with them, and defending them against predators (Wright 2006; Nel & Maas 2013). In the Kalahari, weaning is prompted after the first rains and subsequent flush of insects.
Ecosystem and cultural services: Several bird species apparently benefit from this species during winter, as they consume termites which are dug up by the Bat-eared Foxes (Stenkewitz & Kamler 2008).
- Ecosystem service: influence vegetation structure by digging.
- Ecosystem service: influence seed dispersal through seasonal consumption of fruits (Kamler et al. 2020).
- Ecosystem service: influence disease dynamics as possible vectors of a number of viruses (such as rabies and canine distemper virus).
- Cultural service: skin use, plus various other body parts used in traditional zootherapeutic preparations.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Bat-eared Foxes may be persecuted as damage-causing animals and will also be subject to accidental persecution where the intended target is another species.
Skins are traded commercially, at the national scale, and they are also offered as trophies in small numbers throughout South Africa, Namibia and Tanzania. Commercial use is very limited, but winter pelts are valued and sold as blankets. Bat-eared Foxes are also kept in captivity in North America, Europe, South Africa and Asia as exotic pets, although never in large numbers.
In general, Bat-eared Foxes do well on game/wildlife farms. Because this species focuses on termite consumption, it is compatible with a broad range of habitats and management strategies. However, their well-being may be inversely affected by high numbers of larger predators, particularly Black-backed Jackals, due to intraguild predation.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Used as bushmeat and traditional medicine (skin, anal glands). | Yes | Trophy hunting and pet trade. |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | – | – | – | – |
| 3. Medicine – human & veterinary | – | – | – | – |
| 10. Wearing apparel, accessories | true | true | – | – |
| 15. Sport hunting/specimen collecting | – | true | – | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Unknown, probably stable.
Threats
In the assessment region, the primary threats are hunting for skins and intended or accidental persecution, although the exact relative impacts need to be quantified. Bat-eared Foxes often fall prey to a variety of raptor species, as well as most larger carnivores. Black-backed Jackals are likely the greatest predator of Bat-eared Foxes (Kamler et al. 2012, 2013b). Jackals may not only have negative impacts on numbers of Bat-eared Foxes (Blaum et al. 2009), but they can also affect the group size and home range size of this species (Kamler et al. 2013b). There is also documented evidence of intraguild predation on Bat-eared Foxes from larger carnivores, such as Lion (Panthera leo) and Leopard (Panthera pardus), and competitive pathways between Bat-eared Foxes and the larger carnivore guild are poorly understood (Curveira-Santos et al 2022).
Climate change is likely to affect distribution patterns, but not necessarily population numbers. This species does not exclusively feed on harvester termites (Klare et al. 2011) and therefore could adapt to locally changing conditions by switching to other food sources, such as other invertebrates or fruits. Bat-eared Foxes are able to quickly move into areas that are rehabilitated or suited to their needs (Rova 2003), probably due to their extensive dispersal abilities (Kamler et al. 2013a). The expansion of agricultural areas (direct anthropogenic change) may have a stronger impact on numbers and distribution, given their persecution by farmers, and the increased potential for diseases such as rabies and canine distemper being spread through contact with domestic dogs (Rova 2003; Kamler & Macdonald 2006; Kamler et al. 2017).
Bat-eared Foxes are also very often killed on roads (Bullock et al. 2011; Periquet et al. 2018). For example, 37 dead individuals (possibly killed over a few weeks’ period) were counted on a single day along a 170 km stretch of road along the R360 towards Upington in the Northern Cape (K. Jumban unpubl. data). Bat-eared Foxes were also one of the most frequently recorded species (third or fourth most frequently recorded depending on observer type) from a nationwide citizen science roadkill scheme (Periquet et al. 2018). Further, between 2009 and 2016, at least 602 Bat-eared Foxes were killed as a result of vehicle collisions across South Africa (EWT unpubl. data). This data was collected on an ad hoc basis and therefore, it is expected that numbers are likely to be higher.
Conservation
Bat-eared Foxes occur in many protected areas within the assessment region, including the Kgalagadi Transfrontier Park, as well as in numerous game ranches and farms throughout the assessment region (with the exception of KwaZulu-Natal).
