
Ansorge’s Free-tailed Bat
Mops ansorgei

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Mops ansorgei – (Thomas, 1913)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – MOLOSSIDAE – Mops – ansorgei
Common Names: Ansorge’s Free-tailed Bat, Ansorge’s Wrinkle-lipped Bat (English), Ansorge se Losstertvlermuis, Ansorge–osstertvlermuis (Afrikaans), Ansorge-Bulldoggfledermaus (German), Molosse d’Afrique du Sud, Molosse d’Ansorge, Tadaride d’Ansorge (French)
Synonyms: Nyctinomus ansorgei Thomas, 1913; Chaerephon ansorgei (Thomas, 1913); Chaerephon rhodesiae Roberts, 1946; Tadarida ansorgei (Thomas, 1913)
Taxonomic Note:
This species was previously listed under Tadarida (Hayman & Hill 1971; Largen et al. 1974; Corbet & Hill 1980), and is physically very similar to Tadarida aegyptiaca, especially in pelage colour, although somewhat smaller in size. However, Koopman (1975) and Freeman (1981) described the relationship between this species and Chaerephon bivitatta and C. bemmeleni, and resultantly included this species under the genus Chaerephon, which was later supported by Meester et al. (1986) and Koopman (1993). Lamb et al. (2011) found strong support for the monophyly of the Chaerephon and Mops taxa (about 17.2 Mya), but not for either of the genera separated. Ammerman et al. (2012) also found support for a Mops – Chaerephon clade, which resulted in paraphyly of Chaerephon, and confirm again that Chaerephon is distinct from Tadarida. A further morphological study by Gregorin and Cirranello (2015) confirmed the paraphyly of Chaerephon but was unable to resolve the relationships with the genus. Simmons and Cirranello (2020) include all the Chaerephon species in the genus Mops. No subspecies have been identified.
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Naidoo, T.1, Richards, L.R.1, Balona, J.2 & da Silva, J.M.3
Reviewers: Richardson, E.J.4
Institutions: 1Durban Natural Science Museum, 2Gauteng and Northern Regions Bat Interest Group, 3South African National Biodiversity Institute 4Independent Consultant at Richardson & Peplow Environmental
Previous Assessors and Reviewers: Monadjem, A., Taylor, P., Cohen, L., Jacobs, D., MacEwan, K., Richards, L.R., Schoeman, C. & Sethusa, T.
Previous Contributors: Roxburgh, L., Raimondo, D. & Relton, C.
Assessment Rationale
The species is widely but sparsely distributed across the northeastern regions of the assessment region and has been recorded predominantly from protected areas. The estimated extent of occurrence is 106,515 km2. It occurs in hard-to-reach places and is difficult to sample owing to its fast, high-flying habits, but the population is suspected to be stable. There are no known major threats to the species, and it is thus listed as Least Concern.
