
African Straw-coloured Fruit Bat
Eidolon helvum

2025 Red list status
Near Threatened
Regional Population Trend
Unknown
Change compared
to 2016
Uplisted
Overview
Eidolon helvum – (Kerr, 1792)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – PTEROPODIDAE – Eidolon – helvum
Common Names: African Straw-coloured Fruit-bat, Giant Fruit-bat, Straw-coloured Flying Fox, Straw-coloured Fruit Bat, Yellow Fruit-bat (English); Geel vrugtevlermuis (Afrikaans)
Synonyms: Vespertilio vampirus ssp. helvus Kerr, 1792; Xantharpyia leucomelas (Heuglin 1862); Xantharpyia leucomelas (Fitzinger in von Heuglin & Fitzinger, 1866) [nomen nudum]; Cynonicteris straminea (Bocage 1895); Rousettus stramineus (Haagner 1920); Eidolon helvum ssp. sabaeum (Anderson, 1907)
Taxonomic notes: Three subspecies have been previously described: Eidolon helvum dupreanum (from Madagascar), E. h. helvum (from the African continent) and E. h. sabaeum (from Arabia) (Skinner & Chimimba 2005).
Red List Status: NT, Near Threatened D1 (IUCN version 3.1)
Assessment Information
Assessors: Richardson, E1., Lotter, C2. & da Silva, J.M.3.
Reviewer: Richards, L.R4.
Institutions: 1Richardson & Peplow Environmental, 2Inkululeko Wildlife Services (Pty) Ltd, 3South African National Biodiversity Institute, 4Durban Natural Science Museum.
Previous Assessors and Reviewers: Monadjem, A., Cohen, L., Jacobs, D., MacEwan, K., Richards, L, Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Roxburgh, L., Raimondo, D., Page-Nicholson, S. & Davies-Mostert, H.
Assessment Rationale
The large-bodied and gregarious African Straw-coloured Fruit-bat occurs widely across the lowland rainforest and savannah regions of Africa. In the assessment region, it has been patchily recorded across the central plateau and along the coasts of South Africa. There is a (possibly seasonal) colony in Tembe Elephant Park in northern KwaZulu-Natal and the finding of a non-volant juvenile in the port of Durban in 2019 suggests they are breeding within the assessment region. The closest confirmed breeding colony is located in Marromeu in central Mozambique. This species exists in modified landscapes and is often recorded in urban areas. The species is in serious decline throughout Africa and is listed as Near Threatened internationally (nearly meets criterion A2d), with an estimated loss of 25–30% over the past 15 years (three generations; generation length = 5 years; Pacifici et al. 2013) due to harvesting pressure for bushmeat and traditional medicine. With only one colony known in the assessment region, and certainly less than 1,000 individuals this species would qualify for Vulnerable status. However, as they are strong flyers a rescue effect from Mozambique would be likely and we thus list this species as Near Threatened. With the growing number of wind farms in South Africa and elsewhere on the continent, the impact of wind turbines on this species must be carefully monitored and adequately mitigated where necessary throughout its range – particularly where roosts of this species occur.
Regional population effects: Within the assessment region, it is either an irregular visitor, or possibly a regular migrant at the edge of its range but is not known to breed within the region. Large-scale feeding and migratory movements have been documented (Richter & Cumming 2008) and thus rescue effects are likely possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: There is one known colony in the assessment region with less than 1,000 individuals.
