African Four-striped Grass Rat
Rhabdomys dilectus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Rhabdomys dilectus – (De Winton, 1897)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Rhabdomys – dilectus
Common Names: African Four-striped Grass Rat (English)
Synonyms: No Synonyms
Previously listed as synonym of R. pumilo. The R. dilectus group, was first identified based on mitochondrial dna (Rambau et al. 2003). At least two subspecies compose this species, R. dilectus dilectus and R. d. chakae.
Taxonomic Note:
Rambau et al. (2003) recommended the recognition of two sub-species based on cytogenetic and mitochondrial sequence data, R. dilectus dilectus and R. d. chakae. Rhabdomys d. chakae is endemic to southern Africa occurring in South Africa, Eswatini and Lesotho. R. d. dilectus comprises several mitochondrial lineages and chromosomal races (Castiglia et al. 2011, Sabuni et al. 2018). To date, all these taxa are morphologically cryptic. Specifically, in South Africa the distribution of R. d. dilectus is a mosaic interspersed with pockets of R. d. chakae populations. The South African R. d. dilectus type extends into Zimbabwe, while other taxa of R. d. dilectus are found in eastern Africa and Angola (Castiglia et al. 2011, Sabuni et al. 2018, Krásová et al. 2021). The two mesic subspecies (R. d. chakae and R. d. dilectus) have shorter tails than the two arid species (R. pumilio and R. bechuanae). At the southwestern limit of its range, R. d. chakae was found in sympatry with R. pumilio coastal B in Fort Beaufort (du Toit et al. 2012), Grahamstown (Coetzer & Grobler 2018) and the Birha Coastal region in the Eastern Cape Province (Ganem et al. 2020). The urban areas of the Gauteng Province do not seem to limit the distribution of the two subspecies of R. dilectus, resulting in several localities between Pretoria and Johannesburg where the two taxa co-occur (le Grange et al. 2015; Ganem et al. 2020), although R. d. chakae may be the most common taxon in the province. Sympatric areas between the two subspecies were reported in the North West province of south Africa, while sympatry between R. dilectus and R. bechuanae were documented in several locations of the Free State Province (Ganem et al. 2020). Ongoing studies involving nuclear genome markers suggest that the two R. dilectus subspecies probably hybridise (Ganem G. personal communication).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Ganem, G.1 & da Silva, J.M.2
