
African Striped Weasel
Poecilogale albinucha

2025 Red list status
Near Threatened
Regional Population Trend
Decreasing
Change compared
to 2016
No Change
Overview
Poecilogale albinucha – (Gray, 1864)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – MUSTELIDAE – Poecilogale – albinucha
Common Names: African Striped Weasel, African Weasel, Snake Weasel, Striped Weasel, White-naped Weasel (English), Slangmuishond (Afrikaans), Nakêdi (Setswana), Inyengelezi (Zulu) Kappeniltis (German), Poecilogale à nuque blanche (French), Weissnackenwiesel (German)
Synonyms: africana, bechuanae, doggetti, flavistriata, lebombo, transvaalensis
Taxonomic Note:
Six subspecies have been proposed (Meester et al. 1986), but their validity is dubious as they are based on appearance only (Stuart & Stuart 2013). Divergence dates of the African Striped Weasel are estimated at between 2.2 to 2.7 million years ago from Ictonyx sp. and its ancestors that originated from Eurasian colonisations (Koepfli et al. 2008).
Red List Status: NT – Near Threatened
Assessment Information
Assessors: Smith, K.1, Power, R.J.2, da Silva, J.M.3 & Do Linh San, E.4
Reviewer: Smith, C.5
Institutions: 1University of Pretoria, 2North West Provincial Government, 3South African National Biodiversity Institute, 4Sol Plaatje University, 5Endangered Wildlife Trust
Previous Assessors & Reviewers: Child, M.F., Rowe-Rowe, D., Birss, C., Wilson, B., Palmer, G., Stuart, C., Stuart, M., West, S., & Do Linh San, E.,
Previous Contributors: Roxburgh, L., Skowno, A., Avenant, N. & Page-Nicholson, S.
Assessment Rationale
This enigmatic species occurs at naturally low densities but is also severely threatened by loss of productive habitats and hunting for the traditional medicine trade. Although this species has a wide range, it is not abundant. It is a specialist predator of small mammals and has a high metabolic rate, which means it can only exist in habitats containing adequate numbers of prey. Such habitats are being lost or drastically transformed to grow food, cash crops or commercial forestry plantations. This is compounded by overgrazing that reduces the cover on which the African Striped Weasel’s prey species rely. Human population expansion has increased the number of dogs that often kill African Striped Weasels or compete for food. Similarly, Black-backed Jackal (Lupulella mesomelas) densities have increased markedly in these areas, which may also have a significant impact.
We estimated that only 7,138 km2 of the species’ area of occupancy is within natural habitat outside of potential harvesting pressure which, combined with it being a naturally low-density species, may mean there are fewer than 10,000 mature individuals. The average rate of rural expansion in all core provinces (every province besides the Northern Cape) since 2000 is 10 ± 13%. The estimated three-generation period for this species is 9 years. If we assume that rural settlement expansion is a proxy for harvesting pressure, then the African Weasel satisfies Near Threatened C1, as a 10% decline over a 10-year period is likely. Further field studies and surveys to determine current area of occupancy, density estimates, home range size and habitat preference, and severity of traditional medicine trade on this species are desperately needed. This species should be reassessed as soon as new data become available.
Regional population effects: There is suspected to be little dispersal of individuals from neighbouring countries or not on the scale to impart a significant rescue effect. In dry times, the arid Limpopo river valley may function as a barrier of sorts with the rest of the African population. For the most part there appears to not be a substantial enough population along the northern periphery of the species national range.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No Change
Red List Index
Red List Index: No change
Recommended citation: Smith K, Power RJ, da Silva JM & Do Linh San E. 2025. A conservation assessment of Poecilogale albinucha. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species ranges from southwestern Uganda and Kenya to the Western Cape in South Africa (Stuart & Stuart 1998, 2013; Skinner & Chimimba 2005). It has been recorded from nearly sea level to 2,300 m asl in Malawi (Medland & Dudley 1995). Most of the available records come from southeast South Africa, as this is where most research has been conducted (Rowe-Rowe 1992; Stuart & Stuart 2013). A range expansion may have occurred in southwest South Africa, based on documented road kills (Stuart & Stuart 1998). Because of its secretive nature it has probably been overlooked in many areas (Stuart 1981), especially in light of records from sites that were previously considered unsuitable (Stuart and Stuart 2013). It is also often confused with the Striped Polecat (Ictonyx striatus), which may lead to overestimation of its distribution in some cases (Stuart 1981; Child et al. 2016; Nieman et al. 2020).
Within the assessment region, it occurs along the west coast from Garies southward to the top of the escarpment into the western and southern Cape coastal belt, east and northeast Northern Cape, and all other provinces. It occurs predominantly in moist eastern grasslands (Skinner & Chimimba 2005; Child et al. 2016). In the 1970s, it was only known from the Eastern Cape, Northern Cape, and in the Western Cape, as far east as Sedgefield (Stuart 1981). The species was recorded in the 1970s in the Sehlabathebe National Park of Lesotho (Lynch and Watson 1990), and some more recent records have been made in this country (Figure 1).
Incidental sightings and road kill specimens in the Western Cape indicate a widespread distribution in the Southwest Fynbos bioregion within the Kogelberg and Boland Mountain regions, the Swartland Shale Renosterveld and Cederberg Sandstone Fynbos bioregions within the Cederberg Mountain regions, the Cape Flats Sand Fynbos and Cape Flats Dune Sandveld bioregions, the De Hoop Limestone Fynbos and South Outeniqua Sandstone Fynbos bioregions, extending to the Lamberts Bay Strandveld bioregion (C. Birss unpubl. data, In: Child et al. 2016). Recently, the species has been found to still occurs in the Boland Mountain Complex area, though at a low reporting rate, and they were reported mostly from the East Hawequas area (Nieman et al. 2020). In a leopard-focussed study at the De Hoop Nature Reserve, during 2020, Striped Weasel were detected at 5% of camera traps (Hargey 2022). The species seems to still be present in the Western Cape and it may even be the new stronghold for the species (Figure 1).