Education aimed at improving public awareness of this species is seen as a primary conservation intervention. Awareness campaigns targeting landowners and farmers to the fact that this species primarily eats termites and does not damage livestock is paramount. Similarly, the public should be discouraged from buying bat-eared fox pelts or, at least, a sustainable and well-monitored trade should be established.
The species is a particularly high road-collision risk in some areas (EWT unpubl. data). In the Kalahari, sign boards were used to warn visitors travelling to the Kgalagadi Transfrontier Park to reduce their speed. It was later determined through interviews that the majority of Bat-eared Fox incidents were actually from local residents who were more familiar with the roads and travelled at faster speeds after sunset. However, such signage is used internationally as a standard mitigation measure and can be effective in raising general public awareness if used correctly (Bond & Jones 2013).
Recommendations for land managers and practitioners:
- Monitoring population trends of bat-eared foxes.
- Reducing persecution as damage-causing animals through improved local knowledge.
- Maintain short-growth grassland through suitable grazing regimes.
- Managing numbers of Black-backed Jackals, especially in areas without apex predators, may benefit this species. However, it is important further research is conducted on this before management plans to reduce Black-backed Jackal numbers are implemented.
Research priorities:
In southern Africa, a recent study documented important aspects of Bat-eared Fox ecology, such as home range size, habitat use, seasonal movements, mortality, dispersal patterns of adults and young, and formation of breeding pairs (Kamler et al. 2012, 2013a,b, 2017). However, there is a conspicuous lack of information about both abundance and population trends in this species across its range. Overall, the following research priorities have been identified:
- Quantification of population trends and causes for population fluctuations, including the potential influence of climate change scenarios on population abundance.
- Predictive modelling of the species’ distribution under varying climate change scenarios.
- Quantification of large-scale population structure.
- Quantification of dispersal capabilities.
- Effects of disease on population dynamics.
- Role of Bat-eared Foxes in disease transmission.
- The effect of road mortalities on local populations and at a national level.
- The dispersal of young and the formation of new breeding pairs.
- The causal factors for differences in home range size in different localities, group size and changes in density as a function of food availability are poorly known.
- The impacts of awareness campaigns and whether they can effectively reduce persecution by landowners and farmers.
- The impact of the absence of apex predators on farmland.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Report road mortalities to roads@ewt.org
- Create conservancies by dropping fences between properties and holistically managing large areas.
Bibliography
Blaum, N., Tietjien, B., and Rossmanith, E. 2009. Impact of livestock husbandry on small- and medium-sized carnivores in Kalahari Savannah Rangelands. Journal of Wildlife Management 73: 60-67.
Bond ARF, Jones DN. 2013. Wildlife warning signs – public assessment of components, placement and designs to optimise driver response. Animals 3: 1142–1161.
Bullock KL, Malan G, Pretorius MD. 2011. Mammal and bird road mortalities on the Upington to Twee Rivieren main road in the southern Kalahari, South Africa. African Zoology 46: 60–71.
Coe, M. J. and Skinner, J. D. 1993. Connections, disjunctions and endemism in the eastern and southern African mammal faunas. Transactions of the Royal Society of South Africa 48: 233-256.
Coetzee, C.G. 1977. Order Carnivora. Part 8. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-42. Smithsonian Institution Press, Washington, DC, USA.
Granell-Ruiz, M., Norén, K., Kalthoff, D.C., Le Roux, A. and Dalerum, F. 2021. Genetic variation between and within two populations of bat-eared foxes (Otocyon megalotis Desmarest, 1822) in South Africa. African Zoology, 56(3), pp.165-172.
Grilo C, Bissonette JA, Santos-Reis M. 2009. Spatial–temporal patterns in Mediterranean carnivore road casualties: consequences for mitigation. Biological Conservation 142: 301–313.
Kamler JF, Gray MM, Oh A, Macdonald DW. 2013. Genetic structure, spatial organization, and dispersal in two populations of bat-eared foxes. Ecology and Evolution 3: 2892–2902.
Kamler JF, Macdonald DW. 2006. Longevity of a wild bat-eared fox. South African Journal of Wildlife Research 36: 199–200.
