African Clawless Otter
Aonyx capensis

2025 Red list status
Near Threatened
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Aonyx capensis – (Schinz, 1821)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – MUSTELIDAE – Aonyx – capensis
Common Names: African Clawless Otter, Cape Clawless Otter (English), Groot-otter (Afrikaans), Qibi, Thene (Sesotho), Ntsini (Swati), leNyibi (Tswana), Nivho, Tshphu (Venda), iNtini (Xhosa), umThini (Zulu)
Synonyms: No Synonyms
Taxonomic Note:
Wozencraft (2005) regarded this species as conspecific with the congeneric Congo Clawless Otter (Aonyx congicus). The two are here retained as distinct species (see van Zyll de Jong 1972, Wozencraft 1993, Somers and Nel 2013). Coetzee (1977) recognised three subspecies A. c. capensis (including coombsi and angolae), from southern Africa, W Zambia, Angola, Gabon, Nigeria and throughout West Africa; A. c. hindei (including helios), from Uganda to Eastern Democratic Republic of Congo as far south as Northern Zambia; and A. c. meneleki, from Ethiopia. Only one subspecies, A. c. capensis, has been recognised within the assessment region (Meester et al. 1986), reaching as far north as Zambia and Angola (Skinner and Chimimba 2005).
Red List Status: NT – Near Threatened, C2a(i) (IUCN version 3.1)
Assessment Information
Assessors: Power, R.J.1, Nicholson, S.2 & da Silva, J.M.3
Reviewer: Do Linh San, E.4
Contributor: Patel, T.2
Institutions: 1North West Provincial Department’s Directorate of Biodiversity Management, 2Endangered Wildlife Trust, 3South African National Biodiversity Institute, 4Sol Plaatjie University
Previous Assessors: Okes, N., Ponsonby, W. & Rowe-Rowe, D.
Previous Reviewers: Avenant, N. & Somers, M.J.
Previous Contributors: Nicholson, S., Selier, J., Child, M.F. & Do Linh San, E.
Assessment Rationale
African Clawless Otters are widespread, but patchily distributed, within the assessment region. The most likely population estimate ranges from 21,500 to 30,276 individuals, of which 11,825–19,377 can be considered mature (using 55–64% mature structure) (numbers based on 2016 assessment). However, systematic density estimates are needed to more accurately estimate population size, and this species should be reassessed once such data are available. While most subpopulations may currently be stable, future trends in coastal and riverbank development, combined with climate change, may see significant declines for this species. Local declines are beginning to be documented, for example, there is some evidence that a river site in the Drakensberg, KwaZulu-Natal (KZN), has experienced a 75% decrease in African Clawless Otter density from 1993 to 2010. Similarly, there are estimated to be three times as many otters in peri-urban than urban areas in Gauteng Province, indicating that disturbed rivers are less suitable for this species. Both examples illustrate reduction in abundance with riparian habitat transformation, pollution, and disturbance. A decline in habitat suitability is also expected. For example, in South Africa’s dry interior, increased pumping of water from persistent pools in ephemeral rivers is expected to lead to more river stretches being drier for longer periods, impacting on prey availability, dispersal and social interactions between otter groups/local subpopulations. While the construction of more weirs may create more bodies of permanent or near permanent water, these impact negatively on river ecosystems and are therefore also expected to impact on the African Clawless Otter subpopulations. The effect of damming thus needs to be researched. Similarly, canalisation of rivers will negatively impact otter occurrence, possibly due to a reduction in both food (due to a lack of substrate) and shelter (due to a lack of riparian vegetation). Further research such as collaring and tracking, behavioural studies, and habitat disturbance impact studies are necessary to monitor potential local declines from across the species’ range. Establishment of long-term monitoring sites to measure subpopulation trends, and implementation of appropriate effective management, are essential to prevent a further decline in the number of individuals. As the population is not estimated to be much more than 10,000 mature individuals, no subpopulation is likely to have over 1,000 mature individuals and there is an inferred continuing decline in occupancy, the listing of Near Threatened C2a(i) remains. However, contemporary density estimates are required from across the species’ range to calculate overall population size more accurately. Similarly, establishing long-term monitoring sites will enable estimation of population trends in different regions. Once these data are available a reassessment should be done. Riverside, wetland and coastal habitats must be protected to allow otters sufficient breeding and foraging environments necessary for them to persist and disperse between habitats. Occupancy sign surveys provide a rapid, cost-effective method for method for monitoring changes in otter occupancy that could be implemented by the relevant management authorities at local or provincial scales.
Regional population effects: The range is continuous with the rest of Africa and as they can travel long distances there is suspected to be dispersal between regions. There is a possibility for dispersal from neighbouring regions along watercourses and coastlines, but it is unknown if this is significant enough to lead to rescue effects.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Power RJ, Nicholson S & da Silva JM. 2025. A conservation assessment of Aonyx capensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The African Clawless Otter is the most widely distributed otter species in Africa, with a range extending from Senegal and Mali throughout most of West Africa to Sudan and Ethiopia, and then southwards throughout East Africa to the southern tip of South Africa. They are absent from the Congo basin, where they are replaced by the Congo Clawless Otter (Aonyx congicus), the two species being sympatric in Uganda and Rwanda (Somers and Nel 2013).
The contemporary distribution of the African Clawless Otter is likely not that different to the past distribution. The historical distribution baseline has always been the Eastern Cape (Lynch 1989), Western Cape, and even tributaries of rivers in the upper Karoo of the Northern Cape, and the Orange river (Stuart 1981). In 1839, an Otter was reported from the Kuruman area of the Northern Cape (Skead 2011), suggesting a wider past distribution which may have been related to fluctuations in rainfall over time. They have occurred throughout the northeast of South Africa (Skinner & Chimimba 2005; Nel & Somers 2007), including Kwazulu-Natal (Pringle 1977; Rowe-Rowe 1992a) and Free State (Lynch 1983). The species is known from Lesotho (Lynch 1994) and Eswatini (Monadjem 1998). The species is presently widely distributed through most of South Africa (Nel & Somers 2007; Okes et al. 2016).
The African Clawless Otter occurs in all Bioregions of the North West Province (Power et al. 2019), and marginally so in the edge of the Kalahari in the west, at their range extremities (Nel & Somers 2007), where there may have even been a recession in the distribution edge there. In earlier times, the species was observed on many occasions in the southern parts of the province, within the greater Vaal River catchment (Power 2014). The species occurs in Limpopo and is widely distributed along its river systems in the Soutpansberg (Haring et al. 2023), with a few occurrences along the Limpopo itself (Okes et al. 2016). It is not clear why there is a dearth of occurrence records in the Waterberg, especially considering the expected good quality of water there. The species occurs at Verloren Vallei in Mpumalanga at low prevalence (Loock & Swanepoel 2019; Majelantle et al. 2021), and elsewhere on the Highveld of the province (Majelantle et al. 2021), and occurs in the Lowveld, notably along the Sabie and Crocodile. They occur throughout Gauteng, even occurring where rivers traverse urban areas (Ponsonby & Schwaibold 2019). Occurrences are sparse throughout the Free State (Okes et al. 2016), but they seem to be prevalent along the boundary rivers of the Orange and Vaal. In KwaZulu-Natal, they are likely widespread (Okes et al. 2016), and present in the Drakensberg foothills (Majelantle et al. 2021), as well as Zululand coastal areas (Nicolaides 2023). They are widespread in the Eastern Cape, even in the Karoo areas. In the Western Cape, they occur throughout, but mostly the coastal zone (Okes et al. 2016), and in the valleys of the Cape Fold Mountains too (Nieman 2018). Regionally, in the Cape Peninsula, the species has been found near urban areas (Okes & O’Riain 209) but are generally localised to preferred habitat (Okes & O’Riain 2017). Since the species dependent on sources of fresh water (Verwoerd 1987; Van Niekerk et al. 1998), populations using the marine environment will always occur at river mouths or other freshwater inlets along the coastline (Van Niekerk et al. 1998).