In the Eastern Cape, there does not seem to be many recent records (Figure 1). In 2005, the species was recorded as content in jackal scats in the Great Fish Nature Reserve in the Eastern Cape (Do Linh San et al. 2009). Later on, and more recently in the Mountain Zebra National Park in the Eastern Cape, one detection was made in 2018 (Smith et al. 2023).
They have been observed in southeast Namibia (C. Stuart & M. Stuart pers. Obs., In: Child et al. 2016) and Stuart and Stuart (1990) reported a road kill from the South African side of Kgalagadi Transfrontier Park in 1989, which may indicate a wider distribution in Botswana and eastern Namibia, but it seems to have recently been recorded from the area since (see Figure 1). Striped Weasel have been found in jackal scats at the Rooipoort private Nature Reserve in the Northern Cape (Klare et al. 2010), adding to the indirect evidence required to confirm the species (cf Do Linh San et al. 2009).
In the dry, western-most range of the species, it has been said to lead an increasingly subterranean existence. This may be a behavioural response to avoid extreme temperatures and reduce water requirements. Almost without exception, the few records that have been reported from this region indicate the presence of mole-rats (B. Wilson unpubl. data, In: Child et al. 2016). It should be considered that the species may well have an inquilistic relationship with mole-rats and that, in the arid areas of their range, they thus have a sympatric distribution. It is therefore conceivable that the distribution of the species is slightly more widespread than reported, but remains nevertheless extremely fragmented and the population densities extremely low. In these areas, the loss of any habitat for mole-rats is thus likely to result in the loss of habitat and available prey base for the weasels too (Child et al. 2016).
The species has never been recorded in the Kruger National Park (see Figure 1). Many other areas of the country have not reported sightings for many years, except for the Western Cape (Figure 1). In Mpumalanga’s Verloren Vallei Nature Reserve near Dullstroom, none were recorded in 2000 camera trap nights in 2019 (Loock & Swanepoel 2019), despite this being described as suitable habitat for the species. Elsewhere in the province a record was obtained (Figure 1) and there have been no recent records in Limpopo (Figure 1). Similarly, there have been few recent records in Gauteng (Figure 1), the dearth of the species at the Faraday Market (Whiting et al. 2016), may suggest an absence of them in the region, or that they are indeed at very low regional densities.
In a mammal inventory study ending in August 2013, of the 2715 cumulative trap nights throughout the North West, no Striped Weasel was recorded (Power et al. 2019). This was ascribed to it being a low density, cryptic species (Rautenbach 1982; Rowe-Rowe 1992; Skinner & Chimimba, 2005), and realistically one would have to be reliant on opportunistic records of this species for one to make any headway in listing this species (Power et al. 2019). The species was thus listed as a probable occurrence, and its occurrence at least in 2019 was uncertain (Power et al. 2019). However, of late it appears that a record has emerged in what appears to be Madikwe area (see Figure 1). However, in the province, a roadkill occurrence study which commissioned from April 2020 to February 2024, involving 162, 614 km travelling, which did not turn up any records of the Striped Weasel (NWPG 2024). This points still to the overall scarcity of the species at least in this province.
In the Free State in the 1990s, the Striped Weasel was said to occur on the Tussen-die-Riviere Nature Reserve of the Free State based on signs (Watson 2006), but this would not be construed as strong evidence. The species was recorded in the Eastern Free State, and after recovery from a period of prior cattle farming (Buschke 2016). There seem to have been a few recent records in the Free State (Figure 1).
The species had been recorded recently from Mkhuze Game Reserve in northern KwaZulu-Natal (T. Bodasing pers. comm. 2014, In: Child et al. 2016). The lack of current records, however, may be due to lack of search effort. Further field surveys were needed to determine its occurrence. However, extensive surveys with camera traps have in fact been done to date in the Maputaland area and the species has not been detected (Ramesh et al. 2017; Pretorius 2019; Da Rosa 2019). In various Maputaland reserves on carnivore focussed camera trap surveys, no Striped Weasel were detected in 2014 with as many as 4920 camera trap days (Ramesh et al. 2017), then a year later, neither on 4985 trap nights (Pretorius 2019). Also, at the same time, in the Phinda area, near Mkuze, a study focussing on small mammals and carnivores, found no African Striped Weasels, either despite as much effort as between 11,520–17,280 camera trap nights (Da Rosa 2019). Altogether, just over 27, 000 trap nights across three different studies in the region revealed no sign of the species, which suggest an absence or very low densities. The species was reported up until the 1970s near Ubombo (Pringle 1977).
