African Civet
Civettictis civetta

2025 Red list status
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – VIVERRIDAE – Civettictis – civetta
Common Names: African Civet, Civet (English), Siwetkat, Afrikaanse Siwet, Afrika-siwet (Afrikaans), Insimba (Ndebele), Tsaparangaka (Sesotho), Lifungwe, Imphicanadloti (Swati), Fungwe (Tsonga), Tsaparangaka, Tshipalere, Tshipalore, Tshipalôre, Tshipa-nôre (Tswana), Dhatshatsha, Dzamatamanga, Dzambarananga, Dzambaranwaha, Dzhatshatsha, Linyanganwaha (Venda), Inyhwagi (Xhosa), iQaqa (Zulu), Afrika Zibetkatze (German), Civette africaine, Civette d’Afrique (French)
Synonyms: No Synonyms
Taxonomic Note:
This species was formerly considered to be congeneric with Asian civets of the genus Viverra. It was first included in Civettictis by Pocock (1915) and retained in that genus by several authors, including Ray (1995, 2013), Kingdon (1997) and Wozencraft (2005), but others, such as Ellerman et al. (1953) and Coetzee (1977), continued to include it in Viverra. Although several subspecies have been recorded, their validity remains questionable (Rosevear 1974; Coetzee 1977; Meester et al. 1986).
Red List status: LC – Least Concern
Assessment Information
Assessors: Venter, J.A.1 & da Silva, J.M.2
Reviewers: Power, R.J.3 & Do Linh San, E.4
Institutions: 1Nelson Mandela University, 2South African National Biodiversity Institute, 3North West Provincial Government, 4Sol Plaatjie University
Previous Assessors and Reviewers: Swanepoel, L.H., Camacho, G., Power, J., Amiard, P. & Do Linh San, E.
Previous Contributors: Gaubert, P., Daniel, W., Ray, J.C., Madikiza, Z.J.K. & Relton, C.
Assessment Rationale
The African Civet (Civettictis civetta) is assessed as Least Concern on account of its broad distribution in the Savanna Biome, ecological flexibility, and occurrence in numerous protected areas (including Kruger National Park). Recent camera-trap work indicates robust subpopulations in Limpopo’s Waterberg, Soutpansberg and Alldays regions, the northern Bushveld of the North West, and the Greater Lydenburg area of Mpumalanga; although extensive surveys suggest possible declines in northern KwaZulu-Natal, an apparent south-western edge expansion in the North West likely balances these losses, yielding an overall stable regional outlook. Localised pressures include roadkill, snaring associated with expanding informal settlements (civets are prone to snares), incidental poisoning during carnivore control, and unsustainable trophy hunting practises; the rapid spread of predator-proof fencing on game farms may further restrict movements and fragment populations. While civets are widely traded as bushmeat elsewhere in Africa, exploitation within the assessment region appears lower but could rise if other sources diminish; traditional-medicine use is poorly quantified. Priority actions are to monitor population trends and legal offtakes, quantify traditional-medicine use, assess fence permeability, and implement targeted snare-removal along footpaths near settlements.
