
Angolan Hairy Bat
Cistugo seabrae

2025 Red list status
Vulnerable
Regional Population Trend
Unknown
Change compared
to 2016
Uplisted
Overview
Cistugo seabrae – Thomas, 1912
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – CISTUGIDAE – Cistugo – seabrae
Common Names: Angolan Hairy Bat, Angolan Wing-gland Bat, Angolan Winggland Bat, Seabra’s Wing-gland Bat (English), Angola-langhaarvlermuis (Afrikaans)
Synonyms: Cistugo seabrai Thomas, 1912 [orth. error]; Myotis seabrai (Thomas, 1912)
Taxonomic Note:
The species has historically been included in the genus Myotis (Family: Vespertilionidae), but molecular studies show that the genus is distinct from all other Vespertilionidae (Stadelmann et al. 2004) and is distinct enough to be placed in its own family, Cistugidae (Lack et al. 2010). Given their similarity, the taxonomic relationship between C. seabrae and C. lesueuri largely remained controversial (Corbet & Hill 1980; Meester et al. 1986; Simmons 2005; Kearney 2013; Monadjem et al. 2020;). They are presently recognised as two distinct species that can be separated based on mitochondrial DNA sequence data and cranial morphometry (Taylor et al. 2024).
Red List Status: VU, Vulnerable D1 (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1 & da Sila, J.M.2
Reviewer: Howard, A.3
Institutions:1Durban Natural Science Museum, 2South African National Biodiversity Institute, 3University of the Free State
Previous Assessors and Reviewers: Jacobs, D., MacEwan, K., Cohen, L., Monadjem, A., Richards, L, Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Roxburgh, L., Raimondo, D., Page-Nicholson, S. & Relton, C.
Assessment Rationale
This species is known from six locations in South Africa, in the northwestern Northern Cape Province. Its extent of occurrence is estimated at 11,414 km2, with a population of fewer than 1,000 individuals suspected. It thus qualifies for Vulnerable D1. The species has a low wing loading suggesting that dispersal capability may limited. It occupies desert and semi-desert areas that receive < 100 mm of precipitation per annum, within the regional assessment area. It is potentially threatened by mining activities and by the expansion of wind farm sites in the Northern Cape, but there is no evidence for decline as yet. However, these potential threats should be monitored because, if the population is shown to be declining, this species will qualify for a more threatened listing.
Regional population effects: This species is present in Namibia, albeit it considered rare (T. Laverty, pers. comm.) and the population is suspected to be continuous due to connected habitat and low human population pressure. Thus, although it has a relatively low wing loading (Schoeman & Jacobs 2008), we assume rescue effects are possible.
Reason for change
Reason(s) for Change in Red List Category from the Previous Assessment: The species remains known from only a limited number of localities (n = 6) in the assessment region; this despite recent sampling efforts in the known occurrence area/s. Arid-adapted species, such as C. seabrae are expected to exhibit range contraction in response to increasing aridity coupled to climate change. The species low dispersal capability and recent evidence for genetic structuring between South African and nearest neighbour colonies, suggest that over time, gene flow and hence rescue efforts may be limited. Although typically not a high-risk species (MacEwan et al. 2020), the expansion of wind farms and the associated footprint may pose a threat. The population has <1,000 mature individuals.
