
Banana Bat
Afronycteris nanus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Afronycteris nanus – (Peters, 1852)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Afronycteris – nanus
Common Names: Banana Pipistrelle, Banana Bat, Banana Pipistrelle Bat, Pipistrelle Naine (English), Piesangvlermuis (Afrikaans), Pipistrelle naine, Pipistrelle naine aux ailes brunes (French)
Synonyms: Vespertilio nanus Peters, 1852, Vesperugo pusillulus Peters 1870, Vesperugo stampflii, Vesperugo pagenstecheri, Vesperus pusillus, Pipistrellus minusculus, Pipistrellus culex, Pipistrellus nanus (Peters, 1852), Pipistrellus nanus australis, Pipistrellus abaensis, Pipistrellus fouriei Thomas 1926, Pipistrellus africanus meesteri Kock 2001, Neoromicia nanus
Taxonomic Note:
Kearney et al. (2002) stated that the formerly referred Pipistrellus nana belonged to the genus Neoromicia but nana also shared characteristics with Hypsugo and Pipistrellus (Monadjem et al. 2010b). As such, Helbig et al. (2010) suggested that nana needs to be transferred to a new genus entirely. Subsequent research placed it in a newly erected genus Afronycteris (Monadjem et. al. 2020a). Some authorities consider africana as the oldest name for this species. However, in Decision 2120, the International Commission on Zoological Nomenclature (ICZN) ruled that the name Pipistrellus nanus (currently Afronycteris nanus) should be given precedence over africana, whenever the two are considered to be synonyms. Although it has been suggested that the specific epithet be spelled nana to align with the gender of the genus, the term nanus is a noun and therefore remains unchanged regardless of the genus (Mammal Diversity Database 2025).
Evidence suggests that A. nanus constitutes a species complex (Koubínová et al. 2013; Taylor et al. 2022); however, a continent-wide integrative taxonomic study is needed to clarify the evolutionary relationships among its populations.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1 Balona, J.2 & da Silva, J.M.3
Reviewers: Smith, C.4, Richardson, E.J.5
Institutions: 1Durban Natural Science Museum, 2Gauteng and Northern Regions Bat Interest Group,3South African National Biodiversity Institute, 4Endangered Wildlife Trust, 5Independent Consultant at Richardson & Peplow Environmental
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., Cohen, L., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Relton, C., Raimondo, D. & Child, M.F.
Assessment Rationale
Listed as Least Concern in view of its wide distribution throughout Sub-Saharan Africa, its occurrence in many protected areas in the assessment region (including Great Limpopo Transfrontier Park, Greater Mapungubwe Transfrontier Conservation Area and the iSimangaliso Wetland Park), its tolerance of disturbed habitats, its relative abundance in the eastern regions of the assessment region compared to other species, and because there are no major identified threats that could cause widespread population decline. This species has an EOO of 320,073 km2 and the population size is estimated to exceed 10,000 individuals.