The species appears to still be present in the southern parts of the assessment region, from central KwaZulu-Natal, across the Free State and Northern Cape, with what appears to be a stronghold in the Western Cape (see Map). Child et al. (2016) calculated the proportion of the estimated area of occupancy affected by harvesting. As home range size for the weasel is unknown, they buffered both weasel distribution points and “huts” (as a proxy of rural development measured using Eskom Spot Building Count from 2011) by a radius of 10 km. They then summed the buffered weasel points to “sample” area of occupancy (AOO), which they estimated as 50,518 km2. They then used 2013 land cover data (GeoTerraImage 2015) to estimate the amount of natural land currently contained within the AOO, which they estimated at 36,506 km2. Finally, they subtracted the area of the natural AOO that intersects the buffered rural villages, and thus within harvesting distance, which equated to only 7,138 km2 natural AOO not within 10 km of a village. This represented an 86% reduction in effective AOO. However, this was a snapshot sample and has no time period over which to measure the rate of change.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 2300
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
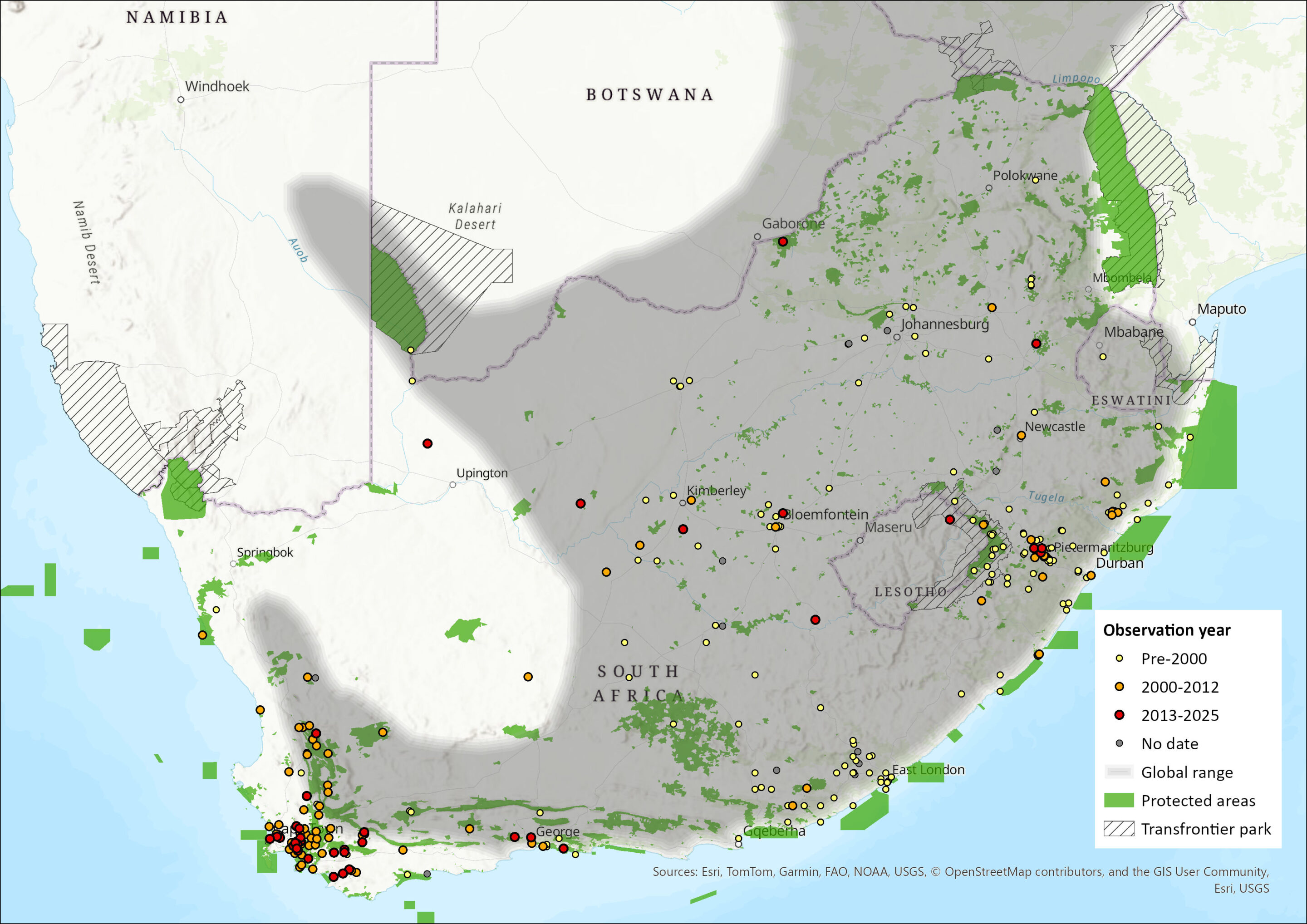 Figure 1. Distribution records for African Striped Weasel (Poecilogale albinucha) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for African Striped Weasel (Poecilogale albinucha) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
It is important to consider a species’ ecological and biological traits to determine the likely mechanisms of climate change impact and quantify these using expert knowledge (Foden & Young 2016). An increase in shrub cover as predicted by many climate change models (Foden and Young 2016), which may affect the species if grasslands are to be affected, as too, if its prey populations are to be affected too.
Population Information
It is rare to uncommon. For example, in the Free State, the lack of observation records (from multiple sources) suggests it is the scarcest of mammal species (N.L. Avenant unpubl. data, In: Child et al. 2016) and this may still be the case. In the Northern Cape, there are two historical records (from the Kalahari and from Schmidtsdrif) and only three other records added in the last eight years of the previous assessment (B. Wilson unpubl. data, In: Child et al. 2016). However, this may be an artefact of increased research effort as well as increased awareness around the species in the farming communities. As such, the status of the species in the Northern Cape remains unclear. Similarly, the Eastern Cape Parks and Tourism Agency had only one record (in 2006) in the period 2006–2013 (D. Peinke unpubl. data, In: Child et al. 2016). Recently, in a multi-species camera trap survey at the Eastern Cape’s Mountain Zebra National Park consisting of 19 camera traps, the African Striped Weasel was detected once in 7900 camera trap days (Smith et al. 2023), which suggests a low abundance. Similarly, in the Western Cape, in the De Hoop Nature Reserve, during a camera trap survey, the species was detected twice on a survey which spanned 2457 trap nights during the period September and December 2020 (Hargey 2022). In the Blaauwberg Nature Reserve and an adjacent coastal residential estate approximately 25 km north of Cape Town, the species was detected once and twice on camera traps in 2019, respectively, during a survey that collectively spanned 1555 trap nights (Ross 2022). Based on a questionnaire approach, a high relative abundance was predicted in the East Hawequas Management Area of the Boland Mountain Complex (Nieman et al. 2021).