In 2019, in the Nuweveld Mountains of the Western Cape Karoo, African Clawless Otter seemed to disappear, likely due to the intensified drought in the region at the time, causing a reduction in both the length of streams and quantity of water bodies such as dams and natural pools (Schurch et al. 2021).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 3000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
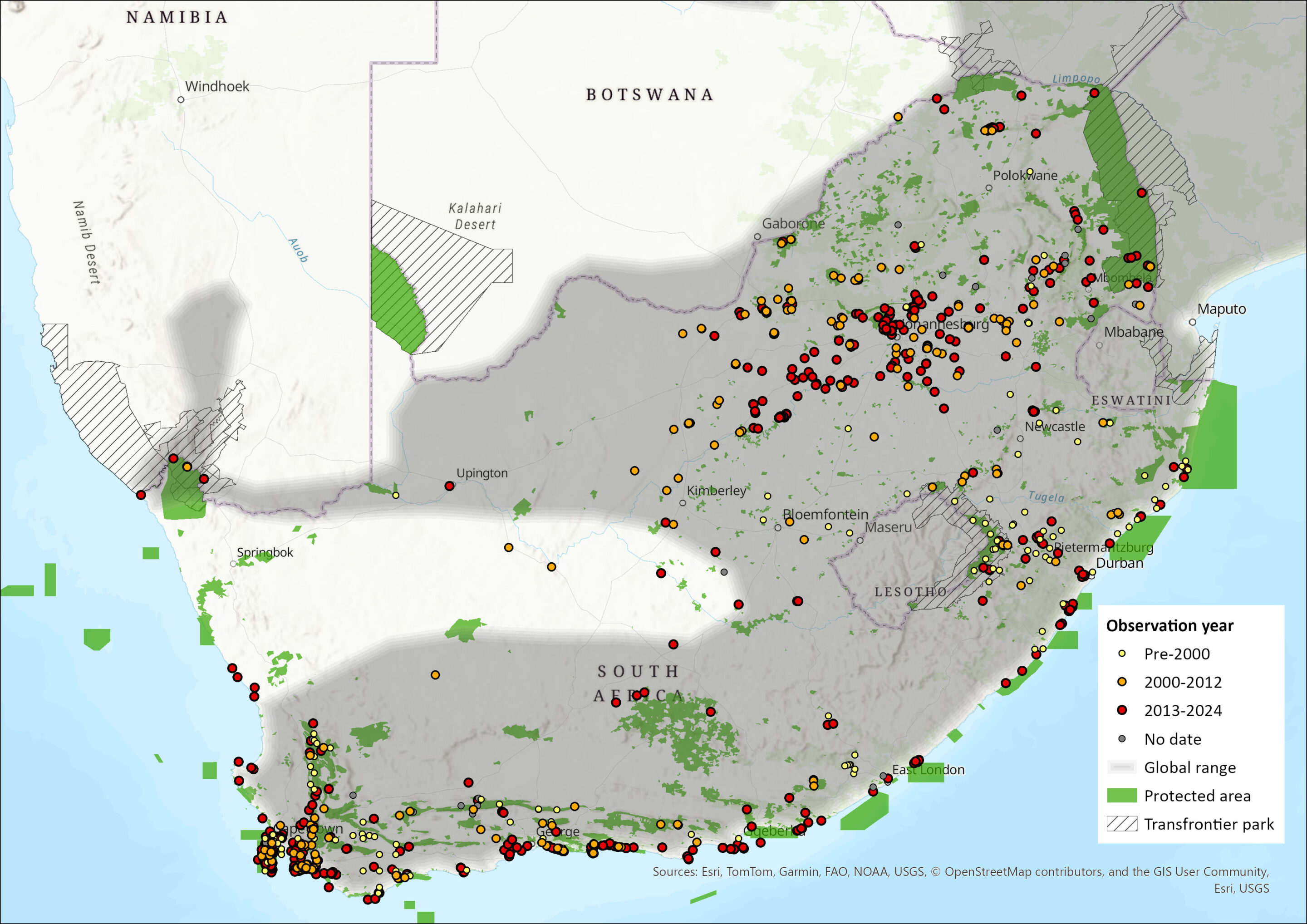
Map
Figure 1. Distribution records for African Clawless Otter (Aonyx capensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
In the arid parts of the North West, the African Clawless Otter locally occurs in the Eastern Kalahari Bioregion (Power et al. 2019), and a concern was raised that relict populations in ephemeral streams would become genetically isolated should dry conditions prevail for a sufficient period, and local extinctions could take place (Power 2014). In the Eastern Cape, during a drought period during the 1990s, the diet of African Clawless Otter then (Somers & Purves 1996), could give insight into the foraging behaviour of this species should such conditions prevail again. The species still fed on normal prey, but they had a high prevalence of insects in their diet, while still maintaining dietary separation among the other sympatric semi-aquatic carnivores (Somers & Purves 1996).
It is important to consider a species’ ecological and biological traits to determine the likely mechanisms of climate change impact and quantify this using expert knowledge (Foden & Young 2016), and to use the best available empirical information (Cianfrani et al. 2018). In an attempt to assess how otters can be impacted by climate change, Cianfrani et al. (2018) considered components of species’ intrinsic sensitivity to climate change (climatic niche specialisation and marginality) together with components of extrinsic exposure (changes in range extent, fragmentation, coverage of protected areas, and human footprint) to develop an integrated vulnerability index to climate change for the world’s freshwater otters. As top freshwater predators, otters are among the most vulnerable mammals, with most species being threatened by habitat loss and degradation. All dimensions of climate change exposure were based on present and future predictions of species distributions. For a given species, climate change can have both positive and negative effects on different components of extrinsic exposure, and that measures of species’ sensitivity are not necessarily congruent with measures of exposure. The global vulnerability index predicted that all freshwater otters would be negatively affected by climate change, and the most impacted species would be the three African species, including the African Clawless Otter (Cianfrani et al. 2018). A modelled climate change scenario would result in a 7% decline in the African range for the African Clawless Otter by 2050 (Cianfrani et al. 2018). Specifically, when forecasting for the year 2050, most of the South African range is stable for the species for the near future, but a range recession would take place in the arid Northern Cape (Cianfrani et al. 2018). Furthermore, under a future climate change regime, for African Clawless Otter, the protection coverage was predicted to decrease from -8% to -22%. Therefore, the impact of range shrinkage may be exacerbated by the decreasing coverage of protected areas and by ranges shifting towards more human-impacted areas (Cianfrani et al. 2018).
Given aridity is expected to be prevalent, especially in the hinterland of South Africa (Foden & Young 2016), less water provision from the catchments may mean less water flow to the oceans. Since the species is dependent on sources of fresh water, reduced stream flow and drying up of rivers may negatively affect continued species occurrence along the coastline (Van Niekerk et al. 1998). On top of this, is the low genetic diversity of the species, suggesting these animals may not be able to cope with future environmental changes (Ponsonby et al. 2019), though this may be of regional extent only.
However, the forecast distribution may not appear that severe as most of the range appears to be stable (Cianfrani et al. 2018), and the perennial Orange river population will likely buffer any range losses in the arid west of the country. Since the species is a generalist, opportunistic feeder, it is likely that it will be able to adapt to potential prey species and abundance changes associated with future anthropogenically driven changes (Jordaan et al. 2015). This behavioural plasticity may locally counter wider systemic range contractions.
Population Information
In earlier times, in the Garden Route National Park, along the Tsitsikamma coastline of the Eastern Cape, the mean population density estimate in 1977 was one otter per 2 km (Van der Zee 1982), then 5 years later it was similarly found to be 1.9 km of coast (Arden-Clarke 1986). Further west, on the Betty’s Bay coastline, a population density of one otter per 2 km of coastline was suggested (Verwoerd 1987), approximately the same as found at the Tsitsikamma coastline in the now Garden Route National Park (Van der Zee 1982; Arden-Clarke 1986; Verwoerd 1987). In the Western Cape later on, based on the recovery of radioactive scats, Somers (2001) gives an estimate of 1.5 individuals / km of river.
Extensive population survey work has been done in the Kamberg-Loteni sections of the now Maloti-Drakensberg Park on African Clawless Otter from the 1970s (Rowe-Rowe 1992b), through 1990s (Perrin & Carugati 2006), and up until 2010 (Khubeka et al. 2013; Rowe-Rowe 2016). In 1994, species relative abundance was 1 Otter per 1.25 km of river in the Loteni and Mooi rivers, at Loteni and Stillerust near Kamberg, respectively (Perrin & Carugati 2006). Nearby, the density was 1 Otter per 2.5 km of river in the Polela river at Cobham, as well as the Mooi river flowing through the ‘Hatchery area’, and in farmlands outside the protected area (Perrin & Carugati 2006). The relative abundance of African Clawless outside the protected area was half that of the Protected area (Perrin & Carugati 2006). In an earlier time, for the same Stillerust area, up until 1974, the Otter density per km, was one otter/ 0.7- 0.84 km (Rowe-Rowe 1992b), so a minor decline had already been evident (Perrin & Perugati 2006). This decline was more drastic later on, and only 25% of otter sign was found there in 2010 when compared to earlier (Kubheka et al. 2013; Rowe-Rowe 2016). In stark contrast, when comparing to the earlier baselines in the reserve at the ‘hatchery area’, the abundance of sign was the same as before (Kubheka et al. 2013), and density even higher, i.e one otter/ 1-1.7 km (Rowe-Rowe 2016). More recently, in the same Park in the Drakensberg, the density of the species at the Cobham section was 2.1±0.6/km2 (Majelantle et al. 2021), though it is unclear how one would compare this conventional density to the earlier linear density estimates (cf Rowe-Rowe 1992; Perrin & Carugati; Kubheka et al. 2013; Rowe-Rowe 2016), and it may even be similar.
In Mpumalanga, in another reserve, the density of African Clawless Otter in the Verloren Vallei Nature Reserve was 0.7±0.2/km2 (Majelantle et al. 2021). Regionally nearby, the density of the species, at the relatively transformed, Millstream Farm was 8.2±2.3/km2 (Majelantle et al. 2021). In KwaZulu-Natal, using random encounter models, the density of African Clawless Otter in the Umlalazi Nature Reserve was 3.26 otters/ km2 (Nicolaides 2023). Similarly, nearby, at the more disturbed Zini Fish Farm in Mtunzini it was 5.47 Otters/ km2 (Nicolaides 2023).
Interestingly, this recent research has shown higher abundances of African Clawless Otter in more disturbed, yet likely artificially enriched habitats for the species, where the abundance may be between 1.7 (Nicolaides 2023) to between 3.9-11.7 times higher (Majelantle et al. 2021). This contrasts to earlier research, where populations outside protected areas were half that of the Protected areas (Perrin & Carugati 2006). In Gauteng, using sign abundance, there was neither a statistical difference between the abundance of this species in urban and peri-urban areas of Gauteng (Ponsonby & Schwaibold 2019). Although no numbers can be availed, in the North West it seemed the species presence in river systems in not a good indicator of river health as they seemed most abundant in the Vaal river (Power 2014), as since river turbidity may be inconsequential, as they forage by feel, and not by sight, they may be more abundant in the middle and lower reaches of rivers, due to higher food biomass (Nel & Somers 2007).
Although reported widely in Lesotho (Lynch 1994), especially in the Lesotho Highlands, no density estimates are known. Instead, densities of latrine sites are commonly reported on during environmental impact assessments (for example, du Plessis et al. 2014).
A population size of 14,000 individuals was calculated from 26,000 km of river and 2,000 km of coast (density estimated at 1 individual / 2 km river or coast) (Friedmann & Daly 2004). Following this, it was estimated that the population most likely ranges from 21,500 to 30,276 individuals, of which 11,825–19,377 can be considered mature (using 55–64% mature structure) (Okes et al. 2016). However, population size could be as high as 50,433 (27,738–32,277 mature individuals).