Regional population effects: The African Civet’s range is continuous with the rest of its African range and there are no major barriers to this species’ dispersal. We therefore assume that there is dispersal across regional boundaries, especially across the Great Limpopo Transfrontier Park (GLTP), and across the Limpopo Province with Botswana and Zimbabwe. However, within the assessment region, an increase in predator-proof fencing across the game ranching areas in Limpopo, Mpumalanga and North West provinces will severely obstruct civet dispersal.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Venter JA & da Silva JM. 2025. A conservation assessment of Civettictis civetta. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
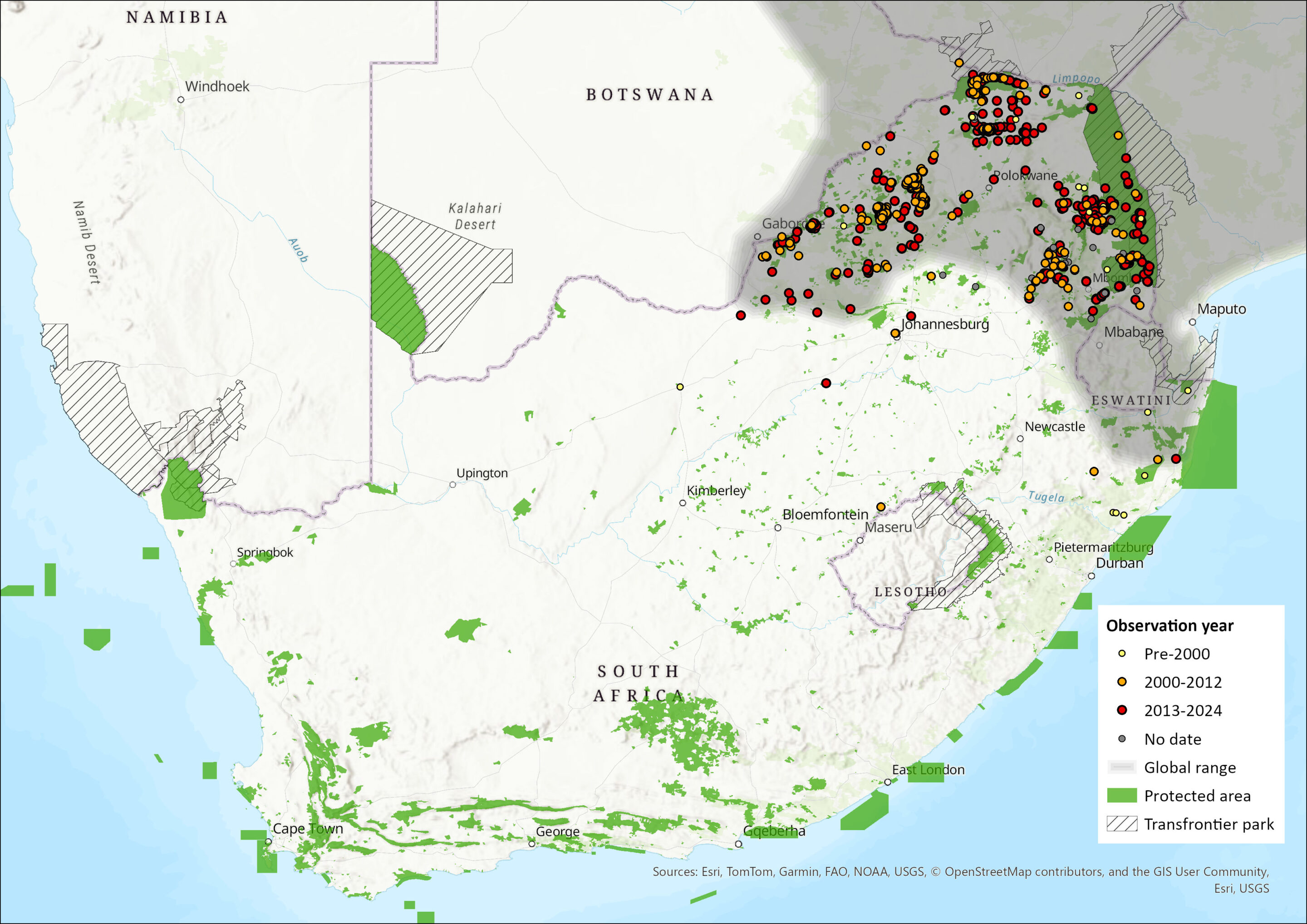
Regional Distribution and occurrence
Geographic Range
The African Civet is widely distributed across Africa, ranging from Senegal and Mauritania eastwards to southern Sudan, Ethiopia, Djibouti, and southern Somalia, and extending southwards through all countries to northeastern Namibia, northern and eastern Botswana, and northeastern South Africa (Ray 2013). It is also present on Zanzibar Island (Pakenham 1984; Stuart & Stuart 1998) and São Tomé Island (Dutton 1994). The species occurs from near sea level up to 5,000 m asl on Mt. Kilimanjaro (Moreau 1944).
Within the assessment region, the species occurs in the northern parts of South Africa, including northern KwaZulu-Natal, Mpumalanga, Limpopo, and the northern North West Province (Skinner & Chimimba 2005). Its dispersal routes are unknown but are presumed to be contiguous within its range. Camera-trapping across Limpopo and Mpumalanga indicates that the species is relatively common (L. Swanepoel unpubl. data), but its distribution and abundance in other provinces remain uncertain. Recent records also document its occurrence in the Free State, although these may reflect escapees from game farms rather than natural populations (N.L. Avenant pers. comm. 2016). In the North West Province, a recent camera-trap record from the Molopo near Mafikeng represents the most southerly record in the province since the previous assessment and corresponds with several other new records further south than those documented a decade earlier (see Map).