Red List Index
Red List Index: Uplisted
Recommended citation: Richardson E, Lötter C & da Silva JM. 2025. A conservation assessment of Eidolon helvum. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This fruit-bat is broadly distributed across the lowland rainforest and savannah zones of Africa from Senegal in the west, through to South Africa in the south and Ethiopia in the east (possibly ranging into Djibouti and southern Eritrea). This species is sparse or absent in large areas of the Horn of Africa, central East Africa, and elsewhere (Bergmans 1990). It is a migratory species in parts of its range (such as southern Africa); in West Africa, populations migrate from the forest north into the savannah zone during the major wet season. There are numerous individual records from the central plateaus of South Africa and Namibia, and it has been widely recorded in Zimbabwe, Zambia, Mozambique, Malawi and Democratic Republic of the Congo (Monadjem et al. 2020). The closest confirmed breeding colony to South Africa is at Marromeu, central Mozambique (Cotterill 2001). There is also a large colony that roosts in the Botanical Gardens in Maputo (Mozambique). There is a massive colony at Kasanka National Park, Zambia, where 1.5 million non-breeding individuals congregate, with significant local impacts on the ecology of the landscape (Monadjem et al. 2020). In the assessment region, the species has been recorded from the Central Bushveld and East Kalahari Bushveld bioregions of South Africa, but records have also been reported from Gauteng, North West, Free State, KwaZulu-Natal, Eastern Cape, Western Cape, and Northern Cape provinces. It is frequently found along the coasts.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0m (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 3,869m (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
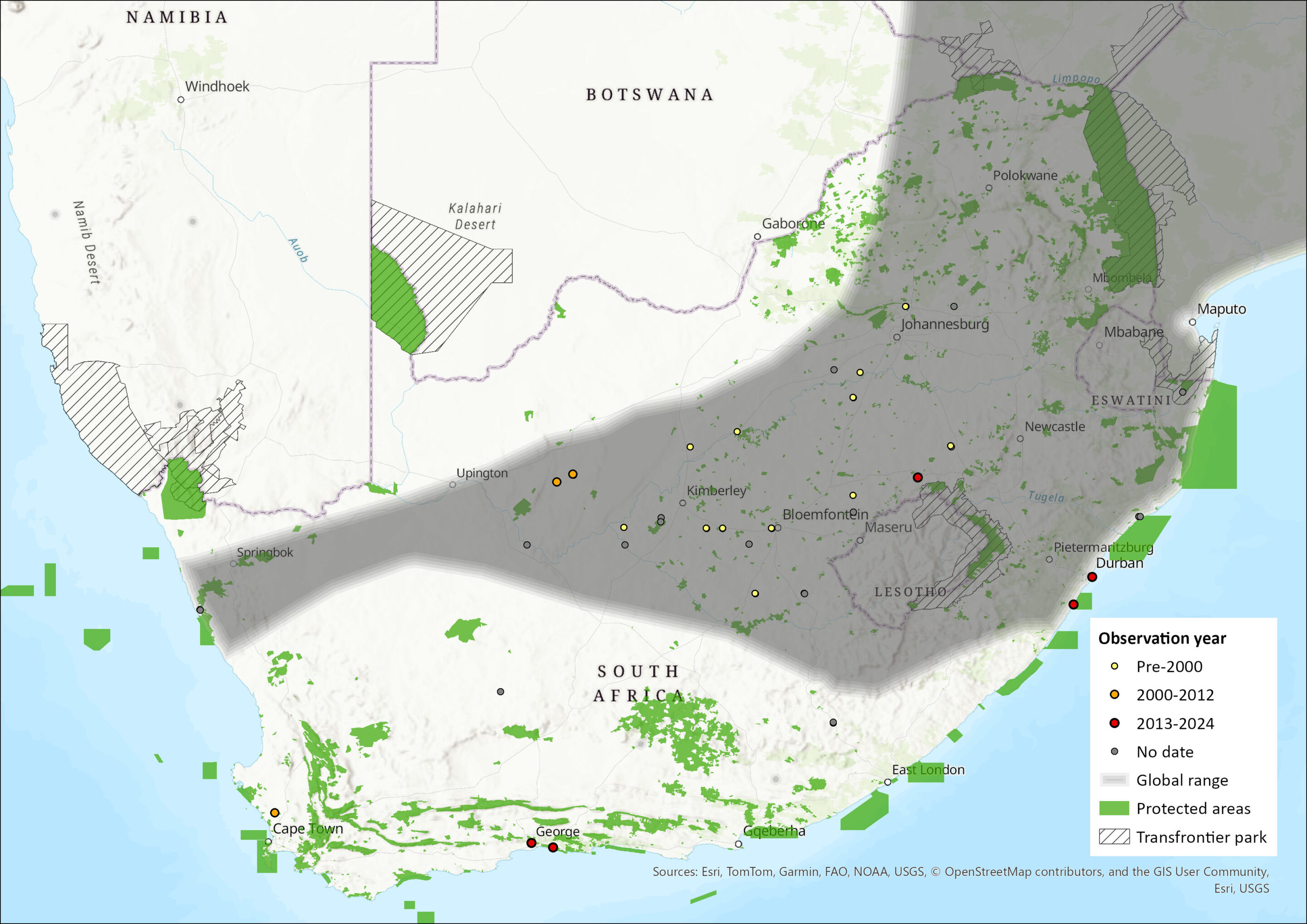 Figure 1. Distribution records for African Straw-coloured Fruit-bat (Eidolon helvum) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for African Straw-coloured Fruit-bat (Eidolon helvum) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Cape Verde Islands | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Presence Uncertain | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Presence Uncertain | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Sao Tome and Principe | Extant | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
Research suggests that climate change could result in negative demographic responses among Pteropodid Old World fruit bats (Festa et al. 2022). More specifically, the African Straw-coloured Fruit Bat could be impacted by climate change due to changes in fruit and flower availability (Sherwin et al. 2012). However, the increasing number of individual migrants of this species in South Africa, as well as the semi-permanent colony in Tembe Reserve in northern KwaZulu-Natal, might suggest that their range is shifting southwards.