Methods used to estimate densities mostly relied on sign surveys carried out at localised scales and thus current systematic density estimates on a national scale are needed to calculate the current mature population size more accurately. Long-term monitoring sites should be established to allow comparative studies to be produced and evidence amassed for a possible net decline in population size over three generations (see Kubheka et al. 2013; Rowe-Rowe 2016), estimated to be a 13-year period (Pacifici et al. 2013), especially as future trends in coastal and riverbank development, combined with climate change, may see significant declines for this species. Many areas may be suffering local declines or extinctions already due to unfettered development or ecosystem modification.
Population Information
Current population trend: Declining
Continuing decline in mature individuals: No
Number of mature individuals in population: 11,825-19,377
Number of mature individuals in largest subpopulation: < 1,000
Number of subpopulations: Unknown
Severely fragmented: No. They can move long distances (> 30 km) through sandy, waterless habitat (an individual tracked by spoor) and cross saddles in mountains between watersheds (a collared individual) (Nel & Somers 2007).
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While a comprehensive population genetic study on this species has not been undertaken, a localised study has been. The genetic diversity of this species in Gauteng was found to be low (mean observed heterozygosity, Ho=0.309) (Ponsonby et al. 2019), with no genetic structure identified between rivers (Ponsonby et al. 2019), which is interesting given the different catchments which converge in the province, with an intervening watershed spanning an urban area. This suggests the species is capable of dispersing between catchments.
The movement of the African Clawless Otter is likely not affected by physical barriers in urbanised areas (Ponsonby et al. 2019), and their highly localised linear habitat may facilitate ease of movement. Consequently, it is possible the species exists as a single metapopulation within the assessment region.
Because the genetic diversity of the species in Gauteng is low, these animals may not be able to cope with future environmental changes (Ponsonby et al. 2019). It may be different elsewhere in the country, and it will only be known if a similar study were conducted.
While effective population size (Ne) estimates are not available, they can be inferred based on the available population size estimates. At least 11,825–13,760 mature individuals and up to 27,738–32,277 mature individuals are expected to occur within the assessment region (see Population Section). Applying a 0.1-0.3 Ne/Nc conversion ratio yields Ne between 1,183 and 9,683 individuals, far exceeding the 500-threshold expected for a healthy and stable population.
Given similarity in the scats of African Clawless Otter and Spotted-Necked Otter, genetic methods are availed to discriminate the species apart (Madisha et al. 2015). Novel mitochondrial genomes are described for the African Clawless Otter. Only 19 out of the 36 genes showed some level of variation between it, and the Spotted-Necked Otter. Such variations may provide guidance in selecting gene regions during marker development for phylogenetic assessments (Madisha et al. 2019).
Habitats and ecology
African Clawless Otters are predominantly aquatic and seldom found far from permanent water. Fresh water is an essential habitat requirement, not only for drinking but also for rinsing their fur. As otters do not have a subcutaneous layer of fat like most other aquatic mammals, they rely on their dense fur for thermoregulation (Van Niekerk et al. 1998; Okes et al. 2016).
African Clawless Otter occur in a wide variety of aquatic habitats, from freshwater lakes to even episodic rivers in arid areas, as long as sufficient food is available (Nel & Somers 2007). The species also utilises the marine littoral zone (Nel & Somers 2007). They are not dependent on the broadly described vegetation type necessarily (Skinner & Chimimba 2005; Nel & Somers 2007), as long as there is sufficient refuge cover (Verwoerd 1987; Perrin & Carugati 2000b; Somers & Nel 2004b; Nel & Somers 2007), and foraging habitat, that confers food availability (Verwoerd 1987; Somers & Nel 2004b; Nel & Somers 2007).
Fresh water availability is an obvious habitat requirement for this semi-aquatic species (Skinner & Chimimba 2005 Nel & Somers 2007), which is especially important for marine environments, where fresh water access is a necessity (Verwoerd 1987; Van Niekerk 1998). However, the species is not dissuaded by turbid waters, as they forage using feel and forage in the shallows near riverbank edges (Nel & Somers 2007).
Rocky substrates to rivers are important to African Clawless Otter, which is probably related to high food biomass (Nel & Somers 2007). Similarly, dense reed beds are a preferred habitat to African Clawless Otter (Nel & Somers 2007; Ponsonby & Schwaibold 2019), and are probably related to high food biomass (Nel & Somers 2007). This conforms to the resource dispersion hypothesis, and total home-range length was correlated with reed bed (high food density patch) proximity (Somers & Nel 2004b).
African Clawless Otters prefer dense riverine vegetation habitats covered with dense vegetation, (Verwoerd 1987; Perrin & Carugati 2000b). The species avoids areas of short grass (Perrin & Carugati 2000b), and select tall grass alongside rivers (Perrin & Carugati 2000b; Ponsonby & Schwaibold 2019). Locally, however they may select open grassed areas for their latrine sites (Nicolaides et al. 2024). Holts of the species were located adjacent to river banks, oxbow lakes or dam shores (Perrin & Carugati 2000b), and preferred habitats are wetlands (Perrin & Carugati 2000b; Okes & O’Riain 2017; Webster et al. 2021) and estuaries (Okes & O’Riain 2017).
African Clawless Otters used habitats to avoid human disturbance at fine scales in the Western Cape (Verwoerd 1987; Okes & O’Riain 2017), and in Gauteng (Ponsonby & Schwaibold 2019). In the urbanised Cape Peninsula, the species presence was negatively influenced by proximity to canalised sections of rivers, and areas that were heavily degraded by human activity (Okes & O’Riain 2017). Up country, in Gauteng, the species utilised urban areas differently to peri-urban areas, which was suggestive of a response to human activity (Ponsonby & Schwaibold 2019). In the urban areas, they avoided human settlements, and selected areas with cover such as reeds, tall grass and trees (Ponsonby & Schwaibold 2019). Specifically, two grass species (Aristida junciformis subsp. junciformis and Hyparrhenia hirta), were associated with otters. So too, were two tree species (Rhus lancea and Acacia dealbata) which were more common at sites of otter presence, while fewer signs of otter were present near Celtis africana, where the latter tree is bare underneath affording no cover (Ponsonby 2018). Concealment from humans appeared to be paramount (Ponsonby & Schwaibold 2019). In the peri-urban areas of Gauteng, several similar variables were associated with positive signs for otters, namely reed beds, tall grass and any tree amount of tree cover, while human settlements were less important there. Furthermore, they selected mild to medium slopes of the riverbanks, and where rivers were wide (30–40 m), and the water was deep (1–2 m) (Ponsonby & Schwaibold 2019).
In the Telperion Nature Reserve, on the Highveld on the periphery of Gauteng, African Clawless Otter were detected in both moist grassland near wetlands and denser riparian habitats (Webster et al. 2021). In the Soutpansberg, African Clawless Otter use was higher in areas with pools, rocky riverbanks and areas with a stream width of at least 2 m, to over 5 m in width (Haring et al. 2023). In coastal Kwazulu-Natal, African Clawless Otter latrine sites were always near water and at the ecotone of different vegetation types, while at the finest scale these latrine sites were in relatively flat, open clearings (Nicolaides et al. 2024, also see Perrin & Carugati 2000b).
Elsewhere, they are found in diverse habitats, from impoundments, estuaries, and mangroves to arid conditions of the upper Doring River in the Western Cape a (Nel & Somers 2007; Somers & Nel 2013); they are also found in many seasonal or episodic rivers in the Karoo, such as the Sak, Vis, Riet, Seekoei and Gamka Rivers, provided suitable-sized pools persist (Nel & Somers 2007; Somers & Nel 2013; Prinsloo 2014). They have even been recorded up to 2,900 m asl in the Lesotho Highlands (Lynch 1994; Avenant & du Plessis 2012). African Clawless Otters have been found in towns and cities (Ponsonby & Schwailbold 2019), and can occupy rivers with high pollution and eutrophication levels (Somers & Nel 2013).
They are predominantly crepuscular, meaning they are mostly active at dawn and dusk (Somers & Nel 2004a). The African Clawless Otter is mostly nocturnal (Verwoerd 1987; Nicolaides 2023), while some populations are active in the mornings after dawn (Webster et al. 2021). Human disturbance however, may prompt them to be more nocturnal (Verwoerd 1987; Nicolaides 2023), so as to avoid dogs, people and disturbance (Okes et al. 2016).
The African Clawless Otter exhibits a wide variety of prey items in its diet (for example, Rowe-Rowe 1977; Somers & Purves 1996; Jordaan et al. 2015), this species shows functional responses to temporal or geographical patterns, and can switch between prey items (for example, between fish, crabs, frogs, insects or combinations thereof), thus facilitating a wide distribution (Nel & Somers 2007).
Crabs are by far the most important prey species to African Clawless otter in most freshwater (Rowe-Rowe 1977; Ligthart et al. 1994; Somers & Purves 1996; Perrin & Carugati 2000a; Somers & Nel 2003; Parker et al. 2005; Ponsonby 2018) and some marine environments (Somers 2000b; Jordaan et al. 2015). Fish are often the second most important prey item to these otters, and in freshwater systems, are regionally important prey to African Clawless Otter but seem to be utilised seasonally, and particularly during winter (Perrin & Carugati 2000a; Somers & Nel 2003), and are less important in some areas (Parker et al. 2005). Exotic trout Salmo spp are preyed upon where they occur (Rowe-Rowe 1977; Perrin & Carugati 2000a), though considering their availability, they are not as important as touted (Jordaan et al. 2019). Other exotic fish species such as the Large Mouth Bass Micropterus salmoides may be more important to their diet (Watson & Lang 2003; Parker et al. 2015). Interestingly, in Groonvlei estuary, Western Cape, since crabs are absent, a dietary shift in the species resuls in fish being the primary food source there (Watson & Lang 2003). Other prey, African Clawless Otters feeds upon are reported are frogs (Rowe-Rowe 1977; Perrin & Carugati 2000a; Parker et al. 2005), insects (Rowe-Rowe 1977; Ligthart et al. 1994; Somers & Purves 1996; Wastson & Lang 2003; Ponsonby 2018), birds (Rowe-Rowe 1977; Ponsonby 2018) and small mammals (Ponsonby 2018). Rowe-Rowe (2019) questioned the importance of mammal remains, given the low prevalence between 0-2% of diet studies examined (Rowe-Rowe 2019). It was suggested that the species does not naturally prey on or eat mammals, and that the presence of mammal remains in scats (spraints) can be attributed to incidental ingestion by the otter, or be the scats of a similar species, the water mongoose Atilax paludinosus (Rowe-Rowe 2019). Several differences were observed between the diet of African Clawless Otter in Gauteng compared to other areas. The most striking is the lack of amphibian remains which may be related to the degraded state of the habitats there (Ponsonby 2018).