Although Power (2014) estimated a 36% contraction in extent of occurrence (EOO) in the North West Province since 1983, this area represents the species’ range edge, where expansions and contractions are difficult to measure accurately. Historically, civets were reported from the Magaliesberg but appear to be absent there since the 1980s (Rautenbach 1978), while at the same latitude further west they remain present in the Marico Biosphere Reserve (see Map). In the North West Province, the species has not been recorded south of 25°30′S, but it is common in Madikwe Game Reserve and Borakalalo National Park, though rare in Pilanesberg National Park (Power 2014). In Limpopo, strong populations are found in the Waterberg and Soutpansberg/Vhembe districts across both protected and farming landscapes. In KwaZulu-Natal, the species appears to have a patchy distribution, with rare detections in reserves such as Phinda and Thanda (L.H. Swanepoel unpubl. data). Notably, recent extensive camera-trapping in several Zululand reserves, including Tembe Nature Reserve, failed to detect the species despite it being within their known range (Pretorius 2019). In Eswatini (formerly Swaziland), Skinner & Chimimba (2005) and Monadjem (1998) record the species from the middleveld and Lubombo regions, particularly in the lower eastern areas (Monadjem & Mahlaba 2000). It does not occur in Lesotho (Lynch 1994).
Overall, the African Civet remains present in the northern savannas of South Africa, with stable populations in Limpopo and Mpumalanga, localized occurrences in KwaZulu-Natal, and possible range expansion along the western margin in the North West Province.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m
Elevation Upper Limit (in metres above sea level): 5000 m
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for African Civet (Civettictis civetta) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: No
FAO Area Occurrence
FAO Marine Areas: No
Climate change
Although specific CCVA studies are lacking for African Civet, Red List criteria can capture climate-related extinction risk through observed symptoms (e.g., population or range reductions) and can be informed by trait-based expert assessment where models are unavailable. For a nocturnal, omnivorous mesopredator of wooded savannas, plausible mechanisms include altered fruit and invertebrate availability under warming/drying and changed fire regimes; increased disease/parasite risk; and indirect effects via prey, competitors and habitat structure. We judge near- to mid-term impacts as low–moderate plausibility, localised scope (wooded savanna zones), and low–moderate severity given the species’ dietary breadth, with risk potentially elevated where habitat fragmentation limits movement or climate refugia access. We recommend periodic reassessment with SDMs to quantify projected range change.
Population
Within the assessment region, density estimates vary across land uses. In the Waterberg of Limpopo, spatial mark–recapture models estimated African Civet densities at 14.11 ± 4.15 individuals / 100 km² in protected areas without African Lions (Panthera leo) (e.g. Lapalala Wilderness), 11.39 ± 5.52 individuals / 100 km² in game farming areas (e.g. the Waterberg Mountains), and 6.42 ± 1.99 individuals / 100 km² in protected areas with lions (e.g. Welgevonden Private Game Reserve) (Isaacs et al. 2022). These findings concur with earlier camera-trap studies in Limpopo, which estimated 10.1 ± 0.56 and 9.04 ± 0.82 individuals / 100 km² in Mogalakwena Game Reserve and 14.18 ± 0.12 individuals / 100 km² in Moyo Conservation Project Reserve (both in the Alldays area; Amiard 2014). Similarly, densities of ~15 individuals / 100 km² were reported in the Soutpansberg (A. Thomissen unpubl. data). By contrast, farmland areas such as Platjan, Limpopo, supported lower densities of 3.6 ± 0.81 individuals / 100 km² (Faure et al. 2022).
In Mpumalanga, camera-trap studies in the mountainous Greater Lydenburg area revealed healthy populations with home ranges of 3–5 km² (G. Camacho unpubl. data). Population density at Thaba Tholo Wilderness Reserve was estimated at 12.52 individuals / 100 km² (Amiard 2014), consistent with other findings.
Overall, these estimates indicate that civet populations remain healthy in protected areas, but densities are lower outside them, reflecting patterns observed in many large carnivores. Multiple anthropogenic factors likely contribute to local declines. First, evidence from Waterberg studies (Isaacs 2016; Isaacs et al. 2022) suggests top-down regulation by large carnivores (sensu Prugh et al. 2009), where high predator densities in small, fenced reserves may suppress civet populations through mesopredator suppression. Second, snaring is an important source of mortality, particularly in rural landscapes of Mpumalanga and Venda (L.H. Swanepoel & G. Camacho unpubl. data). Civets are especially vulnerable as they regularly use footpaths and scavenge carrion, increasing the risk of being caught in bushmeat snares. For example, in the Vhembe District Municipality of Limpopo, a 60-day camera-trap survey (1,060 trap nights across a 1,500 ha area) recorded no civets near villages or urban areas, suggesting they are removed directly or indirectly (Williams et al. 2018). Civets are also frequently present on livestock farms, where they may be incidentally killed during poisoning campaigns targeting larger carnivores.
Finally, there is currently no data on legal removals of civets through local or international trophy hunting. However, expanding human settlements along the edges of protected areas (Wittemyer et al. 2008) are expected to exacerbate threats and could drive localized subpopulation declines or even extirpations. More research is needed to establish overall population trends within the assessment region.
