
Angolan Free-tailed Bat
Mops condylurus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Mops condylurus – (A. Smith, 1833)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – MOLOSSIDAE – Mops – condylurus
Common Names: Angolan Free-tailed Bat, Angolan Mops Bat, Knob-tailed Mops Bat, Knob-tailed Nyctinome (English), Angola-losstertylermuis, Angolese losstertvlermuis (Afrikaans), Molosse d’Angola, Tadaride d’Angola (French)
Synonyms: Nyctinomus condylurus A. Smith, 1833; Mops angolensis Peters, 1870; Mops fulva Thomas, 1908; Mops occidentalis Monard, 1939; Mops orientis G.M. Allen & Loveridge, 1942; Mops osborni J.A. Allen, 1917; Mops wonderi Sanbron, 1936; Tadarida condylura (A. Smith, 1833)
Taxonomic Note:
Many subspecies have been described, but none are recognised here due to the variability in pelage colour and size known to occur within only one locality (Skinner & Chimimba 2005). The sparse-haired band across the shoulders separates Mops from Tadarida and most Chaerephon species (Monadjem et al. 2020). The similar-sized Chaerephon ansorgei shows a shoulder band but has a black throat unlike the pale upper parts in M. condylurus; and Mops midas is far larger (forearm length ≥ 60 mm) (Monadjem et al. 2020).
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1, Howard, A.2 & van der Meulen, K.R.2
Reviewer: Naidoo, T.1
Institutions: 1Durban Natural Science Museum, 2University of the Free State
Previous Assessors and Reviewers: Monadjem, A., Cohen, L., Jacobs, D., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Roxburgh, L., Raimondo, D., Nicholson, S.K. & Relton, C.
Assessment Rationale
Listed as Least Concern in view of its wide distribution (estimated extent of occurrence in the assessment region is 212,332 km2), presumed large population, several colonies that exceed 100 individuals, and it being recorded from several protected areas. It is also able to utilise modified habitats and can occupy a broad range of environments. Savannah habitats in the assessment region are fairly well protected. Although persecuted in certain regions, there is no evidence of decline in species numbers. The potential threat of wind farms should be monitored for its impacts on this species as it is categorised as a “high-risk” species.