Regional population effects: This species has been commonly recorded in Zimbabwe and genetic exchange between individuals occurring within and outside of the assessment region is suspected.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Naidoo T, Richards LR, Balona J & da Silva JM. 2025. A conservation assessment of Mops ansorgei. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Mops ansorgei occurs across much of eastern sub-Saharan Africa through to the Kruger National Park and northern KwaZulu-Natal (ACR 2024), with the core of its range being Zimbabwe (Monadjem et al. 2020). It has not been recorded from Botswana. A possible geographically isolated population is restricted to western Angola (Monadjem et al. 2020). A habitat model suggests that suitable conditions occur in southern Mozambique (Monadjem et al. 2020). Within the assessment region it is found sparsely in the Limpopo, Mpumalanga, and KwaZulu-Natal provinces, recorded mostly from protected areas.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 400
Elevation Upper Limit (in metres above sea level): 2000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
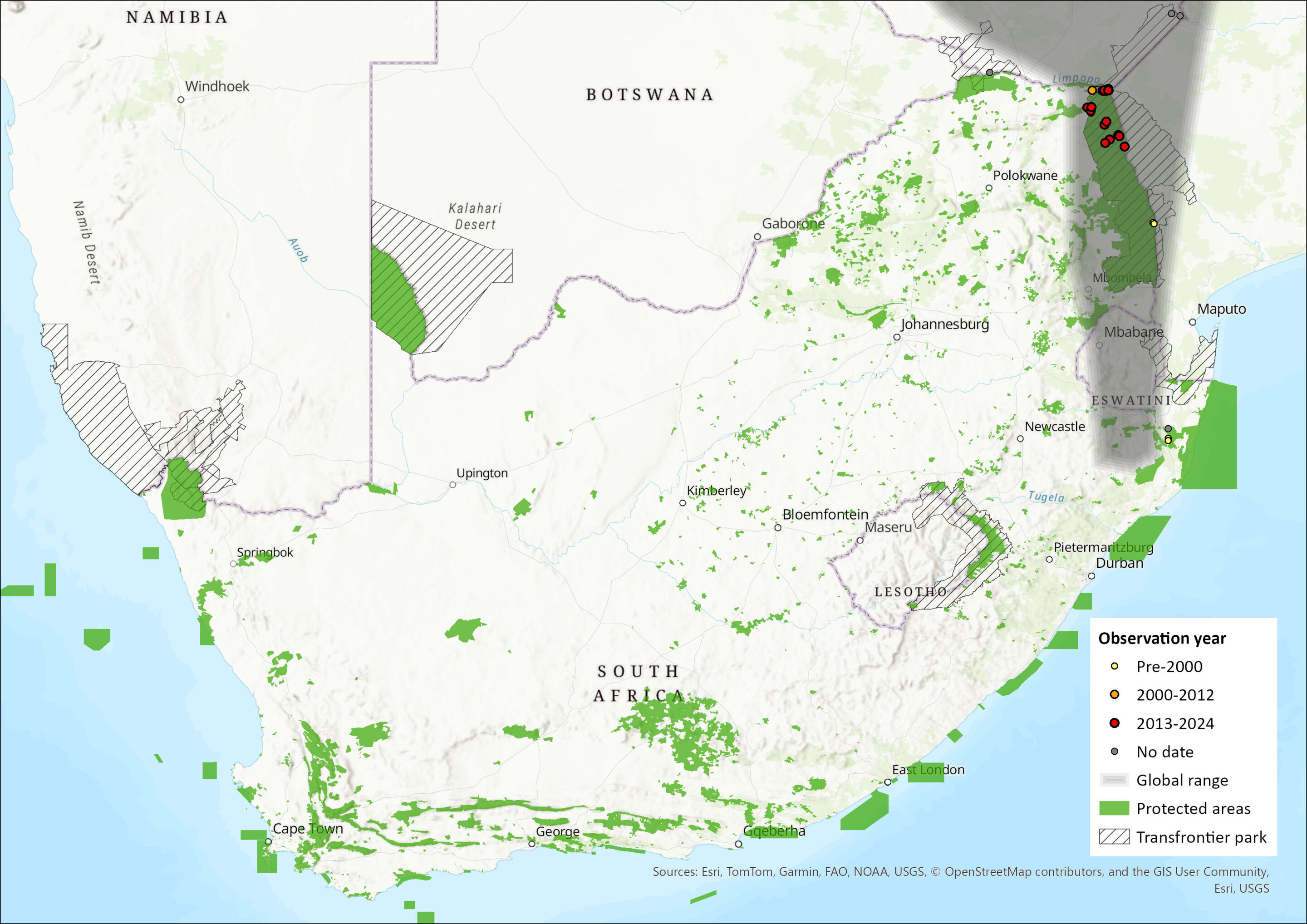 Figure 1. Distribution records for Ansorge’s Free-tailed Bat (Mops ansorgei) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Ansorge’s Free-tailed Bat (Mops ansorgei) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Mozambique | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | – |
| Zambia | Extant | Native | – | Resident |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Studies of bat species have shown that environmental suitability is associated with temperature, precipitation, topographic variables, vegetation cover and waterway presence (Koch et al. 2020, Lee et al. 2012 and Uusitalo et al. 2024). However, the specific effects of climate change have not been documented for this species. Mops ansorgei is associated with savanna woodland habitats of the extreme eastern, and northeastern regions of South Africa. It is therefore likely that increased drought frequency in some of the semi-arid savannas may impact the vegetation and insect prey base of this species (likely to be moths as is the case for similar-sized large molossids) (Goana et al. 2024). This is turn may result in distributional changes and altered roosting patterns.