Reviewer: Pillay, N.3
Contributor: Patel, T.4
Institutions: 1Montpellier University,2South African National Biodiversity Institute,3University of the Witwatersrand,4Endangered Wildlife Trust
Previous Assessors and Reviewers: Du Toit, N., Pillay, N., Ganem, G. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Mondajem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
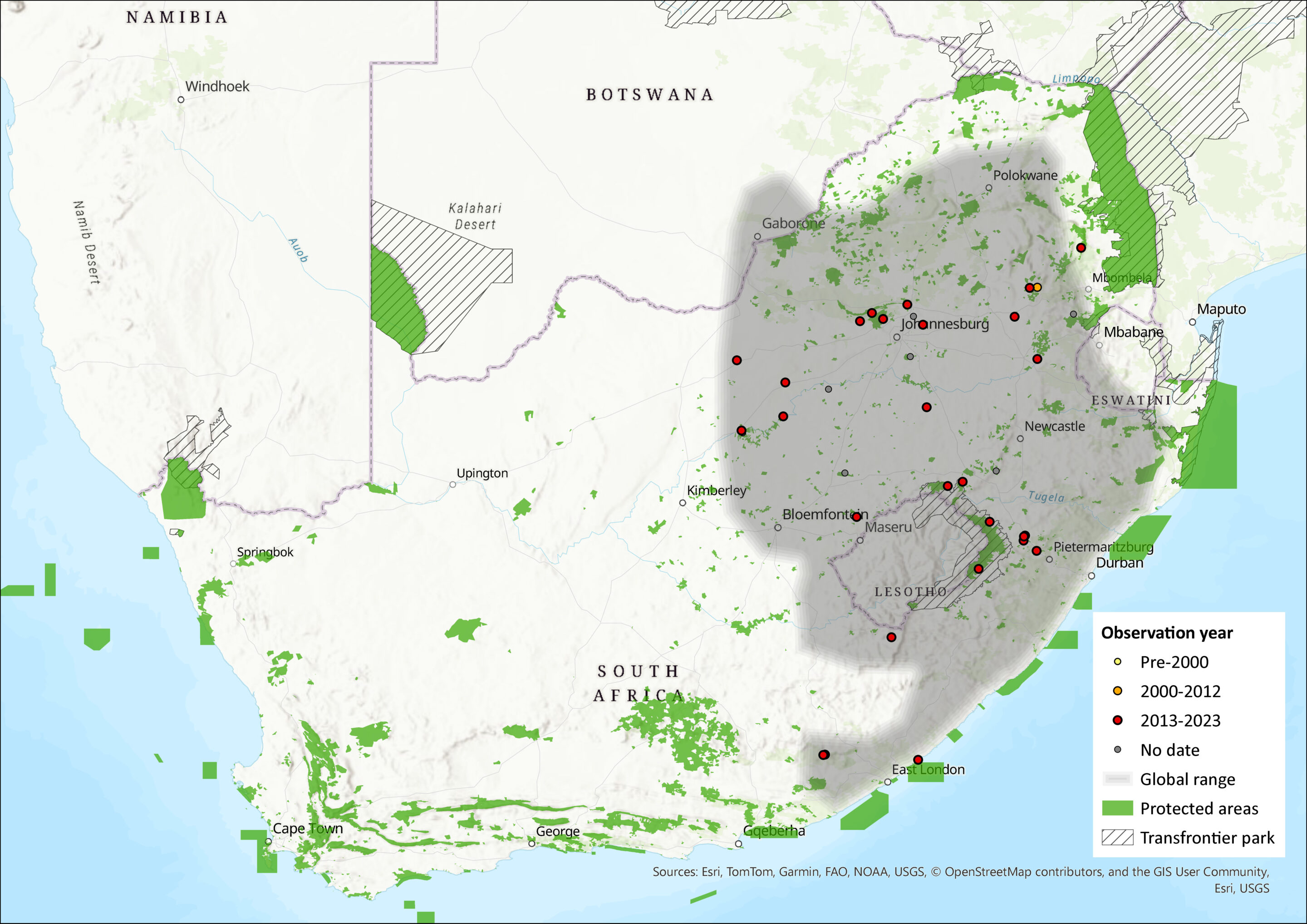
Rhabdomys dilectus has a wide distribution across the northeastern interior and coastal regions of South Africa, as well as Lesotho, Eswatini, Mozambique, Angola, Zimbabwe and eastern Africa. It occurs in many protected areas and is currently listed as Least Concern.
Regional population effects: R. d. chakae has a continuous distribution in the Grassland biome of Lesotho, Eswatini and the central-eastern parts of South Africa, suggesting that many source populations can potentially contribute dispersers over its distribution range, which may not be the case for R. d. dilectus given its limited and patchy distribution (Ganem et al. 2020). In the western limits of its distribution R. d. dilectus distribution overlap with R. bechuanae, and may suffer from competition in a climatic zone, semi-arid, to which it is less adapted (Ganem et al. 2020; Keilani et al. 2024).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Redlist Index
Red List Index: No change
Recommended Citation: Ganem G & da Silva JM. 2025. A conservation assessment of Rhabdomys dilectus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Rhabdomys spp. are broadly distributed across the assessment region. Their extent of occurrence is wide, while the distribution at the local scale is probably patchy and discontinuous. It cannot be stipulated with certainty whether the current distribution differs significantly from the historical distribution.