African Clawless Otters can exhibit substantial behavioural plasticity when it comes to foraging and are opportunistic feeders that are likely able to adapt to potential prey species and their changing abundances (Jordaan et al. 2015). In the marine environment fish are an important food source (Somers 2000a; b; Emmerson & Philip 2004; Jordaan et al. 2015), and especially during winter it seems (Somers 2000b). Other important prey there are lobster (Emmerson & Philip 2004; Jordaan et al. 2015), octopus (Van der Zee 1981; Verwoerd 1987; Somers 2000b) and various molluscs (Emmerson & Philip 2004; Jordaan et al. 2019), and even shark (Jordaan et al. 2019). African Clawless Otters in the marine littoral zone are opportunistic feeders with a good ability to capitalise on available prey (Van der Zee 1981), with seasonal differences exhibited in the diet (Somers & Nel 2003, but see Jordaan et al. 2015). In freshwater ecosystems, when compared to other similar species, the African Clawless otter may be said to have a narrower trophic niche (Perrin & Carugati 2000a), though this is ecologically dependent, as they are documented to also have a wider trophy niche, elsewhere, in particular in disturbed ecosystems (Ponsonby 2018).
African Clawless Otters have been radio-tracked for the purposes of knowing their home-ranges in the Eastern Cape’s Garden Route National Park’s Tsitsikamma section in the Eastern Cape (Arden-Clarke 1986), as well as rivers in the Western Cape (Somers & Nel 2004a). A radio-tracked adult male on the Tsitsikamma coastline had a minimum linear home range size of 19.5 km of coast with a centrally located 12 km core area (Arden-Clarke 1986).In the same site, the minimum home range size of an adult female was 14.3 km of coast, with a 7.5 km core area (Arden-Clarke 1986). The known home ranges of four adult males overlapped completely. Further west, radio-collared African Clawless Otters’ total range length varied from 4.9 to 54.1 km and core length ranged from 0.2 to 9.8 km (Somers & Nel 2004). Total area of water used by African Clawless Otters varied between 4.9 and ~1062 ha, and core areas ranged from 1.1 to ~140 ha (Somers & Nel 2004). The pattern of home-range use by a female otter was suggestive of territoriality (Somers & Nel 2004a). Male otters had overlapping home ranges, both with other males (Arden-Clarke 1986; Somers & Nel 2004a) and other females (Somers & Nel 2004a). Males were observed foraging together, indicating a clan-type of social organisation (Arden-Clarke 1986).
Ecosystem and cultural services:
Globally, other otter species are considered to be indicators of water quality—for example, in both North American rivers (North American River Otter Lontra canadensis) (Mayack 2012) and coastlines (Sea Otter Enhydra lutris) (Jessup et al. 2004)—or sentinels of environmental health; for example, in Europe (European Otter Lutra lutra) (Chadwick 2007; Lemarchand et al. 2011). Mayack (2012) suggested that the otter’s wide distribution, opportunistic and predatory nature, plus their flexibility in habitat and diet, allow them to serve as a useful upper level consumer component in ecosystem monitoring. Similarly, the African Clawless Otter may be a useful flagship species to highlight the importance of river health, and potentially provide information on the contaminant levels in African urban rivers as European Otters do in Europe. However, it is not yet known whether African Clawless Otters have an important role in the freshwater ecosystems.
Indicators of healthy ecosystems: Trace residues of Anticoagulant Rodenticides have been discovered in African Clawless Otters from the Cape Peninsula (Serieys et al. 2019). Exposure in this species would be via secondary routes of exposure via a vector, and it is suspected that otters may be secondarily exposed via invertebrate prey, or alternatively, directly exposed via contaminated water (Serieys et al. 2019), though given the small amounts, it is uncertain how important the species can be in this.
Nutrient cycling: Otters in African savanna systems, specifically the Bankenveld of the South African Highveld-Bushveld edge, have been hypothesised, through their spraints at latrines, to contribute to processes of nutrient cycling via deposition of aquatically-derived nitrogen in soils. However, the uptake thereof is not realised in the vegetation. It is suggested that aquatically-derived nutrients made available in soils are not being utilised by the surrounding vegetation (i.e leaching) and are of no apparent benefit to plant growth or ecosystem productivity. The role that semi-aquatic predators, such as otters play in facilitating nutrient transfer of limited nutrients in the Bankenveld was not substantial, and thus it is suggested that nutrients are acquired in this terrestrial system through alternative pathways. This pattern of limited nutrient transfer may be specific to the Bankenveld, or sandy savanna systems studied, and the role of aquatic predators in transferring nutrients in other systems (e.g. loamy or clay) (Conradie et al. 2019), or across South Africa may be more substantial elsewhere.
Biological control: Given that the African Clawless Otter can prey upon some fish species such as trout (Rowe-Rowe 1977; Perrin & Carugati 2000a), large mouthed bass (Watson & Lang 2003; Parker et al. 2015) and Oreochromis mossambicus (Watson & Lang 2003), they may be a sort of biological control agent for these species. This is particularly important in the Bloukrans River where the predation on bass has buffered predation any predation upon the threatened Sandelia bainsii (Parker et al. 2015).
Palaeoecology studies: Long-term, site-specific stability in African Clawless Otter diet suggests that monitoring the diet of otters could provide some useful information on the status of shoreline faunal communities (Jordaan et al. 2015).
Tourism: Otters do have the potential to increase ecotourism in the area, and also to contribute financially to the poverty stricken local Dwesa community of the Eastern Cape (Dumalisile et al. 2005), and they may even be able to serve as flagship or umbrella species.The prestigious Otter Trail on the Tsitsikamma coastline is named after this species.
A recent children’s booked entitled ‘Wild adventures with Lazzy the otter’ written by Danelle Murray may have some importance with youth interests in the species.
In the Lesotho Highlands, inhabitants report that African Clawless Otters are commonly used for traditional medicine, clothes, hats, and as food (Avenant 2004).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.7. Forest -> Forest – Subtropical/Tropical Mangrove Vegetation Above High Tide Level | – | Suitable | – |
| 1.8. Forest -> Forest – Subtropical/Tropical Swamp | – | Suitable | – |
| 4.6. Grassland -> Grassland – Subtropical/Tropical Seasonally Wet/Flooded | – | Marginal | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Suitable | – |
| 5.2. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent/Irregular Rivers/Streams/Creeks | – | Suitable | – |
| 5.3. Wetlands (inland) -> Wetlands (inland) – Shrub Dominated Wetlands | – | Marginal | – |
| 5.4. Wetlands (inland) -> Wetlands (inland) – Bogs, Marshes, Swamps, Fens, Peatlands | – | Marginal | – |
| 5.5. Wetlands (inland) -> Wetlands (inland) – Permanent Freshwater Lakes (over 8ha) | – | Suitable | – |
| 5.6. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent Freshwater Lakes (over 8ha) | – | Marginal | – |
| 5.7. Wetlands (inland) -> Wetlands (inland) – Permanent Freshwater Marshes/Pools (under 8ha) | – | Suitable | – |
| 5.8. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent Freshwater Marshes/Pools (under 8ha) | – | Marginal | – |
| 5.13. Wetlands (inland) -> Wetlands (inland) – Permanent Inland Deltas | – | Suitable | – |
| 5.14. Wetlands (inland) -> Wetlands (inland) – Permanent Saline, Brackish or Alkaline Lakes | – | Marginal | – |
| 5.15. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent Saline, Brackish or Alkaline Lakes and Flats | – | Marginal | – |
| 5.16. Wetlands (inland) -> Wetlands (inland) – Permanent Saline, Brackish or Alkaline Marshes/Pools | – | Marginal | – |
| 5.17. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent Saline, Brackish or Alkaline Marshes/Pools | – | Marginal | – |
| 9.10. Marine Neritic -> Marine Neritic – Estuaries | – | Suitable | – |
| 12.5. Marine Intertidal -> Marine Intertidal – Salt Marshes (Emergent Grasses) | – | Marginal | – |
| 13.4. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Coastal Brackish/Saline Lagoons/Marine Lakes | – | Marginal | – |
| 13.5. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Coastal Freshwater Lakes | – | Suitable | – |
| 15.1. Artificial/Aquatic & Marine -> Artificial/Aquatic – Water Storage Areas (over 8ha) | – | Marginal | – |
| 15.2. Artificial/Aquatic & Marine -> Artificial/Aquatic – Ponds (below 8ha) | – | Suitable | – |
| 15.3. Artificial/Aquatic & Marine -> Artificial/Aquatic – Aquaculture Ponds | – | Suitable | – |
| 15.9. Artificial/Aquatic & Marine -> Artificial/Aquatic – Canals and Drainage Channels, Ditches | – | Marginal | – |
Life History
Generation Length: 4.4 years (Pacifici et al. 2013)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial, Freshwater (=Inland waters), Marine
.
General Use and Trade Information
Individuals used to be taken from the wild for use in zoos and aquariums (Okes et al. 2016), and it is no longer a threat, as it is either not happening or very rarely so.
Trade in body parts for commodity:The skins of the African Clawless Otter have been traded in South Africa (Nieman 2018), Lesotho (Avenant 2004) and countries to the north of us (Ray et al. 2005). The skins are decorative and can be sold for about R700 (Nieman 2018), and otter fur is very soft and it can possibly make a comfortable kaross of sorts.