All of the Pteropodids are susceptible to extreme weather conditions, especially heat waves (see, for example Matthew et al. 2022) and increased deaths associated with increasing numbers and severity of these may seriously impact species numbers.
Population information
This is a common species across much of its range, forming large colonies of thousands to even millions of individuals (Sørensen & Halberg 2001). Within colonies they may form clusters of up to 100 animals, although this clustering is not always evident in large colonies (Mickleburgh et al. 2008). For example, in western Kenya, total counts of bats at three identified roosts varied from 7,000 to 48,000 individuals (Webala et al. 2014). It is relatively well represented in museums, with over 100 specimens examined in Monadjem et al. (2020). Population size and trend is unknown for the assessment region but is assumed to be under 1,000 individuals. However, the collection of pregnant females from Chiniziwa, central Mozambique and Mutare, eastern Zimbabwe (Cotterill 2001), as well as the presence of a non-volant juvenile in KwaZulu-Natal (Bat Interest Group of KwaZulu-Natal data), affirm that this species does breed in southern Africa.
In West and Central Africa this species is declining due to harvesting for bushmeat (Mickleburgh et al. 2008). Although no declines have been recorded for the population in the assessment region, this should be investigated. With the growing number of wind farms in South Africa and elsewhere on the continent, the impact of wind turbines on this species must be carefully monitored and adequately mitigated where necessary throughout its range, and particularly where roosts of this species occur.
Current population trend: Unknown
Continuing decline in mature individuals? Populations are known to be declining in much of the species’ range but without adequate monitoring numbers cannot be assessed in the region.
Extreme fluctuations in the number of subpopulations: (Not specified)
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown. This is a highly migratory species, so it is not possible to define subpopulations.
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Severely fragmented: No severe fragmentation suspected due to its highly migratory nature.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
No population genetic study has been conducted on this species in relation to the assessment region; however, given its large size, migratory nature and ability to inhabit a wide variety of habitats, it is expected that the species exists as a single metapopulation within the assessment region. The effective population size cannot be estimated due to the lack of population estimates in the region.
Habitats and ecology
African Straw-coloured Fruit-bats are gregarious and live in colonies of several hundred individuals (Skinner & Chimimba 2005; Monadjem et al. 2020). This adaptable species has been recorded from a wide range of habitats: it is commonly found in tropical rain forest, including evergreen forest habit ats, coastal (including mangrove) forest and riverine forest, through moist and dry savannah woodland mosaics (Mickleburgh et al. 2008). Populations can persist in human-modified habitats, and the species is often recorded in urban areas, such as wooded city parks (ACR 2024). Colonies are rarely found in protected areas or in forests, but frequently near human habitations especially gardens, probably because there are fruit trees nearby (Webala et al. 2014). In Kenya, only a few colonies of African Straw-coloured Fruit-bats are known. This is wide-ranging species and individuals have been recorded foraging at distances of up to 59 km from their roosts (Monadjem et al. 2020), with one migrating bat recorded to have moved 370 km in one night and a cumulative 2,518 km in 149 days (Richter & Cumming 2008).
As their name suggests, the species primarily feeds on fruits (wild and cultivated) and flowers (Monadjem et al. 2020). While no information on their diet is available within the assessment region, it assumed that their diet is consistent with other African populations. In southern Africa, the seasonal appearances and disappearances of African Straw-coloured Fruit-bats likely reflect responses of these bats to changing food supplies (Richter and Cumming 2008).
Ecosystem and cultural services: The role of frugivorous bats is crucial in ecosystems as these species perform key functions as pollinators and seed dispersers (Fujita & Tuttle 1991; Hodgkison et al. 2003). For example, Hodgkison et al. (2003) found that 13.7 % of trees in a botanical survey of a 1 ha old-growth forest were partially dependent on bats for pollination and seed dispersal. This species has been shown to retain ingested seeds for long periods and to traverse large distances, making it an important seed disperser in tropical Africa (Abedi-Lartey et al. 2016). In West Africa, the African Straw-coloured Fruit bat is a critically important seed dispersal agent for the economically important and threatened timber tree, the African Teak (Milicia excelsa) (Daïnou et al. 2012).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.7. Forest -> Forest – Subtropical/Tropical Mangrove Vegetation Above High Tide Level | – | Suitable | – |
| 1.8. Forest -> Forest – Subtropical/Tropical Swamp | – | Unknown | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Marginal | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: Forearm mean = 11.93 ±1.03 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Forearm mean = 11.25 ±1.30 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): Female forearm length = 13.50 cm; Male forearm length = 13.0 cm (Monadjem et al. 2020)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Richter and Cumming (2008) documented large-scale feeding and migratory movements of four bats from the Kasanka colony which were radio-tracked using satellite telemetry. These individuals foraged at distances of up to 59km from their roost. One migrating bat moved 370km in one night, and one individual travelled a cumulative 2 518km in 149 days (Monadjem et al. 2020).