Within the R. dilectus clade, R. d. chakae, which is endemic to Lesotho and Eswatini, and occurs in the eastern regions of South Africa (including parts of the Free State and the North West provinces) from the Soutpansberg Mountains in the north to the Eastern Cape Province in the south (Ganem et al. 2020; Matamba et al. 2021); while R. d. dilectus occurs in northern South Africa northwards through Zimbabwe, Malawi to East Africa, Angola, as well as Mozambique. The two subspecies are mostly parapatric (Happold 2013) with several cases of sympatry within the species as well as with R. bechuanae and R. pumilio clade B (Ganem et al. 2020). At the southwestern limit of its range, R. d. chakae was found in sympatry with R. pumilio coastal B in Fort Beaufort (du Toit et al. 2012), Grahamstown (Coetzer & Grobler 2018) and the Birha Coastal region in the Eastern Cape Province (Ganem et al. 2020). The urban areas of the Gauteng Province do not seem to limit the distribution of the two subspecies of R. dilectus, resulting in several localities between Pretoria and Johannesburg where the two taxa co-occur (le Grange et al. 2015; Ganem et al. 2020), although R. d. chakae may be the most common taxon in the province. Sympatric areas between the two subspecies were reported in the North West province of south Africa, while sympatry between R. dilectus and R. bechuanae were documented in several locations of the Free State Province (Ganem et al. 2020). Ongoing studies involving nuclear genome markers suggest that the two R. dilectus subspecies probably hybridise (Ganem G. personal communication).
Further molecular research and vetting of museum specimens is needed to more accurately delineate the distribution ranges of the various species.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): R. d. dilectus: 1100 (South Africa); R. d. chakae: 1200
Elevation Upper Limit (in metres above sea level): R. d. dilectus: 1360 (South Africa); R. d. chakae: 1600
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for African Four-striped Grass Rat (Rhabdomys dilectus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: (Not specified)
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
As the distribution of R. d. dilectus is patchy, different explanations were proposed: 1) R. d. dilectus expanding its South African distribution that would have been formerly restricted to the north-east of the country (in the moist vegetation type of the Savannah biome) into the Grassland biome of Gauteng, the North West and Free State provinces; and/or 2) R. d. chakae dispersed into, and established in, areas formerly occupied by R. d. dilectus in Gauteng Province and further west. These two scenarios involve expansion of the two taxa but at different times. Two arguments may favour the second scenario. Indeed, although mismatch analysis of COI haplotypes distribution of the two subspecies indicates that both taxa experienced expansion, R. d. chakae has a continuous distribution in the Grassland biome of Lesotho and the eastern parts of South Africa, suggesting that many source populations can potentially contribute dispersers over its distribution range, which may not be the case of R. d. dilectus given its limited and patchy distribution (from Ganem et al. 2020). Moreover R. d. dilectus western range limit overlap that of R. bechuanae in a relatively semi-arid regions that experience frequent drought. R. bechuanae being more adapted (sensu lato) to dry conditions than R. d. dilectus (Keilani et al. 2024; Ganem et al. 2020), there is a risk that its western distribution might shrink due to R. bechuane progress eastward.
Population
No national abundance information is available for this species and. A comparative study between the Rhabdomys spp. in the arid Succulent Karoo (R. Pumilio) and those in a moist grassland habitats of South Africa (R. d. chakae) found that, as a result of increased rainfall levels, R. d. chakae in grassland regions exhibited much longer breeding seasons (Schradin and Pillay 2005). Additionally, high annual survival rates of R. pumilio resulted in significantly higher population densities compared to R. d. chakae (Schradin and Pillay 2005). In other parts of South Africa, densities of 35–93 individuals / ha, and 30–300 individuals / ha were recorded in the Highveld grasslands (R. dilectus, Brooks 1974).