Population Information
In the assessment region this species is suspected to be uncommon as it is difficult to capture, and most records appear to be isolated (Skinner and Chimimba 2005). However, colonies outside of the assessment region have been recorded as consisting of hundreds of individuals (Mickleburgh et al. 2008). This species is well represented in museums from within the core of its range in Zimbabwe, where more than 250 individuals were examined in Monadjem et al. (2020).
Current population trend: Stable
Continuing decline in mature individuals? Not determined, but unlikely
Extreme fluctuations in the number of subpopulations: Not known
Continuing decline in number of subpopulations: Not known
All individuals in one subpopulation: Suspected, based on ecomorphology and dispersal capability
Number of mature individuals in largest subpopulation: Not determined, but can roost in large colonies elsewhere in Africa
Number of Subpopulations: One, comprised of different colonies
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Not known, but unlikely)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Not known
Probability of extinction in the wild within 100 years: Not known
Population Genetics
No population-level genetic study has been conducted on this species; however, dispersal capability is thought to be high based on the species’ high wing loading as is seen with other similar-sized molossids (Taylor et al. 2012). Consequently, it is expected to exist as a metapopulation, possibly well connected to populations in neighbouring countries. Due to limited information on the overall population size within the assessment region. it is not possible to estimate effective population size (Ne) using proxy methods. A population genetic study incorporating samples within and outside of the assessment region would be highly informative in understanding the genetic health of this species.
Habitats and ecology
This species inhabits dry woodland savannah habitats, usually near to rugged hills and mountains with rocky cliffs (Monadjem et al. 2020). Natural roost sites include narrow cracks in rocks, especially on cliff faces (Cotterill and Fergusson 1993), but is known to roost in buildings, roofs of houses, mine adits and expansion joints of bridges (Cotterill, 2013; Monadjem et al. 2020). The availability of natural roost sites is considered a primary habitat requirement for this species (Skinner and Chimimba 2005). Coleoptera, Trichoptera and Lepidoptera constitute most of this species’ diet (Fenton 1985). It is a gregarious species and roosts communally, usually in small to medium-sized groups (Allen et al. 1917). In Zimbabwe, reproductive studies have shown that it generally produces a single young in late November, with a second birth peak occurring in April (at the end of the warm, wet season) (Monadjem et al. 2020).
Ecosystem and cultural services: As this species is an aerial insectivore, it may contribute to controlling insect populations that destroy crops (Boyles et al. 2011, Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus result in a decrease in pesticide use.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Marginal | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 6. Rocky areas (eg. inland cliffs, mountain peaks) | – | Suitable | – |
Life History
Generation Length: Suspected to be 1-3 years
Age at Maturity: Female or unspecified: Approximately 1 year based on available data for other molossid species
Age at Maturity: Male: Approximately 1 year based on available data of other molossid species
Size at Maturity (in cms): Female: Mean total length = 10.60 ±0.61 cm; Mean forearm length = 4.60 ±0.15 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean total length = 11.60 ±0.73 cm; Mean forearm length = 4.72 ±0.28 cm (Monadjem et al. 2020)
Longevity: Not known
Average Reproductive Age: Approximately 1 year based on available data of other molossids
Maximum Size (in cms): Not known
Size at Birth (in cms): Not known
Gestation Time: Presumed to be approximately 3 months
Reproductive Periodicity: Seasonal polyoestry with young born in the austral summer and mid-autumn (Cotterill 2013)
Average Annual Fecundity or Litter Size: Presumed to be 1 pup
Natural Mortality: Preyed upon by Barn owls (Tyto alba) and Eagle-owls (Bubo capensis) (Cotterill 1992)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown
Congregatory: Yes, in other parts of Africa
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region. However, in some parts of West Africa it is utilised (and possibly even over-utilised) as bushmeat (Mickleburgh et al. 2008).