Red List Index
Red List Index: Uplisted
Recommended citation: Richards LR & da Silva JM. 2025. A conservation assessment of Cistugo seabrae. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This southern African species ranges from the type locality of Moçâmedes in southwestern Angola, southwards through western Namibia (the four localities of Berseba, Huab River mouth, Lüderitz district, and 70 km west of Maltahohe) (Herselman 1980; Skinner and Chimimba 2005) and six locations in the Northern Cape province of South Africa (Augrabies Falls National Park, Goebag, Goodhouse, Khamkirri, Springbok, Steyerskraal; Monadjem et al. 2020).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,871 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
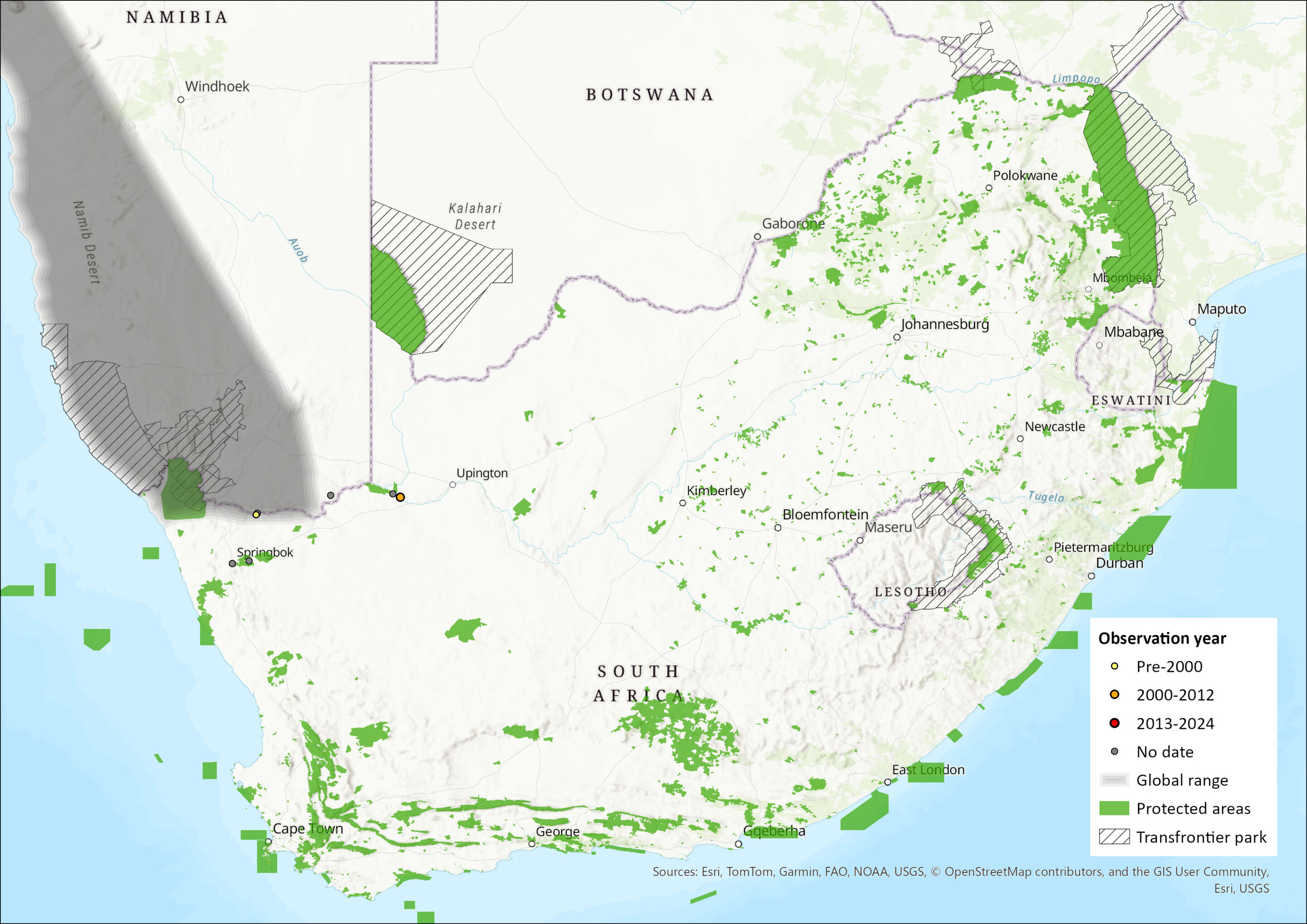 Figure 1. Distribution records for Angolan Hairy Bat (Cistugo seabrae) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Angolan Hairy Bat (Cistugo seabrae) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
The species is classified as arid-adapted and is reported from localities within the assessment regions with a mean annual rainfall of <100 mm (Roberts 1951, Skinner and Chimimba, 2005). According to the predictive models of Taylor et al. (2024), precipitation of the wettest month is a postulated limiting factor in the distributional range of the species. It is therefore expected that this seldomly encountered bat species may be negatively impacted (e.g. physiological stress, leading to increased mortality and/or reduced fecundity, see Festa et al. 2023), by the effects of climate change. Conversely, the species has shown adaptation to both natural and anthropogenic foraging and roosting sites; such habitats may become increasingly important to the species and potentially other arid-adapted species, as climate change progresses.