Regional population effects: Their range is continuous with Mozambique and Zimbabwe through the Great Limpopo and Greater Mapungubwe Transfrontier Conservation Areas. However, it has low wing loading (Aldridge and Rautenbach 1987; Schoeman & Jacobs 2008), so rescue effects are uncertain.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR, Balona J & da Silva JM. 2025. A conservation assessment of Afronycteris nanus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Banana Bat is widely distributed throughout most of sub-Saharan Africa. It ranges from Senegal in the west, through West and Central Africa, to Eritrea, Ethiopia and Somalia in the east; from here its range spreads southwards to southeastern South Africa. The two records reported from the Kirindy Centre de Formation Professionelle Forestière Forest and from Zombitse in Madagascar (Goodman & Langrand 1994) are believed to be misidentifications (Goodman et al. 2015). It appears to be absent, or largely absent, from northern Kenya, Namibia, southern Botswana, western Zimbabwe, Lesotho and much of South Africa, but it is possible that it occurs more widely in these areas than is currently known, and further field studies are needed to investigate its distribution (ACR 2024). Within the assessment region, it occurs widely in the wetter eastern and northern parts of the region but is absent from the arid southwest (Monadjem et al. 2010b). It has been recorded from as far south as Cape Morgan in the Eastern Cape, north through KwaZulu-Natal, Eswatini, southern Mozambique and the lowveld of South Africa where it occurs in the Greater Mapungubwe and Great Limpopo Transfrontier Conservation Areas. Predictive niche models suggest a wider distribution within the Eastern Cape province of South Africa and Mozambique (Monadjem et al. 2010b).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 4,207 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
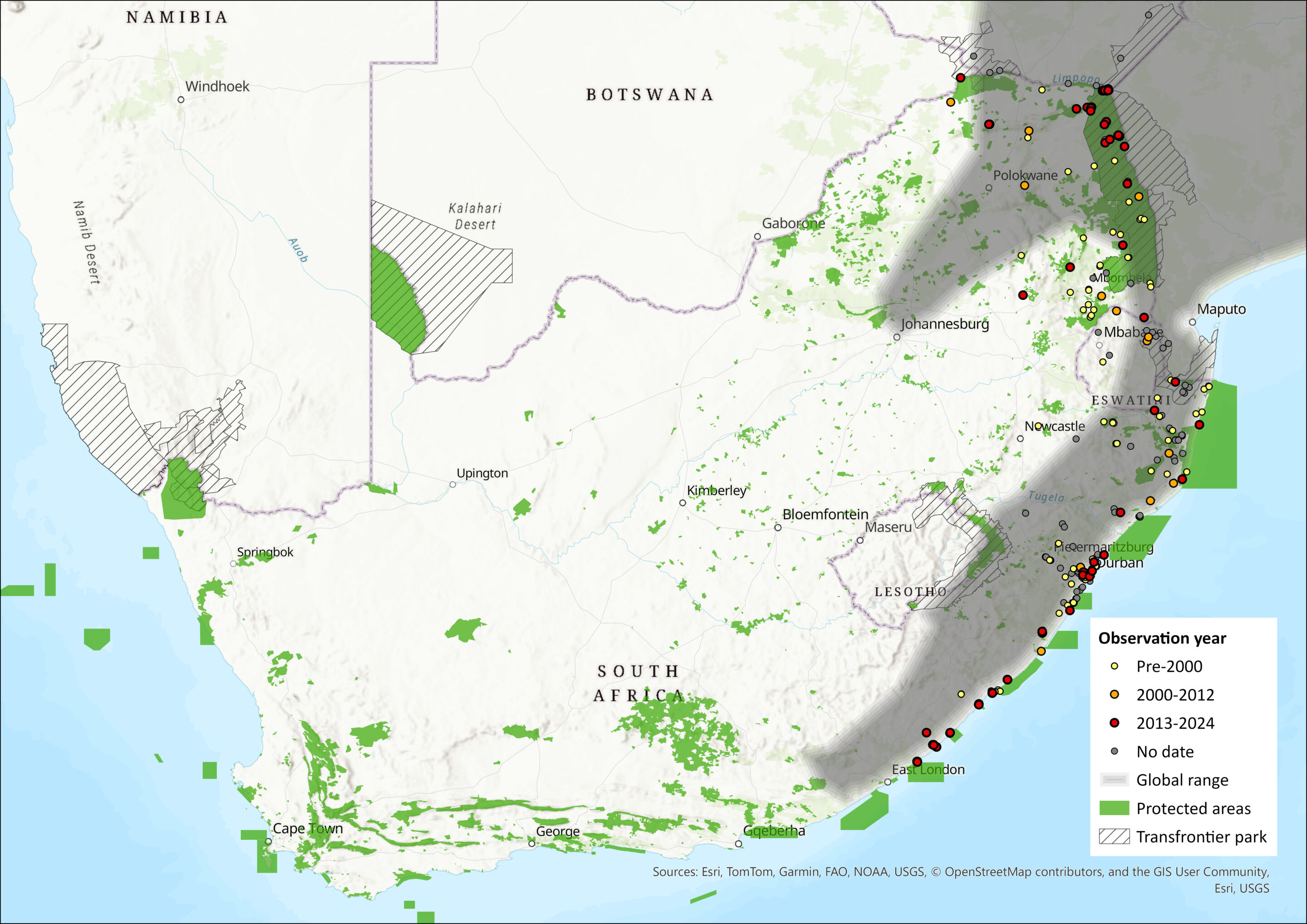 Figure 1. Distribution records for Banana Bat (Afronycteris nanus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Banana Bat (Afronycteris nanus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Presence Uncertain | Native | – | – |
| Liberia | Extant | Native | – | – |
| Madagascar | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Sudan | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
There are no studies on the impacts of climate change on this species. It is a widespread, adaptable species and thus likely to be relatively tolerant of a wide range of climatic conditions. Rainfall in the species range in South Africa is predicted to increase by 10%, with an increase in intense rainfall events, while temperatures are also predicted to increase in line with global averages, with drought indexes also increasing (Engelbrecht et al. 2024). Currently there is not enough information to predict the impacts of climate change on this species.