Congregatory: Within the assessment region this species is represented by one (possibly seasonal) colony and visiting or vagrant individuals. Outside the assessment region, the Kasanka roost site sees an influx of 1.5 million non-breeding bats between November and January (Sørensen & Halberg 2001).
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be utilised or traded within the assessment region, but it is used for bushmeat and medicine in Central and West Africa where it is one of the most frequently consumed mammals in the region (Mickleburgh et al. 2008). Kamins et al. (2011) estimate, based on interviews with 551 Ghanaians, that a minimum of 128,000 African Straw-coloured Fruit-bats are sold per annum.
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | true | – |
| 3. Medicine – human & veterinary | true | true | true | – |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no recorded threats in the assessment region. However, due to the flight behaviour and migratory movements of this species, it is categorised as having a “High” risk of fatality from wind turbines (MacEwan et al. 2020). Given this, and the growing number of wind farms in Africa, especially South Africa, the impact of wind turbines on this species must be carefully monitored and adequately mitigated where necessary throughout its range – particularly where roosts of this species occur. To date, there has been no known fatality of this species at a wind farm in South Africa (Doty and Martin 2013; MacEwan 2016; Aronson 2022). However, at a wind farm in West Africa, which was constructed near known sizeable roosts of this species, many turbine fatalities of these bats occurred within just the first year of operation. If fatalities of this species remain unmitigated at certain wind farms, this will severely impact affected roosts and populations, and potentially the species as a whole.
African Straw-coloured Fruit-Bats are the most heavily harvested bat for bushmeat in West and Central Africa, where they are threatened by hunting for food and medicinal use, as well as by severe deforestation The species is also persecuted because it often damages fruit plantations (Webala et al. 2014; Monadjem et al. 2020). Large colonies can be very noisy, and roosting sites of large colonies can be messy, sometimes causing defoliation to the trees in which they roost (Kunz 1996). As tree density is an important factor in roost-site selection (Webala et al. 2014), removal of roost trees could impact this species especially outside protected areas. More recently, people are afraid of large concentrations bats due to Ebola outbreaks in Central and West Africa and may remove important roost trees to evict the colonies.
Conservation
This species is present in only a few protected areas across its entire range, with a large roosting colony in Kasanka National Park, Zambia. African populations are also protected under the Convention on the Conservation of Migratory Species of Wild Animals (CMS) since 2001 (Appendix II). Within the assessment region, it has been recorded from two protected areas: Ndumo Game Reserve (Skinner & Chimimba 2005), and Tembe Elephant Park (C Hanekom, pers comm), both in KwaZulu-Natal, which, together with other protected areas in the region may be important refuge habitats for the species (K. Richardson pers. comm. 2016). Throughout its range, conservation measures to mitigate severe deforestation in many parts of Africa are needed. The highest priority is to limit harvesting to sustainable levels (Mickleburgh et al. 2008). However, this threat needs to be investigated within the assessment region where no direct interventions are possible without first identifying and protecting key roost sites through systematic surveys and monitoring. For example, the Kasanka population depends on a functional network of roosting and foraging sites (intact fruiting woodlands) throughout Zambia and DRC (Monadjem et al. 2020). As this species may be threatened by roost tree clearance and direct persecution and/or harvesting, identifying such a functional network of sites for protected area expansion within the assessment region is a priority. Additionally, an education and community outreach programme in local schools and communities was proposed for the long-term conservation of viable populations in western Kenya (Webala et al. 2014), which could potentially be an intervention in the assessment region, depending on the severity of threats.
Additionally, the impact of wind farming should be monitored to determine if there is any impact on this species within the assessment region, and to devise effective mitigation at wind farms elsewhere in Africa where this species is known to be impacted.
Recommendations for land managers and practitioners:
- Identify and protect important roost sites.