It is said high abundances are reached in moist higher rainfall grasslands (Stuart & Stuart 2013), such as the grasslands of KwaZulu-Natal (Rowe-Rowe 1992). It is probably only common in areas where there are perennially dense rodent populations (Stuart & Stuart 2013). It is currently not possible to estimate population size because, although widespread, they are difficult to observe and there are no density estimates available. However, as all available data sources still indicate a sparsely distributed and scarce population, indicating a small overall population size, it is conceivable that there are still fewer than 10,000 mature individuals within the assessment region. Field studies are needed to determine area requirements, population composition, numbers, and trends. At present, we infer the population to be declining for the following reasons:
- Between 1972 and 1977, surveys of small carnivores were performed in KwaZulu-Natal (Rowe-Rowe 1978a). In a second survey, conducted 20 years later (Rowe-Rowe 1992), declines in both numbers and occupancy were reported. This was inferred to be caused by loss of habitat to food crops and commercial forestry and declines in habitat quality from overgrazing and erosion. Considering the ongoing trend in habitat loss (Jewitt et al. 2015), a declining population is likely to be ongoing too.
- A useful index of abundance was given by Cunningham and Zondi (1991). In their comprehensive survey of wildlife use in traditional medicines in KwaZulu-Natal, they concluded that the African Striped Weasel was the province’s most threatened species. Dealers reported that the species, which is highly prized in traditional medicine (Rowe-Rowe 1990, 1992; N.L. Avenant unpubl. Data, In: Child et al. 2016), was becoming scarcer as there were fewer animals in the wild. Thus, population decline is inferred by data presented in Cunningham and Zondi (1991) and others (see Use and Trade), and it may also be applicable to the northern provinces, i.e. Gauteng where none were reported in the 2000s (see Whiting et al. 2016). The average rate of rural expansion in all core provinces (every province besides the Northern Cape), between 2000 and 2013 was 10 ± 13% (GeoTerraImage 2015). Generation length is estimated as 3 years (Stuart et al. 2015), which yields a 9-year three-generation period. Thus, if we construe rural settlement expansion as a proxy for harvesting pressure and subsequent population decline, we can infer a > 10% population decline over a 10-year period.
- Corroborating this, reporting rate has declined from 89 in total from 1991–2001, to 49 in total from 2002–2013. These data are a composite of museum records, provincial and SANParks recordings and individual camera trap data. However, the Western Cape data show the opposite trend, where most of the data were collected post-2000, and there has been a steady regional increase from this region (Figure 1). It is expected that the decline in other provinces is ongoing from the previous assessment.
- An intensive survey in North West Province from 2010 to 2013 revealed no records (Power et al. 2019), compared to the 1970s when there were records obtained for the same regions (Rautenbach 1982), and despite extensive road-killed based surveys (NWPG 2024), and other surveys, none could be availed.
- Notably, given the extensive carnivore focussed camera trap surveys in the Maputaland part of Zululand in Northern Kwazulu-Natal, all of which commenced after 2015, and were done independently, yet none of them reported the species (see Ramesh et al. 2017; Da Rosa 2019; Pretorius 2019) This suggests a regional decline at least in this area (cf Rowe-Rowe 1992).
Although inferring exact decline from the data described above is unreliable because observer effort is unequal in time and space, we feel the data are robust enough to indicate some form of decline is still taking place. However, contrary to the overall trend, limited data from the Western Cape may suggest a stable subpopulation there at least. Relatively large numbers are collected dead on the roads, picked up on camera traps and reports from suburbia are also not infrequent (for example, Somerset West, West Stellenbosch and Durbanville) (C. Birss unpubl. data, In: Child et al. 2016) and are still reported (Nieman et al. 2021). The transformation from fynbos to grassland habitat and the removal of larger predators may have facilitated this trend. However, this may also be a “western anomaly”, perhaps caused by climate change, which only applies to a relatively small area of the total distribution. The anomaly between the Western Cape increase in reporting rate and the rest of country may also be explained by the relative lack of rural settlements in the Western Cape (and thus potentially lower harvesting rates) compared to the rest of the country. More research is needed to provide evidence for these interesting hypotheses (Child et al. 2016).
Population Information
Current population trend: Decreasing in part of its range but stable in the Western Cape.
Continuing decline in mature individuals: Yes
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While the species has been examined in a phylogenetic context (Koepli et al. 2008; Sato et al. 2012; Gray et al. 2022), to date no population genetic study has been undertaken. It is possible the population exists as a single metapopulation within the assessment region. Assuming no subpopulations have gone extinct within the assessment region, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – could be estimated at 1.0 (all populations remaining). A fine scale population genetic study incorporating individuals from neighbouring countries would be needed to confirm this. Given that fewer than 10,000 individuals are thought to exist in the assessment region, and that little to no dispersal is expected between neighbouring countries, a gross estimate of the effective population size (Ne) for the region can be quantified. Assuming a 0.1-0.3 Ne/Nc conversion ratio, the projected Ne is likely less than 3000, which is above the Ne 500 threshold, below which would signal concern for the genetic health of the species. Based on this information, the GBF’s headline genetic indicator – proportion of populations with an Ne > 500 would be 1/1 population above Ne 500, equating to a score of 1.0. As noted with genetic structure above, it is highly recommended that a population genetic study be undertaken to validate this indicator value and provide a more accurate measure of the genetic health of the species.
Habitats and ecology
African Striped Weasels are mainly found in savannah and grassland habitats, although it probably has a wide habitat tolerance and has been recorded from lowland rainforest, semi-desert grassland, fynbos (with dense grass), and intensively managed and altered landscapes such as pine plantations and residential estates (Larivière & Jennings 2009; Stuart & Stuart 2013, Ross 2022). The species has been reported in the following vegetation types: Kalahari Thornveld, Karoo, Grassveld, Fynbos, Forest fringe (Stuart 1981), and Fish River valley bushveld (Stuart 1981; Do Linh San et al. 2009). The area where the African Striped Weasel was recorded in jackal scat was a semi-open habitat called, typified by Noorsveld, composed of Searsia spp. and Scutia myrtina bushclumps and a karroid herbaceous layer (Do Linh San et al. 2009).