Population Information
In suitable habitats, this species is considered common (Happold 1987). They are well-represented in museums, with more than 500 individuals examined in Monadjem et al. (2020b). While this has been used as a proxy of abundance, it is not necessarily a true reflection of current abundance. In a recent study in the Durban region, this species was the most commonly recorded of 16 species along the Umbilo River (Naidoo et al. 2011), and acoustic surveys conducted in the greater Durban region of South Africa indicate relatively high activity levels, particularly in well-vegetated areas and near water bodies (L. Richards, unpublished data). The species is also recorded within green spaces in the Durban CBD (L. Richards, unpublished data). In the non-breeding season, males roost singly but form harem groups during the breeding season (Monadjem et al. 2020b and references therein). Lactating females roost in maternity groups or alone with their young (Monadjem et al. 2020 and references therein).
Current population trend: Stable, as evidence by a recent phylogeographic study (Moir et al. 2020).
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: Yes, however there is some haplotypic structuring between South African samples and those from Eswatini (Moir et al. 2020)
Number of mature individuals in population: >10,000 individuals
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: The existence of subpopulations has not been confirmed, as no phylogeographic or landscape genetic studies have been conducted on this species within the assessment region.
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unlikely given the species’ broad distribution and large estimated population size.
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unlikely given the species’ broad distribution and large estimated population size.
Probability of extinction in the wild within 100 years: Unlikely given the species’ broad distribution and large estimated population size.
Population Genetics
While the species is widely distributed across Africa, a recent phylogenetic study showed bats within South Africa, form a distinct genetic clade (Taylor et al. 2022). This clade is thought to exist as a single metapopulation across the country. Moir et al. (2020) reported evidence of genetic structuring among sampled populations in the eastern regions of South Africa, likely attributable to philopatry. Distinct haplotype structuring was observed between samples from South Africa and those from Eswatini. Overall, the species exhibited high haplotype and nucleotide diversity, supporting the notion of a large stable metapopulation within the assessment region, yet signatures of historical population expansion (around ~191 kya) were evident.
Because of its wide distribution and it being the most common species in some areas, it is assumed that greater than 10,000 individuals exist within this metapopulation.