- Development of wind farms must strictly avoid encroachment into the prescribed buffers around roosts and other important habitat for fruit bats (see MacEwan et al. 2020), especially since current options for mitigating fatalities at wind farms are more limited for fruit bats compared to insectivorous bats. Although not applicable within the assessment region, in Zambia, Mozambique, and elsewhere in Africa, wind farm development should not be permitted within a 40 km or larger radial zone around the largest roosts of this species (comprising >20,000 individuals).
- Potential future turbine-fatalities of this species must be reduced with interventions such as curtailing turbines at low wind speeds.
Research priorities:
- Systematic surveys to identify additional colonies, key roosting sites and potential breeding sites.
- Research into the migratory and dispersal patterns within the assessment region, as studies on dispersal have been done only in Zambia (for example, Richter & Cumming 2008), and including southern African samples in population genetic studies.
- Research to assess the efficacy of different mitigation methods to reduce fruit bat mortality at wind farms. For example, it is not known whether turbine blade painting (to mitigate avian fatalities) is beneficial or detrimental for fruit bats.
- Possible future wind farm carcasses of this species could be used to obtain greater insight into the biogeography, genetics, and ecology of this species in the assessment region.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Abedi-Lartey M, Dechmann DKN, Wikelski M, Scharf AK, Fahr J. 2016. Long-distance seed dispersal by straw-coloured fruit bats varies by season and landscape. Global Ecology and Conservation 7: 12–24.
Aronson, J. 2022. Current state of knowledge of wind energy impacts on bats in South Africa. Acta Chiropterologica 24: 221-238.
Bergmans, W. 1990. Taxonomy and biogeography of African fruit bats (Mammalia, Megachiroptera).
Cotterill, F. P. D. 2001. New records for two species of fruit bats (Megachiroptera: Mammalia) in southeast Africa, with taxonomic comments. Durban Museum Novitates 26: 53-56.
Daïnou K, Laurenty E, Mahy G, Hardy OJ, Brostaux Y, Tagg N, Doucet J-L. 2012. Phenological patterns in a natural population of a tropical timber tree species, Milicia excelsa(Moraceae): Evidence of isolation by time and its interaction with feeding strategies of dispersers. American Journal of Botany 99: 1453–1463.
Doty AC, Martin AP. 2013. Assessment of bat and avian mortality at a pilot wind turbine at Coega, Port Elizabeth, Eastern Cape, South Africa. New Zealand Journal of Zoology 40: 75–80.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S., 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Fujita MS, Tuttle MD. 1991. Flying foxes (Chiroptera: Pteropodidae): threatened animals of key ecological and economic importance. Conservation Biology 5: 455–463.
Hodgkison R, Balding ST, Zubaid A, Kunz TH. 2003. Fruit bats (Chiroptera: Pteropodidae) as seed dispersers and pollinators in a Lowland Malaysian Rain Forest. Biotropica 35: 491–502.
Kamins AO, Restif O, Ntiamoa-Baidu Y, Suu-Ire R, Hayman DT, Cunningham AA, Wood JL, Rowcliffe JM. 2011. Uncovering the fruit bat bushmeat commodity chain and the true extent of fruit bat hunting in Ghana, West Africa. Biological Conservation 144: 3000–3008.
Kunz TH. 1996. Obligate opportunistic interactions of Old-World tropical bats and plants. In: Hasan Z, Akbar Z (ed.), Conservation and Faunal Biodiversity in Malaysia, pp. 37–65. Penerbit Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia.
MacEwan KL. 2016. Fruit bats and wind turbine fatalities in South Africa. African Bat Conservation News 42: 3–5.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association. South Africa.
Matthew, M et al. (2022) Estimating flying-fox mortality associated with abandonments of pups and extreme heat events during the austral summer of 2019–20. Pacific Conservation Biology 28, 124-139
Mickleburgh S, Hutson AM, Bergmans W, Fahr J, Racey PA. 2008. Eidolon helvum.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Richter, H.V. and Cumming, G.S. 2008. First application of satellite telemetry to track African straw-coloured fruit bat migration. Journal of Zoology 275: 172-176.
Sherwin, H.A., Montgomery, W.I., and Lundy, M.G. 2012. The impact and implications of climate change for bats. Mammal Review. 2012. 10.1111/j.1365-2907.2012.00214.x.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, England.
Sørensen, U. G. and Halberg, K. 2001. Mammoth roost of nonbreeding straw-coloured fruit bat Eidolon helvum (Kerr, 1792) in Zambia. African Journal of Ecology 39: 213–215.
Webala PW, Musila S, Makau R. 2014. Roost occupancy, roost site selection and diet of straw-coloured fruit bats (Pteropodidae: Eidolon helvum) in western Kenya: the need for continued public education. . Acta Chiropterologica 16: 85–94.