Preferred habitats are grassy; for example, the few records from arid southwestern Africa are associated with semi-desert grassland (Stuart & Stuart 2013). African Striped Weasels may reach their highest densities in moist grasslands (Rowe-Rowe 1992) and are also found in montane grassland (Rowe-Rowe 1992; Medland & Dudley 1995). Roadkills have been collected from areas of pastures and cultivated fields (Monadjem 1998; Stuart et al. 2015).
They are predominantly nocturnal but are occasionally seen during the day. They may make use of existing rodent burrows but are also good burrowers themselves. As such, soil texture may also be an important habitat determinant (Rowe-Rowe 1992). This species’ body shape, elongated with short, powerful limbs, makes it ideally suited to a subterranean lifestyle. It has relatively poor eyesight, probably because it spends most of its activity periods underground, and hunts using smell rather than sight. This poor eyesight may be one of the reasons that many of the specimens obtained for museum collections are in fact roadkill victims (Child et al. 2016).
This species is a specialist predator, feeding only on warm-blooded vertebrates, and there are no formal records of them feeding on invertebrates, amphibians or reptiles (Rowe-Rowe 1978b; Stuart & Stuart 2013). They mainly feed on rodents (often catching them in their burrows), moles and shrews. Birds, on the ground, are taken opportunistically (Rowe-Rowe 1978b). They can prey on rodents up to their own size (Skinner & Chimimba 2005). Rowe-Rowe (1978b) mentions a 500 g juvenile rabbit killed by one.
Interestingly, females do not appear to experience oestrus (Rowe-Rowe 1975, 1978c). They are generally solitary animals, widely dispersed, so the chances of an oestrus female encountering an adult male are very small. However, they do indulge in prolonged copulation that lasts between 65 and 72 minutes (Rowe-Rowe 1978c), which may induce ovulation (Rowe-Rowe 1975) and represent an adaptation to a solitary lifestyle. Based on Mustelid energetics, smaller, elongated sexually dimorphic mustelids exhibit intrasexual territoriality (Powell 1979), which suggests that the species is likely to be territorial.
Ecosystem and cultural services: As African Striped Weasels generally occur at low densities, they do not necessarily keep rodent populations under control, however only local abundances of them, may ever reach a potential of being a useful ecosystem service in this regard, though this is rarely attainable. Since the African Striped Weasel is said to be cunning, it is believed to help as a good luck charm, particularly in court cases (Simelane and Kerley 1998).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: 3.5 years
Age at Maturity: Female: 14 months
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 32 days
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
The species is used in traditional medicine and is easily captured when encountered. Cunningham and Zondi (1991) regarded this species as one of the most heavily hunted animals for the traditional medicine trade. The high demand for this species was corroborated by Ngwenya (2001) who found it was the second most sought after species in KwaZulu-Natal. Skins are commonly used by traditional healers and sangomas as a good luck charm (Simelane and Kerley 1998; Stuart et al. 2015). They have also been found in traditional medicine markets in the Eastern Cape and Gauteng (Simelane & Kerley 1998; Whiting et al. 2011), but not so later on in Gauteng (Whiting et al. 2016). They are also highly prized in traditional medicine in Lesotho (Lynch 1994; N.L. Avenant pers. comm. 2015). They have recently appeared in traditional medicine markets of the Western Cape (Nieman et al. 2018).
In some areas of South Africa, there is an overall clientele consisting of 60–80% of South African citizens who seek cures from wild animals. In the Western Cape, there has been an increase in medicinal use trade (Nieman 2018). A skin piece of the striped weasel can protect livestock from evil spirits (Nieman et al. 2018) and can sell for R3000 (Nieman 2018). The paws of the striped weasel can unify livestock (Nieman et al. 2018) and can sell for R1200 (Nieman 2018). The entire carcass of the striped weasel can protect against evil spirits (Nieman et al. 2018), and can sell for up to R2000 (Nieman 2018).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Traditional medicine | Yes | Traditional medicine |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 3. Medicine – human & veterinary | true | true | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
Habitat loss
Habitat loss is perhaps the greatest threat to this species. Being a specialist feeder on small mammals, occurring at low density, and an apparent association with habitats that are being altered or lost, places this species at risk within the assessment region. Large tracts of grasslands, where rodent densities are highest, have been ploughed to grow food or cash crops (maize, sugarcane, bananas, pineapples, vegetables and livestock fodder) while moist grassland is being increasingly converted to commercial timber plantations (Rowe-Rowe 1990, 1992; Child et al. 2016). In the North West in 2013, the ecological capacity of game on game farms was determined and of 609 game farms, 30% had exceeded their capacities, while this may be a small fraction, of those that did, the average was a 46% excess but ranged up to 3.75 times the capacity in the most overstocked site (Power 2014). This local overstocking on ranches, could have had regional level implications in terms of habitat degradation, i.e. loss of cover. This is only speculated at and could possibly be inferred. The species was recorded on a Free State farm, and after recovery from a period of prior cattle farming (Buschke 2016), which points to well managed rangelands being a prerequisite.