Trade for medicinal use and beliefs: Body parts of the African Clawless Otter do have a value in the medicinal uses trade and are sold in KwaZulu-Natal (Cunningham & Zondi 1991), Eastern Cape (Simelane and Kerley 1998) Gauteng (Whiting, Williams and Hibbits 2016) and the Western Cape provinces (Nieman 2018).
The species appeared in 15% of herbalist shops in the Eastern Cape (Simelane and Kerley 1998).
In some areas of South Africa, there is an overall clientele consisting of 60 – 80% of South African citizens (Nieman 2018). In the Western Cape, there has been an increase in medicinal use trade (Nieman 2018). In traditional healer communities in the Western Cape, body parts, mostly skins of the African Clawless Otter appeared in 41.2% of communities Nieman 2018).
The skins of this otter are believed to protect against evil spirits and can be sold for between R400–R2000 (Nieman 2018). The viscera can be used for epilepsy treatment, and can be sold for R400–R3000 (Nieman 2018). The skull is believed to be used to predict the future, spiritual enlightenment and to even cause misfortune in someone, and can be sold for R 1600 (Nieman 2018). Even otter scats have value, and are believed to increase crop yield and can sell for R 2000 (Nieman 2018), and it may be that the latter by-product may be the sustainable of the many.
In the Lesotho Highlands, inhabitants report that otters are commonly used for traditional medicine, clothes, hats, and as food (Avenant 2004). In Eswatini, otters are used for loin skins, and by traditional healers (Okes et al. 2016).
Trophy hunting: The Professional Hunters register was examined for 2016 until 2023, and there were no records of off-take, though there were a few in 2015. The species is thus not in demand for hunting.
The species is listed on Appendix II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) due to their similarity to sister species Aonyx congicus, the Congo Clawless Otter, which is hunted for fur and bushmeat in Cameroon and Congo, but more threatened (Ray et al. 2005).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Skins are used by traditional healers, commonly used for traditional medicine, clothes, hats and bushmeat. | Yes | Individuals are taken from the wild for use in zoos and aquariums. However, this is not suspected to have a negative effect on the population. |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
| 3. Medicine – human & veterinary | true | – | – | – |
| 10. Wearing apparel, accessories | – | – | – | – |
| 13. Pets/display animals, horticulture | – | true | true | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: Commercial use is not suspected to have a negative effect on the population.
Threats
Habitat
In some parts of their range, their habitat has been either drastically changed or lost, following bush clearing, deforestation, overgrazing, siltation, expansion of human settlements, draining of wetlands, water extraction or denudation of riparian vegetation (Rowe-Rowe 1995; Nel et al. 2007; CSIR 2010).
In KwaZulu-Natal, the burning of vegetation on riverbanks was detrimental to African Clawless Otter as it removed cover and negatively affected the availability of potential holt sites of the species (Perrin & Carugati 2000b). In KwaZulu-Natal, Rowe-Rowe (1992a) identified the primary threat as the increasing human population, leading to adverse alteration of freshwater habitats and riparian vegetation. Negative effects usually arise from increased agricultural activity, such as overgrazing with increased soil run-off, and commercial afforestation (for example, Jewitt et al. 2015).
Since the species is dependent on sources of fresh water, reduced stream flow resulting from increased human activities upstream can negatively affect continued African Clawless Otter occurrence along the coastline (Van Niekerk et al. 1998).
Occupancy modelling in the Cape Peninsula across a transformed landscape suggests that otters can tolerate certain levels of human impact and can inhabit areas in close proximity to roads (although road collisions are a threat in these areas) and in areas of high human population density (Okes & O’Riain 2017).
Analysis of habitat use within rivers, however, suggests a higher probability that otters prefer non-canalised sections of river that are not heavily polluted (Okes & O’Riain 2017). More data regarding the level of degradation of riparian areas the otters can successfully tolerate is needed. Research in South America has shown that the Southern River Otter (Lontra provocax) is able to tolerate moderate levels of change to riverbank vegetation so long as there is very little human activity along the floodplain (Medina-Vogel et al. 2003).
Other predicted threats in the Highlands are overgrazing of riverbanks, and the burning of riverbanks and wetland areas (for grazing) (see Perrin & Carugati 2000b). Occasionally, they are accidentally caught and drowned in gill nets and fish traps (Rowe-Rowe 1990).
The faecal GlucoCorticoid Metabilite (fGCM) concentrations of Africa Clawless Otters from transformed areas were significantly higher than those from otters in natural areas (Majelantle et al. 2020). These preliminary results suggest that African clawless otters may have increased adrenocortical activity that could be due to conditions linked to living in a transformed environment. This indicated that factors related to human dominated landscapes could inflict stress on African clawless otters, reflected by an increase in their adrenocortical activity (Majelantle et al. 2020).
Pollution
In earlier times, in KwaZulu-Natal, scats of African Clawless Otter contained high concentrations of Organochlorine and PolyChlorinated Biphenyls (PCB) contaminants. However, overall levels of contamination were said to be low and unlikely to pose a threat to otter populations at the localities sampled (Mason & Rowe-Rowe 1992).
Acid pollution from coal mining, pesticides from farmlands and waste from urban landfill sites are also observed as a threat (Mason & Rowe-Rowe 1992; Okes et al. 2016). Later on, in Gauteng, heavy metal analysis showed that there may be potential for arsenic and mercury to bioaccumulate through the species’ food chain as these heavy metals were detected at lower concentrations in the otter’s scat than in its food source, suggesting a high risk for the otter as these contaminants are not being eliminated (Clark 2018).
Urban expansion and industrialisation are responsible for the pollution of streams and rivers and for the loss of habitat. However, the impacts of habitat loss, increased riparian activity and disturbance on otter populations are largely unknown from many areas (Okes et al. 2016). However, a study done in 2010 changed this (Kubheka et al. 2013). Kubheka et al (2013) reported on the decline in otter relative abundance in a previously pristine habitat and speculate on its causes. They surveyed sign of African Clawless Otter along a river in the Maloti-Drakensberg Park, KwaZulu-Natal. The area, Stillerust, lies downstream of a commercial dairy farm and a rural village–subsistence farming area. The data collected were compared with two previous surveys, carried out in 1972–1974 and 1993–1994 (see Rowe-Rowe 1992b; Perrin & Carugati 2006), and were also compared with a pristine section of river in the nature reserve, upstream of the farming areas. At Stillerust, the number of spraint (scat) sites found, for the otter species, approximated only 25% of those recorded in the earlier studies. In the pristine section of the river, the amount of sign and abundance of otters were similar to those recorded in 1993 (i.e Perrin & Carugati 2006). Assessments of stream biota and water quality analyses revealed negative changes in the river below the pristine area, down to, and including, Stillerust. Visible signs of pollution were evident. Since the 1970s, the rural village population has increased eightfold, and the commercial farm has changed from an extensive livestock enterprise to an intensive dairy farm (Kubheka et al. 2013).
Internationally, increases in pollution have led to a dramatic decrease in local otter populations (Kruuk 2006). Current research on otters in the Cape Peninsula has found that certain persistent organic pollutants are present in the tissue of otters in urban areas (Serieys et al. 2019). More research however is required on the effects of pollution on otters and their associated sources of food in South Africa as there is a gap in knowledge relating to the threat faced by otters from degradation of riparian areas.
Another problem arising from human activity along rivers is pollutants entering rivers causing some species in the rivers to die-off, leading to a decrease in the biotic diversity of the rivers (Grimm et al. 2008), ultimately resulting in a shift in predator-prey dynamics. More research is required on the potential impacts pollution and climate change may have on the otter’s prey base. No research has yet been conducted on the threshold for African Clawless Otter survival along disturbed rivers, though there have been some abundance estimates undertaken and it looks like they are fairly tolerant (Majelantle et al. 2021).
Pesticides
Trace residues of Anticoagulant Rodenticides have been discovered in African Clawless Otters from the Cape Peninsula (Serieys et al. 2019). Small residues concentrations of brodifacoum, bromadiolone, difenacoum were found in the specimens examined (Serieys et al. 2019). Of the samples examined 44% had exposure to these anticoagulant rodenticides (Serieys et al. 2019). Exposure in this species would be via secondary routes of exposure via a vector, and it is suspected that otters may be secondarily exposed via invertebrate prey, or alternatively, directly exposed via contaminated water (Serieys et al. 2019), though they can also consume small mammals in urban environments (Ponsonby 2018), and directly ingest this.
Persecution – direct killing
African Clawless Otter are persecuted for alleged poultry killing, such as ducks (Stuart 1981).
In South Africa, they are sometimes blamed for killing sheep or goats, and the fact that the species does not kill mammals should be used to prevent unnecessary persecution (Rowe-Rowe 2019). Since this species is known to prey upon trout where they occur (Rowe-Rowe 1977), they do come into conflict with recreational fishing destination that stock the species (De Vos & McIntyre 2024), or those that simply breed them (Nieman 2018; De Vos & McIntyre 2024). These conflict areas are mostly in Mpumalanga (De Vos & McIntyre 2024), and increasingly also in the Western Cape (Nieman 2018). Of landowners in the Boland area of the Western Cape, 5% report damage from this otter species, and of these, they have low tolerance toward the species, and 60 % would resort to lethal control (Nieman 2018).
De Vos and McIntyre (2024) investigated this human-otter conflict through a questionnaire aimed at owners and managers of fly-fishing properties in South Africa. While perceptions of losses varied, all respondents reported losses to predators. Mitigation measures, including lethal control, were employed mostly against otters. Of respondents, a third of them reportedly used lethal control. For preventative mitigation in recreational fishing, 36% used non-lethal mitigation such as exclusion for instance (De Vos & McIntyre 2024).
Given that fly-fishing properties potentially provide valuable habitat for protected species such as otters, the results there is a need for improved mitigation measures and exploring alternative options of potentially generating income from tourism associated with the presence of charismatic predators (De Vos & McIntyre 2024).