Regional population effects: Colonies from within the assessment region are continuous with those occurring in Mozambique and Zimbabwe. It has high wing-loading (Norberg & Rayner 1987), and thus dispersal capacity is assumed to be sufficient for rescue effects.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR, Howard A & van der Meulen KR. 2025. A conservation assessment of Mops condylurus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Angolan Free-tailed Bat has a broad distribution across much of sub-Saharan Africa, occurring across a range of habitat types (Skinner & Chimimba 2005). Its range extends from Senegal, The Gambia and Mali in the west, to Sudan, Ethiopia and Somalia in the east; southwards as far as eastern South Africa and Eswatini and is largely absent from the Congo basin (Monadjem et al. 2016). It has not been recorded from Namibia (ACR 2023). Within the assessment region, it has been recorded from KwaZulu-Natal, Eswatini, and through the Limpopo province and the greater Kruger National Park region, from where its range extends to Zimbabwe, northern Botswana, Zambia, Malawi, Democratic Republic of the Congo, eastern Angola and Mozambique (Monadjem et al. 2020). Estimated extent of occurrence in the assessment region is 212,332 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
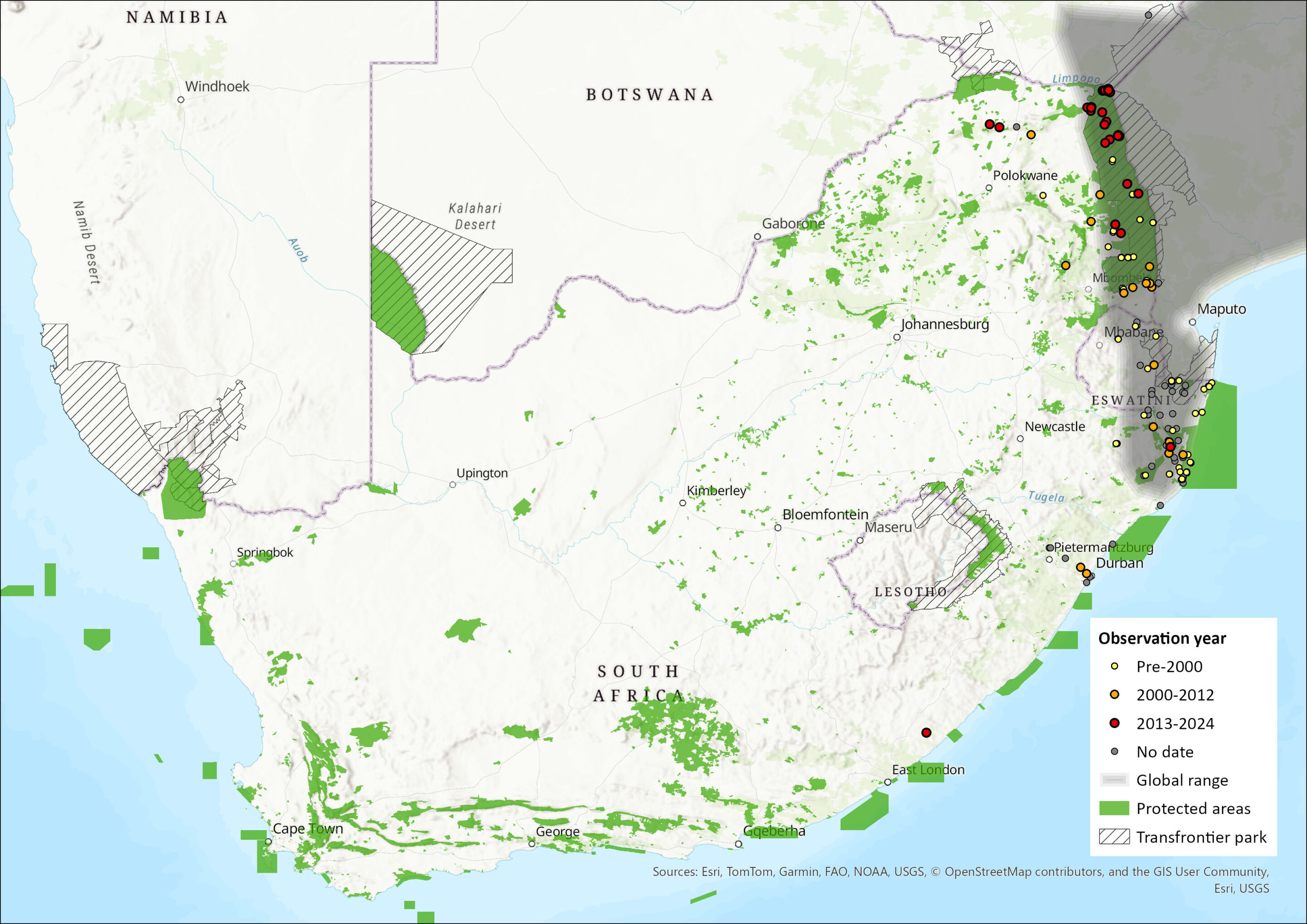 Figure 1. Distribution records for Angolan Free-tailed Bat (Mops condylurus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Angolan Free-tailed Bat (Mops condylurus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: (Not specified)
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
No formal study on the direct effects of climate change has taken place, however, due to the increasing temperature predicted for the eastern region (Mbokodo et al. 2020) the energetic costs of thermoregulation is likely to increase with dehydration becoming more common (Buffenstein et al. 1999; Vivier and van der Merwe, 2007). Roost preferences will likely shift to select for within-roost temperatures of 35-40°C to maintain basal metabolism (Vivier and van der Merwe 2007).
Population Information
The species is considered globally common (ACR 2024) and is abundant within its range across eastern South Africa (Monadjem et al. 2020). This species roosts communally in groups ranging from just a few individuals to thousands (Monadjem et al. 2020).