Prey loss via Habitat loss
Furthermore, overgrazing, particularly in subsistence farming areas, causes declines of small mammals by removing ground cover (Rowe-Rowe and Lowry 1982; Child et al. 2016). Loss of a prey base is likely to affect this species significantly more than other because they are long and thin and well-adapted for entering burrows or confined spaces to kill their prey, but this means they have a higher metabolic rate (owing to a greater proportion of surface area to body mass) than conventionally shaped and stockier animals (Rowe-Rowe 1978a). Males need about 95 g of prey / day and females need 67 g / day (Rowe-Rowe 1978a). During pregnancy (one month) and lactation (two months), the amount of food needed increases, as well as the 5-6 weeks before the young can kill on their own (Rowe-Rowe 1978c). If females raise one litter per year, food requirements are greater for at least 4 months. Thus, the annual prey requirements of Rhabdomys-sized (c. 35g) small mammals would be 1000-1100 /year for males and 730-1100 /year for females. A pair of African Weasels needs about 2000 murids / year and are thus highly dependent on intact habitat and healthy prey populations (Rowe-Rowe 1978c).
The Highveld Gerbil (Gerbilliscus brantsii) is a known problem species in maize cultivation areas of the North West Province (Power 2014). Farmers in the Ottosdal area of the North West commonly controlled the numbers of the problem rodent species, especially the Highveld gerbil (Foster, McIntyre & Haussmann 2022). Out of 44 Ottosdal farmers interviewed, to control the species 28 used chemical methods, 12 mechanical and 9 biological means of control (Foster et al. 2022). Given African Striped Weasels were reported near Barberspan in the past (Rautenbach 1982), and that they do prey on rodents (Rowe-Rowe 1978b), local losses of this species, and perhaps ingestion of poisoned rodents might have precipitated a decline in this species in the area. This is merely inferred and suspected. General anthropogenic disturbance. Between 2004 and 2007, of all carnivores admitted to a Durban-based rehabilitation centre in KwaZulu-Natal, most were orphans, and the African Striped Weasel was occasionally admitted (Wimberger & Downs 2010). However, there was no information on the proximal threats.
Domestic dogs
With increasing human populations in rural areas there are also increases in the numbers of dogs which compete with African Weasels for food and often kill them (Rowe-Rowe 1990, 1992). More recently, dogs have been observed either disturbing or killing weasels in the Western Cape (Nieman et al. 2020). Although not specifically aimed at the species, a mesocarnivore study in KwaZulu-Natal surmised that domestic dogs did impact on the abundance of some species (Pretorius 2019), and conceivably they would be affected too.
Inflated abundances of native mesopredators
High abundances of Black-backed Jackal owing to mesopredator release (Pretorius 2019), may impact upon the African Striped Weasel via predation. The African Striped Weasel has appeared in the scats of Black-backed Jackal, though at low frequencies (<2%) in the Eastern Cape (Do Linh San et al. 2009) and Northern Cape (Klare et al. 2010). Although jackals may have an effect on weasels (Bagniewska and Kamler 2013), this threat may not be significant as many smaller carnivores coexist with larger mesopredators by employing fine-scaled temporal avoidance behaviours (Smith et al. 2023).
Traditional medicine trade
Additionally, Cunningham and Zondi (1991) regarded this species as one of the most-used animals in traditional medicine, and in parts of its range the skins are commonly used by traditional healers and sangomas as a good luck charm (Cunningham and Zondi 1991; Simelane and Kerley 1998). However, this does not necessarily mean that the species is over-exploited but may rather reflect declining numbers in the wild. Any African Weasel encountered in KwaZulu-Natal, though, is likely to be killed for personal use or to sell to traditional healers (Cunningham and Zondi 1991; Child et al. 2016). In the Western Cape, Striped Weasel appeared in 23.5% of communities which traded with animal parts (Nieman 2018). Animal body parts, such as the striped weasel were increasingly more important in South Africa (Nieman et al. 2018). In the Western Cape at least, the Striped Weasel has a Cultural Significance Index (CSI) of 2.25, which is relatively low amongst several others that are a lot more important such as the Leopard Panthera pardus for instance which scored at 15 (Nieman 2018). In the Western Cape, the threat score based on a Rapid Vulnerability Assessment (RVA) was 4 for the Striped Weasel, which compared to the Leopard which was 10 (Nieman 2018) and was quite low. The threat of trade for the species is thus emerging in the Western Cape, though it may not be high enough yet to threaten the population.
Roadkill
Road kills do occur in the species (Stuart and Stuart 1990). Although road collisions were listed as a threat in the 2004 assessment, there was no evidence this is a serious threat to African Weasels. There have been very few incidents and there were no recorded incidents on the EWT Road Collision database (Child et al. 2016), though several newer records are of road kills (see Map). This threat might simply be low due to the lower abundance of them.
Conservation
This species is present in several protected areas across its range, which should be comprehensively documented. The following protected areas have recorded the species: Kgalagadi Transfrontier Park (Stuart and Stuart 1990), Tussen-die-Riviere Nature Reserve (Lynch and Watson 1996), Sehlabethebe Nature Reserve (Lynch and Watson 1990), Rooipoort Nature Reserve (Klare et al. 2010), Great Fish River Nature Reserve (Do Linh San et al. 2009), Oribi Gorge Nature Reserve and Maloti Drakensberg Park (Pringle 1977; Rowe-Rowe 1992), and several others. Recently, records show their occurrence in the Mountain Zebra National Park (Smith et al. 2023), and De Hoop Nature Reserve (Hargey 2021). Given their occurrence in the Boland Mountain Complex greater area (Nieman et al. 2021), the Hottentots-Holland Nature Reserve and Kogelberg Nature Reserve are likely important conservation areas, as is the Cedarberg Wilderness Area (see Map).
Outside protected areas, land-use planning should continue to conserve grassland habitats through protected area expansion, conservancy formation or stewardship schemes. Reducing agricultural intensification to conserve grassland habitats is likely to be a successful intervention for this species. For example, the species was recorded in 2015 on a former dairy farm in the Free State where the last cattle were removed in 2004 (Buschke 2016).