In the North West, this otter species has been found to be a problem with Anglersat Bloemhof dam, where they raid keep nets, and these problems seem to be exacerbated on busier periods when more people are present (ie. Fishing competitions) (Power 2014). However, in the latter case, tolerance has to occur as it is a protected area.
Domestic dogs
Feral dog hunting packs have been implicated in African Clawless Otter mortality in parts of the Western Cape (Nieman et al. 2020). In the Lesotho Highlands, the impacts of dogs and cattle herders are expected to be less on otters than on most other mammals along rivers, due to the otter’s ability to escape in and through the water, and their nocturnal activity patterns (Avenant 2004; Avenant & du Plessis 2012). Higher up in the mountains, where streams are smaller/easier to cross, dogs may be a threat (Okes et al. 2016).
Roads
The threat of roadkill mortalities on the African Clawless Otter is largely unknown (Ray et al. 2005). Road kills of African Clawless Otter have been reported from the Western Cape from the 1990s (Verwoerd 1987) to the present (Serieys et al. 2019). In the Cape Peninsula, 6 of 9 African Clawless Otter opportunistically obtained for sampling were obtained from roadkill (Serieys et al. 2019).
In the North West, since 2019, two road kills of African Clawless Otter have been recorded, and they were both on the edges of protected areas, specifically Barberspan and Molemane Reserves (North West Directorate of Biodiversity Management, unpublished records, 2019-2023). There thus may be local importance in this, especially if source populations are being impacted.
Body parts uses trade
Otter body parts for commodities, such as the skin, and body parts have been in demand in mostly countries north of South Africa (Ray et al. 2005), and increasingly more so in South Africa (Nieman 2018). The African Clawless Other has Cultural Significance Index (CSI) of 4, which is moderate amongst several others that are a lot more important such as the Leopard (Panthera pardus) for instance (Nieman 2018).
Problem animal control off takes
The species is controlled to protect use of other resources. Some farmers persecute this species as a perceived competitor for fish stocks, especially subsistence rural dwellers (De Vos & McIntyre 2024). Additionally, some farmers that (for example) farm with geese (for down) regard them as a threat to poultry (Stuart 1981), thus consider them pests. For example, fisheries managers blame trout declines on otter predation and competition with trout for food, even though scat analysis revealed that only 1% of otter faeces contained the remains of trout and their diets overlapped by only 17% (Butler 1994; Butler & Marshall 1996).
Summary of threats
Threat categories have been assessed for various carnivore species in Africa, and each species is differentially impacted by a set of ten external threats commonly encountered by African carnivores (Ray et al. 2005). Each species was subjected to a scoring process for each threat that assessed the relative severity (its effect on the species), urgency (timescale over which it is most likely to occur), probability (likelihood of occurrence), and geographical extent of the threat through the species’ range in Africa. The threat score for the African Clawless Otter was 68.5, and of 20 carnivores in Africa it ranked 6th (Ray et al. 2005). This pan-African assessment suggests the species to be relatively at risk.
More recently, a Rapid Vulnerability Assessment (RVA) found that the species may face potential regional endangerment in the Boland area of the Western Cape (Nieman 2018). This was a synergistic assessment of species’ vulnerability to the combined impacts of the various hunting practices, and subsequently found that the African Clawless Otters inter alia experience a high potential endangerment (Nieman 2018).
Habitat loss and degradation are the main threat to the African Clawless Otter (Okes et al. 2016), and the use for medicinal and belief purposes may be an emerging threat.
Conservation
For otter species distributed more widely, conservation challenges become more complex. Additional factors, such as a species’ population status, extent and enforcement of legal protections, and local environmental threats also influence how conservation strategies should be and can be implemented effectively (Brooks & Serfass 2019). African Clawless Otters are present in a number of protected areas across their range, both in the interior (Rowe-Rowe 1992a; Avenant 1997; Avenant & Watson 2002; Watson 2006; Perrin & Carugati 2006; Rowe-Rowe 2016; Majalantle et al. 2021) and along the coast (Arden-Clarke 1986; Nicolaides 2023). The main interventions revolve around riparian and coastline protection and enforcement of existing legislation (Okes et al. 2016). Important protected areas for the species are the following: Garden Route National Park, Table Mountain National Park, Maloti-Drakensberg Park, isiMangaliso Wetland Park, Golden Gate Highlands National Park, Mokala National Park, Augrabies Falls National Park, Wolwespruit Nature Reserve, Bloemhof dam Nature Reserve and the Kruger National Park.
African Clawless Otters occur both inside and outside protected areas (Perrin & Carugati 2000b), and densities outside protected areas may even be higher regionally (Majelantle et al. 2021), and locally so (Nicolaides 2023).
They are able to utilise resources available in novel, human-impacted environment, which does not appear to be affecting their movement (Ponsonby et al. 2019), habitat utilisation, diet (Ponsonby 2018), and neither their occurrence or abundance (Ponsonby & Schwaibold 2019; Majelantle et al. 2021; Nicolaides 2023).
It was suggested in the past that urban development in the Bettys’ Bay area of the Western Cape be more restricted to designated areas along the coast as part of a management plan for the area (Verwoerd 1987), and it would appear that given the governance of the province, this would be the case. Work on the effects of vegetation removal along rivers in South America have shown that the local species, Lontra provocax, will still frequent areas with moderate vegetation removal but only if human activity is not present as there is not enough cover to remain concealed from human detection (Medina-Vogel et al. 2003). The development of game reserves/national parks along rivers inhabited by African Clawless Otters would be beneficial as these areas would protect habitat along rivers and prevent the removal of necessary vegetation types. However, as is seen in the work by Kubheka et al. (2013), pollution entering rivers upstream from reserves still makes its way into the reserves, which will impact the otters.
Biodiversity stewardship opportunities of key sites should be sought. For example, stricter regulations and enforcement on development along riverbanks and coastlines needs to be implemented. Monitoring and mitigation of pollution (chemical and physical) of rivers needs to be implemented or increased. Education and awareness campaigns that position this species as an indicator of river health, and thus prestige for the landowner, should be promoted and tested as a conservation intervention. In the Lesotho Highlands, a limit to the number of dogs and stricter enforcement of no grazing in conservation areas may help. The use of extension services to restore riparian habitats, such as the Working for Water Programme, should continue to be used in key watersheds within the species’ range.
Structures like the Ramsar wetland convention should be seen as ways to communicate conservation concerns for otter species as well as other water-based protections that carry greater legal standing (Brooks & Serfass 2019). A large Global Environment Facility (GEF) 8 funded project will commence soon run by Agricultural Research Council (ARC) soon, will cover three major peatland ecosystems where the work will focus on restoration, monitoring and research on the following systems: Molemane/Molopo (North West), Gouritz (Western Cape), and Maputaland (KwaZulu-Natal), and monitoring on this species may be considered as part of this work.
Control of exotic fish
Given the dominance of alien fish in otter diet in some cases (Watson & Lang 2003; Parker et al. 2005), it would be interesting and important to note the influence of applications of this policy on removals on these species on the diet and abundance of clawless otters in Groenvlei for instance, where they are dependent on these alien species (Watson & Lang 2003).
Translocations and rehabilitation
During 2018, a rehabilitated African Clawless Otter was translocated by the Wild4Life rehabilitation centre to Millvale Golf Estate, in Rustenburg, South Africa (61 km apart), and it had registered elevated levels of fGCM (Majelantle et al. 2020), suggesting it was a stressful process.
In the North West, between 2013 and 2023, there have been three reports where Problem Animal Control officers had to deal with the species. These were all prior to 2016, and involved one relocation, one euthanasia of an injured one, and the third case was one where it was a case of misidentification of a leopard that was confirmed to be this otter species. Of interest, in the first three months of 2024, two different incidences of otters raiding khoi ponds in residential homes has been reported, and the culprits have been captured and relocated (North West Directorate of Biodiversity Management, unpublished records, 2013-2024).
Monitoring
Monitoring freshwater discharge points along coastlines would indicate whether otters were present in a general area or not, which may be a time-saving method of determining otter distribution on a large scale. Monitoring only mouths of rivers is possible, as most otter spraints and couches were located very close to fresh water there (Van Niekerk et al. 1998).
Given similarity in the scats of African Clawless Otter and Spotted-Necked Otter, genetic methods are availed to discriminate the species apart (Madisha et al. 2015). One can positively differentiate between the two otter species and the ability to accurately identify species using partial DNA will be beneficial in understanding numerous aspects of the behaviour and ecological importance of this species in its environment (Madisha et al. 2015).
In order for African Clawless Otters to persist on the peninsula, conservation authorities must maintain wetland, estuarine and river habitat in close proximity to MPAs (Okes & O’Riain 2017).
Recommendations for land managers and practitioners:
- Nature conservation officers should cooperate with agricultural extension officers to give coordinated advice in veld management, agricultural practices and river care.
- Landowners should be encouraged to reduce stocking rates and take down fences, as, due to the large home range of the species (Arden-Clarke 1986; Somers & Nel 2004a, it can cover large distances when searching for fresh water and fences will only fragment their habitat.
- Adopt non-lethal mitigation (i.e exclusion) to protect trout stocks in dams, or domestically kept high-value fish such as khoi.
- Ensure adequate protection of poultry, especially ducks, and enclose them during the crepuscular and nocturnal hours.
Research priorities:
- Determine what limits otter populations.
- Determining the role of African Clawless Otters on river ecosystem functioning.
- Develop effective long-term monitoring programmes for this species in different habitat types.
- Determine, using genetics, accurate long-term population density and structure estimates across various habitats.
- Members of the IUCN/SSC Otter specialist group must be encouraged to conduct research and publish their findings, in particular in their bulletin.
- Future studies using radio telemetry to accurately assess otter movement through the urban environment will greatly improve upon predictions made on landscape use (Ponsonby 2018).
- Human-otter conflict should be given attention, particularly in rural areas where fishing is a primary source of income and otters are regarded as competitors and pests (Nicolaides 2023; De Vos & McIntyre 2024).