Kamler JF, Rostro-García S, Macdonald DW. In press. Seasonal changes in the social behaviour and movements of bat-eared foxes in South Africa: disease implications. Journal of Mammalogy 98 (in press).
Kamler, J. F., Stenkewitz, U., Klare, U. Jacobsen, N. F., and Macdonald, D. W. 2012b. Resource partitioning among cape foxes, bat-eared foxes, and black-backed jackals in South Africa. Journal of Wildlife Management 76: 1241-1253.
Kamler, J. F., Stenkewitz, U., and Macdonald, D. W. 2013. Lethal and sublethal effects of black-backed jackals on cape foxes and bat-eared foxes. Journal of Mammalogy 94(295-306).
Kamler, J.F., Rostro-García, S. and Macdonald, D.W. 2017. Seasonal changes in social behavior and movements of bat-eared foxes in South Africa: disease implications. Journal of Mammalogy, 98(5), pp.1426-1433.
Kamler, J.F., Klare, U. and Macdonald, D.W. 2020. Seed dispersal potential of jackals and foxes in semi-arid habitats of South Africa. Journal of Arid Environments, 183, p.104284.
Kingdon, J. 1977. East African Mammals: An Atlas of Evolution in Africa. Volume IIIA (Carnivores). Academic Press, London, UK.
Klare U, Kamler JF, Macdonald DW. 2011. The bat-eared fox: a dietary specialist? Mammalian Biology 76: 646–650.
Kuntzsch V, Nel JAJ. 1992. Diet of bat-eared foxes Otocyon megalotis in the Karoo. Koedoe 35: 37–48.
Kurberg L. 2005. The effect of grazing on bat-eared foxes, and how farmers in Namibia perceive the bat-eared fox. M.Sc. Thesis. Uppsala University.
Maas, B. 1993a. Bat-eared fox behavioural ecology and the incidence of rabies in the Serengeti National Park. Onderstepoort Journal of Veterinary Research 60: 389-393.
Maas, B. 1993b. Behavioural ecology and social organisation of the bat-eared fox in the Serengeti national Park, Tanzania. D. Phil. Thesis, University of Cambridge.
Macdonald IAW. 1982. The influence of short term climate fluctuations on the distribution of savannah organisms in southern Africa. M.Sc. Thesis. University of Natal.
Mackie, A. J. and Nel, J. A. J. 1989. Habitat selection, home range use, and group size of bat-eared foxes in the Orange Free State. South African Journal of Wildlife Research 19: 135-139.
Nel, J. A. J. 1993. The bat-eared fox: a prime candidate for rabies vector? Onderstepoort Journal of Veterinary Research 60: 396-397.
Nel, J. A. J. and Maas, B. 2013. Otocyon megalotis Bat-eared Fox. In: Kingdon, J. & Hoffmann, M. (ed.), Mammals of Africa. Volume 5. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 78-81. Bloomsbury, London.
Nel, J. A. J., Mills, M. G. L. and Van Aarde, R. J. 1984. Fluctuating group size in bat-eared foxes (Otocyon m. megalotis) in the south-western Kalahari. Journal of Zoology (London) 203: 294-298.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Renda S, Le Roux A. 2017. Sensory ecology of prey detection in the bat-eared fox (Otocyon megalotis). Behaviour 154: 227–240.
Rova E. 2003. Estimating the Rehabilitation of Former Farmlands into Wildlife Areas Using the Bat-eared Fox as an Indicator Species. Committee of Tropical Ecology, Minor Field Study 94. Uppsala University, Uppsala, Sweden.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stenkewitz U, Kamler JF. 2008. Birds feeding in association with bat-eared foxes on Benfontein Game Farm, South Africa. Ostrich 79: 235–237.
Waser, P.M. 1980. Small nocturnal carnivores: ecological studies in the Serengeti. African Journal of Ecology 18: 167-185.
Wright HWY, Gray MM, Wayne RK, Woodroffe RB. 2010. Mating tactics and paternity in a socially monogamous canid, the bat-eared fox (Otocyon megalotis). Journal of Mammalogy 91: 437–446.
Wright HWY. 2006. Paternal den attendance is the best predictor of offspring survival in the socially monogamous bat-eared fox. Animal Behaviour 71: 503–510.