Population Information
Current population trend: Unknown
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: No. They have a broad habitat tolerance and can exist in agricultural and rural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
To date, only one phylogenetic study has been conducted on this species, which reported that the Civettictis–Viverra clade, to which the African Civet belongs, diverged from Viverricula approximately 16.2 million years ago (Gaubert & Cordeiro-Estrela 2006). Although no population genetic data are currently available, the species appears to occur as a single metapopulation within the assessment region, with a largely contiguous distribution that is likely connected to populations in neighbouring countries. Under the Convention on Biological Diversity’s population genetic indicators, this suggests that all populations are maintained (one continuous metapopulation; PM indicator = 1/1 = 1.0). While density estimates exist for several localities (see Population section), the absence of an estimate of Area of Occupancy (AOO) prevents calculation of a total census size for this metapopulation. As a result, the effective population size (Ne 500 indicator) cannot be quantified at present.
Habitats and ecology
African Civets occupy a wide variety of habitats, particularly closed environments such as secondary forest, woodland, and bush. They are generally absent from arid regions, except where riverine systems provide sufficient cover and resources. In South Africa, the species occurs mainly within the Savannah Biome, with marginal extension into parts of the Grassland Biome. Wooded landscapes are preferred over open grasslands, and riverine areas with tributaries and rocky outcrops are especially favoured. In Eswatini, they are recorded from broadleaved woodland (Monadjem & Mahlaba 2000). Civets typically rest during the day in dense vegetation such as thickets and tall grasses, among tangled roots or under logs, and occasionally in burrows dug by other animals (Ray 2013).
The species is an omnivorous and opportunistic forager (Ray & Sunquist 2001; Bekele et al. 2008b; Amiard 2014), consuming both wild and cultivated foods. Diet items include cereals (maize, wheat, barley) and domestic fruits (e.g., bananas, figs, olives; Bekele et al. 2008b). They readily feed on toxic prey such as millipedes (Smithers & Wilson 1978) and scorpions, while wild fruits—particularly Grewia spp.—and invertebrates such as millipedes (Archispirostreptus gigas) dominate their diet in the Limpopo and Musina regions (Amiard 2014; Amiard et al. 2015). Vertebrates (rodents, birds, carrion) are eaten less frequently. By ingesting and dispersing large numbers of seeds, civets likely play an important ecological role as seed dispersers in savanna ecosystems (Amiard et al. 2015).
African Civets are largely terrestrial, nocturnal, and solitary, except during the breeding season when small groups may be observed. Movement ecology studies from Ethiopia report variation in home range size: a subadult male in Bale Mountains National Park used 11.1 km² (Admasu et al. 2004), while at Wondo Genet an adult male (0.74 km²) and subadult female (0.82 km²) had much smaller ranges, moving at an average of 326 m/hour and travelling 1.3–4.2 km nightly (Ayalew et al. 2013). No spatial ecology studies have yet been conducted in southern Africa.
When disturbed, civets often move slowly and may freeze or lie down motionless (Skinner & Chimimba 2005).
Ecosystem and cultural services: Civet species play an important role in seed dispersal, especially in forests of Asia and probably Africa too (Pendje 1994 but see Abiyu et al. 2015). Civets play an important role in dispersing seeds of key plant species (Amniard et al 2015). Additionally, African Civets have historically been the main animal species from which a musky scent could be extracted and used in perfumery (see Use and Trade).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
Life History
Generation Length: 6 Years (Do Lihn San et al. 2019)
Age at Maturity: Female or unspecified: ~12 months (first litters ~14 months).
Age at Maturity: Male: ~7 months (213 days; captive data)
Size at Maturity (in cms): Female: Adults are 67–89 cm head–body (tail 34–47 cm); sexes similar
Size at Maturity (in cms): Male: (Not specified)
Longevity: Up to 28 years in captivity (wild unknown).
Average Reproductive Age: ~1.2 years (first breeding around 14 months in captivity)
Maximum Size (in cms): Head–body up to ~89 cm (tail to ~47 cm; shoulder height ~40–52 cm).