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
No major threats have been identified for this species within the assessment region, although minor threats such as human disturbance at roost sites and the climatic impacts associated with global climate change have been identified for other bat species and may similarly impact this species. In parts of West Africa this species may be vulnerable to overexploitation as a food source (Mickleburgh et al. 2008). There are also historic records from northeastern Democratic Republic of Congo of people suffocating bucket-loads of Ansorge’s Free-tailed Bats to death in their roosting sites (Allen et al. 1917).
With wind energy installations moving into parts of KwaZulu-Natal and Mpumalanga, this could pose a future threat to Ansorge’s Free-tailed Bat as it is an open-air forager (Baerwald et al. 2008, Cryan and Barclay 2009, Rydell et al. 2010, Rollins et al. 2012), and is categorised as high risk for turbine related fatalities (MacEwan et al. 2020). Bats are attracted to wind farm sites which often tend to have higher insect densities at well-lit turbines (Voigh et al. 2024). When bats fly near to turbine blades, they either collide directly with the blade or they experience barotrauma. Barotrauma is tissue damage caused by rapid excessive changes in air pressure near turbine blades (Baerwald et al. 2008, Rydell et al. 2010). Additionally, this species may face persecution when roosting in the roofs and crevices of buildings, even in protected areas.
Conservation
Within the assessment region, this species has been found in the Kruger National Park, Mapungubwe National Park, Vhembe Biosphere Reserve, and Mkhuze Game Reserve in KwaZulu-Natal. No specific conservation efforts are necessary at present, although they are likely to benefit from enhanced protection of key roost sites and awareness of the species ecological value in controlling insect populations.
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes.
Research priorities:
- Field surveys are needed to identify further subpopulations and delimit its distribution range more accurately.
- The reproductive and feeding ecology of Ansorge’s Free-tailed Bat requires further investigation.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution. Ansorge’s Free-tailed Bat closely resembles the Egyptian Free-tailed Bat (Tadarida aegyptiaca) but is slightly smaller in size and lacks the unusually flattened skull seen in T. aegyptiaca (Rautenbach 1997).
Bibliography
ACR. 2024. African Chiroptera Report 2023. Van Cakenberghe, V.and Seamark, E.C.J. (Eds.) African Chiroptera Project, Pretoria. i – xviii; 1 – 1210 pp.
Allen JA, Lang H, Chapin J. 1917. The American Museum Congo Expedition collection of bats. Bulletin of the American Museum of Natural History 37: 405-478.
Ammerman, L. K., D. N. Lee and T. M. Tipps. 2012. First molecular phylogenetic insights into the evolution of free-tailed bats in the subfamily Molossinae (Molossidae, Chiroptera). Journal of Mammalogy, 93 (1): 12 – 28. doi: http://dx.doi.org/10.1644/11-MAMM-A-103.1
Baerwald EF, D’Amours GH, Klug BJ, Barclay RM. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18: 695–696.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Corbet, G.B. and Hill, J.E. 1980. World list of mammalian species. British Museum, London, UK.
Cotterill FPD, Fergusson RA. 1993. Capturing free-tailed bats (Chiroptera: Molossidae): the description of a new trapping device. Journal of Zoology 231: 645–651.
Cotterill, F.P.D. 2013. Tadarida ansorgei Ansorge’s Free-tailed Bat. Pp 494-495 in Happold, M. & Happold, D.C.D. (eds) Mammals of Africa: Volume IV. Bloomsbury Publishing, London.
Cryan PM, Barclay RM. 2009. Causes of bat fatalities at wind turbines: hypotheses and predictions. Journal of Mammalogy 90: 1330–1340.
Fenton MB. 1985. The feeding behaviour of insectivorous bats: echolocation, foraging strategies, and resource partitioning. Transvaal Museum Bulletin 21: 5–19.
Freeman, P.W. 1981. A Multivariate Study of the Family Molossidae (Mammalia, Chiroptera): Morphology, Ecology, Evolution. Fieldiana: Zoology 7: 1-173.
Gregorin, R. and A. Cirranello. 2015. Phylogeny of Molossidae Gervais (Mammalia: Chiroptera) inferred by morphological data. Cladistics: The International Journal of the Willi Henning Society, 32 (1): 2 – 35 (for 2016). doi: 10.1111/cla.12117.