Current population trend: Stable
Continuing decline in mature individuals? Not likely
Number of subpopulations: Unknown, but 30 colonies
Extreme fluctuations in the number of subpopulations: Unknown, but not likely
Continuing decline in number of subpopulations: No evidence for decline
All individuals in one subpopulation: Yes (suspected based on ecomorphology and dispersal capabilities)
Number of mature individuals in largest subpopulation: Undetermined but likely to be >2000
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Not likely
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Not likely
Probability of extinction in the wild within 100 years: Unknown
Population Genetics
No population genetic study has been conducted on this species within the assessment region, however, due to its wide distribution and dispersal capabilities, in addition to them often existing in large colonies, it is expected the species exists as a single, genetically healthy population (Ne > 500 based on some colonies numbering in the thousands (Monadjem et al. 2020).
Habitats and ecology
The Angolan Free-tailed Bat is commonly associated with both moist and dry savannah habitats, but also occasionally occurs at the edges of woodland (Monadjem et al. 2016). Natural roost sites include narrow crevices in rock faces and caves, as well as hollows in trees (Fenton et al. 1994), and sites that allow for a free vertical drop as they take flight, are preferred (Verschuren 1957). This species has also taken to roosting in anthropogenic structures, particularly roofs of houses, in the expansion joints of bridges (Monadjem et al. 2020) and within mine shafts (Skinner & Chimimba 2005). In the assessment region, the species has been recorded from the Mopane Bioregion, Central Bushveld, Lowveld, Mesic Highveld Grassland, Indian Ocean Coastal Belt and Sub-Escarpment Savanna bioregions.
Within hot, dry summer conditions in South Africa, the Angolan Free-tailed Bat reveals a preference for temperature zones of 35–40°C within the roost to maintain basal metabolism and minimise energetic costs associated with thermoregulation (Maloney et al. 1999). They are capable of utilising a broad range of climatic and environmental (semi-arid to mesic) conditions using adaptive thermoregulation and osmoregulation, and will enter a state of torpor during both summer and winter conditions (Bronner et al. 1999; Buffenstein et al. 1999; Maloney et al. 1999; Vivier & van der Merwe 2007).
The species is considered an open-air forager. Individuals from the assessment region feed primarily on Lepidoptera, Diptera, and Hemiptera (Bohmann et al. 2011), while Kenyan conspecifics mostly consume Coleoptera species (Whitaker & Mumford 1978). Individuals commonly forage throughout the night, departing from the roost site at dusk and only returning at dawn (Monadjem et al. 2020). They can cover a maximum distance of approximately 5 km from the roost each night (Noer et al. 2012).
Female Angolan Free-tailed Bats are considered seasonally polyoestrus, exhibiting a bimodal pattern of reproduction with two birth seasons from early September to early May (Vivier & van der Merwe 1997; Monadjem et al. 2020). The gestation period is 85 days, and lactation occurs for about 50–60 days after birth (Vivier & van der Merwe 1997). The breeding season of males in Mpumalanga was found to occur for a duration of 9 months from June to February, peaking in August/September, and then again in November/December (Vivier & van der Merwe 1996).
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations (Boyles et al. 2011; Kunz et al. 2011; Taylor et al. 2017, 2018). Bats often prey on insect pest species that destroy crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus result in a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: From 1 year onwards as seen with other members of this family (Vivier & van der Merwe 1997)
Age at Maturity: Male: From 1 year onwards as seen with other members of this family (Vivier & van der Merwe 1997)
Size at Maturity (in cms): Female: Mean total length = 10.8 ±0.75 cm; Mean forearm length = 4.59 ±0.18 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean total length = 11.30 ±0.81 cm; Mean forearm length = 4.70 ±0.16 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: 1 year
Maximum Size (in cms): Female forearm length = 4.96 cm; Male forearm length = 5.10 cm (Monadjem et al. 2020)
Size at Birth (in cms): (Not specified)
Gestation Time: 85 days (Vivier & van der Merwe 1996, 1997)
Reproductive Periodicity: Two birthing seasons between September and May (Vivier & van der Merwe 1996, 1997)
Average Annual Fecundity or Litter Size: One
Natural Mortality: Unknown
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Capable of covering ~ 5 km per night
Congregatory: Yes
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region.