Size at Birth (in cms): Not reported; birth mass ~300–380 g
Gestation Time: ~60–71 days (mean ~65 d; some sources to 60–81 d)
Reproductive Periodicity: Polyestrous; typically 2–3 litters/year
Average Annual Fecundity or Litter Size: Mean litter size ~2.5; ~2.2 litters/year (captive averages)
Natural Mortality: (no robust wild annual mortality estimates located)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: No
Systems
System: Terrestrial
General Use and Trade Information
In addition to their prevalence in bushmeat markets in West and Central Africa (see Threats), African Civets are economically important for their perineal gland secretion (civet musk or “civet”), which has been harvested for centuries as a fixative (“civetone”) in the perfume industry (Randall 1979; Bekele et al. 2008a; Wondmagegne et al. 2011). Although synthetic alternatives have been available for nearly 70 years (Rosevear 1974), civetone remains a significant export commodity in several countries, particularly Ethiopia, and to a lesser extent Niger and Senegal (Ray 1995, 2013; Abebe 2003; Ray et al. 2005). Between 1985 and 1997, “civiculture” (civet farming) generated annual revenues of approximately US$150,000–835,000 in Africa. In Ethiopia, more than 200 registered civet farmers capture wild civets and maintain several thousand males in captivity for musk production, as males produce greater quantities and higher-quality musk (Kumera 2005). Breeding in captivity has not yet been attempted (Tolosa & Regassa 2012). Only about 2% of civet musk is consumed domestically, with the remainder exported, primarily to France (85%) for perfume, and in smaller quantities to Arabian countries for medicinal uses and to India for the tobacco industry (Girma 1995; Tamiru 1995). To date, civet farming is not practiced within the assessment region.
In South Africa, African Civets are increasingly hunted for trophies, especially in Limpopo. CITES export data indicate that an average of 104 ± 37 (n=1464) hunting trophies, 7 ±4 (n=95) personal effect and 3 ±3.5 (n=30) commercial effects were exported annually between 2010 and 2023. The South African Professional Hunter register database indicate an average of 46 ± 22 (n=645) animals hunted during the same period. Given the estimated densities outside protected areas, this harvest is currently thought to have low impact, although quotas do not account for additional non-hunting mortality or local hunting related mortality. Civet skins have also been confiscated in the Northwest Province (Power 2014), suggesting some local trade, although likely limited due to the species’ marginal distribution in the area.
Information on the use of African Civets in the traditional medicine trade within the assessment region is scarce. While the species is widely used for medicinal purposes in other parts of Africa, it appears to be uncommon in South Africa. For example, a survey of Johannesburg’s Faraday market recorded very few civet products (Whiting et al. 2013).
Wildlife ranching may support civet conservation by maintaining suitable habitats; however, two risks exist. First, carnivore persecution on game ranches can result in accidental civet deaths, particularly during poisoning campaigns aimed at larger predators. Second, the increasing use of predator-proof fencing to protect valuable game species may restrict civet movements between properties, potentially leading to population fragmentation and localised inbreeding.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | – | – | – |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 6. Other chemicals | – | – | true | – |
| 10. Wearing apparel, accessories | true | true | – | – |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major range-wide threats to African Civets within the assessment region, but several emerging pressures have been identified since the last assessment. Civets are sometimes poisoned directly by landowners for livestock or crop protection, or indirectly through persecution programmes targeting other damage-causing carnivores. Because they are nocturnal and frequently use roads and footpaths, civets are particularly vulnerable to snares and vehicle collisions. In South Africa, roadkill is a significant cause of mortality, especially in Mpumalanga and Limpopo, where they ranked among the top three mammal roadkill species in the Greater Mapungubwe Transfrontier Conservation Area; 16 individuals were recorded killed in just 120 days, suggesting annual mortalities of up to 50 individuals locally (Collinson 2013; Collinson et al. 2015).
The species is also subject to trophy hunting, but it is unknown whether current quotas are sustainable or detrimental to populations. Further research is required to evaluate the impact of trophy harvests, especially in combination with incidental mortalities.
Across the broader African range, civets are commonly exploited for bushmeat. They are frequently sold in markets, particularly in West and Central Africa, and are among the most abundant species traded in southeastern Nigeria (Angelici et al. 1999; Ray 2013). In Eswatini, they have also been reported in the bushmeat trade (Monadjem 1998). While not a prevalent food source in South Africa, exploitation may increase if traditional bushmeat species become scarce.
African Civets host a wide variety of parasites, including gastrointestinal helminths, cestodes, blood parasites, and ectoparasites such as ticks and fleas (Ahmed et al. 2017). They are incidental hosts of rabies (Markotter et al. 2013; Umoh et al. 1983; Sabeta et al. 2020) and have experienced fatal outbreaks of canine distemper virus (CDV) in captivity (Chaiyasak et al. 2015). Bacterial pathogens such as Salmonella and Klebsiella have also been recorded in civets and other viverrids (Wicker et al. 2017). While disease is not currently a major driver of decline, it may increase mortality risk where populations are fragmented or in close contact with humans.
Finally, evidence suggests that high densities of reintroduced large carnivores, particularly Lions, may suppress civet populations through mesopredator regulation (Isaacs et al. 2022).
Overall, roadkill represents the most significant documented threat to African Civets in the assessment region, although incidental mortality from snares, persecution, poisoning, and pressures in agricultural landscapes also contribute to local declines.