Habitats and ecology
Although this species has been recorded at elevations up to 4,207 m above sea level elsewhere in its range (Monadjem et al. 2024), within the assessment region and specifically in the Soutpansberg, it has not been observed above 1,031 m asl (Weier et al. 2019a). It is associated with well-wooded habitats such as riparian vegetation and forest patches, especially in the proximity of water (Monadjem & Reside 2008). It has also been recorded from lowland and montane tropical moist forest, dry and moist savannah, and similar wooded areas. They also occur in disturbed habitats, including logged forests (Monadjem et al. 2020b) and polluted riverscapes as well as wastewater treatment plants in the Durban region (Naidoo et al. 2011, 2013, 2016). Classified as an ‘urban-adaptor’ species (Schoeman 2015), they are frequently recorded in urban environments, including green spaces within the Durban CBD (L. Richards, unpublished data). They appear to be particularly common in localities with banana or Strelitzia plants (Skinner & Chimimba 2005). The rolled-up, terminal leaves of these plants are frequently used as roosting sites, although the species has also been recorded in the leaves of other plants and roofs (especially those made of thatch or palm leaves) (Taylor 2000; Skinner & Chimimba 2005; Monadjem et al. 2020b). They have thickened thumb and hindfoot pads that are thought to assist with their upright roosting position within furled leaves. They typically occupy a furled leaf only when the entrance opening measures between 7 and 24 cm; such roosts are abandoned or switched once the leaf unfurls, usually within 1–3 days (Van Cakenberghe & Happold, 2013). They usually roost singly or as small colonies of two to six individuals (Skinner & Chimimba 2005). Group composition varies considerably over the annual cycle and is linked to reproductive status (van der Merwe & Stirnemann 2009).
It is an insectivorous clutter-edge forager, characterised by low wing loading (Aldridge and Rautenbach 1987; Schoeman and Jacobs 2008), with a diet consisting predominantly of small Coleoptera, Lepidoptera and to a lesser extent Diptera (Fenton et al. 1977; Fenton & Thomas 1980; Schoeman 2006). They are most active after sunset for about 5 hours, while, after midnight, activity declines rapidly (Monadjem et al. 2010a). They forage by slow-hawking and exhibit highly manoeuvrable flight and are capable of easily navigating confined spaces (Van Cakenberghe and Happold 2013). Flight patterns are characterised by multiple banks and twists (Van Cakenberghe and Happold 2013).
Studies in KwaZulu-Natal found that mating occurs in May and sperm is stored in the female until mid-August when ovulation takes place (LaVal & LaVal 1977). In southern and central Africa, this species is considered seasonally monoestrous (LaVal & LaVal 1977; Happold & Happold 1990, 1996; Bernard et al. 1997).
Ecosystem and cultural services: As this species is insectivorous, it contributes to controlling insect populations that damage crops (Boyles et al. 2011; Kunz et al. 2011 – see below). Ensuring a healthy population of insectivorous bats can thus decrease the need for pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Unknown | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
| 18. Unknown | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: One-year (Bernard et al. 1997)
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean forearm length = 3.11 ±0.17 cm (Monadjem et al. 2020b)
Size at Maturity (in cms): Male: Mean forearm length = 2.98 ± 0.17 cm (Monadjem et al. 2020b)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): Female maximum forearm length = 3.40 cm; male maximum forearm length = 3. 40 cm (Monadjem et al. 2020b)
Size at Birth (in cms): No available measurement data, but neonates weigh approximately 1 g (Van Cakenberghe and Happold 2013)
Gestation Time: Approximately three months (LaVal and LaVal 1977)
Reproductive Periodicity: Exhibit seasonal monoestry, with matin occurring in May, followed by period of sperm-storage prior to ovulation and fertilisation that coincides with the onset of spring (LaVal and Laval 1977; Bernard et al. 1997). Births take place in November and early December (LaVal and LaVal 1977).
Average Annual Fecundity or Litter Size: Typically give birth to twins (Shortridge 1934; LaVal and LaVal 1977; Bernard et al. 1997)
Natural Mortality: Prey item for many predators including Bat Hawks (Macheiramphus alcinus (Allen et al. 1917) and Grey-headed Bush Shrikes (Malaconotus blanchoti, Van Jaarsveld 1988).
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Likely to be largely sedentary on account of low wing loading (poor dispersal capability) and the results of banding studies where the 30% of banded individuals were recovered (Van Cakenberghe and Happold 2013).