National Commercial Value: No
International Commercial Value: No
End Use: N/A
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
No major species-specific threats have been identified for this species. However, they are likely to be vulnerable to pressures identified for other bat species. With wind energy installations potentially moving into parts of KwaZulu-Natal, this could pose a future threat to the Angolan Free-tailed Bat as it is an open-air forager (Baerwald et al. 2008; Cryan & Barclay 2009; Rydell et al. 2010; Rollins et al. 2012), and is categorised as high risk for turbine related fatalities (MacEwan et al. 2020). Bats are attracted to wind farm sites which often tend to have higher insect densities at well-lit turbines (Voight et al. 2024). When bats fly near to turbine blades, they either collide directly with the blade or they experience barotrauma. Barotrauma is tissue damage caused by rapid excessive changes in air pressure near turbine blades (Baerwald et al. 2008; Rydell et al. 2010). Additionally, this species may face persecution when roosting within the roofs and crevices of buildings (Monadjem et al. 2016), even in protected areas. As this species often roosts in caves, it may face the threat of human disturbance, as many local traditional ceremonies and tourism activities take place in caves.
Conservation
This species is present in a number of protected areas, such as Vhembe Biosphere Reserve, Mapungubwe National Park (MNP), Venetia Limpopo Nature Reserve (VNR), Kruger National Park, Ndumo Game Reserve, Tembe Elephant Park, Isimangaliso Wetland Park and Hluhluwe-iMfolozi Game Reserve. No direct conservation actions are currently needed for the species, but it is likely to benefit from the direct protection of roost sites.
Recommendations for land managers and practitioners:
- Protection of roosting sites, and the provision of bat boxes, to limit the persecution of this species when roosting in roofs and buildings.
Research priorities:
- Monitoring the potential impact of wind farming on Angolan Free-tailed Bat populations within KwaZulu-Natal.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report 2023. Van Cakenberghe, V. and Seamark, E.C.J. (Eds.) African Chiroptera Project, Pretoria. i – xviii; 1 – 1210 pp.
Baerwald EF, D’Amours GH, Klug BJ, Barclay RM. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18: 695–696.
Bohmann, K., Monadjem, A., Lehmkuhl Noer, C., Rasmussen, M., Zeale, M.R., Clare, E., Jones, G., Willerslev, E., Gilbert, M.T. 2011. Molecular diet analysis of two African free-tailed bats (Molossidae) using high throughput sequencing. PloS one 6(6):e21441.
Boyles JG, Cryan PM, McCracken GF, Kunz TH. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Bronner GN, Maloney SK, Buffenstein R. 1999. Survival tactics within thermally-challenging roosts: heat tolerance and cold sensitivity in the Angolan free-tailed bat, Mops condylurus. South African Journal of Zoology 34: 1–10.
Buffenstein R, Bronner GN, Maloney SK. 1999. Seasonal and daily variation in blood and urine concentrations of free-ranging Angolan free-tailed bats (Mops condylurus) in hot roosts in South Africa. South African Journal of Zoology 34: 11–18.
Cryan PM, Barclay RM. 2009. Causes of bat fatalities at wind turbines: hypotheses and predictions. Journal of Mammalogy 90: 1330–1340.
Fenton, M. B., Rautenbach, I. L., Smith, S. E., Swanepoel, C. M., Grossell, J. and van Jaarsveld, J. 1994. Raptors and bats: threats and opportunities. Anim. Behav 48: 9–18.
Lehmkuhl Noer, C., Dabelsteen, T., Bohmann, K., Monadjem, A. 2012. Molossid bats in an African agro-ecosystem select sugarcane fields as foraging habitat. African Zoology 47(1):1-1.