Conservation
The African Civet occurs in numerous protected areas across its northern range within the assessment region, including the Greater Mapungubwe Transfrontier Conservation Area, Marakele National Park, Blyde River Canyon Nature Reserve, Pilanesberg National Park, Madikwe Nature Reserve, and Kruger National Park, which holds the largest protected subpopulation. The national population is likely connected to those in Botswana, Zimbabwe, and Mozambique. In Botswana, the species is listed on CITES Appendix III. Guidance for sustainable musk harvest, if this practice were ever to become relevant in South Africa, is available in Abebe (2003).
Educational and awareness campaigns should be developed to reduce persecution by landowners and to improve the species’ profile among the general public. The trophy hunting industry should be closely monitored, particularly with respect to the age, sex, and location of harvested animals. Formation of conservancies should be incentivised to promote connectivity, while the use of permeable fences—such as those tested in Namibia with rubber tyre modifications to allow wildlife movement (Weise et al. 2014)—should be trialled locally. Roadkill monitoring is also recommended to assess ecological impacts on local populations (Collinson et al. 2015), noting that habitat use and movement patterns will influence the location and effectiveness of mitigation measures.
Recommendations for Land Managers and Practitioners:
- No dedicated management plan is currently required, but local monitoring programmes should be established to track population size and trends.
- Monitor the impacts of trophy hunting, trade, traditional medicine use, and bushmeat hunting.
- In reserves with Lions, stocking should be kept at conservative levels to minimise unnatural mesopredator suppression of civet populations.
Research Priorities:
- Estimate population sizes in protected and non-protected areas across the species’ range, using by-catch data (e.g. Panthera’s annual Leopard (Panthera pardus) surveys).
- Assess the impact of harvest on population persistence (e.g. comparing hunted vs. non-hunted areas).
- Document population demographics.
- Study spatial ecology, particularly the impacts of predator-proof fencing on movement and population structure.
- Evaluate the role of African Civets in seed dispersal as an ecosystem service.
- Quantify the prevalence of civet body parts in traditional medicine markets.
- Assess the impacts of snaring on populations.
- Ongoing research which includes studies on population density in the species distribution range.
Encouraged Citizen Actions
- Report sightings and roadkill on virtual museum platforms such as iNaturalist and MammalMAP, especially outside protected areas.
- Landowners are encouraged to form conservancies or employ permeable fences to maintain connectivity for civets.
Bibliography
Abebe, Y.D. 2003. Sustainable utilization of the African Civet (Civettictis civetta) in Ethiopia. In: B.W. wa Musiti (ed.), 2nd Pan-African Symposium on the Sustainable Use of Natural Resources in Africa, pp. 197-207. IUCN, Gland, Switzerland and cambridge, UK.
Abiyu A, Teketay D, Glatzel G, Gratzer G. 2015. Tree seed dispersal by African civets in the Afromontane Highlands: too long a latrine to be effective for tree population dynamics. African Journal of Ecology 53: 588–591.
Admasu, E., Thirgood, S.J., Bekele, A. and Laurenson, M.K. 2004. A note on the spatial ecology of African civet Civettictis civetta and common genet Genetta genetta in farmland in the Ethiopian Highlands. African Journal of Ecology 42: 160-162.
Ahmed, I., Sibhat, B., & Abera, B. 2017. Gastrointestinal and blood parasites of African civet (Civettictis civetta) in Limmu, Southwestern Ethiopia. Asian Journal of Animal Sciences, 11(4), 194–201. https://scialert.net/fulltext/?doi=ajas.2017.194.201
Amiard, P. 2014. Ecology of the African Civet (Civettictis civetta) in Three Different Vegetation Types of South Africa: Study of the Population Density, the Habitat Use and the Diet. Master Thesis, University of Reims Champagne-Ardenne, Reims, France.
Amiard, P.J., Kruger, C.V., Mullers, R.H. and Schipper, J., 2015. The diet of African Civet Civettictis civetta in two vegetation types of the Savannah biome in South Africa. Small Carnivore Conservation, 52, pp.4-12.
Angelici, F.M., Luiselli, L., Politano, E. and Akani, G.C. 1999. Bushmen and mammal fauna: A survey of the mammals traded in bush-meat markets of local people in the rainforests of southeastern Nigeria. Anthropozoologica 30: 51-58.
Anonis, D.P. 1997. Animal notes in perfumery: civet and civet compounds. Perfumer and Flavourist 22: 44-47.
Ayalew, B., Afework, B. and Balakrishnan, M. 2013. Home range and movement patterns of African civet Civettictis civetta in Wondo Genet, Ethiopia. Small Carnivore Conservation 48: 83-86.