Congregatory: Reported to congregate when foraging, however, the composition of such foraging groups has not been further investigated (Van Cakenberghe and Happold 2013). Within the assessment region, between 1-8 individuals may roost together within a furled leaf (e.g. banana or strelitzia) or clumps of leaves (LaVal and LaVal 1977). In Malawi, females with young would roost separately to males, while at other times groups of 1 male with 1-10 females would be encountered (Happold and Happold 1996). A supposed maternity colony of 150 bats roosting in a thatched roof was observed in the Kruger National Park (LaVal and LaVal 1977); however, this atypical behaviour may be indicative of a cryptic diminutive species Afronyteris cf. helios (Van Cakenberghe and Happold 2013).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region. The species has been recorded agroecosystems including sugarcane fields (Mtsetfwa et al. 2018) and macadamia orchards (Weier et al. 2019). It is known to consume hemipteran pest species, inclusive of stink bugs, that are a major threat to the macadamia industry (Weier et al. 2019b).
Local Livelihood: None that has been reported
National Commercial Value: A recognised contributor to reducing pest insect populations within agroecosystems, including macadamia orchards.
International Commercial Value: A recognised contributor to reducing pest insect populations within agroecosystems, including macadamia that is a crop of considerable economic importance as a major export product.
End Use: None that has been reported
Is there harvest from captive/cultivated sources of this species? None that has been reported
Harvest Trend Comments: N/A
Threats
There appear to be no major threats to this widespread and seemingly adaptable species, which can persist in modified habitats. However, there is ongoing habitat loss from agricultural transformation, especially in KwaZulu-Natal (Jewitt et al. 2015), which may reduce the insect prey base for this species. Domestic cats represent a significant predation threat to bats in urbanised environments, including species such as the Banana Bat (based on grounded bat records from Bats KZN and Durban Natural Science Museum specimen records). Moreover, in urban areas, indiscriminate vegetation clearance eliminates critical foraging and roosting habitats. Water pollution, particularly contamination by heavy metals, may have long-term physiological impacts on Banana Bats foraging in affected areas. These impacts include haematological and genotoxic effects (Naidoo et al. 2015) and may also impair non-enzymatic antioxidant capacity (Hill et al. 2018) and affect mitochondrial function, leading to reduced energy utilization (Hill et al. 2016).
Conservation
This species occurs in many protected areas within the assessment region including large reserves like Great Limpopo Transfrontier Park, Greater Mapungubwe Transfrontier Conservation Area, iSimangaliso Wetland Park, Ndumo Game Reserve, Tembe Elephant Park, Phinda Private Game Reserve, uMkhuze Nature Reserve and Hluhluwe-iMfolozi Game Reserve. No direct conservation interventions are necessary for the species at present. However, outside of protected areas, it would benefit from holistic land management that reduces pesticide use and conserves buffer strips of natural vegetation to sustain insect biomass.
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes and maintain buffer strips of natural vegetation.
- Conserve and maintain remaining coastal forest habitats along the eastern seaboard, which remain a stronghold for the species within the assessment region.
- Reduce water and agricultural pesticide and herbicide run-off into waterways.
Research priorities:
- A wide-scale integrative taxonomy study is necessary to unravel the evolutionary history of this species complex.
- Further phylogenetic studies are required to resolve the species boundaries and clarify evolutionary relationships between A. nanus, A. helios and A. cf. helios.
- Studies on habitat specificity and distributional constraints are essential to accurately delineate the species’ range within the assessment region.
- Monitoring of known subpopulations should be performed to establish population size and trend.
- Quantifying the severity of local threats, inclusive of climate change, is necessary.
- Further research is required to assess long-term, population-level physiological effects of heavy metal contamination and other environmental pollutants.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
- Deposit any deceased specimen material in a recognized museum or established biorepository.
- Inspect garden vegetation, particularly wild banana and strelitzia plants, for roosting bats before undertaking any pruning.
- Keep domestic cats indoors at night.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Aldridge HD, Rautenbach IL. 1987. Morphology, echolocation and resource partitioning in insectivorous bats. The Journal of Animal Ecology 1:763-78.
Allen, J.A., Lang, H. and Chapin, J.P. 1917. The American Museum Congo Expedition collection of bats. Bulletin of the American Museum of Natural History: v. 37, article 18.