Kunz TH, Braun de Torrez E, Bauer D, Lobova T, Fleming TH. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Maloney SK, Bronner GN, Buffenstein R. 1999. Thermoregulation in the Angolan free-tailed bat Mops condylurus: a small mammal that uses hot roosts. Physiological and Biochemical Zoology 72: 385–396.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Monadjem A, Cotterill F, Hutson AM, Mickleburgh S, Bergmans W. 2016. Mops condylurus. e.T13838A22075340.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Noer L, C., Dabelsteen, T., Bohmann, K. and Monadjem, A. 2012. Molossid bats in an African agro-ecosystem select sugarcane fields as foraging habitat. African Zoology, 47(1), pp.1-11.
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Rollins KE, Meyerholz DK, Johnson GD, Capparella AP, Loew SS. 2012. A forensic investigation into the etiology of bat mortality at a wind farm: barotrauma or traumatic injury? Veterinary Pathology Online 49: 362–371.
Rydell J, Bach L, Dubourg-Savage M-J, Green M, Rodrigues L, Hedenström A. 2010. Mortality of bats at wind turbines links to nocturnal insect migration? European Journal of Wildlife Research 56: 823–827.
MacEwan, K., Sowler, S., Aronson, J., and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities – ed 5. South African Bat Assessment Association
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Taylor, P.J., Schoeman, M.C. and Monadjem, A. 2013. Diversity of bats in the Soutpansberg and Blouberg Mountains of northern South Africa: complementarity of acoustic and non-acoustic survey methods. South African Journal of Wildlife Research-24-month delayed open access, 43(1), pp.12-26.
Taylor, P.J., Matamba, E., Steyn, J.N.K., Nangammbi, T., Zepeda-Mendoza, M.L. and Bohmann, K. 2017. Diet determined by next generation sequencing reveals pest consumption and opportunistic foraging by bats in macadamia orchards in South Africa. Acta Chiropterologica, 19(2), pp.239-254.
Taylor, P.J., Grass, I., Alberts, A.J., Joubert, E. and Tscharntke, T. 2018. Economic value of bat predation services–A review and new estimates from macadamia orchards. Ecosystem Services, 30, pp.372-381.
Taylor, P.J., Nelufule, M., Parker, D.M., Toussaint, D.C. and Weier, S.M. 2020. The Limpopo River exerts a powerful but spatially limited effect on bat communities in a semi-arid region of South Africa. Acta chiropterologica, 22(1), pp.75-86.
Verschuren J. 1957. Exploration du Parc National de la Garamba, 7, Chiroptères. Institute des Parcs Nationeaux du Congo Belge, Bruxelles.
Vivier L, van Der Merwe M. 1997. Reproduction in the female Angolan free-tailed bat, Tadarida (Mops) candylura (Microchiroptera: Molossidae), in the eastern Transvaal, South Africa. Journal of Zoology (London) 243: 507–521.
Vivier L, van der Merwe M. 1996. Reproductive pattern in the male Angolan free-tailed bat, Tadarida (Mops) condylura (Microchiroptera: Molossidae) in the Eastern Transvaal, South Africa. Journal of Zoology 239: 465–476.
Vivier L, van der Merwe M. 2007. The incidence of torpor in winter and summer in the Angolan free-tailed bat, Mops condylurus (Microchiroptera: Molossidae), in a subtropical environment, Mpumalanga. South Africa. African Zoology 42: 50–58.
Voigt, C.C., Bernard, E., Huang, J.C.C., Frick, W.F., Kerbiriou, C., MacEwan, K., Mathews, F., Rodríguez-Durán, A., Scholz, C., Webala, P.W. and Welbergen, J. 2024. Toward solving the global green–green dilemma between wind energy production and bat conservation. BioScience, 74(4), pp.240-252.
Whitaker, J.O. and Mumford, R.E. 1978. Foods and ectoparasites of bats from Kenya, East Africa. Journal of Mammalogy, 59(3), pp.632-634.