Population information
The Angolan Hairy Bat appears to avoid mist nets and is thus rarely recorded. Subsequently, it is poorly represented in museums, with just 26 specimens examined in Monadjem et al. (2020). There are likely to be fewer than 1,000 individuals occurring within the assessment region.
Current population trend: Unknown, but there is no evidence of a decline.
Number of mature individuals in population: Unknown, but likely to be less than 1,000.
Number of mature individuals in largest subpopulation: Uncertain, but < 1,000 mature individuals.
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: See below
Number of Subpopulations: Individuals within the assessment region are assigned to a “southern clade”, whereas those individuals from Namibia upwards to Angola comprise a “northern clade’ (Taylor et al. 2024; see below).
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
No population genetic study has been conducted on the species; however, an integrated phylogenetic study using mitochondrial DNA (incorporating acoustic, morphological and ecological data) found clear intraspecific differentiation, identifying a northern and southern clade, which diverged in the mid-Pleistocene (Taylor et al. 2024). The northern clade extends from southern Angola to Namibia and therefore is outside of the assessment region, while the southern clade is restricted to a small area of the Namaqualand-Richtersveld Ecoregion south of the Orange River in the Northern Cape, South Africa. The break coincides closely with the lower Orange River valley (Taylor et al. 2024). It is expected that this clade exists as a metapopulation within the region, but this will need to be confirmed using a fine-scale genomic assessment to investigate the current population structure and diversity within the species.
Given that the total population within the assessment region is estimated at less than 1, 000 mature individuals, and at least 5,000 individuals are generally required for the population to be considered genetically healthy and stable (i.e., Ne > 500; using a conservation Ne/Nc conversion ratio of 0.1 – 0.1*5000 = 500). This suggests that Ne may be well below the Ne 500 threshold. Again, this inference would need verification using genetic analyses.
Habitats and ecology
Very little is known about the natural history of this species and there are few museum specimens available. All the localities from which they have been collected are arid with a mean annual rainfall of less than 100 mm (Skinner & Chimimba 2005). Specimens have been netted in riverine vegetation along dry riverbeds (Seamark & Kearney 2006) or close to open water (ACR 2024).
The Angolan Hairy Bat has short and broad wings with low wing loading (5.7 N.m-2) and low aspect ratio (6.9) (Schoeman & Jacobs 2008). This species is an insectivorous, clutter-edge forager. Diptera, Trichoptera, Hemiptera and Coleoptera were recorded in their diet in Goodhouse (Northern Cape Province) (Schoeman 2006). Roberts (1951) observed the species circling low around trees and bushes, snatching insects from leaves at dusk.
Ecosystem and cultural services: As this species is insectivorous, it plays an important role in controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Bats often prey on the insect species which destroy crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus result in a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 6. Rocky areas (e.g. inland cliffs, mountain peaks) | – | Suitable | – |
| 8.3. Desert -> Desert – Cold | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Unknown | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean forearm length = 3.3 ±1.2 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean forearm length = 3.3 ±2.0 cm (Monadjem et al. 2020)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Maximum forearm length; Females: 3.5 cm; Males = 3.5 cm (Monadjem et al. 2020)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: Unknown
Average Annual Fecundity or Litter Size: Unknown
Natural Mortality: Unknown
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown
Congregatory: Unknown
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence that this species is traded or utilised for commercial or subsistence use.
Local Livelihood: Not specified
National Commercial Value: The species has been observed foraging within groves of orange trees, gleaning small insects from the leaves (Roberts 1951; Kearney 2013), indicating that individuals may contribute to natural insect pest control in agro-ecosystems.