Bernard, R.T.F., Happold, D.C.D. and Happold, M. 1997. Sperm storage in a seasonally reproducing African vespertilionid, the banana bat (Pipistrellus nanus) from Malawi. Journal of Zoology (London) 241: 161–174.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Engelbrecht, F.A., Steinkopf, J., Padavatan, J. and Midgley, G.F., 2024. Projections of future climate change in Southern Africa and the potential for regional tipping points. In Sustainability of Southern African Ecosystems under Global Change: Science for Management and Policy Interventions (pp. 169-190). Cham: Springer International Publishing.
Fenton, M.B., Boyle, N.G.H., Harrison, T.M. and Oxley, D.J. 1977. Activity patterns, habitat use, and prey selection by some African insectivorous bats. Biotropica 9: 73–85.
Fenton, M.B. and Thomas, D.W. 1980. Dry-season overlap in activity patterns, habitat use, and prey selection by sympatric African insectivorous bats. Biotropica 12: 81–90.
Goodman, S.M. and Langrand, O. 1994. Inventaire biologique forêt de Zombitse. Recherches Pour le développement. Série Sciences Biologiques: 1–106.
Goodman, S.M., Rakotondramanana, C.F., Ramasindrazana, B., Kearney, T., Monadjem, A., Schoeman, M.C., Taylor, P.J., Naughton, K. and Appleton, B. 2015. An integrative approach to characterize Malagasy bats of the subfamily Vespertilioninae Gray, 1821, with the description of a new species of Hypsugo. Zoological Journal of the Linnean Society 173: 988–1018.
Happold, D.C.D. and Happold, M. 1990. The domiciles, reproduction, social organisation and sex ratios of the banana bat Pipistrellus nanus (Chiroptera, Vespertilionidae) in Malawi, central Africa. . Zeitschrift für Säugetierkunde 55: 145–160.
Happold, D.C.D. and Happold, M. 1996. The social organization and population dynamics of leaf-roosting banana-bats, Pipistrellus nanus Chiroptera, Vespertilionidae), in Malawi, east-central Africa. . Mammalia 60: 517–544.
Happold, D.C.D. 1987. The Mammals of Nigeria. Oxford University Press, London, UK.
Helbig, M.T., Datzmann, T., Mayer, F. and Fahr, J. 2010. Molecular phylogeny of African “pipistrelle” bats (Vespertilionidae) suggests new clades, rearrangement of genera, and extensive cryptic diversity within species. Pages 168–169 in Horácek I, Benda P, editors. 15th IBRC – the Conference Manual: Programme, abstracts, list of participants. Volume of abstracts of the 15th International Bat Research Conference. Prague, Czech Republic.
Hill K, Van Aswegen S, Schoeman MC, Claassens S, Jansen van Rensburg P, Naidoo S, Vosloo D. 2016. Foraging at wastewater treatment works affects brown adipose tissue fatty acid profiles in banana bats. Biology Open 5(2):92-9.
Hill K, Schoeman MC, Vosloo D. 2018. The brains of bats foraging at wastewater treatment works accumulate arsenic, and have low non-enzymatic antioxidant capacities. Neurotoxicology 1;69:232-41.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kearney, T.C., Volleth, M., Contrafatto, G. and Taylor, P.J. 2002. Systematic implications of chromosome GTG-band and bacula morphology for southern African Eptesicus and Pipistrellus and several other species of Vespertilioninae (Chiroptera: Vespertilionidae). Acta Chiropterologica 4: 55–76.
Koubínová D, Irwin N, Hulva P, Koubek P, Zima J. 2013. Hidden diversity in Senegalese bats and associated findings in the systematics of the family Vespertilionidae. Frontiers in Zoology10(1):48.
Kunz TH, Braun de Torrez E, Bauer D, Lobova T, Fleming TH. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
LaVal RK, LaVal ML. 1977. Reproduction and behaviour of the African banana bat Pipistrellus nanus. Journal of Mammalogy 58: 403–410.