Conservationists should also work with farmers and rural communities to reduce overgrazing and retain ground cover to sustain the prey base of African Striped Weasels (Child et al. 2016).
Conservationists should also raise the public profile of this species and increase sightings submissions to improve our knowledge of its distribution. For example, in 2004, a private conservation group, Royal Hembe Conservation Group, on the north coast of KwaZulu-Natal started using the African Striped Weasel as a flagship species to promote care and sound management of declining coastal habitats. One of their members, a zoo curator, initiated a captive breeding programme for the species at Mitchell Park Zoo, Durban (Child et al. 2016). At a Durban-based rehabilitation centre, several Striped Weasels have been admitted for rehabilitation over the years (Wimberger and Downs 2010). However, we have not seen any publications or reports on post-release monitored individuals that have been rewilded.
Recommendations for land managers and practitioners:
- Sustain natural prey diversity by retaining ground cover, and to avoid overstocking of rangelands.
- Systematic monitoring projects should be established to formally assess population trend.
- Ezemvelo KZN Wildlife has listed the African Striped Weasel as a key species for continued monitoring and each staff member keeps a booklet to record sightings. Similarly, CapeNature has it listed as a “priority species” and all sightings and specimens are collected. This should be established in other provinces, i.e. North West.
Research priorities:
- Long-term surveys are necessary to establish baseline population data and density to gauge the effectiveness of conservation efforts.
- Consider also assessing study areas where the species was absent (see Ramesh et al. 2017; Da Rosa 2019; Loock & Swanepoel 2019; Pretorius 2019), particularly if it is an area within the EOO.
- Quantifying the severity of threats, including habitat loss and levels of harvesting.
- Determining whether the species is a habitat generalist or a specialist of grasslands.
- Field studies are needed to evaluate area requirements, population composition and specific habitat tolerance (for example, prey availability, soil texture, and cover). This includes determining densities and ecological adaptations in arid areas.
- The genetics of the southern African population/s should be assessed to determine whether there is any gene flow and establish whether there are any Evolutionarily Significant Units (ESUs). Dispersal and mobility information, which could be obtained from genetic studies, may assist in understanding what is happening with the Western Cape population to understand patterns of gene flow and connectivity between populations. Genetic samples are being collected and are currently housed with the University of Stellenbosch, to understand regional differences with a view to possible future reintroductions.
- Understanding the effects of climate change in reducing suitable grassland habitat.
- Investigate the hypothesis of the species being more of a subterranean species – are we looking for it in the wrong places?
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP). This includes reporting road casualties to the Endangered Wildlife Trust Road Watch application.
Bibliography
Avenant, N.L. and du Plessis, J.J. 2008. Sustainable small stock farming and ecosystem conservation in southern Africa: a role for small mammals? Mammalia 72: 258-263.
Bagniewska JM, Kamler JF. 2014. Do black-backed jackals affect numbers of smaller carnivores and prey? African Journal of Ecology 52: 546–567.
Buschke FT. 2016. A camera trap survey of nocturnal mammals on former farmland in the eastern Free State Province, South Africa, 10 years after removing livestock. African Journal of Ecology 54: 514–518.
Child MF, Rowe-Rowe D, Birss C, Wilson B, Palmer G, Stuart C, Stuart M, West S, Do Linh San E. 2016. A conservation assessment of Poecilogale albinucha. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Cunningham, A.B. and Zondi, A.S. 1991. Use of animal parts for the commercial trade in traditional medicines. Institute of Natural Resources, University of Natal, Pietermaritzburg, South Africa.
Da Rosa BRP. 2019. Optimizing Small Mammal Relative Abundance Measures Using Non-Invasive Sampling and Assessment of its Contribution to Occupancy Modelling of Small Carnivores in Dry Woodland Savannah of South Africa. Master’s thesis, University of Lisbon, Lisbon.
Do Linh San E, Malongwe NB, Fike B, Somers MJ, Walters M. 2009. A diverse autumn diet without dominant prey for opportunistic black-backed jackals Canis mesomelas. Wildlife Biology in Practice. 5(2):96-103.
Foden WB, Young BE. 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Foster IB, McIntyre T, Haussmann NS. 2022. Understanding the relationship between farmers and burrowing mammals on South African farms: are burrowers friends or foes?. Pages 69-81. In: Kerr RB, Pendergrast TL, Smith BJ, Liebert J, Eds. Rethinking Food System Transformation. Springer, New York.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Hargey, A. 2022. Estimating leopard density in a coastal protected area of the Western Cape, South Africa. Msc thesis, University of Cape Town, Cape Town.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Klare UN, Kamler JF, Stenkewitz UT, Macdonald DW. Diet, prey selection, and predation impact of black‐backed jackals in South Africa. The Journal of Wildlife Management. 2010 Jul;74(5):1030-1041.
Koepfli KP, Deere KA, Slater GJ, Begg C, Begg K, Grassman L, Lucherini M, Veron G, Wayne RK. 2008. Multigene phylogeny of the Mustelidae: resolving relationships, tempo and biogeographic history of a mammalian adaptive radiation. BMC biology. 6:1-22.
La Grange MR. 2011. The rate and projected impact of development, with emphasis on mining, on biodiversity in the Mpumalanga Province, South Africa. Honours Thesis. University of Pretoria.
Loock D, SwanepoeL L. 2019. Assessing local patterns in carnivore occupancy and richness by camera trapping: a monitoring of diversity at Verloren Vallei Nature Reserve, Dullstroom, Mpumalanga. Wildlife & Resource Association (WRA), Bloemfontein.
Larivière, S. and Jennings, A.P. 2009. Family Mustelidae (Weasels and relatives). In: D.E. Wilson and R.A. Mittermeier (eds), Handbook of the Mammals of the World. 1. Carnivores, pp. 564-656. Lynx Edicions, Barcelona, Spain.