Gaona FP, Delabye S, Potocky P, Govorov V, Cuda J, Foxcroft LC, Garlacz R, Hejda M, MacFadyen S, Pyrcz T, Pyskova K. 2024. Climate-driven vegetation characteristics shape phytophagous and carnivorous insect diversity in South African savannahs. bioRxiv. 2024:2024-07.
Hayman, R.W. and Hill, J.E. 1971. Order Chiroptera. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 73. Smithsonian Institution Press, Washington, D.C., USA.
Koopman, K.F. 1975. Bats of the Sudan. Bulletin of the American Museum of Natural History 154(4): 355-444.
Koch, L. K., Cunze, S., Kochmann, J., & Klimpel, S. 2020. Bats as putative Zaire ebolavirus reservoir hosts and their environmental suitability in Africa. Scientific Reports, 10(1): 14268. https://doi.org/10.1038/s41598-020-71226-0 Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Lamb, J. M., T. M. C. Ralph, T. Naidoo, P. J. Taylor, F. Ratrimomanarivo, W. T. Stanley and S. M. Goodman. 2011. Toward a molecular phylogeny for the Molossidae (Chiroptera) of the Afro-Malagasy region. Acta Chiropterologica, 13 (1): 1 – 16. doi: 10.3161/150811011X578589.
Largen, M.J., Kock, D. and Yalden, D.W. 1974. Catalogue of the mammals of Ethiopia 1. Chiroptera. Monitore Zoologico Italiano Supplemento 16(5): 221–298.
Lee, D. N., Papeş, M., & Van Den Bussche, R. A. 2012. Present and potential future distribution of common vampire bats in the Americas and the associated risk to cattle. PLoS One, 7(8): e42466. https://doi.org/10.1371/journal.pone.0042466
MacEwan, K., Sowler, S., Aronson, J., and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities – ed 5. South African Bat Assessment Association
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Mickleburgh, S., Hutson, A.M., Bergmans, W. and Cotterill, F.P.D. 2008. Hipposideros vittatus. (Accessed: 16 February 2016).
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Ed., University of Witwatersrand Press, Johannesburg.
Rautenbach N. 1997. Ansorge’s free-tailed bat Chaerephon ansorgei. Struik Publishers, Cape Town, South Africa.
Rollins KE, Meyerholz DK, Johnson GD, Capparella AP, Loew SS. 2012. A forensic investigation into the etiology of bat mortality at a wind farm: barotrauma or traumatic injury? Veterinary Pathology Online 49: 362–371.
Rydell J, Bach L, Dubourg-Savage MJ, Green M, Rodrigues L, Hedenström A. 2012. Bat mortality at wind turbines in northwestern Europe. Acta Chiropterologica 12(2):261-74.
Sherwin HA, Montgomery WI, Lundy MG. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Simmons, N. B. and A. L. Cirranello. 2020. https://batnames.org/explore.html. Accessed: 10 May 2020.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, England.
Taylor PJ, Goodman SM, Schoeman MC, Ratrimomanarivo FH, Lamb JM. 2012. Wing loading correlates negatively with genetic structuring of eight Afro-Malagasy bat species (Molossidae). Acta Chiropterologica, 14(1):53-62.
Taylor PJ, Schoeman MC, Monadjem A. 2013. Diversity of bats in the Soutpansberg and Blouberg Mountains of northern South Africa: complementarity of acoustic and non-acoustic survey methods. South African Journal of Wildlife Research 43(1):12-26.
Uusitalo RJ, Jackson RT, Lunn TJ, Korhonen EM, Ogola JG, Webala PW, Sironen TA, Forbes KM. 2024. Current and future environmental suitability for bats hosting potential zoonotic pathogens in rural Kenya. Ecology and Evolution 14(6): e11572.
Voigt, C.C., Bernard, E., Huang, J.C.C., Frick, W.F., Kerbiriou, C., MacEwan, K., Mathews, F., Rodríguez-Durán, A., Scholz, C., Webala, P.W. and Welbergen, J. 2024. Toward solving the global green–green dilemma between wind energy production and bat conservation. BioScience, 74(4): 240-252.