Population Information
Current population trend: Unknown
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
A phylogenetic study of the genus Rhabdomys did not find support for the two subspecies of R. dilectus (based on one mitochondrial marker, CO1) despite apparent clustering (Ganem et al. 2020). A more fine scale study using nuclear genomic markers could help provide taxonomic clarity, and inform the population genetic structure within this species.
Very preliminary genetic data on R. d. dilectus indicate low genetic diversity (Ganem et al. 2020). Together with evidence for its discontinuous distribution in South Africa, suggests that its conservation status may need revision.
A study of R. d. chakae in Eastern cape and southern Kwazulu-Natal compared three main forest groups (coastal, mistbelt, and scarp forests), showing shallow sequence divergences ranging from 0.001–0.015 (Kimura 2-parameter model) and very little population genetic structure (FST= 0.071787) (Matamba et al. 2021).
Habitats and ecology
R.dilectus individuals were sampled in grasslands (wet and dry), Kikuyu (Pennisetum clandestinum) pastures and disturbed grasslands in Umvoti Vlei Conservancy, KwaZulu-Natal Province (Fuller and Perrin 2001). R. dilectus favours mesic habitats within the Grassland and Savanna Biomes and relies strongly on ground cover (Ganem et al. 2012, Meynard et al. 2012, Dufour et al. 2015). When R. d. dilectus co-occur sympatrically with R. bechuanae it is found to thrive and nest in semi-arid low quality shrubby habitats (Dufour et al. 2015, 2019) R. dilectus are diurnal and crepuscular omnivores with a diet of seeds, fruits, green plant material, Acacia pods, Pinus bark and arthropods (Fuller and Perrin 2001). They are primarily granivorous, but depending on the season, are known to be opportunistically omnivorous, occasionally consuming insects (Perrin and Curtis 1980). Research suggests that the mesic-adapted R. d. chakae is mostly solitary (Schradin and Pillay 2005), however R. d. dilectus was found to nest either solitarily or in groups with little cohesions (Dufour et al. 2019).
The breeding season commences in August (Schradin and Pillay 2005), and extends until March/April in different parts of their range, due to spatiotemporal variation in rainfall patterns (Schradin and Pillay 2005). The start of the breeding season is dependent on a combination of food availability, temperature and rainfall (Brooks 1974; Perrin 1980a, 1980b; Rowe-Rowe and Meester 1982). Young are generally born in the summer months between September and April (Smithers and Wilson 1979, Perrin 1980b, Rowe-Rowe and Meester 1982, Nel et al. 1984, David and Jarvis 1985, Wirminghaus and Perrin 1992, Lynch 1994). A mean gestation period of 25.4 days has been suggested (Brooks 1974, 1982). Litter size averaged 5.8 (range, 3–8) in Lesotho (Lynch 1994), 4.6 (range, 3–7) in the Drakensberg (Rowe-Rowe and Meester 1982), 5.9 (range, 2–9) in Gauteng (Brooks 1974), and 6.1 (range, 1–11) in KwaZulu-Natal (Taylor 1998).
Ecosystem and cultural services: Rhabdomys species is particularly important forage species for diurnal raptors, snakes and small mammals, including Caracal (Caracal caracal), Black-backed Jackal (Lupulella mesomelas), African Wildcat (Felis silvestris) and several species of mongoose (De Graaff 1997), as they are one of the few diurnal rodent species. Since different species of the complex have different ecological requirements, they may be used as bio-indicators of changes in aridity. R. dilectus is thought to rely strongly on vegetation cover (Meynard et al. 2012; Dufour et al. 2015).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Rhabdomys is currently only being utilised for research and museum collections. It is an important research model to assess ecological diversification, and responses to enhanced dry conditions.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
R. d.dilectus patchy distribution across its range in South Africa, put it into risk of local extinction with reduced probability of recolonisation from nearby source populations.
Conservation
The species is located in many protected areas throughout its range, in provincial reserves of the Free State, Gauteng, the Northwest province, Limpopo and Gauteng. No conservation interventions are deemed necessary for R. d. chakae, however the patchy distribution of R. d. dilectus may need further attention.