Lynch CD, Watson JP. 1990. The mammals of Sehlabathebe National Park, Lesotho: species accounts. Navorsinge van die Nasionale Museum: Researches of the National Museum. 6(12):530-43.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Medland, R.D. and Dudley, C.O. 1995. Natural history notes: sightings of the African striped weasel, Poecilogale albinucha, in Nyika National Park. Nyala 18: 42-43.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Minnie L, Gaylard A, Kerley GI. 2016. Compensatory life-history responses of a mesopredator may undermine carnivore management efforts. Journal of Applied Ecology 53: 379-387.
Monadjem A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41(2): 45-59.
Ngwenya MP. 2001. Implications of the medicinal animal trade for nature conservation in KwaZulu-Natal, Report No. NA/124/04. Ezemvelo KZN Wildlife.
Nieman WA. 2018. Culture, Conflict, and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the HumanWildlife Interface. Msc thesis, University of Stellenbosch, Stellenbosch.
Nieman, W.A., Leslie, A.J. and Wilkinson, A., 2018. The Use of Animals and Animal-Derived Constituents in African Traditional Medicine and Other Cultural Applications: Townships in the Western Cape Province. Culture, Conflict and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human-Wildlife Interface, p.215.
Nieman, W.A., Wilkinson, A. and Leslie, A.J. 2020. Farmer attitudes and regional risk modelling of human–wildlife conflict on farmlands bordering the Boland Mountain Complex, South Africa. African Journal of Wildlife Research, 50(1), pp.36-54.
Nieman WA, Schultz BC, Wilkinson A, Leslie AJ. 2021. Stakeholders’ perceptions of mammal occurrence and abundance on agricultural properties bordering the Boland Mountain Complex, South Africa. African Zoology 56(2): 104-116.
NWPG (North West Provincial Government) 2024 The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Powell RA. 1979. Mustelid spacing patterns: variations on a theme by Mustela. Zeitschrift für Tierpsychologie 50(2):153-65.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Power RJ, Van Straaten A, Schaller R, Mooke M, Boshoff T, Nel HP. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Pretorius, M. 2019. Mesocarnivores in Protected Areas: ecological and anthropogenic determinants of habitat use in northern Kwa-Zulu Natal, South Africa. Msc thesis, University of Cape Town, Cape Town.
Pringle JA. 1977. The distribution of mammals in Natal. Part 2. Carnivora. Annals of the Natal Museum 23(1): 93-115.
Ramesh T, Kalle R, Downs CT. 2017. Staying safe from top predators: patterns of co-occurrence and inter-predator interactions. Behavioral Ecology and Sociobiology. 71:41.
Rautenbach, I.L. 1982. The mammals of the Transvaal. Ecoplan Monograph 1:1-211.
Ross, M.D. 2022. Mammalian and avian diversity in a coastal nature reserve and an adjacent eco-estate. MSc thesis, University of Cape Town, Cape Town.
Rowe-Rowe DT, Lowry PB. 1982. Influence of fire small mammal populations in the Natal Drakensberg. South African Journal of Wildlife Research 12: 130–139.
Rowe-Rowe DT. 1978. The small carnivores of Natal. Lammergeyer 25: 1–48.
Rowe-Rowe DT. 1978a. Comparative prey capture and food studies of South African mustelines (Carnivores: Mustelidae). Mammalia 42: 175–196.
Rowe-Rowe DT. 1978b. Reproduction and post-natal development of South African mustelines (Carnivora: Mustelidae). Zoologica Africana 13: 103–114.
Rowe-Rowe, D.T., 1978c. Reproduction and post-natal development of South African mustelines (Carnivora: Mustelidae). African Zoology, 13(1), pp.103-114.
Rowe-Rowe DT. 1982. Biology of Natal Mustelids. M.Sc. Thesis. University of KwaZulu-Natal.
Rowe-Rowe DT. 1990. The African Weasel: A Red Data Book Species in South Africa. Mustelid and Viverrid Conservation.
Rowe-Rowe DT. 1996. Agonistic behaviour of male striped weasels. . Lammergeyer 44: 1–5.
Rowe-Rowe, D.T. 1992. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Schoeman, F., Newby, T.S., Thompson, M.W. and Van den Berg, E.C. 2013. South African national land-cover change map. South African Journal of Geomatics 2(2): 94-105.
Simelane T.S., Kerley G.I.H. 1998. Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research 28: 121–126.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smith K, Venter JA, Peel M, Keith M, Somers MJ. 2023. Temporal partitioning and the potential for avoidance behaviour within South African carnivore communities. Ecology and Evolution. 13(8):e10380.
Stuart, C. and Stuart, T. 1998. New distribution records for Poecilogale albinucha and Rhynchogale melleri in southern Africa. Small Carnivore Conservation 18: 21.
Stuart, C. and Stuart, T. 2013. Poecilogale albinucha African Striped Weasel. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. Volume V: Carnivores, Pangolins, Equids and Rhinoceroses, Bloomsbury Publishing, London, UK.
Stuart, C., Stuart, M. and Do Linh San, E., 2015. Poecilogale albinucha. The IUCN Red List of Threatened Species 2015: e. T41662A45215258 [online]
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Stuart, C.T. and Stuart, M.D. 1990. Striped weasel, Poecilogale albinucha, recorded from the Kalahari Gemsbok National Park. Koedoe 33: 112-113.
Whiting, M.J., Williams, V.L. and Hibbitts, T.J. 2011. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Journal of Zoology 284: 84-96.
Whiting MJ. Williams VL, Hibbits TJ. 2016. A picture of health? Animal use and the Faraday traditional medicine market, South Africa. Journal of Ethnopharmacology. 179:265-73.Wimberger K, Downs CT. 2010. Annual intake trends of a large urban animal rehabilitation centre in South Africa: a case study. Animal Welfare. (4):501-13.
