Angoni Vlei Rat
Otomys angoniensis

2025 Red list status
Least Concern
Regional Population Trend
Increasing
Change compared
to 2016
No Change
Overview
Otomys angoniensis – Wroughton, 1906
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Otomys – angoniensis
Common Names: Angoni Vlei Rat (English), Angoni-vleirot (Afrikaans)
Synonyms: Otomys maximus Roberts, 1924
Taxonomic Note:
Unpublished molecular and morphometric data show that East and southern African Otomys angoniensis may be distinct species (Monadjem et al. 2015). While O. a. maximus has been regarded as a separate species, it is provisionally included in O. angoniensis due to mitochondrial DNA similarity (S. Maree unpubl. data) and morphometric overlap (Bronner & Meester 1988). Three subspecies are recognised (Taylor 2013).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.1 & da Silva, J.M.2
Reviewer: Oosthuizen, M.3
Institutions:1University of the Free State,2South African National Biodiversity Institute,3University of Pretoria
Previous Assessors and Reviewers: Taylor, P., Baxter, R. & Child, M.F.
Previous Contributors: Avenant, N., Avery, M., MacFadyen, D., Monadjem, A., Palmer, G. & Wilson, B.
Assessment Rationale
This species is listed as Least Concern in view of its wide distribution within the assessment region, observed range expansions in Limpopo and North West provinces, ability to survive in modified habits, and because there are no threats likely to cause range-wide population decline. The savanna habitat is well-protected and thus this species is not expected to be significantly impacted by habitat loss. Conversely, climate change is projected to expand its area of occupancy by 30–123% between 1975 and 2050, mirroring the expansion of savanna habitat in the region. For example, evidence suggests it is usurping the grassland-adapted species O. auratus in the Soutpansberg Mountains, due to shifts in vegetation structure (Taylor et al. 2016).
Regional population effects: It is a widespread species existing in largely intact habitats. Thus, the regional population is presumably connected.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Taylor P & da Silva JM. 2025. A conservation assessment of Otomys angoniensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
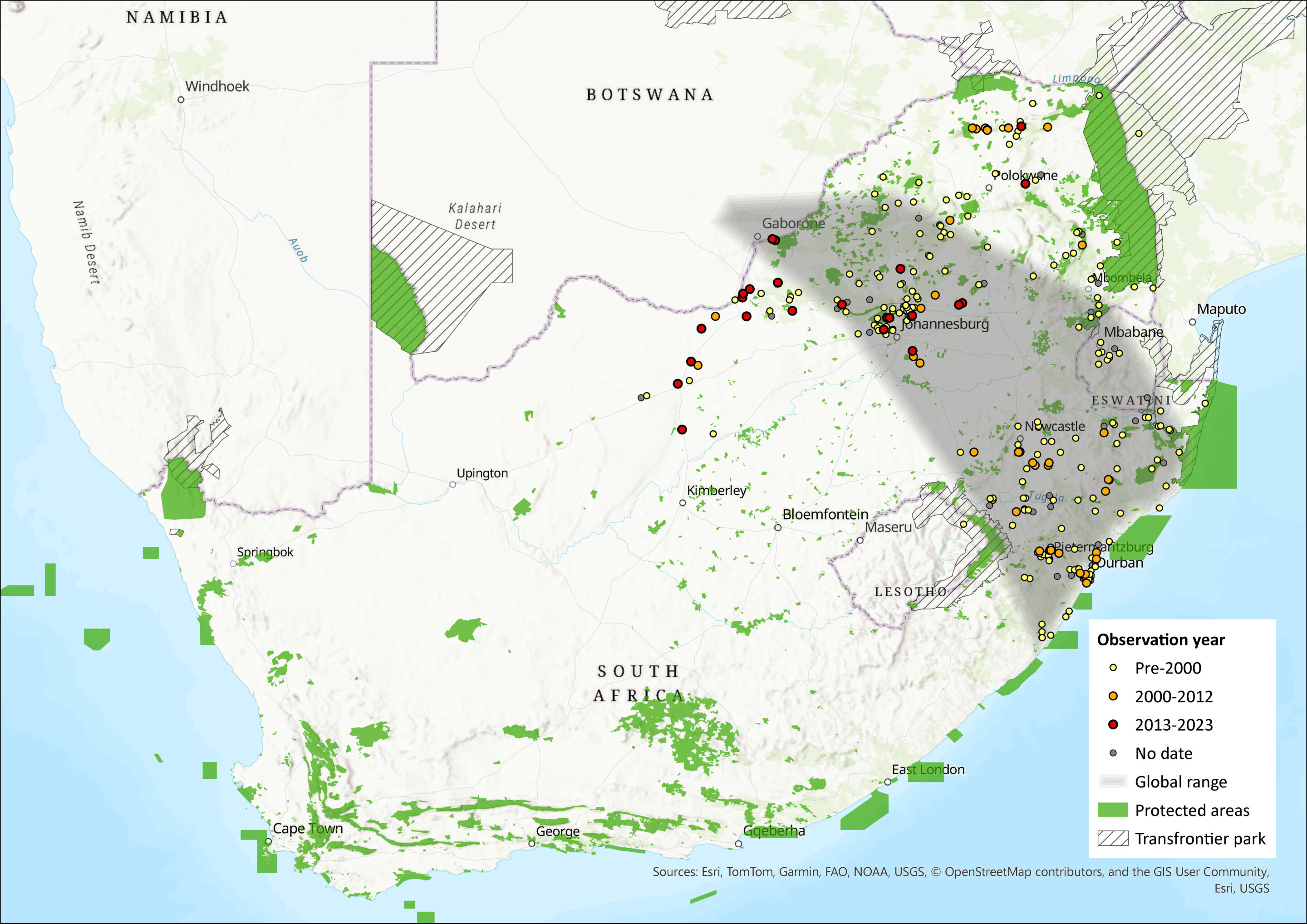
This is the most widespread of all Otomys species, occurring in mesic savannahs in southern and central Africa, as well as the foothills of major mountain ranges in Malawi, the western slopes of the Albertine Rift, Uganda, Kenya and northern Tanzania (Monadjem et al. 2015). Similarly, in the Drakensberg Range, it occurs on the lower slopes in savanna habitats. It occurs in the northern and northeastern provinces of South Africa, including Eswatini. Records from the Eastern Cape (De Graaff 1981; Bronner & Meester 1988) are not corroborated by museum specimens (Taylor 2013).