Recommendations for land managers and practitioners:
- R. dilectus may benefit from suitable land management: landowners should leave corridors of grassland between grazed areas and decrease stocking rates.
Research priorities:
- Factors inducing R. d. dilectus patchiness
- Species geographical distribution, morphometrics, genotyping of populations and taxonomic assessment. This includes vetting of existing museum specimens.
- Species social and general biology.
- Ecology and mechanisms of coexistence with other Rhabdomys species.
- Continued research into the status of the species with genomic nuclear markers.
There is a long-term ongoing collaboration between WITS University (Prof. N. Pillay), National Museum Bloemfontein (Dr N. L. Avenant) and Montpellier University France (Prof. G. Ganem), addressing the distribution of Rhabdomys species, investigating species interaction in contact zones, population dynamics, and adaptation to arid conditions. This collaboration involves extensive field work, genomic studies, behavioral ecology and ecophysiology.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP), especially outside protected areas. However, due to their morphological similarities, identification to species level may prove difficult, especially in zones of sympatry.
Bibliography
Brooks, P.M. 1974. The ecology of the four-striped field mouse, Rhabdomys pumilio(Sparrman, 1984), with particular reference to a population on the Van Riebeeck Nature Reserve, Pretoria. D.Sc. Thesis. University of Pretoria.
Brooks, P.M. 1982. Aspects of the reproduction, growth and development of the four-striped field mouse, Rhabdomys pumilio (Sparrman, 1784). Mammalia 46: 53–64.
Castiglia, R., Solano, E., Makundi, R.H., Hulselmans, J., Verheyen, E. and Colangelo, P. 2012. Rapid chromosomal evolution in the mesic four-striped grass rat Rhabdomys dilectus (Rodentia, Muridae) revealed by mtDNA phylogeographic analysis. Zoological Systematics and Evolutionary Research 50: 165–172.
Coetzer, W.G. and Grobler, J.P. 2018. Identifying Rhabdomys museum specimens following taxonomic changes: use of short COI sequences. Vertebrate Zoology, 68, pp.191-197.
David J.H.M. and Jarvis J.U.M. 1985. Population fluctuations, reproduction and survival in the striped fieldmouse Rhabdomys pumilio on the Cape Flats, South Africa. Journal of Zoology 207: 251–276.
De Graaf, G. 1997. Striped mouse Rhabdomys pumilio. In: G. Mills and L. Hes (eds), The Complete Book of Southern African Mammals, Struik Publishers, Cape Town, South Africa.
Dufour, C.M., Meynard, C., Watson, J., Rioux, C., Benhamou, S., Perez, J., Du Plessis, J.J., Avenant, N., Pillay, N. and Ganem, G. 2015. Space use variation in co-occurring sister species: response to environmental variation or competition? PloS One 10(2): e0117750. DOI: 10.1371/journal.pone.0117750.
Dufour, C.M.S., Pillay, N., Avenant, N., Watson, J., Loire, E. and Ganem, G. 2019. Habitat characteristics and species interference influence space use and nest-site occupancy: implications for social variation in two sister species. Oikos 128: 503-515. 10.1111/oik.05357
du Toit , N., Jansen van Vuuren , B., Matthee, S. and Matthee, C.A. 2012. Biome specificity of distinct genetic lineages within the four-striped mouse, Rhabdomys pumilio (Rodentia: Muridae) from southern Africa with implications for taxonomy. Molecular Phylogenetics and Evolution 65: 75–86.
Fuller, J.A. and Perrin, M.R. 2001. Habitat assessment of small mammals in the Umvoti Vlei Conservancy, KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 31: 1-12.