Mammal Diversity Database. 2025. Afronycteris nanus (W. C. H. Peters, 1851) Banana Serotine. https://mammaldiversity.org/taxon/1005700
Moir, M., Richards, L.R., Cherry, M.I. and Rambau, R.V. 2022. Demographic responses of forest-utilizing bats to past climate change in South Africa. Biological Journal of the Linnean Society 130(4):850-68. Monadjem A, Reside A. 2008. The influence of riparian vegetation on the distribution and abundance of bats in an African savanna. Acta Chiropterologica 10: 339–348.
Monadjem, A., Ellstrom, M., Maldonaldo, C. and Fasel, N. 2010a. The activity of an insectivorous bat Neoromicia nana on tracks in logged and unlogged forest in tropical Africa. African Journal of Ecology 48: 1083–1091.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman, M.C. 2010b. Bats of Southern and Central Africa: A Biogeographic and Taxonomic Synthesis. University of the Witwatersrand Press, Johannesburg, South Africa.
Monadjem A, Demos T., Dalton D., Webala P., Musila S., Kerbis Peterhans J., Patterson B. 2020a. A revision of Pipistrelle-like bats (Mammalia: Chiroptera: Vespertilionidae) in East Africa with the description of new genera and species, Zoological Journal of the Linnean Society.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman, M.C. 2020b. Bats of Southern and Central Africa: A Biogeographic and Taxonomic Synthesis, Second Edition. University of the Witwatersrand Press, Johannesburg, South Africa.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Mtsetfwa, F., McCleery, R.A. and Monadjem, A. 2018. Changes in bat community composition and activity patterns across a conservation-agriculture boundary. African Zoology 1;53(3): 99-106.
Naidoo S, Mackey RL, Schoeman CM. 2011. Foraging ecology of insectivorous bats (Chiroptera) at a polluted and an unpolluted river in an urban landscape. Durban Museum Novitates 34: 21–28.
Naidoo S, Vosloo D, Schoeman MC. 2013. Foraging at wastewater treatment works increases the potential for metal accumulation in an urban adapter, the banana bat (Neoromicia nana). African Zoology 48(1):39-55.
Naidoo S, Vosloo D, Schoeman MC. 2015. Haematological and genotoxic responses in an urban adapter, the banana bat, foraging at wastewater treatment works. Ecotoxicology and Environmental Safety 114:304-11.
Naidoo S, Vosloo D, Schoeman MC. 2016. Pollutant exposure at wastewater treatment works affects the detoxification organs of an urban adapter, the Banana Bat. Environmental Pollution 208:830-9. Schoeman MC, Jacobs DS. 2008. The relative influence of competition and prey defenses on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS One 3: e3715.
Schoeman MC. 2006. The relative influence of competition and coevolution on the community structure of insectivorous bats in southern Africa. Ph.D. Thesis. University of Cape Town.
Schoeman MC. 2015. Light pollution at stadiums favors urban exploiter bats. Animal Conservation 19(2):120-30. Sherwin HA, Montgomery WI, Lundy MG. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Van Cakenberghe, V. and Happold, M. 2013. Pipistrellus nanus Banana pipistrelle; pp 639-643 in Happold M, Happold DCD (eds) Mammals of Africa: Volume IV Hedgehogs, Shrews and Bats. Bloomsbury Publishing, London
van der Merwe, M. and Stirnemann, R.L. 2009. Group composition and social events of the banana bat, Neoromicia nanus, in Mpumalanga, South Africa. South African Journal of Wildlife Research 39: 48–56.
Van Jaarsveld, J. 1988. African Goshawks and European Hobbies bat-hawking. Gabar 3: 29-31.
Weier, S.M., Linden, V.M., Gaigher, I., White, P.J. and Taylor, P.J. 2019a. Changes of bat species composition over altitudinal gradients on northern and southern aspects of the Soutpansberg mountain range, South Africa. Mammalia 81(1):49-60.
Weier, S.M., Moodley, Y., Fraser, M.F., Linden, V.M., Grass, I., Tscharntke, T. and Taylor, P.J. 2019b. Insect pest consumption by bats in macadamia orchards established by molecular diet analyses. Global Ecology and Conservation18:e00626.