- Given the ever-increasing anthropogenic development along coastlines, it is imperative that research studies assess and monitor how African Clawless Otters adjust their population densities and activity time to exploit or avoid anthropogenically augmented landscapes (Okes et al. 2016; Majelantle et al. 2021; Nicolaides 2023).
- The adaptability and plasticity of African Clawless Otters should be investigated given the rapid and continued human encroachment and development along coastlines and riverbanks (Jordaan et al. 2019; Nicolaides 2023).
- Further research is needed to determine the long-term impacts on otters persisting in degraded ecosystems (Okes & O’Riain 2017).
- Aside from studying new study sites, continued population monitoring should take place at the Kamberg-Loteni area of the Maloti-Drakensberg Park, aiming at being as comparable as possible with the methodologies of those studies (see Rowe-Rowe 1992; Perrin & Carugati 2006; Kubheka et al. 2013; Rowe-Rowe 2016).
Encouraged citizen actions:
- Avoid building properties on riverbanks and ensure Environmental Authorisations are done for any development near rivers
- Visit and pay to stay at ecotourism destinations that offer trips to search and see otters.
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
- In some urban areas near rivers and wetlands, citizens can become involved in the local WESSA affiliated Friends of the River groups which help to ensure the conservation of these systems.
- Citizens can partake in beach and coastal clean ups.
- Supervise hunting dogs in farmlands to prevent unnecessary otter deaths. Similarly, dog walkers should be mindful of their dogs when walking on beaches in the early morning and late evening, when this species is often active.
- Involving citizens in the monitoring of the urban African Clawless Otter population has complemented standardized occupancy surveys and provided additional benefits. In addition to alleviating the pressure on local authorities to allocate resources to routine monitoring, citizen involvement provides an opportunity to gather supplementary data on behaviour as well as threats to the species; and can shed light on the potential dispersal routes, and promote awareness and encourage coexistence with urban adapted wildlife (Okes & O’Riain 2019).
Bibliography
Arden-Clarke CHG. 1986. Population density, home range size and spatial organization of the Cape Clawless Otter, Aonyx capensis, in a marine habitat. Journal of Zoology 209: 201-211.
Avenant MF. 2010. Challenges in using fish communities for assessing the ecological integrity of non-perennial rivers. Water SA 36.
Avenant NL, Watson JP. 2002. Mammals recorded in the Sandveld Nature Reserve, Free State province, South Africa. Navorsinge van die Nasionale Museum, Bloemfontein 18: 1-12.
Avenant NL, du Plessis JJ. 2012. Letšeng Expansion Project: Project Kholo. Mammal Specialist Report. Environmental Resources Management (Southern Africa) Pty Ltd.
Avenant NL. 1997. Mammals recorded in the QwaQwa National Park (1994-1995). Koedoe 40: 31-40.
Avenant NL. 2004. Conserving Mountain Biodiversity in Southern Lesotho: Mammal report. United Nations Development Plan, Lesotho.
Brooks RP, Serfass TL. 2019. Aquatic Habitat Classifications: Implications for Otter Conservation. Proc.14th Int. Otter Congress, IUCN Otter Spec. Group Bull. 36 (A): 3 – 12.
Butler, J.R.A. 1994. Cape Clawless Otter conservation and a trout river in Zimbabwe: A case study. Oryx 28: 276-282.
Butler JRA, du Toit JT. 1994. Diet and conservation status of Cape Clawless Otters in eastern Zimbabwe. South African Journal of Wildlife Research 24: 41.
Butler, J.R.A. and Marshall, B.E. 1996. Resource use within the crab-eating guild of the upper Kairezi River, Zimbabwe. Journal of Tropical Ecology 12: 475-490.
Carugati C. 1995. Prey and area requirements of otters (Aonyx capensis and Lutra maculicollis) in the Natal Drakensberg. M.Sc. Thesis. University of KwaZulu-Natal, Pietermaritzburg, South Africa.
Chadwick EA. 2007. Post mortem study of otters in England and Wales 1992–2003. Environmental Agency, Rio House, Waterside Drive, Aztec West, Almondsbury, Bristol.
Cianfrani C, Broennimann O, Loy A, Guisan A. 2018. More than range exposure: Global otter vulnerability to climate change. Biological Conservation 221: 103-113.
Clark KC. 2018.Assessing the potential risk of accumulation of selected freshwater contaminants in urban and peri-urban otters. Msc thesis, University of the Witwatersrand, Johannesburg. Conradie SR, Hall G, Somers MJ, McIntyre T. 2019. Limited animal-facilitated nutrient transfer across an aquatic–terrestrial interface in a southern African savanna. African Journal of Wildlife Research 49(1): 52-61.
CSIR. 2010. A CSIR perspective on water in South Africa – 2010. Centre for Scientific and Industrial Research, Pretoria, South Africa.
Cunningham, A.B. and Zondi, A.S. 1991. Use of animal parts for the commercial trade in traditional medicines. Institute of Natural Resources, University of Natal, Pietermaritzburg, South Africa.
De Luca, D.W. and Mpunga, N.E. 2005. Small carnivores of the Udzungwa Mountains: presence, distribution and threats. Small Carnivore Conservation 32: 1-7.
De Vos M, McIntyre T. 2024. Stakeholder perceptions of human–predator conflict in the South African fly‐fishing industry. African Journal of Ecology 62(1), p.e13188.
Dumalisile L, Somers M, Walters M, Nel J. 2005. Tourists’ willingness to pay to view otters along the Wild Coast, South Africa: a potential for increased ecotourism.TD: The Journal for Transdisciplinary Research in Southern Africa 1(1): 97-106.
Du Plessis J, Avenant N, Putsane T. 2014. Mammals of the Katse Dam catchment. Specialist report for Contract 1273: Biological Resources Monitoring within Phase 1 of the LHWP Catchments 2013–14. Anchor Environmental Consultants to the Lesotho Highlands Development Authority.
Emmerson W, Philip S. 2004. Diets of Cape Clawless Otters at two South African coastal localities. African Zoology 39(2): 201-210.
Erasmus, B.F.N., van Jaarsveld, A.S., Chown, S.L., Kshatriya, M. and Wessels, K.J. 2002. Vulnerability of South African animal taxa to climate change. Global Change Biology 8: 679-693.
Foden WB, Young BE. 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM. 2008. Global change and the ecology of cities. Science 319: 756-760.
Haring C, Weier S, Linden B. 2023. Distribution and Habitat Preference of Cape Clawless Otters (Aonyx capensis) and Water Mongooses (Atilax paludinosus) in the Soutpansberg, South Africa. IUCN Otter Spec. Group Bull. 40(1): 26-38.
Hendrix, C.S. and Glaser, S.M. 2007. Trends and triggers: climate, climate change, and civil conflict in Sub-Saharan Africa. Political Geography 26: 695-715.
Jacques H, Reed-Smith J, Somers MJ. 2015. Aonyx capensis. The IUCN Red List of Threatened Species 2015.
Jessup DA, Miller M, Ames J, Harris M, Kreuder C, Conrad PA, Mazet JA. 2004. Southern sea otter as a sentinel of marine ecosystem health. EcoHealth 1: 239–245.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Jordaan RK, McIntyre T, Somers MJ, Bester MN. 2015. An assessment of spatial and temporal variation in the diet of Cape Clawless Otters (Aonyx capensis) in marine environments. African Journal of Wildlife Research 45: 342–353.
Jordaan RK, Somers MJ, Hall G, McIntyre T. 2019. Plasticity and specialisation in the isotopic niche of Cape clawless otters foraging in marine and freshwater habitats. Mammalian biology 98: 61-72.
Kruuk, H. 2006. Otters: ecology, behaviour and conservation. Oxford University Press, Oxford.
Kubheka, S.P., Rowe‐Rowe, D.T., Alletson, J.D. and Perrin, M.R. 2013. Possible influence of increased riparian activity (stream modification and agricultural intensification) on abundance of South African otters. African Journal of Ecology 51: 288-294.
Larivière S. 2001. Aonyx capensis. Mammalian Species 671: 1–6.
Lemarchand C, Rosoux R, Berny P. 2011. Semi aquatic top-predators as sentinels of diversity and dynamics of pesticides in aquatic food webs: the case of Eurasian Otter (Lutra lutra) and Osprey (Pandion haliaetus) in Loire River Catchment, France. In: Stoytcheva M (ed.), Pesticides in the Modern World – Risks and Benefits., pp. 289-310. InTech, Croatia.
Ligthart MF, Nel JAJ, Avenant NL. 1994. Diet of Cape Clawless Otters in part of the Breede River system. South African Journal of Wildlife Research 24:38-39.
Loock D, SwanepoeL L. 2019. Assessing local patterns in carnivore occupancy and richness by camera trapping: a monitoring of diversity at Verloren Vallei Nature Reserve, Dullstroom, Mpumalanga. Wildlife & Resource Association (WRA), Bloemfontein.
Lynch CD. 1983. Mammals of the Orange Free State. Memoirs van die Nasionale Museum, Bloemfontein 18: 1-218.
Lynch CD. 1989. The mammals of the north-eastern Cape Province. Memoirs van die Nasionale Museum, Bloemfontein 25: 1-116.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Madisha MT, Ponsonby D, Schwaibold U, Kotzé A, Jansen R, Brettschneider H, Dalton DL. 2015. Differentiation of two South African otter species (Aonyx capensis and Lutra maculicollis) from spraint based on partial CytB primer sets. Global Ecology and Conservation 4: 8-13.
Madisha MT, Du Plessis M, Kotzé A, Dalton DL. 2019. Complete mitochondrial genomes of the African clawless (Aonyx capensis) and spotted necked (Hydrictis maculicollis) otter: structure, annotation, and interspecies variation. Mitochondrial Dna Part B 4(1): 1556-1557.
Majelantle TL, McIntyre T, Ganswindt A. 2020. Monitoring the effects of land transformation on African clawless otters (Aonyx capensis) using fecal glucocorticoid metabolite concentrations as a measure of stress. Integrative Zoology 15(4): 293-306.