It has mostly an allopatric distribution with O. auratus overlapping only at the interface of the Grassland and Savannah biomes, around 1,000 m asl in KwaZulu-Natal and Eswatini and 1,300 m asl in Limpopo (Monadjem 1998; Taylor 1998; Nengovhela 2014). However, a recent range expansion has been documented in the Soutpansberg Mountains, Limpopo where it has expanded from the foothills to the peaks and has displaced O. auratus (savanna species replacing grassland species) due to vegetation transition from grassland to thicket (Taylor et al. 2016). Similarly, range expansions have been documented in North West Province where Power (2014) confirmed local records at both S. A. Lombard and Bloemhof Nature Reserves, where previous intensive surveys in the areas did not reveal this species (Newbery 1995). Range expansion is projected to continue due to expansion of savanna habitats in response to climate change. Taylor et al. (2016) used MaxEnt models to determine an increase in area of occupancy between 30% (with limited dispersal) to 123% (with unrestricted dispersal) between 1975 and 2050.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Angoni Vlei Rat (Otomys angoniensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Presence Uncertain | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Some populations may expand their range with climate change (as in the Soutpansberg, Taylor et al. 2016), but it does not do well in degraded habitats. Climate change is projected to expand its area of occupancy by 30–123% between 1975 and 2050, mirroring the expansion of savanna habitat in the region.
Population Information
It is common to abundant in suitable habitats (Taylor 2013) and, given its currently expanding range, we suspect there are over 10,000 mature individuals within the assessment region and the population is likely to be increasing. More field surveys and long-term monitoring should be undertaken to confirm this. In Kenya, a population was recorded at a density of 30 animals / ha (Taylor 2013).
Population Information
Current population trend: Increasing
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic studies have been conducted on this species; however, it is expected to exist as a single metapopulation due to being a fairly abundant species, capable of utilizing varied habitats. Based on the population size assumptions (see above), the effective population size (Ne) is thought to exceed the Ne 500 threshold (i.e., > 1000 assuming a 0.1 Nc/Ne conversion ration).
Habitats and ecology
It occurs in well-watered savanna grassland (associated with wetlands), seasonally flooded grassland and wetlands generally below 1,000 m asl. It is usually found near water, but drier habitats can be occupied during the wet season (Taylor 2013). It can be found in modified habitats, such as in pastureland but usually not when livestock is present. Distinctive trails are made through dense grass and used by other small mammals (Taylor 2013).
Where O. auratus and O. angoniensis co-occur, the former is associated with sedges and grasses adapted to densely vegetated wetlands with wet soils, while the latter is associated with plants that typically grow in the drier margins of wetlands (Davis 1973). More recently, at Telperion Nature Reserve, O. auratus was found in a single wetland area, whereas O. angoniensis was found everywhere, thus, O. angoniensis is more flexible in terms of habitat, and they also occur in the wetter areas and co-occur with O. auratus (M. Oosthuizen pers. comm.).