Ganem, G., Dufour, C.M.S., Avenant, N.L., Caminade, P., Eiseb, S.J., Tougard, C. and Pillay, N. 2020. An update on the distribution and diversification of Rhabdomys sp. (Muridae, Rodentia). Journal of Vertebrate Biology 69(2): 20013.1–17. https://doi.org/10.25225/jvb.20013
Happold, D.C.D. 2013. Rhabdomys pumilio Four-striped Grass Mouse. In: D.C.D. Happold (ed.), Mammals of Africa vol. III: Rodents, Hares and Rabbits, pp. 545–547. Bloomsbury, London.
Keilani, H., Avenant, N., Caminade, P., Pillay, N. and Ganem, G. 2024. Do sister species respond similarly to dry conditions? An ecophysiological approach in natura, BioRxiv. https://doi.org/10.1101/2024.03.11.583554
Krásová, J., Mikula, O., Bryja, J., Baptista, N.L., António, T., Aghová, T. and Šumbera, R. 2021. Biogeography of Angolan rodents: The first glimpse based on phylogenetic evidence. Diversity and Distributions, 27(12), pp.2571-2583.
Le Grange, A., Bastos, A.D., Brettschneider, H. and Chimimba, C.T. 2015. Evidence of a contact zone between two Rhabdomys dilectus (Rodentia: Muridae) mitotypes in Gauteng province, South Africa. African zoology, 50(1), pp.63-68.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Matamba, E., Richards, L.R., Cherry, M.I. and Rambau, R.V. 2021. DNA barcoding of the mesic adapted striped mouse, Rhabdomys dilectus in the Eastern Cape and KwaZulu-Natal provinces of South Africa. Vertebrate Zoology, 71, pp.503-515.
Meynard, C.N., Pillay, N., Perrigault, M., Caminade, P. and Ganem, G. 2012. Evidence of environmental niche differentiation in the striped mouse (Rhabdomys sp.): inference from its current distribution in southern Africa. Ecology and Evolution 2: 1008–1023.
Nel, J.A., Rautenbach, I.L., Els, D.A. and De Graaff, G. 1984. The rodents and other small mammals of the Kalahari Gemsbok National Park. Koedoe 27: 195–220.
Perrin MR, Curtis BA. 1980. Comparative morphology of the digestive system of 19 species of Southern African myomorph rodents in relation to diet and evolution. South African Journal of Zoology 15: 22–33.
Perrin MR. 1980b. The breeding strategies of two coexisting rodents, Rhabdomys pumilio and Otomys irroratus: with a brief review of some pertinent life history ideas. Acta Oecologica. Oecologia Generalis (France) 1: 383–410.
Perrin, M.R. 1980a. The feeding habits of two co-existing rodents, Rhabdomys pumilio (Sparrman, 1784) and Otomys irroratus (Brants, 1827). Acta Oecologica-Oecologia Generalis 1: 71–89.
Rambau, R.V., Robinson, T.J. and Stanyon, R. 2003. Molecular genetics of Rhabdomys pumilio subspecies boundaries: mtDNA phylogeography and karyotypic analysis by fluorescence in situ hybridization. Molecular Phylogenetics and Evolution 28: 564–575.
Rowe-Rowe, D.T. and Meester, J. 1982. Population dynamics of small mammals in the Drakensberg of Natal, South Africa. Zeitschrift für Säugetierkunde 47: 347–356.
Sabuni, C., Aghová, T., Bryjová, A., Šumbera, R. and Bryja, J. 2018. Biogeographic implications of small mammals from Northern Highlands in Tanzania with first data from the volcanic Mount Kitumbeine. Mammalia, 82(4), pp.360-372.
Schradin, C. and Pillay N. 2005. Demography of the striped mouse (Rhabdomys pumilio) in the succulent karoo. Mammalian Biology 70: 84–92.
Smithers, R.H.N. and Wilson, V.J. 1979. Check List and Atlas of the Mammals of Zimbabwe Rhodesia. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Wirminghaus, J.O. and Perrin, M.R. 1992. Diets of small mammals in a southern African temperate forest. Israel Journal of Zoology 38: 353–361.