International Commercial Value: No
End Use: N/A
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
At present, there are no major or measurable threats to this species. However, being an arid-adapted species, the effects of climate change inclusive of increased aridity in desert and semi-desert regions where the species occurs, is likely to result in physiological stress. Additionally, it may be threatened by the expansion of wind farm infrastructure in the Northern Cape (Baerwald et al. 2008), specifically between Springbok and Kleinsee. It is, however, considered to have a low risk of collision risk with wind turbines (MacEwan et al. 2020).
Lastly, it may be locally threatened by mining operations in some parts of its range (ACR 2024).
Conservation
This species is present with the Richtersveld National Park and Augrabies Falls National Park. Although no direct conservation interventions are necessary at present, the impact of wind farming should be monitored to determine population decline (if any) for this species. Additionally, conservation planning and engagement with both the mining and wind energy sectors are needed to mitigate population and habitat loss and disturbance. Occurrence records based on acoustic data should be reported to SANBI and the above listed assessors. Further studies on the utilisation of agro-ecosystems, such as citrus orchards (Roberts, 1951; Kearney 2013), by the species ought to be prioritised.
Recommendations for land managers and practitioners:
- Known roosts should be overlain on a map with existing and planned wind farm sites to identify key sites for protection.
- Engagement with the mining and wind energy sectors to mitigate future impacts on this species and its roosting sites.
- Promote the retention of disused sheds and barns that are used as night roosts by the species.
Research priorities:
- Field surveys, in combination with acoustic sampling, and niche modelling are needed to more accurately delimit the distribution of this species.
- Further genetic sampling to bolster existing phylogenetic data sets are needed.
- Further studies are needed into the reproductive and feeding ecology of this little-known species.
- To determine how best to mitigate wind farm sites to decrease mortalities of insectivorous bats in turbines
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Limit disturbance to roost sites, including night roosts.
Bibliography
ACR. 2024. African Chiroptera Report 2024. AfricanBats, African Chiroptera Project, i-xix. Pretoria.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Baerwald, E.F., D’Amours, G.H., Klug, B.J. and Barclay, R.M. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18:695–696.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332:41–42.
Corbet, G.B. and Hill, J.E. 1980. A world list of mammalian species. British Museum (Natural History) and Cornell University Press, London, UK and Ithaca, USA.
Kearney, T. 2013. Cistugo seabrae Angolan Wing-gland bat; pp 687-688 in Happold, M. and Happold, D.C.D. (eds) Mammals of Africa: Volume IV. Bloomsbury Publishing, London.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223:1–38.
Lack, J.B., Roehrs, Z.P., Stanley, C.E., Ruedi, M. and Van Den Bussche, R.A. 2010. Molecular phylogenetics of Myotis indicate familial-level divergence for the genus Cistugo (Chiroptera). Journal of Mammalogy 91(4): 976–992.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5:1–359.
MacEwan, K., Sowler, S., Aronson, J., and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities – ed 5. South African Bat Assessment Association
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Roberts, A. 1951. The Mammals of South Africa. Central New Agency, Johannesburg, South Africa.
Schoeman, M. C. 2006. The relative influence of competition and coevolution on the community structure of insectivorous bats in southern Africa. PhD thesis, University of Cape Town, Cape Town.
Schoeman, M. C. and Jacobs, D. S. 2008. The relative influence of competition and prey defenses on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS ONE 3(11): e3715.
Seamark, E. C. J. and Kearney, T. C. 2006. New distribution of the Angolan wing-gland bat (Cistugo seabrae Thomas 1912). Afr. Bat Conserv. News 7: 2-4.
Simmons, N.B. 2005. Order Chiroptera. Pages 474–475 in Wilson DE, Reeder DM, editors. Mammal Species of the World. The John Hopkins University Press, Baltimore, Maryland, USA.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., Kearney, T.C., Clark, V.R., Howard, A., Mdluli, M.V., Markotter, W., Geldenhuys, M., Richards, L.R., Rakotoarivelo, A.R., Watson, J., Balona, J., and Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate‐driven diversification and a buffer against future climate change in bats. Global Change Biology, 30(6), p.e17344.
Thomas, O. 1912. On a collection of small mammals from the Tsin-ling Mountains, Central China, presented to Mr. G. Fenwick Owen to the National Museum. Annals and Magazine of Natural History, Series 8 10: 395-403.