Ecosystem and cultural services: They are an important prey species (sensu Malan 2001; Taylor 2013).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 5.4. Wetlands (inland) -> Wetlands (inland) – Bogs, Marshes, Swamps, Fens, Peatlands | – | Suitable | – |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 2-3 years in captivity
Average Reproductive Age: 5 weeks for females, 8 weeks for males (Phillips et al 1997)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 37 days (Phillips et al 1997)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: Mean litter size 3.1 (Phillips et al 1997)
Natural Mortality: (Not specified)
Does the species lay eggs? no
Does the species give birth to live young: yes
Does the species exhibit parthenogenesis: no
Does the species have a free-living larval stage? no
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species. However, it can be locally threatened by habitat degradation due to overgrazing by domestic livestock and/or wildlife ranching. Further studies are needed to determine the resilience of this species to land-cover change. Unlike other Otomys species, climate change is projected to increase its area of occupancy along with savanna expansion and extend widely into grassland areas currently occupied by O. auratus (Taylor et al. 2016). For example, habitat change from open grassy plains to dense thicket has seen O. angoniensis completely replace O. auratus at a key zone of sympatry in the Soutpansberg Mountains, Limpopo between 1923 and 2015 (Taylor et al. 2016).
Habitat trend: Increasing. Savanna habitat is largely intact (Driver et al. 2012), and climate change is projected to increase its range (Taylor et al. 2016). However, wetlands are the most threatened ecosystem in South Africa (Driver et al. 2012). The South African National Land-Cover change report found a 32.8% decline in natural wetlands nationally from 1990–2013/14, which is a combination of both genuine wetland loss through anthropogenic activities and the generally drier conditions currently than in 1990 (GeoTerraImage 2015). Wetland loss should thus be monitored for its impacts on this species.
Conservation
This species is present in many protected areas, including Kruger National Park. Although no specific interventions are necessary at present, the following interventions are encouraged that will benefit all small mammals:
- Using previously cultivated areas for development instead of remaining natural areas.
- Land managers should maintain a vegetation buffer around wetlands to reduce impacts of land-use practices (Driver et al. 2012).
- Holistic management of ranchlands: including de-stocking, rotational grazing and buffering wetland vegetation, are encouraged.
Recommendations for land managers and practitioners:
- Land managers should decrease stocking rates to maintain vegetation around wetlands.
- Prioritise old fields for development in systematic conservation planning.
Research priorities:
- Effects of savanna habitat loss and degradation.
- Effects of range expansion on other Otomys species.
Encouraged citizen actions:
- Report vlei rat sightings on virtual museum platforms (for example, iNaturalist and MammalMAP): The feeding signs of this species are easy to detect by short chopped lengths of grass and green moist faecal pellets (Skinner & Chimimba 2005; Taylor 2013).
Bibliography
Bronner GN, Meester JAJ. 1988. Otomys angoniensis. Mammalian Species 306: 1–6.
Davis, R.M. 1973. The ecology and life history of the vlei rat, Otomys irroratus (Brants, 1827), on the van Riebeeck Nature Reserve, Pretoria. Ph.D Thesis. University of Pretoria.
De Graaff, G. 1981. The Rodents of Southern Africa. Butterworths, Durban, South Africa.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
GeoTerraImage. 2015. 1990-2013/14 South African National Land-Cover Change. DEA/CARDNO SCPF002: Implementation of Land-Use Maps for South Africa. Project Specific Data Report.
Malan G. 2001. Otomyine rodent refuge habitat: does it represent habitat quality for pale chanting-goshawk families? South African Journal of Wildlife Research 31: 49–58.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41: 45-59.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Nengovhela A. 2014. Investigating past, present and future distributions of cryptic species of vlei rats (Otomys auratus, O. irroratus s.s. and O. angoniensis) in South Africa, with a focus on Limpopo Province. M.Sc. Thesis. University of Venda.
Nengovhela, A., Baxter, R.M. and Taylor, P.J. 2016. Temporal changes in cranial size in South African vlei rats (Otomys): evidence for the “third universal response to warming.”. Zoology 50: 233-239.
Newbery CH. 1995. Mammal checklist of the provincial nature reserves. North West Parks Board, Rustenburg, South Africa.
Phillips, J., Kearney, T., Pillay, N. & Willan, K. 1997. Reproduction and postnatal development in the Angoni vlei rat Otomys angoniensis (Rodentia, Muridae). Mammalia 61:219-229.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor PJ. 2013. Otomys angoniensis Angoni Vlei Rat. In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits., pp. 577– 578. Bloomsbury Publishing, London, UK.
Taylor, P.J., Nengovhela, A., Linden, J. and Baxter, R.M. 2016. Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia 80: 359–375.