Bahaa-el-din, L., Henschel, P., Aba’a, R., Abernethy, K., Bohm, T., Bout, N., Coad, L., Head, J., Inoue, E., Lahm, S., Lee, M. E., Maisels, F., Rabanal, L., Starkey, M., Taylor, G., Vanthomme, A., Nakashima, Y. and Hunter, L. 2013. Notes on the distribution and status of small carnivores in Gabon. Small Carnivore Conservation 48: 19-29.
Bekele, T., Afework, B. and Balakrishnan, M. 2008a. Scent-marking by the African Civet Civettictis civetta in the Menagesha–Suba State Forest, Ethiopia. Small Carnivore Conservation 38: 29-33.
Bekele, T., Afework, B. and Balakrishnan, M. 2008b. Feeding ecology of the African Civet Civettictis civetta in the Menagesha–Suba State Forest, Ethiopia. Small Carnivore Conservation 39: 19-24.
Chaiyasak, S., Techangamsuwan, S., Rungsipipat, A., Banlunara, W., & Rattanasrisomporn, J. (2015). Fatal canine distemper virus infection in captive African civets (Civettictis civetta) in Thailand. Veterinary Pathology, 52(2), 329–332. https://journals.sagepub.com/doi/full/10.1177/0300985814551580
Coetzee, C.G. 1977. Order Carnivora. Part 8. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-42. Smithsonian Institution Press, Washington, DC, USA.
Collinson WJ. 2013. standardised protocol for roadkill detection and the determinants of roadkill in the Greater Mapungubwe Transfrontier Conservation Area. M.Sc. Thesis. Rhodes University.
Collinson, W.J., Reilly, B.K., Parker, D.M., Bernard, R.T. and Davies-Mostert, H.T., 2015. An inventory of vertebrate roadkill in the greater Mapungubwe Transfrontier conservation area, South Africa. African Journal of Wildlife Research, 45(3), pp.301-311.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Monadjem, A., & Mahlaba, T. A. A. 2000. A mammal survey of the lower Usuthu Basin, Swaziland. UNISWA Research Journal of the Agriculture, Science and Technology, 4, 55-61.
Dutton, J. 1994. Introduced mammals in São Tomé and Príncipe: possible threats to biodiversity. Biodiversity and Conservation 3: 927-938.
Ellerman, J.R., Morrison-Scott, T.C.S. and Hayman, R.W. 1953. Southern African Mammals 1758 to 1951: A Reclassification. British Museum (Natural History), London, UK.
Faure, J. P. B., Swanepoel, L. H., Cilliers, D., Venter, J. A., & Hill, R. A. 2022. Estimates of carnivore densities in a human-dominated agricultural matrix in South Africa. Oryx, 56(5), 774-781.
Gaubert, P. and Cordeiro-Estrela, P., 2006. Phylogenetic systematics and tempo of evolution of the Viverrinae (Mammalia, Carnivora, Viverridae) within feliformians: implications for faunal exchanges between Asia and Africa. Molecular Phylogenetics and Evolution, 41(2), pp.266-278.
Girma, G. 1995. Musk trade and export. Proceedings of the Civet Farming, Musk Production and Trade Workshop: 45-53. Ethiopian Wildlife Conservation Organisation. Addis Ababa, Ethiopia. (In Amharic).
Isaacs L. 2016. Estimating African civet Civettictis civetta (Schreber 1776) density and occupancy in a mosaic of land uses in the Waterberg district, South Africa. M.Sc. Thesis. University of Pretoria.
Isaacs, L., Somers, M. J., & Swanepoel, L. H. 2022. Density of African civets in a moist mountain bushveld region of South Africa. Small Carnivores: Evolution, Ecology, Behaviour, and Conservation, 249-258.
Kingdon, J. 1997. The Kingdon Field Guide to African Mammals (first edition}. Academic Press, San Diego, California, USA.
Kumera, W. 2005. Better handling of African Civet for quality musk extraction. Agriculture and Rural Development Journal 2: 21-34. [In Amharic].
Lynch, C.D., 1994. The mammals of Lesotho: order Carnivora: family Canidae. Navorsinge van die Nasionale Museum: Researches of the National Museum, 10(4), pp.199-209.
Mateos, E., Zerihun, G., Yosef, M. and Megersa, D. 2015. Community attitude towards African Civet Civettictis civetta conservation in eastern sub-catchment of Lake Hawassa basin, Southern Ethiopia. Discovery 27(96): 2-7.
Markotter, W., Kuzmin, I. V., Rupprecht, C. E., & Nel, L. H. 2013. Lyssaviruses and rabies: African civets as incidental hosts. Viruses, 5(10), 2679–2690. https://pmc.ncbi.nlm.nih.gov/articles/PMC7232503/
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Moreau, R.E. 1944. Kilimanjaro and Mount Kenya: Some comparisons with special reference to the mammals and birds; and with a note on Mount Meru. Tanganyika Notes and Records 18: 28-68.