Majelantle TL, Ganswindt A, Jordaan RK, Slip DJ, Harcourt R, McIntyre T. 2021. Increased population density and behavioural flexibility of African clawless otters (Aonyx capensis) in specific anthropogenic environments. Urban Ecosystems 24: 691-699.
Mason, C.F. and Rowe-Rowe, D.T. 1992. Organochlorine pesticide residues and PCBs in otter scats from Natal. South African Journal of Wildlife Research 22: 29-31.
Mayack DT. 2012. Hepatic mercury, cadmium, and lead in mink and otter from New York State: monitoring environmental contamination. Environmental Monitoring and Assessment 184: 2497–2516.
Medina-Vogel, G., Kaufman, V.S., Monsalve, R. and Gomez, V. 2003. The influence of riparian vegetation, woody debris, stream morphology and human activity on the use of rivers by southern river otters in Lontra provocax in Chile. Oryx 37: 422-430.
Meester J., Rautenbach I.L., Dippenaar N. and Baker C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Nel JL et al. 2011. Technical Report for the National Freshwater Ecosystem Priority Areas project. Water Research Commission, Pretoria, South Africa.
Nel JL, Roux DJ, Maree G, Kleynhans CJ, Moolman J, Reyers B, Rouget M, Cowling RM. 2007. Rivers in peril inside and outside protected areas: a systematic approach to conservation assessment of river ecosystems. Diversity and Distributions 13: 341-352.
Nel, J.A.J. and Somers, M.J. 2007. Distribution and habitat choice of Cape Clawless Otters, Aonyx capensis, in South Africa. South African Journal of Wildlife Research 37: 61-70.
Nicolaides SG. 2023. The behavioural ecology of African clawless otters, Aonyx capensis, in KwaZulu-Natal, South Africa. Msc thesis, UNISA, Pretoria.
Nicolaides SG, Mostert TH, McIntyre T. 2024. Latrine site selection by African clawless otters, Aonyx capensis, and their behavior during latrine visitations. Journal of Mammalogy 105(1): 107-121.
Nieman WA. 2018. Culture, Conflict, and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human Wildlife Interface. Msc thesis, University of Stellenbosch, Stellenbosch.
Nieman WA, Wilkinson A, Leslie AJ. 2020. Farmer Attitudes and Regional Risk Modelling of Human–Wildlife Conflict on Farmlands Bordering the Boland Mountain Complex, South Africa. African Journal of Wildlife Research 50: 36. https://doi.org/10.3957/056.050.0036.
Okes NC, O’Riain MJ. 2017. Otter occupancy in the Cape Peninsula: Estimating the probability of river habitat use by Cape Clawless Otters, Aonyx capensis, across a gradient of human influence. Aquatic Conservation: Marine and Freshwater Ecosystems 27(3): 706-716.
Okes NC, O’Riain MJ. 2019. Can opportunistic citizen sightings assist in the monitoring of an elusive, crepuscular mammal in an urban environment?. Urban ecosystems 22: 483-492.
Okes N, Ponsonby DW, Rowe-Rowe D, Avenant NL, Somers MJ. 2016. A conservation assessment of Aonyx capensis. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Parker DM, Burchell RK, Bernard RT. 2005. The diet of Cape Clawless Otters at two sites along the Bloukrans River, Eastern Cape Province, South Africa. African Zoology 40(2): 330-334.
Perrin MR, Carugati C. 2000a. Food habits of coexisting Cape Clawless Otter and spotted-necked otter in the KwaZulu-Natal Drakensberg, South Africa. South African Journal of Wildlife Research 30(2): 85-92.
Perrin MR, Carugati C. 2000b. Habitat use by the Cape Clawless Otter and the spotted-necked otter in the KwaZulu-Natal Drakensberg, South Africa. South African Journal of Wildlife Research 30: 103–113.
Perrin MR, Carugati C. 2006. Abundance estimates of the CapeClawless Otter Aonyx capensis (Schinz 1821) and the spotted necked otter Lutra maculicollis (Lichtenstein 1835) in the KwaZulu-Natal Drakensberg, South Africa. Tropical Zoology 19: 9-19.
Ponsonby DW. 2018. The ecology of otters in an urban environment. Doctoral dissertation, University of the Witwatersrand, Johannesburg.
Ponsonby DW, Schwaibold U. 2019. Country otter, city otter: The distribution patterns of two otter species in an urbanized area of Gauteng, South Africa. African Journal of Ecology 57(1): 148-154.
Ponsonby DW, Thabang Madisha M, Schwaibold U, Dalton DL. 2019. Genetic diversity of African clawless otters (Aonyx capensis) occurring in urbanised areas of Gauteng, South Africa. South African Journal of Science 115(7-8): 1-8.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Power RJ, Van Straaten A, Schaller R, Mooke M, Boshoff T, Nel HP. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Pringle JA. 1977. The distribution of mammals in Natal. Part 2. Carnivora. Annals of the Natal Museum 23(1): 93-115.
Prinsloo HES. 2014. Otters – useful indicators for determining environmental water requirements in non perennial rivers? M.Sc. Thesis. Centre for Environmental Management, University of the Free State.
Rowe-Rowe DT. 1977. Food ecology of otters in Natal, South Africa. Oikos 28: 210–219.
Rowe-Rowe, D.T. 1990. Action plan for African otters. In: P. Foster-Turley, S. MacDonald and C. Mason (eds), Otters: an Action Plan for their Conservation, pp. 41-51. IUCN, Gland, Switzerland.
Rowe-Rowe, D.T. 1992a. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Rowe-Rowe DT. 1992b. Survey of South African otters in a freshwater habitat, using sign. South African Journal of Wildlife 22: 49–55.
Rowe-Rowe, D.T. 1995. Distribution and status of African otters. Habitat 11: 8-10.
Rowe-Rowe DT. 2016. Densities of Otters in the Drakensberg of Kwazulu-Natal, South Africa. IUCN Otter Spec. Group Bull. 33(2): 64-67.
Rowe-Rowe DT. 2019. Does the African (Cape) Clawless Otter eat mammals? IUCN Otter Spec. Group Bull. 36(1): 3-6.
Schurch MP, McManus J, Goets S, Pardo LE, Gaynor D, Samuels I, Cupido C, Couldridge V, Smuts B. 2021. Wildlife-friendly livestock management promotes mammalian biodiversity recovery on a semi-arid Karoo farm in South Africa. Frontiers in Conservation Science 2:6. https://doi.org/10.3389/fcosc.2021.652415
Seaman MT et al. 2010. 2010. Developing a method for determining the environmental water requirements for non-perennial systems. Water Research Commission, Pretoria, South Africa.
Serieys LE, Bishop J, Okes N, Broadfield J, Winterton DJ, Poppenga RH, Viljoen S, Wayne RK, O’Riain MJ. 2019. Widespread anticoagulant poison exposure in predators in a rapidly growing South African city. Science of the Total Environment 666: 581-590.
Simelane TS, Kerley GI. 1998. Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research (4):121-126.
Skead, C.J. 2011. Historical incidence of the larger land mammals in the broader Western and Northern Cape provinces.In: Boshoff A.F., Kerley G.I.H, Lloyd P. (ed.), Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, England.
Somers MJ. 2000a. Foraging behaviour of Cape Clawless Otters (Aonyx capensis) in a marine habitat. Journal of Zoology 252(4): 473-480.
Somers MJ. 2000b. Seasonal variation in the diet of Cape Clawless Otters (Aonyx capensis) in a marine habitat. African Zoology 35(2): 261-268.
Somers, M. J. 2001. Habitat utilization of Cape Clawless Otters Aonyx capensis. Ph.D. Thesis, University of Stellenbosch.
Somers MJ, Nel JAJ. 2004a. Movement patterns and home range of Cape Clawless Otters (Aonyx capensis), affected by high food density patches. Journal of Zoology 262(1): 91-98.
Somers MJ, Nel JA. 2004b. Habitat selection by the Cape Clawless Otter (Aonyx capensis) in rivers in the Western Cape Province, South Africa. African Journal of ecology 42(4): 298-305.
Somers MJ, Purves MG. 1996. Trophic overlap between three syntopic semi-aquatic carnivores: Cape Clawless Otter, spotted-necked otter and water mongoose. African Journal of Ecology 34(158–166.).
Somers, M.J. and Nel, J.A.J. 2013. Aonyx capensis. In: J.S. Kingdon and M. Hoffmann (eds), The Mammals of Africa 5. Carnivores, Pangolins, Rhinos and Equids and Rhinoceros, pp. 104-108. Bloomsbury Press, London, UK.
Van der Zee D. 1981. Prey of the Cape Clawless Otter (Aonyx capensis) in the Tsitsikama Coastal National Park, South Africa. Journal of Zoology 194(4): 467-483.
Van der Zee D. 1982. Density of Cape Clawless Otters Aonyx capensis (Schinz, 1821) in the Tsitsikarna Coastal National Park. South African Journal of Wildlife Research 12: 8–13.
Van Niekerk, C.H., Somers, M.J. and Nel, J.A.J. 1998. Freshwater availability and distribution of Cape Clawless Otter spraints and resting places along the south-west coast of South Africa. South African Journal of Wildlife Research 28: 68-72.
Verwoerd DJ. 1987. Observations on the food and status of the Cape Clawless Otter Aonyx capensis at Betty’s Bay, South Africa. South African Journal of Zoology 22: 33–39.
Watson JP. 2006. Check list of the mammals of Tussen-die-Riviere Provincial Nature Reserve, Free State Province, South Africa. Koedoe 49: 111-117.
Watson LH, Lang AJ. 2003. Diet of Cape Clawless Otters in Groenvlei Lake, South Africa. South African Journal of Wildlife Research 33(2): 135-137.
Webster AB, Pretorius ME, Somers MJ. 2021. The determinants of mesocarnivore activity patterns in highveld grassland and riparian habitats. African Journal of Wildlife Research 51(1): 178-192.
Whiting MJ. Williams VL, Hibbits TJ. 2016. A picture of health? Animal use and the Faraday traditional medicine market, South Africa. Journal of Ethnopharmacology. 179:265-73.