Monadjem, A., 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe, 41(2), pp.45-59.
Pakenham, R.H.W. 1984. The Mammals of Zanzibar and Pemba islands. Printed Privately, Harpenden.
Pendje G. 1994. Fruit consumption and seed dispersal by the African civet Civettictis civetta in Mayombe, Zaire. evue d’Écologie – La Terre et la Vie 49: 107–116.
Pocock, R.I. 1915. On the feet and glands and other external characters of the Viverrinae with the description of a new genus. Proceedings of the Zoological Society of London 1915: 131-149.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Pretorius, M., 2019. Mesocarnivores in Protected Areas: ecological and anthropogenic determinants of habitat use in northern Kwa-Zulu Natal, South Africa. MSc Dissertation, University of Cape Town.
Prugh LR, Stoner CJ, Epps CW, Bean WT, Ripple WJ, Laliberte AS, Brashares JS. 2009. The rise of the mesopredator. BioScience 59: 779–791.
Randall, R.M. 1979. Perineal gland marking by free-ranging African Civets, Civettictis civetta. Journal of Mammalogy 60: 622-627.
Rautenbach, I.L. 1978. The mammals of the Transvaal. Ph.D. Thesis, University of Natal.
Ray, J.C. 1995. Civettictis civetta. Mammalian Species 488: 1-7.
Ray, J.C. 2013. Civettictis civetta African civet. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 255-259. Bloomsbury, London, UK.
Ray, J.C. and Sunquist, M.E. 2001. Trophic relations in a community of African rainforest carnivores. Oecologia 127: 395-408.
Ray, J.C., Hunter, L. and Zigouris, J. 2005. Setting conservation and research priorities for larger African carnivores. Wildlife Conservation Society, New York, USA.
Rosevear, D.R. 1974. The Carnivores of West Africa. Trustees of the British Museum (Natural History), London, UK.
Sabeta, C.T., Marston, D.A., McElhinney, L.M., Horton, D.L., Phahladira, B.M. and Fooks, A.R. 2020. Rabies in the African civet: an incidental host for lyssaviruses?. Viruses, 12(4), p.368. March 18, 2025, at 9:30 AM
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. and Wilson, V.J. 1979. Check List and Atlas of the Mammals of Zimbabwe Rhodesia. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Stuart, C. and Stuart, T. 1998. A note on the herpestids and viverrids of south-eastern Unguja (Zanzibar) Island. Small Carnivore Conservation 18: 16-17.
Tamiru, G. 1995. Civet quality control. Proceedings of the Civet Farming, Musk Production and Trade Workshop: 40-44. Ethiopian Wildlife Conservation Organisation. Addis Ababa, Ethiopia.
Tolosa T, Regassa F. 2012. The husbandry, welfare and health of captive African civets (Vivera civetica [sic]) in western Ethiopia. Poster presented at the 61st International Conference of the Wildlife Disease Association and the 10th Biennal Conference of the European Wildlife Disease Association.
Umoh, J. U., Blenden, D. C., & Durojaiye, O. A. 1983. Rabies in a civet cat (Civettictis civetta) in the Jos Zoo, Nigeria. Tropical Animal Health and Production, 15(2), 113–114. https://www.researchgate.net/publication/19965675
Weise FJ, Wessels Q, Munro S, Solberg M. 2014. Using artificial passageways to facilitate the movement of wildlife on Namibian farmland. South African Journal of Wildlife Research 44: 161-166.
Wicker, L., Canuti, M., & Langwig, K. 2017. Zoonotic bacteria and neglected tropical diseases in viverrids: a review. Zoonoses and Public Health, 64(7), 431–444. https://pmc.ncbi.nlm.nih.gov/articles/PMC7165938/
Williams, S.T., Maree, N., Taylor, P., Belmain, S.R., Keith, M. and Swanepoel, L.H., 2018. Predation by small mammalian carnivores in rural agro-ecosystems: An undervalued ecosystem service? Ecosyst. Serv. 30, 362–371 [online]
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.
Whiting, M.J., Williams, V.L. and Hibbitts, T.J., 2013. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Animals in traditional folk medicine: implications for conservation, pp.421-473.
Wondmagegne, D., Afework, B., Balakrishnan, M. and Gurja, B. 2011. Collection of African Civet Civettictis civetta perineal gland secretion from naturally scent-marked sites. Small Carnivore Conservation 44: 14-18.
Wozencraft, W.C. 2005. Order Carnivora. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World: A taxonomic and geographic reference. Third Edition, pp. 532–628. Smithsonian Institution Press, Washington, DC, USA.